Abstract
This article considers data derived from auto/biographical interviews with 10 participants in the United States and United Kingdom at risk of rare familial early-onset dementias. Previous research utilising such approaches has detailed the impact parental young onset dementia (YOD) has on young people’s familial relationships, and life courses. There is a need to understand the experiences of people at risk of familial dementias, paying attention to decisions around whether to pursue genetic testing and make use of Assisted Reproductive Technologies. Surfacing these narratives is timely given the possibilities for genetic testing which offers challenges and opportunities for the first generation making these choices. Familial dementia and genetic testing feature in intimate relationships due to the impact on dating, couple life courses and reproductive choices. We conceptualise the road to testing – or not – as an intimate practice. Data suggest that wishes to have children or not inform testing decisions and highlight societal ideals around having children and the moral imperatives attached to testing. Findings indicate a need for timely testing, improved awareness in general medical practice, and tailored psychosocial support.
Introduction
Scientific developments have raised the possibility of identifying the genetic causes of a multitude of diseases including cystic fibrosis, cancers, Huntington’s Disease and dementias. In addition, Assisted Reproductive Technologies (ARTs) may halt further inheritance of such diseases through embryo selection. Dementia is the umbrella term for symptoms indicative of deteriorating brain function and encompasses multiple variants with differing presentations. It is thought to affect 55 million people worldwide, with projections in the region of 100 million by 2050 (Alzheimer’s International, 2023). Although commonly equated with memory loss in older people, rare genetic variants of this degenerative and terminal condition can also affect younger people. Variants including frontotemporal, vascular and early-onset familial dementias (Alzheimer’s Society, 2021) are thought to account for 5% of cases and can present from late 20s and early 30s onwards (Andrade-Guerrero et al., 2023). Research is desperately seeking a cure through clinical trials involving human participants (Benzinger, 2025).
Screening can detect the likelihood of developing dementia, with a blood test anticipated within 5 years (Alzheimer’s Research UK, 2024). Concurrent to this is the commodification of genetics and consumer-led technology exemplified by ‘direct-to-consumer’ kits (e.g. 23andme), marketed as a novel way to access genetic information, including ancestry, health and disease. Although media reports suggest their capacity for detecting dementia risk, the complexities of dementia diagnosis means they lack accuracy (Alzheimer’s Society, 2021). They also lack social support such as pre-testing counselling in medical settings. They are neither recognised or recommended by leading dementia organisations (including the Alzheimer’s Association and Alzheimer’s Society), scientists and medics. In addition, research identifies the emotional impact of ‘direct-to-consumer testing’, including the ramifications for families’ relationships, access to donor identities and the importance for supportive, planned disclosure (Gilman et al., 2024). Concerns have previously been raised over the appropriateness of learning about dementias, including familial ones, through such means (Hanganu et al., 2022), highlighting ethical concerns, privacy, health insurance implications, the need for social support and a role for clinicians in interpreting results particularly in light of the lack of treatment or cure (Arias et al., 2022). While DTC testing for dementia was not the focus of the research, it is part of the context. Documenting experiences of testing in clinical settings illuminates the dilemmas that accompany diagnosis, adding to research which considers the implications of DTCtesting.
Beyond clinical studies of hereditary variants of dementia, research has tended to focus on caregiving. Garcia-Toro et al (2020) conducted interviews to understand the experience of caregiving in families with a risk of developing the same early-onset dementia and highlighted anxiety over the disease, rather than caring specifically. However, it should be noted that this research focused on psychological concepts such as anxiety, depression, resilience and self-efficacy. Our study contributes to familial experiences of dementia and genetic testing through a sociological lens. Biographical interviews with participants predominantly in the United States of America, consolidated by a small number in the United Kingdom illuminate experiences of genetic variants of dementia in families. Potential dementia has the capacity to impact life course planning, intimate relationships including immediate kin and beyond, adding to the hardships of parental dementia. Our research is timely as this is the first generation who have the potential to access information about their own risk of familial dementia. Understanding why individuals pursue testing (or not), and how they navigate outcomes for themselves, family members and intimate partners, current and future, provides crucial insights into dementia. Data are viewed through a lens of ‘intimate practices’ which ‘enable, generate and sustain a subjective sense of closeness and being attuned and special to each other’ (Jamieson, 2011). Accordingly, we offer understandings of the impact of health on intimate relationships intertwined with conceptualisations of the life course. Findings suggest the impact of familial dementias on individual’s personal lives, prompting new intimate practices and contesting temporalities for couples and would-be parents. Perspectives on testing are shaped by constructions of families, relationships and the life course. These are of significance for social perspectives on dementia and those supporting individuals affected by familial dementia.
Inheritable disease, relationships and the life course
Research has documented familial relationships in the context of inheritable diseases including breast cancer (Hesse-Biber, 2014), Huntington’s (Cox and McKellin, 1999) and Cystic Fibrosis (Boardman and Clark, 2022). Findings indicate familial connectedness and tensions related to illness disclosure, testing processes and receiving different results (Dimond et al., 2022; Douglas et al., 2009; Maxted et al., 2014). Despite commonalities – notably Huntington’s – there are distinct hardships for each disease. Dementia is unique to conditions which may have preventive treatment options, for example, breast cancer (Dimond et al., 2022). Although ART inhibits the disease at conception, there is no stopping its development beyond this. Furthermore, the conflation of dementia with Alzheimer’s provokes age-related stigma and an emphasis on memory loss, resulting in ignorance to other symptoms and it’s terminal nature. These may impede public sympathy to familial dementias in younger age groups (Low and Purwaningrum, 2020). Predictive testing for Huntington’s has been possible since 1993, compared with 2017 for familial dementias.
Dimond et al (2022) conceptualise ‘entanglements’ which describes relational complexity and that accessing information regarding risk rests on familial relationships for disclosure of illnesses including Huntington’s and breast cancer. Extending this and focusing on dementia, we illuminate the role of couple relationships, rather than direct relations. To deepen understandings of familial dementia and how it impacts relationships, we deploy sociological perspectives on the life course and practices of intimate citizenship. This research contributes couple perspectives to a field dominated by family-as-kin. We also reflect on the implications of insurance-based versus public health care systems and attitudes towards ART.
The life course
The ‘life course’ conceptualises individuals’ experiences along a birth to death continuum, with women’s life courses framed around childbearing years. The approach suggests the cultural significance of particular phases (Elder, 1994). However, established ‘master narratives’ or ‘social scripts’ may be limiting and stigmatising. Perceived ‘failure’ to conform indicated by age, gender and social class can cause anguish (Hall and Sikes, 2020). Modernity has supposedly increased individual agency, weakening ‘standard’ biographies previously shaped by gender, social class, ethnicity, geography (Brannen and Nilsen, 2002), with familial obligations superseded by intimate lives based on choice (Giddens, 1991). This has been notable for women whose life courses have been reshaped by increased paid employment and education. Health conditions, including dementia, have a disruptive impact on biographies (Bury, 1982), altering relationships, perspectives and everyday lives (Hall and Sikes, 2020).
Families and intimacies
Studies of relationships and personal life, illuminate the minutiae of family life, theorised as ‘family practices’. Constructions of families in the Western world typically position the heterosexual married couple with children performing sexual, reproductive, socialisation and economic functions, as the bedrock of society. Trends including divorce, lone parenthood, acceptance of homosexuality and the feminisation of the workforce have been cited as a ‘crisis’ in ‘the’ family (Morgan, 1996). Highlighting the diversity of personal life, Morgan (1996) emphasises the relational dimensions of family life: family is not something we ‘are’ but something we ‘do’, encapsulated as ‘family practices’.
In addition, a shift in focus from families to personal life captures a multitude of relationships and intimacies that transcend immediate kin. Intimacies are ‘practices of close association, familiarity and knowledge’ (Jamieson, 2005: 189). Everyday relationships and intimacies – sharing meals, living together, texting – provoke affinities. Personal life encompasses romantic practices, for example, casual dating, committed unions, relationships, sexualities, a conscious decision to conceive a child. Taken-for-granted practices may be disrupted by an experience such as genetic risk for dementia.
This article is grounded in understandings of personal life, theorised as ‘intimate citizenship’ which denotes the agency exercised in personal and private decisions about intimate lives. Plummer (1995: 151) highlights the ‘control – or not – over one’s body, feelings, relationships, access or not to representations, relationships and public spaces, and socially grounded choices about identities, gender experiences, and erotic experiences’. Roseneil at el (2020) add the ‘pro-creative norm’ suggesting the cultural imperative for biological procreation. Intimate relationships, sexuality and the social world assume procreation as the ‘norm’, feeding into social policy, cultural expectations and representations of families and relationships, culminating in a pro-natal society. These are not merely external forces, but can become internalised, provoking shame, guilt and anxiety for those who do not conform (Roseneil et al., 2020). These dominant, societal pro-creative norms impact on individuals, couples and familial testing decisions.
These theorizations contribute to understandings of personal life and couple relationships, the role of a threatened illness in these, and the impact of medical advancements. Parallels can be drawn with other illnesses, particularly those with a stigmatising impact on intimate life, for example, HIV/AIDS, which has seen improved treatment regimens (Roth et al., 2023), and identification of genetic predispositions for certain cancers and preventive strategies (Kerr et al., 2018). The social manifestation of dementia symptoms indicates specific challenges in terms of attack on personhood and the lack of treatment options (Harrison et al., 2019).
Researching dementia narratives
A narrative approach was deployed to elicit the personal stories of individuals who have a parent with dementia attributed to a genetic variant, and who have the option for genetic testing, whether or not they have undergone the test, regardless of outcome. Narrative methodologies encourage participants to freely tell their story openly, in their own words, with limited prompts. They were complementary to a study that addresses a complex and sensitive subject such as dementia and relationships and have previously been used in research in health (Bury, 2001) and relationships (Phoenix et al., 2021).
The authors have forged a network of academics interested in children and young adults’ experiences of parental dementia having published extensively in a field dominated by patient and spousal experiences. We identified a need for exploratory research grounded in sociology (Mel Hall), lived experience of families and dementia (Pat Sikes) and social work (Caroline Gelman). We sought participants from our respective countries – United States (Caroline) and United Kingdom (Pat & Mel) – to broaden our pool and draw comparisons between health care systems. Access to genetic testing and the repercussions of the outcome differ due to the insurance-based US system and the public NHS health care system in the United Kingdom. A positive result impacts US insurance premiums and access to benefits and services, including life insurance. Those affected may be eligible for clinical trials to offset costs, but this has implications for having children (addressed in the findings). The study was shared by existing networks including Youngtimers, an organisation specifically for persons affected by familial dementias and Dementia UK. Our contacts at these organisations circulated a flyer (Appendix 1) via mailing lists. This strategy yielded eight US participants and two from the United Kingdom but regrettably, an even balance was not met. This may be explained by the Youngtimers explicit familial dementia focus and that our primary contact reached out directly to participants. While acknowledging this limitation of this exploratory study, the small comparative element is a novel contribution to dementia research.
Participants were aged 28 to 55, predominantly in their late twenties/thirties (Table 1). The sample includes individuals who are being tested, considering testing, had opted out at present, or who had received positive or negative results. They were not necessarily undergoing genetic testing – a strength of the research – but identified as part of the familial dementia community, highlighting that not everyone at risk pursues testing. Whether, how, and why individuals faced with similar risks made sense of their situation and testing decisions options was an area for exploration (to be discussed in a forthcoming publication). We did not inquire about participants’ sexual identities but infer from analysis that the sample is heteronormative. This is compounded by the overlaps between genetic testing and ART.
Participant overview.
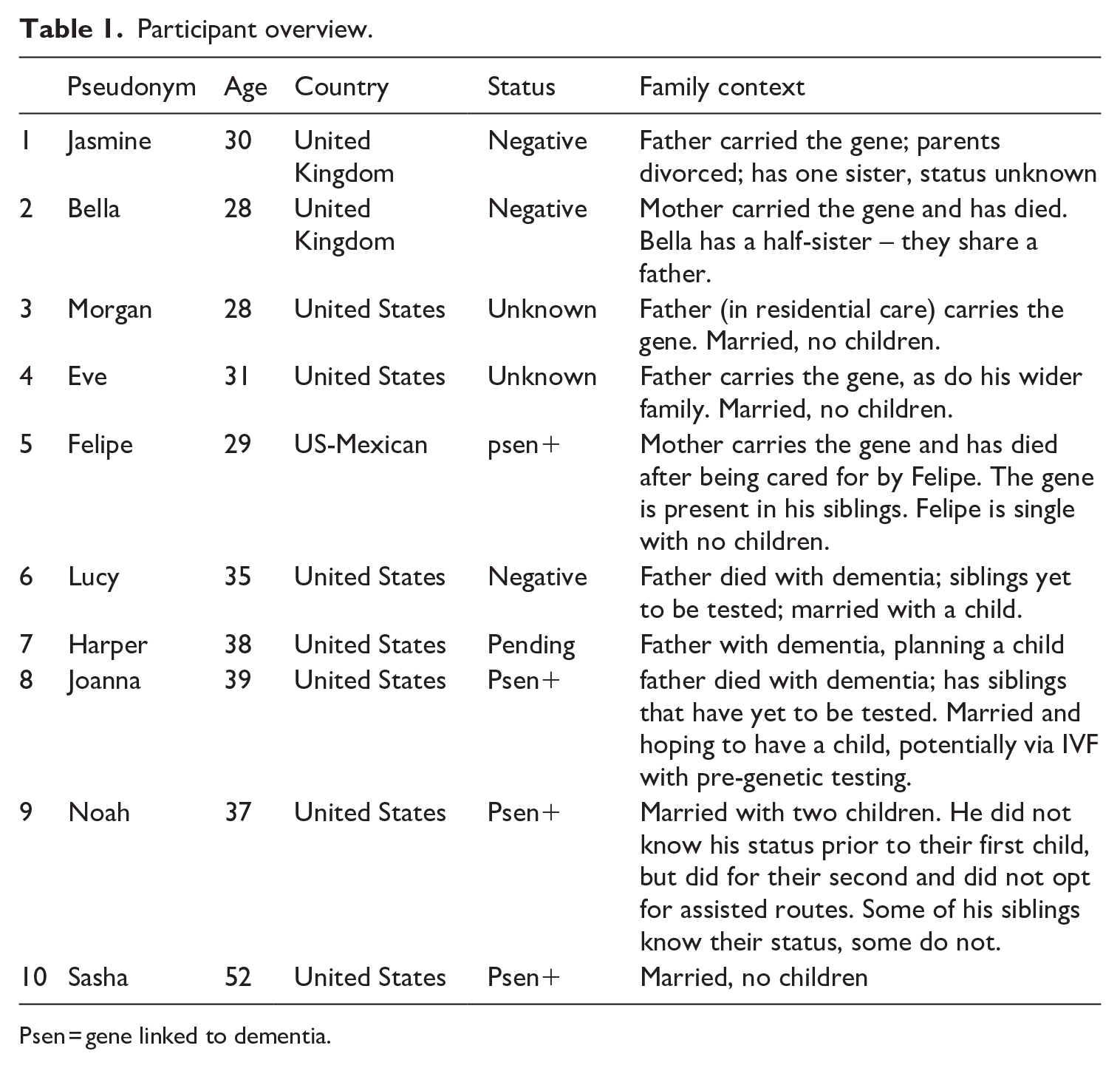
Psen = gene linked to dementia.
To capture perspectives on dementia, genetic testing and results, the narrative approach invited participants to share accounts of their personal and familial experience of dementia. This entailed one or two unstructured conversations lasting 45 minutes to 2 hours. The follow-up design was guided by our previous research to enhance researcher/participant rapport and capture developments over time. While a follow-up interview was offered to all participants, only three engaged with this approach though this reflects participant preferences rather indicative of a study limitation.
The City University New York IRB provided ethical approval. Consent was obtained prior to interviews which were conducted online and transcribed verbatim the research team. Transcripts were read independently by each researcher to perform preliminary analysis, followed by whole team meetings to identify themes in-depth (Braun and Clarke, 2022). This data-driven process used an open approach, allowing for the generation of open codes, code clustering, and thematic coding without imposing a priori themes.
Reflecting on the distinctions between those with negative and positive results has been challenging due to complexities, including that not everyone received their result or planned to pursue testing. Nevertheless, data reveal the complexities of navigating intimate life in the context of familial variants of dementia, which has been hitherto underexplored since research typically tends to patient and carer experience (Chirico et al., 2021). Our analysis unites sociological approaches to personal life and health and illness to understand how relationships are shaped by this condition.
Findings: familial dementia as an intimate practice
Findings explore practices of establishing couple relationships and how dementia impacts these. Narratives illustrate where familial dementia maps onto the temporalities of individual’s lives and their personal relationships which been shaped by the threat of dementia, yielding intimate practices. By the same token, dominant narratives around personal life – for example, pro-natal discourses and ‘good’ parenting governed perspectives on testing. Temporalities were renegotiated because relationships were accelerated by shared experiences, and the advent of testing, correlating with social and biological dimensions of the life course.
Establishing relationships: complexities and opportunities
Personal life tends to be synonymous with couple relationships and in Western contexts, equated with romantic love and choice (May and Nordqvist, 2019). Cultural norms present parameters for couples including a compulsion to pursue or endure romantic relationships presumed to be (hetero)sexual and monogamous. Benchmarks of success include children and marriage. Those who do not conform can encounter stigma (Lahad, 2017) and social injustices, for example, high costs of living for singles (Hall, 2019). ‘Coupling up’ can occur by happenstance or deliberately, for example, online dating. Establishing a relationship necessitates two individuals getting to know one another. Early dating stages may be characterised by sharing life experiences and hopes for the future. Potential dementia may be part of this process, as Felipe describes, When I go on dates and it starts to work out . . . which has happened . . . ‘hey I’m positive with this gene that’s called psen one actually one of the most aggressive genes out there for Alzheimer’s . . . at the age of 40 I’m gonna start declining like my mom’. (Felipe, 29, US)
Felipe illustrates the complexities of dating with knowledge of familial dementia, resonating with research on HIV disclosures in young people’s dating practices (Fair and Albright, 2012) and dating with genetic conditions including BRCA and Huntington’s (Klitzman and Sweeney, 2011) each of which highlighted landmarks and challenges. These include whether to date, how and when to disclose information – as in Felipe’s case – and fear of, or experienced, rejection. There could also be positive aspects. For example, Jasmine – who ultimately tested negative – felt the process had accelerated her relationship with her partner through conversations about having children earlier: It made us a lot closer . . . I noticed how my relationship compared to my friends of the same age has moved a lot quicker than theirs . . . it put in our heads about the having kids thing . . . when I did get the result, we decided we wanted to have kids soon . . . it sparked that conversation. (Jasmine, 30, UK)
Jasmine’s experience illustrates where relationships are infused by temporality. Western culture emphasises the ‘temporal linearity’ and solidification of relationships (Roseneil et al., 2020). Unwritten rules surround the ‘appropriate’ length of time that precede cohabitation and marriage, and – fuelled by pro-natal society – parenthood. Jasmine suggests that normative quantitative measures of temporality were disrupted by dementia. Meanwhile, for Bella, a process like genetic counselling was a milestone which endorsed bonds and, testing confirmed the quality of relationships and partners: The genetic counselling process did make me realise how committed he was to me because of a fear that someone . . . might end up looking after me. We did have a lot of these discussions like, ‘what if it’s positive? Are you just gonna look after me then?’. . . He was like, ‘Well, yeah, I’m not gonna break up with you . . . we’ll work around it’. (Bella, 28, UK)
Jasmine’s and Bella’s accounts suggest a normative relationship pace and that standardised temporalities can be displaced by trials that cement affinities (Layder, 2015). Felipe, Jasmine and Bella illuminate the challenges inherited illness such as dementia can present for dating, for example, knowing when and how to divulge the information and anxieties or anguish of negative responses. However, navigating dementia testing within a couple context also offers potential for closeness and can then be constructed as an intimate practice, addressed below.
Familial dementia as an intimate practice
Cohabitation, marriage and children can be markers for couple relationships. Bella reflected on her relationship with her partner, and familial dementia impacted them both: We ended up talking about 20, 30 years down the line, like care and you just think . . . we are talking about all of this now . . .
While genealogy is a tie that binds kin – materially in the case of genetic disease – the emotional support by intimate partners –with whom we may spend significant time – offers a deep bond, as Joanna reflected on her difficult relationship with her family, compared to her marriage: ‘My husband is now kind of like ‘Screw the world, it’s me and you babe, nobody else. It’s him and I. He’s gonna take care of me’ (Joanna, 39, US).
Partner support had been important throughout the genetic testing process but specifically because of how individuals were constructing the future. Lucy had confided in her then-boyfriend in a way she hadn’t to her wider family – she needed support with her decision: I remember having conversations with my boyfriend at the time being like ‘should I get tested? . . . I talked to my ex-boyfriend’ and he said ‘you live your life like you already have it, so like what’s the downside of knowing?’. (Lucy, 35, US)
This suggests ‘family practices’ (Morgan, 1996) have a relevance beyond families-as-kin to encompass other intimates.
Lucy, Joanna and Bella show that familial dementia can be a specific, meaningful shared experiences for couples. This is compounded by the effect of dementia on social functioning which means it cannot be an individual endeavour (Le Fontaine and Oyebode, 2014). Inherited variants implicate family members (Dimond et al., 2022) but partners too. In addition, the life course is not simply individual but also a couple’s endeavour, especially when key milestones – cohabitation, marriage, and parenthood – entail relationships (May and Nordqvist, 2019). Although couples were not interviewed, individual participants such as Bella, suggest the impact of dementia on couples, referring to ‘we’. Dementia impacted couples’ plans, including financial considerations, pensions and living arrangements but it was the issue of having children where this was most prominent – likely due to the age of participants.
The future permeates relationships: planning the mundane short-term matters such as holidays or meals and substantial decisions such as wills, and health conditions which present dilemmas. As a terminal illness, dementia undermines the future, but equally, makes individuals and couples consider the future in ways others may not. A negative test unfurled Bella’s future and impacted her plans positively, as she no longer felt bound by mortality: I’ve been living with the potential of dying younger, I’ve never considered living past 60 . . . coming up to the age of 30 I was feeling middle aged . . . I’ve gained 30 years . . . anything could happen but at least I’ve got the same chance . . . I mean our plans haven’t drastically changed . . . we still want to move eventually and start a family but . . . we’ve no time pressure. (Bella, 28, UK)
‘Biographical disruption’ (Bury, 1982) theorises the impact of illness on a individual trajectories and identities, relevant to the proposition of dementia. However, as Bella shows, this can be positive. New intimate practices were identified. Bella described finding out about her negative result alongside her partner as a ‘moment’ for them both, and their respective families and friends: Completely shell shocked. We rang people quite soon after . . . my sister and Jamie’s mum and dad. It was really emotional for all his family as well as they had been through it with us and so we were like crying with him on the phone . . . . we just had this day where we get all these like these messages of congratulations . . . we were walking around on Cloud 9. (Bella, 28, UK)
Lucy, whose wider family were at risk, described how she had kept the decision to test between her and her husband: ‘I just kind of wanted to create this nice little protective cocoon for my husband and myself to deal with that information’ (Lucy, 35, US). This intimate relationship perhaps offered a form of support which could centre her as the individual being tested, rather than weighed down by the feelings of others at risk (Dimond et al., 2022). By contrast, Joanna (39, US), opted to find out alone, suggesting testing is also a private practice, ‘I knew the exact date and the time for the phone call, but I didn’t tell [my husband]. . . I wanted to do it. . . in my house alone’.
While imperative to centre persons with dementia, there are ripple effects on intimate relationships where reciprocal and mutual care are considered a given (Hockey and James, 2007). Couples support is necessary as the medical community benefits from the strength of these relationships through social support and medical trial participants to have a consistent ‘study partner’. In common with other participants, Morgan (28, US), had chosen her spouse: ‘My husband will be my study partner, they try to have you keep the same one. . . someone that’s around you a lot’.
Doing it for the kids: testing-as-a-family practice
The themes of testing, planning, and consideration in relationships – especially regarding children or potential children – were prominent, shaped by aforementioned procreative norms. Moralities are attached to parenthood in pro-natal society, accompanied by a moral panic regarding declining birth rates and ageing populations in some territories, stoked by politicians, religious leaders and culture (Hall and Van Hooff, 2024). Women are constructed as mothers-in-waiting, defined by reproductive capacity while children exemplify the future, to the affect of reducing children to prototype adults, rather than social actors in their own right (Hockey and James, 2007). Moralities also surround healthcare, for example, sexual health screening (Mamo et al., 2022). Genetic screening technology has reconceptualised responsibility, generating expectations to access this where available (Leefmann et al., 2017). This could include individual and embryonic testing to conceive children who are clear from dementia.
Participants such as Sasha (52, US) seemed compelled to test for the purposes of future children, rather than themselves: I don’t think I could ever live with myself knowing I had passed it onto my kids.
Child conception was a prominent intimate practice in this data. Extant debate has explored when life begins – for example, around conception and baby loss (Reed et al., 2023). Research highlights the meaning attached to life that does not exist or sadly no longer exists through non-motherhood, childlessness, infertility, miscarriage and stillbirth (Letherby, 2015). The narratives suggest that imagined children are in the frame and that developments in medicine have reconceptualised parental responsibilities to apply family practices to potential children. While they might not yet exist, individuals test out of care for potential children, rather than to enrich their own lives. Bella noted the significance of this: It was funny . . . I was making my life decision kind of based on what would be the best for my kids in the future and at one point I was putting all this pressure on myself to, you know have kids young. (Bella, 28, UK)
This contrasted with Noah whose family had a history of dementia diagnoses stemming back to his grandfather, his father and 2 of his father’s siblings. He and his siblings who had tested positively had two children conceived ‘naturally’ in spite of risk due to financial barriers and stress: We knew it was a 50/50 for me . . . I hadn’t been tested at the time, and we went through the mental exercise of you know whether we really wanted a kid and at the time . . . I was on a graduate stipend, and my wife was in graduate school, and so we had no money . . . Thinking about something like IVF was just not financially on the table . . . even if it was, my wife was hesitant about the you know the stress on her body of going through that . . . Our decision with our daughter, we made very, very consciously . . . There’s a lot of people that have opinions about that. (Noah, 37, US)
Noah was keen to justify his choices though his decision should be viewed according to temporal context. While some had a choice now, it should be acknowledged that this wasn’t always possible. Noah continued, It’s unfathomable to them how you could make a conscious decision . . . it’s kind of a weird transition point right now, because so many are maybe my age or a little bit older . . .
In addition, geographical contexts and culture are relevant. In the United Kingdom, assisted methods may be free at the point of use on the NHS, whereas the US requires health insurance. Furthermore, religious and cultural beliefs around embryos determines the appeal of ART which entails their destruction. Abortion discourses are emotive, particularly in the United States since Roe v Wade was overturned in 2022. It is not merely dementia that presents a ‘hurdle’ to having children but the need to align resources and values.
Focusing on women’s life courses, there are specific implications as dementia testing may correlate with their fertile years. Waiting for treatment in the future was not a luxury Harper could afford: Either there will be a silver bullet or there will be no treatment. In the meantime, I’m going to have kids (Harper, 38, US). This issue was remarked on by Eve: If I still haven’t gotten tested by the time I’m like 45 . . . 50, I mean, I probably would at that point . . . I will have either already decided to have kids anyway, or not. And whether I decide not to because of this or not – I don’t know – it seems like a lot to miss out on having kids because of this, but it seems impossible to have a child and risk passing on these genetics. (Eve, 31, US)
Harper and Eve’s experiences undermine established master narratives. Reproduction as a fundamental life goal is oversimplified: wanting and having children is framed as a binary, and narratives of ambivalence are relatively new (Chapman and Gubi, 2022). For our participants, this was more complex. Eve (US), did not want the test as things stood, but had yet to make a decision regarding children – not simply due to dementia but considering matters including climate change, costs, and instability in the world (Hall, 2019): I’m 31, I’m with my partner for a long time . . . how do you even figure out how you wanna have kids in this . . . crazy world, and like in these crazy housing markets . . . how do you even manage a career and everything? And then . . . [dementia] on top . . . it’s totally overwhelming.
Furthermore, dementia narratives understandably underline the importance of valuing persons with dementia and ensuring dignity of care (Rahman, 2014). However, the dawn of genetic testing and ART does not ameliorate all the challenges of dementia and the imperative for support. Even when the gene has been identified and embryos selected to prevent the gene’s inheritance, children experiencing their own parents’ dementia is inevitable, as Jasmine stated, I didn’t want to have dementia and have kids that were still relatively young. I knew what that had done to me, but I still wanted kids so we would have immediately gone pre-implantation genetic diagnosis route.
ART such as IVF has addressed fertility challenges for large numbers of people. However, barriers include high costs, relatively low success rates, social and biological stressors (Mounce et al., 2022). For Joanna, this was exacerbated by dementia and her perception of a lack of knowledge around IVF and impacts on dementia: We were planning on starting our family . . . We are in rush mode . . . I’m 38, he’s 55 . . . I already have plaque on my brain due to this disease . . . I don’t wanna go through IVF. It’s expensive . . . What if it brings on my symptoms earlier? I don’t know if I should go through IVF . . . injecting your body with a crap tonne of hormones . . . What is that going to do to my brain? There’s not much research on this specific topic –pregnancy, IVF, with the PSEN1 mutation. (Joanna, 38, UK)
Relatedly, dementia research relies on clinical trials which participants had been invited into. However, involvement was contingent on not having children, which was a significant issue because of the timing for women like Lucy and Morgan: ‘They were like, you want to participate in a trial?’ And I’m like, ‘Okay, yeah, sure’. And they’re like, ‘but you can’t have children for four to six years’. And I’m like, as a 30-year-old woman, that’s kind of an issue for me. (Lucy, 35, US) ‘The primary prevention trial. The drug they’re actually going to be testing is the Gant that my dad is taking . . . they’re very hopeful about that, since they’re using the same drug . . . you can’t get pregnant during the study. If you do get pregnant, you’ll have to drop out just cause they don’t know the effects of it . . . That’s something that’s really hard . . . the ages are like the exact childbearing years . . . like somewhere between 25 and 34 to be in it, because you have to be 15 years from the age of onset . . . the whole kid thing . . . kind of like race against time. (Morgan, 28, US)
Morgan who had witnessed firsthand the pain of dementia was keen to contribute to science but this posed a dilemma: ‘Because if we were to have kids now, I would still be able to participate in that study . . . I kind of want to be able to do both’. However, a trial participation had opened up the possibility of biological children for Harper (US) whose IVF would not have been covered by her insurance but was paid for the through trial: I came to the conclusion that the upsides of testing were not there . . . I had no interest in being pregnant . . . I had always been intrigued by the idea of adoption . . . felt very good about that decision . . . we started looking at adoption resources but then there’s this third option of IVF because that was covered by [The State]. I decided to get tested . . . I do have the gene.
So while genetic selection rendered children possible for Jasmine, there were also individuals for whom having biological children remained impossible, or at least posed a dilemma despite genetic selection.
Families were longing for future medical advances. Noah for example, had learned he carried the gene in between having his first and second child, hoped for developments for his own children: We’ve probably 30 to 50 years of scientific advancement that can help out these kids . . . if there’s any of these dominant families that these therapies are gonna help, it’s gonna be ours. It’s a little bit later onset [in our family], so selfishly . . . kind of betting on, hoping that science help.
Meanwhile, Harper argued, I feel very hopeful . . . If there’s any treatment, I’m going to be one of the very first to access it . . . You’re way luckier if you were diagnosed with HIV in 1995 than in 1982, and way luckier in 2005 than 1995. Right now, we’re curing people. So what if that happens when I’m 44 and I’ll start getting routine treatment for a lifetime chronic disease . . . or there will be no treatment. (Harper, 30s, US)
This brings us back to the significance of temporalities at play which may be in flux where living with the threat of inherited dementia is concerned. Experienced in time – people’s life courses – as well as being temporally situated for example, the context being medical advancement currently possible that were not previously possible – this has prompted dilemmas. This resonates with Halberstam’s (2005) conceptualisation of queer time, particularly given the connection to personal life. However, our there is a departure given the lack of cure for management of symptoms. Technology and medicine play a part in revealing information about the risks taken when we enter into relationships – the potential for illness and loss is theoretical, but intimates are forced to confront this in the context of genetic testing. As this data show, it shapes the formation of relationships and families and everyday intimacies.
Discussion and conclusion
Social science literature has established that genetic technologies have changed what it is to be a patient, reshaping agency and spatial aspects. We contribute to literature on dementia in the context of families, describing how individuals negotiate couple relationships, having children or not, and illness impacts. The narratives suggest dementia shaped the establishment of relationships, and that this presents challenges and opportunities. We conceptualise navigating familial dementia together as an intimate practice to be understood through the lens of relationships and personal life, rather than families-as-kin. We illuminate the emotion work and magnitude of the decision over whether to undergo testing and consideration of future-beings, ahead of themselves. Dementia occupies a presence in intimate lives as part of couple conversations and plans – both ordinary and significant. Genetic testing has the potential to shape life course planning and relationships emphasising the need for supportive intimate relationships (Klitzman and Sweeney, 2011). Data suggest that testing decisions are informed by a desire to have children in a pro-natal society and reflect health moralities. These findings offer significance to practice as well as making contributions to understandings of relationships in the context of health broadly and dementia.
Those experiencing dementia are navigating the everyday trials and tribulations of personal life, and may be living and grieving their own life courses and their parents. They may be making important intimate and health-related choices, including whether to undergo genetic screening. There may be financial impacts, particularly in insurance-based health systems as well as ramifications for at-risk kin. This may correspond with caring arrangements for parents. There is a need for timely testing and raising awareness among medical practitioners, particularly in the United Kingdom where GPs gatekeep diagnosis. In addition, other health care providers, including genetic counsellors and social workers may benefit from awareness of the impact of familial dementias on intimate practices and the time sensitive nature.
The rapid development of genetic technology, particularly in terms of pre-symptomatic testing, has the potential to transform health care and the experiences of ‘patients’ and their families. For those at risk of dementia, a serious terminal illness with no cure or treatment which contrasts with the possibilities in terms of genetic technology and genetic knowledge. This underlines the importance of advancing knowledge of social experiences of dementia, for example, how people facing testing can be best supported while there is no cure. Furthermore, medical research is crucial to improved knowledge on dementia. As it is a commitment not to become pregnant during trials, trial participation has consequences for women at specific life stages, showing that material reality and temporality impacts medical advancement and the development of a cure. Trial teams need to be aware of the impact on people’s lives precisely due to the temporality to support those making decisions over whether or not to participate. There are responsibilities in lay contexts too. The availability of commercial testing is developing apace and there is a need to ensure availability of social support and to consider the ethics of this as this will have implications for couple and extended familial relationships. We have elsewhere (Hall and Sikes, 2018; 2020) argued towards a plurality of social scripts and this research, which centres the voices of those impacted, reiterates the need to share real-life stories.
Findings contribute to understandings of how illness is experienced in the context of relationships. What is striking is that when there is the potential for a genetic illness, individuals and couples consider the needs of imagined children and that testing offers an avenue for ‘doing’ and ‘displaying’ family (Finch, 2007; Morgan, 1996) long before children are conceived or born. The theoretical framework drew attention to the choice biography, weakened by cultural expectations around procreation, but this is exacerbated for families aware of dementia risks. However, the condition also brings into being new stages and milestones, for example, testing outcomes, signs of commitment and fertility treatments which are not Hallmark occasions, but nevertheless, occupy significance, which we conceptualise as intimate practices.
There are commonalities with other conditions as discussed earlier. Our narratives corroborate the need for improved support and further research. There are distinctions from some illnesses, specifically a lack of prevention or cure on as with treatments for HIV and genetic cancer such as BRCA (Dimond et al., 2022; Klitzman and Sweeney, 2011). This has been an exploratory study, despite attempts at a broader sample, concentrated in the US rather than the United Kingdom which may impede the generalisations drawn. We would like to expand further on the research, though believe it would have been difficult to have taken a different approach at the time, partly for ethical reasons and partly as a result of the impact of COVID on dementia diagnoses which may have affected recruitment. However, insights into a relatively rare disease, even with a small sample has implications for medics. This is the first familial dementia generation grappling with choices in the certainty of risk owing to genetic developments. It is important to understand their experiences, how they navigate outcomes for themselves, family members and intimate relationships to inform support. The timeliness of the research highlights a potential for revisiting experiences of a range of other genetic conditions in light of medical developments and consumer behaviours – including the widespread availability of tests. They also need to be reconsidered in light of social developments relating to the rise of childfree families and attitudes around this. As Felipe makes the point of the impact of timing on his experience, ‘I feel like I’m the first one in this family history to have the opportunity to be single. Not have any kids, and know that I’m positive with the gene’. Our study indicates the implications for relationships and the benefit of applying a families and relationships lens, rather than concentrating on direct kin. These data highlight a need for further research, including those who are single and those who do not want, or are ambivalent towards having children and the LGBTQ+ community whose experiences are underexplored. Doing so would decentre dominant, narrow conceptualisations of ‘families’ as recommended by sociologists of personal life.
Footnotes
Appendix 1
Do you have a parent diagnosed with a potentially inheritable type of dementia?
We are interested in understanding the impact on children who are now 18 years of age or older of having a parent with this type of dementia and their thoughts about genetic testing in order to better support such individuals and their families.
This research is being led by Caroline Gelman, Ph.D., Associate Professor of Social Work at Hunter College, New York, and Pat Sikes, Ph.D., Emeritus Professor, School of Education, University of Sheffield, England. If you are interested in participating in an interview or have further questions, please contact them at
You will receive $25 in consideration of your time and effort.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by City University of New York, Hunter College (TRADB-51-239).
