Abstract
Recent studies using polymerase chain reaction (PCR) demonstrate that blood and pleural fluid (PF) cultures are inadequate to identify pneumococcus in children with pneumonia and empyema, leading to unnecessary utilization of broad-spectrum, often toxic antibiotics. Identification of pneumococcus facilitates the de-escalation of antibiotics. We compared a point-of-care immunochromatographic test that detects pneumococcal C-polysaccharide antigen (BINAX) performed in PF and urine to cultures (blood and PF) and 2 different PCR tests in PF of 79 children with empyema. BINAX in PF had 100% sensitivity and negative predictive value (NPV) to identify pneumococcus in children with empyema, compared with cultures (sensitivity 20.7%/NPV = 68.8%) or PCR (sensitivity 96.5%/NPV 98%). Urine BINAX demonstrated lower sensitivity (89%) and NPV (93%). BINAX in PF is superior to cultures (PF and blood), identifying pneumococcus in children with empyema. PCR has similar sensitivity and NPV to BINAX but is not readily available.
Introduction
Empyema, defined as the presence of a complex, often loculated collection of pus in the pleural space, is a serious and relatively common complication of community-acquired pneumonia (CAP-Emp) in children that frequently requires lengthy hospitalization.1,2
In the clinical setting of parapneumonic empyema in children, a bacterial pathogen is identified by blood or pleural fluid (PF) culture in < 25% of cases.3,4 Recent studies using polymerase chain reaction (PCR) in blood 5 or PF6 -8 demonstrate significantly improved pathogen identification, particularly Streptococcus pneumoniae, which remains the most common pathogen causing CAP-Emp in children.7,9 -12 However, other pathogens such as Staphylococcus aureus, both methicillin sensitive (MSSA) and resistant (MRSA), and Streptococcus pyogenes, are also frequently identified and often associated with significant morbidity and mortality.7,12 -15 Furthermore, Hemophilus influenzae, Streptococcus spp. (frequently S. intermedius), and anaerobic bacteria are also pathogens found in CAP-Emp. Recently, Mycoplasma pneumoniae, often macrolide resistant, has been reported with increasing frequency and severity in CAP-Emp.16,17 Thus, empirical use of broad-spectrum, potentially toxic antibiotics (eg, vancomycin, third-generation cephalosporins, oxazolidinones, clindamycin, and quinolones), surgical procedures, and prolonged hospitalization are common practices.7,18
Rapid identification of S pneumoniae has been shown to result in early de-escalation of antibiotics, typically to an oral penicillin-based regimen, allowing for shorter hospitalization, enhanced safety, decreased cost, and possible decrease in antimicrobial resistance.4,7,19
The BinaxNOW Streptococcus pneumoniae antigen assay (Abbott Diagnostics, Scarborough, Inc., Scarborough, Maine) (BINAX) is a point-of-care immunochromatographic test that detects the C-polysaccharide antigen in all strains of S pneumoniae. 20 This inexpensive and easy-to-use test can be quickly performed in most clinical laboratories and has been validated for detecting S pneumoniae in urine and cerebrospinal fluid (CSF) samples. 21 Urine BINAX testing is frequently used in adults with suspected invasive pneumococcal disease (IPD), but due to concerns over lack of specificity arising from high-density nasopharyngeal colonization, its use in urine is not recommended in children. 3 BINAX has also been shown previously to accurately identify S pneumoniae in PF samples of adults22,23 and children with CAP-Emp.24-28 The objective of this study was to evaluate the utility of BINAX in PF and urine samples in comparison to standard blood and PF cultures, as well as PCR testing of PF for the timely identification of S pneumoniae in children hospitalized with CAP-Emp.
Methods
Study Design and Population
We conducted a prospective, open-label study to evaluate the microbiologic and diagnostic validity of the BINAX test for identifying S pneumoniae in children > 2 months to < 18 years old who were admitted with CAP-Emp to the Children’s Hospital of Orange County (CHOC) in Orange, California, between December 2018 and August 2023. Patients who lived in chronic facilities or had devices (tracheostomy and gastric tube) or medical conditions (cystic fibrosis and immune deficiencies including patients receiving chemotherapy) that could affect oropharyngeal colonization and etiology of pneumonia were excluded.
Baseline demographic data including age, sex, ethnicity, and race, and immunization status were collected at admission. Per standard of care, blood cultures were obtained at the time of admission, and PF cultures were obtained by thoracentesis, chest tube placement, or video-assisted thoracoscopic surgery (VATS) at the discretion of the treating physician. Urine samples were collected for BINAX testing per study protocol. S pneumoniae isolates from blood and/or PF cultures were sent to the Orange County Health Care Agency for serotyping. Aliquots of PF from every patient were sent to the University of Utah (UofU) for pathogen identification using the BioFire BCID/BCID2multiplex PCR (mPCR) panel (BioFire Diagnostics, Salt Lake City, Utah) and to Baylor College of Medicine (BCM) for pneumococcal lytA real-time PCR (rtPCR) testing. All specimens positive for S pneumoniae by rtPCR testing at BCM were serotyped using a multi-step multiplex PCR (msPCR) for pneumococcal serotype identification. BINAX testing was performed onsite at CHOC on all PF and urine samples.
Statistical Methods
Demographic characteristics of the patient population were summarized using mean (SD) for age and counts with percentages for categorical variables, including age group, sex, ethnicity, and race. Differences in demographic characteristics between positive and negative patients for S pneumoniae were assessed using Chi-square tests for categorical variables and independent t-tests for continuous variables. Immunization status was summarized descriptively for the overall cohort and for the subset of patients with S pneumoniae CAP-Emp, based on data obtained from immunization cards, the California Immunization Registry (CAIR), and parent recall. Patients were considered fully vaccinated if they had received 3 primary PCV doses during the first year of life and a booster between 12 and 15 months; patients who received a supplemental dose of PCV-13 after 2 years of age were also considered fully immunized, independent of previous PCV immunization status, as per recommendations of the American Committee on Immunization Practices. Patients were considered up to date for age if they had received an appropriate number of PCV doses for age at the time of presentation with empyema; not up to date were those who had not completed the number of PCV doses recommended for age at presentation, and patients were considered unimmunized if they had not received any PCV doses. Microbiological etiologies other than S pneumoniae, as identified by culture and PCR testing, were visualized using pie charts displaying the distribution of each pathogen by count and percentage. The diagnostic performance of BINAX testing (both PF and urine) for detecting S pneumoniae, as confirmed by PCR or culture, was evaluated using standard validity metrics: sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV). Statistical analyses were conducted using IBM SPSS Statistics, version 29.0 (IBM Corporation, Armonk, NY), and pie chart visualizations were created in Microsoft Excel for Microsoft 365, version 16.0 (2023 release; Microsoft Corporation, Redmond, WA).
This study was conducted in accordance with the protocol, legal and regulatory requirements, and the general principles set forth in the International Ethical Guidelines for Biomedical.
Research Involving Human Subjects (Council for International Organizations of Medical Sciences, 2022), ICH Guideline for Good Clinical Practice, and the Declaration of Helsinki. Approval of the study was obtained from CHOC’s IRB.
Before participation, written informed consent/assent was obtained from parents/Legally Authorized Representatives and patients (patients 7–11 years old signed an assent; patients ≥12 years old cosigned consent).
Results
During the study period, 80 patients met the inclusion criteria. Of these, 76 were approached for participation; 1 patient declined, and 1 consented but was discharged without PF sampling. A total of 74 patients were included in the final analysis. All patients underwent blood and PF aerobic and anaerobic cultures; all PF specimens were tested by PCR and BINAX; 73 of the 74 patients provided urine, which was tested by BINAX.
Demographics
The mean age of included patients was 6.2 years (SD = 4.5) (Table 1), and age was distributed as follows: 25.7% were < 3 years, 18.9% were 3-4 years, 39.2% were 5-12 years, and 16.2% were 12-16 years. A slightly higher proportion of patients were female (56.8%), and nearly half identified as Hispanic (48.6%). Reported race included White (55.4%), Asian (12.2%), and other (32.4%). Of the 74 patients, 29 (39.2%) tested positive for S pneumoniae by blood/PF culture or PCR. These patients were, on average, younger than those who tested negative (mean = 4.8 years (SD = 3.4) versus 7.1 years (SD = 4.9) (P = .029), although sex, ethnicity, and race distributions were similar between the 2 groups.
Characteristics of the Study Population Described by ± Streptococcus pneumoniae by Either Culture From Blood and/or PF or PCR.
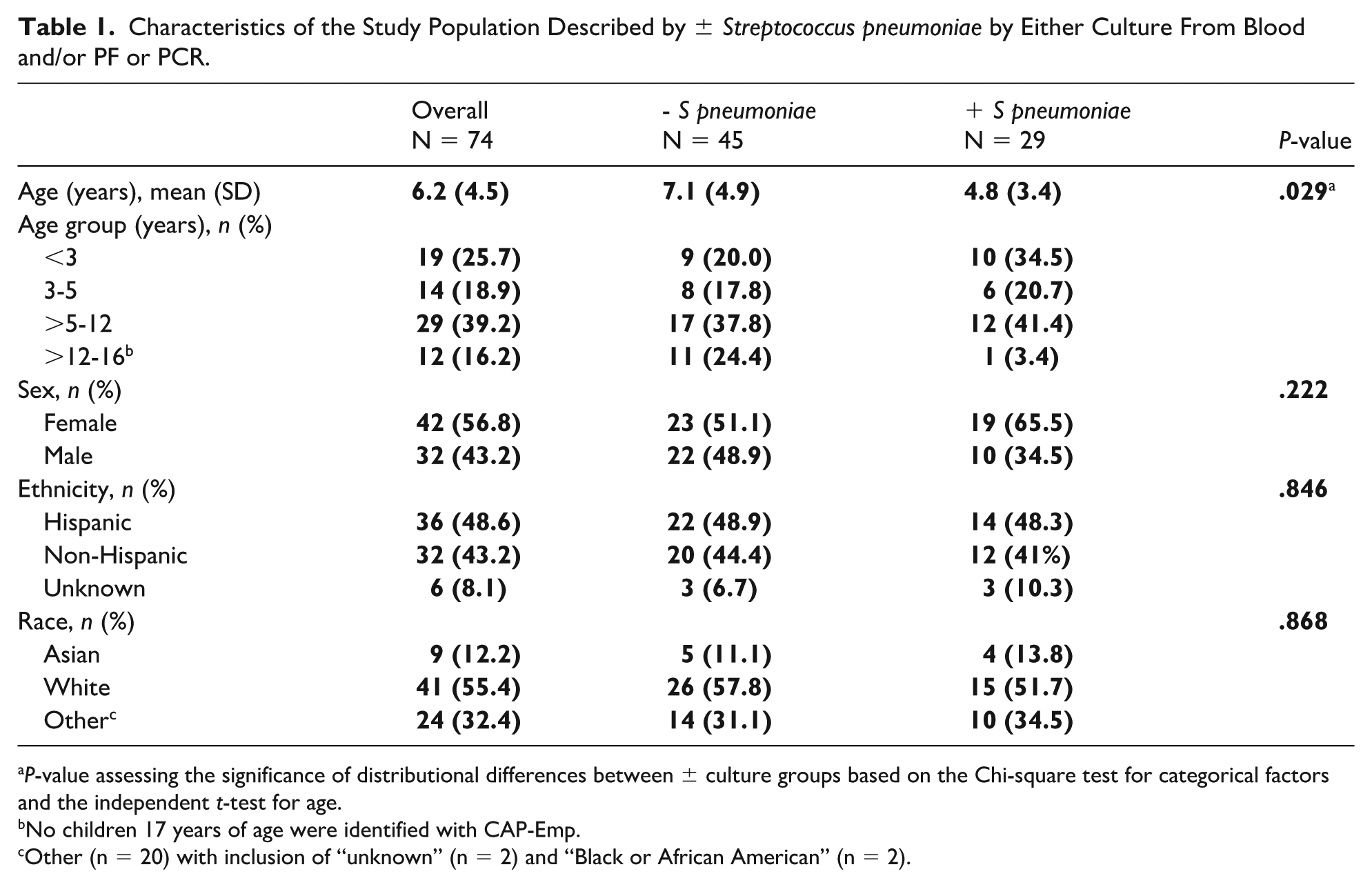
P-value assessing the significance of distributional differences between ± culture groups based on the Chi-square test for categorical factors and the independent t-test for age.
No children 17 years of age were identified with CAP-Emp.
Other (n = 20) with inclusion of “unknown” (n = 2) and “Black or African American” (n = 2).
Immunization Status
Immunization status was obtained on all 74 patients: 5 (6.8%) from an immunization card, 64 (86.5%) from CAIR, and 5 (6.8%) by parent recall. Fifty (67.6%) patients were considered fully vaccinated. Two patients (2.7%) were considered up to date for age, 20 (27%) were not up to date, and 2 (2.7%) were unimmunized. Among the 29 S pneumoniae CAP-Emp patients, 21 (72.4%) were fully immunized (had received at least 4 PCV doses; 18 received all PCV-13 doses and 3 received 4 PCV-7 doses with a PCV-13 booster after 2 years of age).
Microbiologic Results
S pneumoniae was identified in 29/74 patients (39.2%), with 6/29 (20.7%) identified by culture and the remainder by PCR (23/29; 79.3%). mPCR identified S pneumoniae in 23 PF specimens including 5/6 previously identified by blood and/or PF cultures, rtPCR identified S pneumoniae in 5 additional PF specimens; both PCR methods failed to identify S pneumoniae in PF from a patient who had a positive blood culture for S pneumoniae early in the course of her illness, 7 days before PF was obtained, during which time she was receiving antibiotics. Of note, BINAX was positive in PF and urine in this patient. In 20/49 (40.8%) patients with bacterial pathogen identified, other bacteria were identified by culture (n = 16) and/or PF PCR (n = 4), which included Streptococcus spp. (7; 14.3%), S. pyogenes (6; 12.2%), S. aureus (5; 10.2% [4 of which were MRSA]), and H. influenzae (2; 4.1%). Of note, all S. aureus grew from blood and/or PF. The remaining 25/74 (33.8%) patients had no bacterial pathogen identified by culture or PCR. (Figure 1A and B).
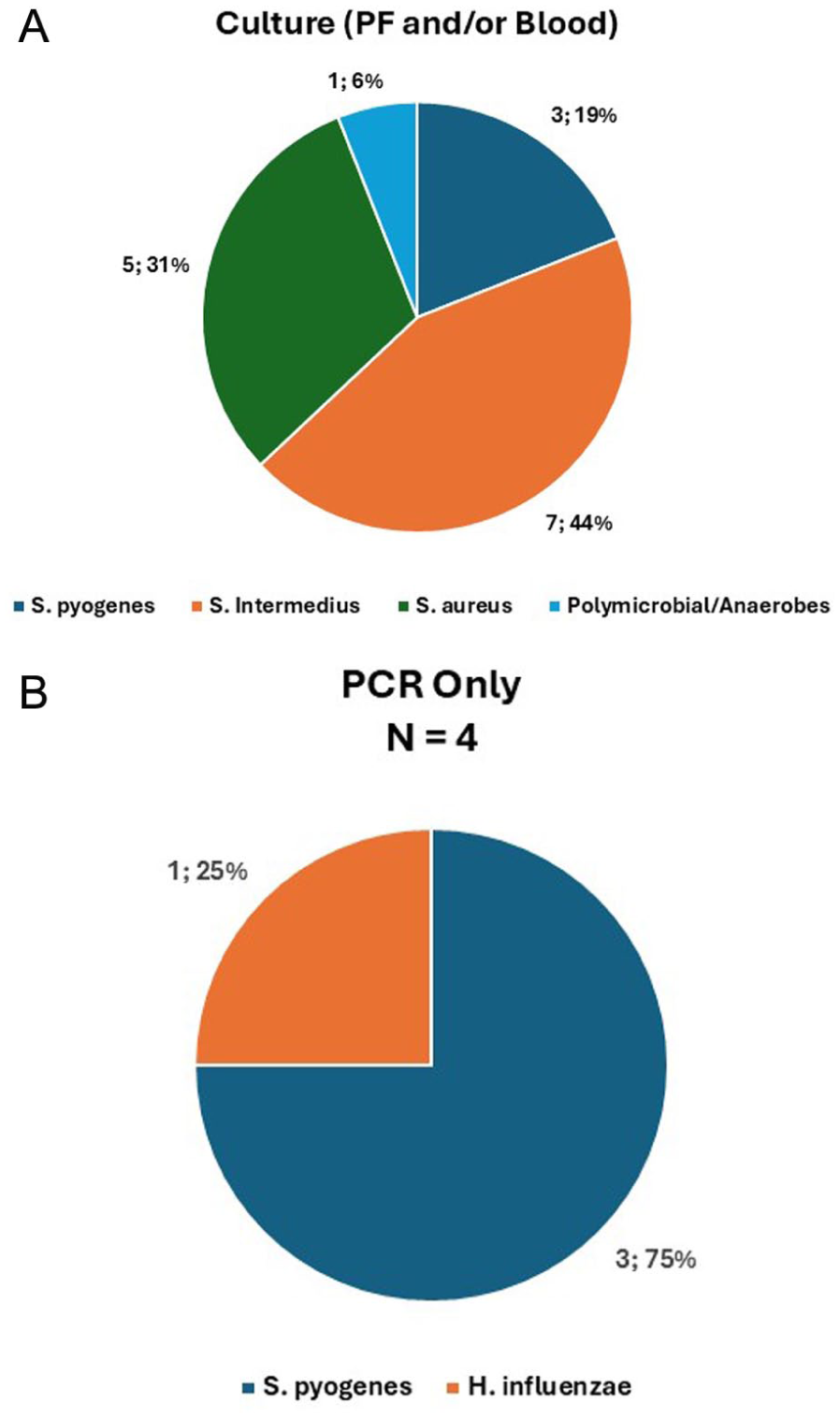
(A) Total Number of Patients With Non-Streptococcus pneumoniae-specific Microbiological Etiologies Established by Culture From Blood and/or Pleural Fluid. (B) Total Number of Patients With Non-S pneumoniae-Specific Microbiological Etiologies Established Only by Pleural Fluid PCR, by Organism.
Results of BINAX Testing
Pleural fluid BINAX
BINAX in PF was positive in 36/74 (48.6%) patients, including all 29 with S pneumoniae and an additional 7 patients in whom S pneumoniae was not identified by any other method (sensitivity = 100%; specificity = 84.4%; positive predictive value (PPV) = 80.6% and NPV = 100%) (Table 2).
Results of PF BINAX Testing in Patients With and Without Streptococcus pneumoniae Infection as Established by Culture or PCR.
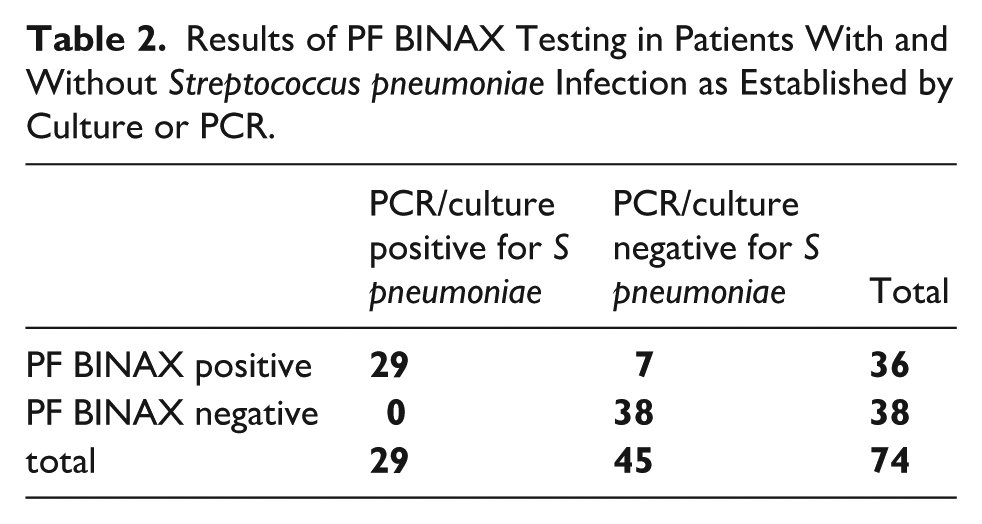
Of the 7 BINAX-positive patients without S pneumoniae, 2 had other Streptococcus spp. identified by PF culture and PCR and 1 had growth of mixed aerobic/anaerobic oropharyngeal flora. The remaining 4 had no bacterial pathogens identified by culture or PCR methods. Three of these patients had been treated with antibiotics for ≥ 7 days (the 4th one had been treated for 5 days) before PF was obtained (Figure 2).
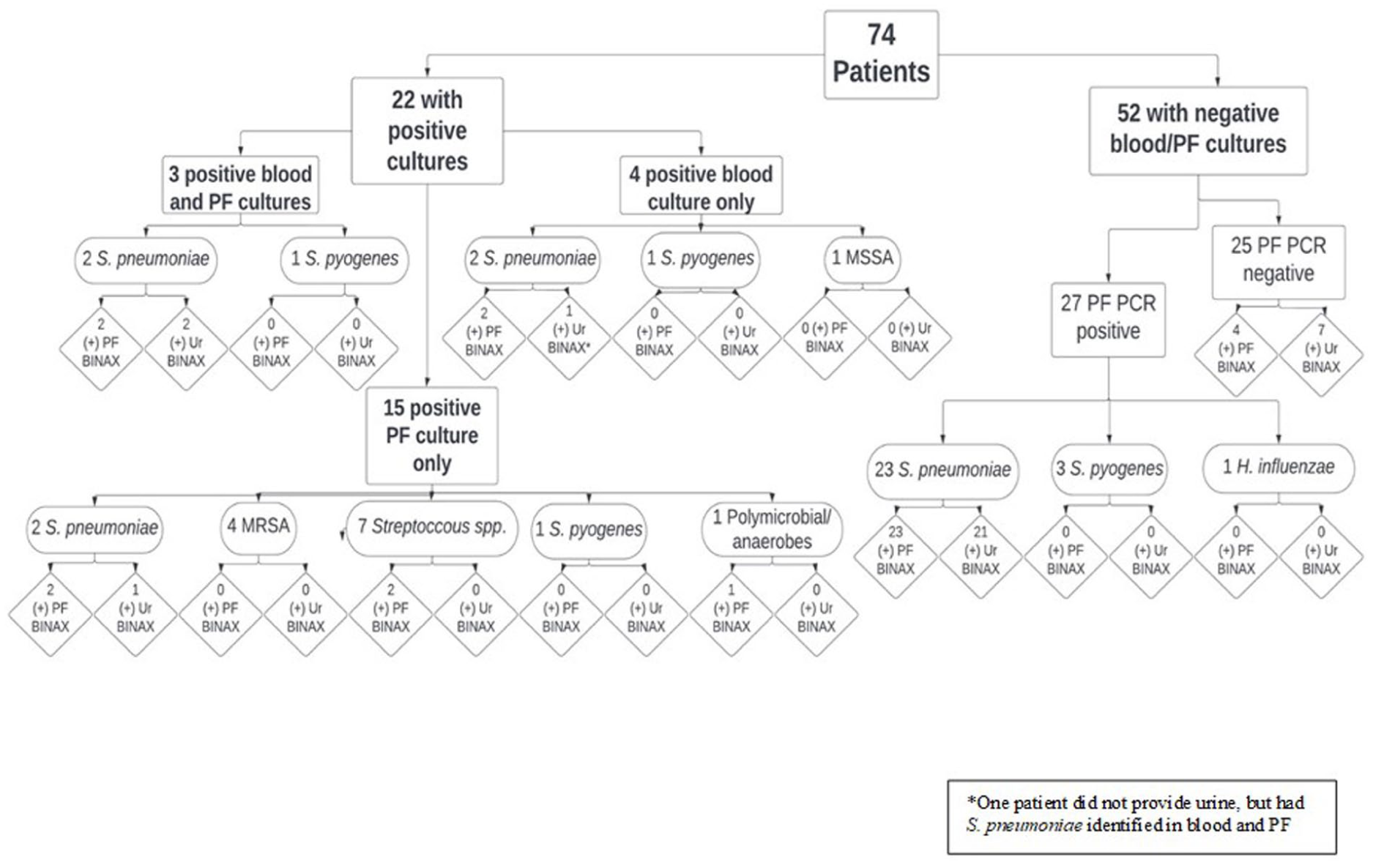
Breakdown of results of blood cultures, PF cultures, PF PCR, PF BINAX, and urine BINAX for all 74 participants.
Urine BINAX
BINAX in urine was tested in 73 patients, which included 28 (96.6%) of the 29 with S pneumoniae identified in blood and/or PF by culture/PCR. BINAX was positive in 32/73 (43.8%) patients including 25/28 (89.3%) S pneumoniae infections. Seven patients out of 45 (15.6%) without identified S pneumoniae infection had a positive urine BINAX (sensitivity = 89%; specificity = 84%; PPV = 78%; NPV = 93%) (Table 3).
Results of Urine BINAX Testing in Patients With and Without Streptococcus pneumoniae Infections as Established by Culture or PCR.
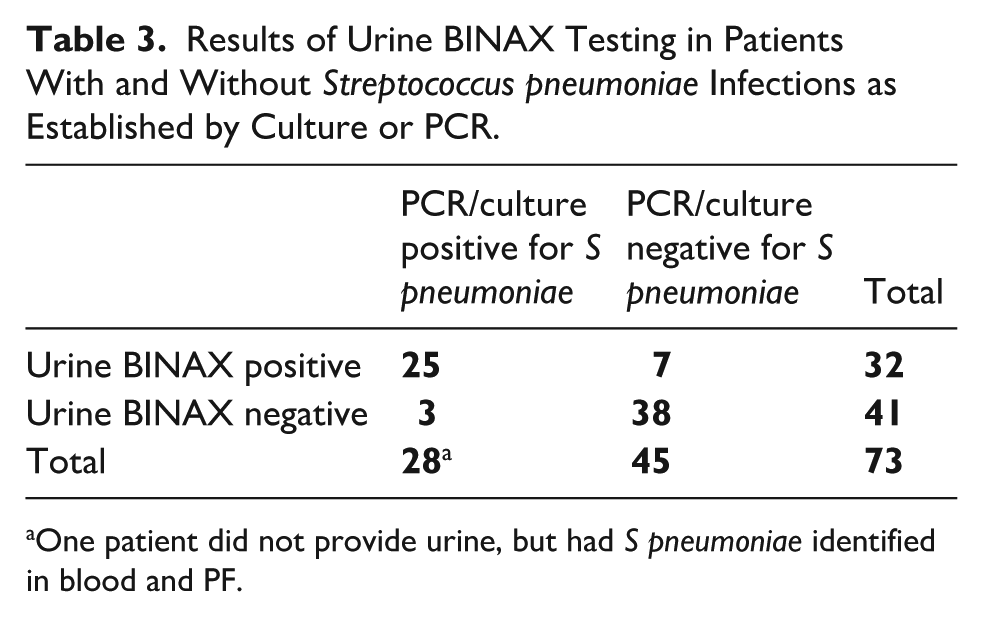
One patient did not provide urine, but had S pneumoniae identified in blood and PF.
Four of these 45 (8.9%) had urine and PF BINAX positive result and had no bacterial pathogen identified by any other method and are described above. (Figure 2)
Discussion
In this study, BINAX in PF was able to accurately identify all cases of S pneumoniae in children with CAP-Emp (100% sensitivity). BINAX had superior sensitivity compared with culture, by which only 6/29 cases were identified (sensitivity 20.7%). It was also more sensitive than mPCR and rtPCR in PF, which identified 23/29 (79.3%) and 28/29 (96.5%) cases, respectively. One child with CAP-Emp, who had a positive S pneumoniae in blood culture on admission to the hospital, had a positive BINAX test in PF and urine after 7 days of antibiotic treatment, while neither the PF PCR method was positive.
BINAX in PF demonstrated moderate specificity (84%) and PPV (81%) when compared with culture and PF PCR combined. Unlike previous studies, we used a multiplex PCR, which identifies other likely pneumonia pathogens in children, in addition to S pneumoniae, improving our specificity analysis. BINAX was positive in 7 children with negative S pneumoniae detection (by culture and PCR), 2 of whom had proven CAP-Emp due to other alpha-hemolytic Streptococcus spp. (1 identified by culture and mPCR and 1 by mPCR only), 1 with growth of mixed aerobic/anaerobic oropharyngeal flora and 4 in whom no bacterial pathogen was identified by culture or PCR. A possible source for false positive BINAX results in PF includes cross-reactivity with other alpha-hemolytic Streptococci. In the present study, Streptococcus intermedius (by culture) and Streptococcus spp. (by PCR) were, as a group, the second most common bacterial pathogen identified, differing from other similar studies. To our knowledge, no previous studies on CAP-Emp have reported cross-reactivity between S pneumoniae and other alpha-hemolytic Streptococci; however, in our study, 3 cases with alpha-hemolytic Streptococcus spp infection had positive PF BINAX suggesting the possibility that the C-polysaccharide-like antigen used as a target in BINAX may cross-react with C-polysaccharide antigen present in other alpha-hemolytic Streptococci including Streptococcus oralis and Streptococcus mitis, which are closely related genetically to S pneumoniae. 29 There are no data on the number of days the PCR assay would remain positive in PF of children receiving antibiotics, but it is possible that C-polysaccharide antigen will remain present and detectable by BINAX in PF while bacterial DNA may be degraded in PF, typically a low Ph environment at body temperature. Four patients, who had been treated with antibiotics for several days before PF was obtained for culture and PCR, had positive BINAX in PF and urine, but no bacterial pathogen was identified by culture or PCR; considering the low rate of positive cultures in patients with proven S pneumoniae CAP-Emp, it is reasonable to think these patients had S pneumoniae empyema. Pharyngeal colonization has been associated with false positive urinary BINAX testing; 20 we have not found reports that pharyngeal colonization could result in false positive BINAX results in PF. There were no cases where S pneumoniae was identified by PCR or culture with a negative PF BINAX result (NPV = 100%).
A recent study by Ho et al 7 highlights the benefits of early identification of S pneumoniae as the pathogen responsible for CAP-Emp; specifically, they report faster optimization of the antibiotic regimen and decreased exposure to MRSA therapy, which was often initiated in their population despite national 3 and institutional recommendations for the initial antibiotic regimen in children with CAP-Emp. Of note, in our study, in all 5 (100%) cases of CAP-Emp caused by S. aureus—including both MSSA (1) and MRSA (4)—the pathogen was recovered in cultures from blood (1 MSSA) and/or PF (4 MRSA). S. pyogenes was identified by culture alone in only 3 (50%) cases. Considering the morbidity and mortality reported with these pathogens, it is common to start vancomycin, clindamycin, or linezolid empirically for treatment of CAP-Emp. Recent reports of an increase in frequency, severity, and macrolide resistance in M pneumoniae pneumonia have resulted in increased use of fluoroquinolones.16,17 When cultures are negative, these antibiotics are often continued for several days or weeks. Fast identification of S pneumoniae would allow rapid antibiotic de-escalation to narrower-spectrum antibiotics.4,7 We used positive BINAX results to successfully discharged to home, patients with negative cultures on appropriate oral anti-pneumococcal antibiotics. No patients had to be readmitted for antibiotic failure.
BINAX in urine demonstrated lower sensitivity (89%) and NPV (93%), this has been recently emphasized by Hamer, DH who found urinary BINAX to be more likely to be positive in children with nasopharyngeal colonization 20 and no more likely to be positive among children with radiographically confirmed pneumonia than among control subjects; similarly, in a different study, Dowell, SF reported BINAX in urine was significantly more likely to be positive among children who were nasopharyngeal carriers of pneumococci. 30 This level of diagnostic performance is insufficient for reliable standalone identification of S pneumoniae infection and does not support its use in de-escalation of broad-spectrum antibiotics in children with CAP-Emp.
Previous studies show PCR in PF significantly increases the microbiological diagnosis of CA-Emp, particularly due to S pneumoniae.6 -8 In our study, S pneumoniae was the most common pathogen, identified in 29 cases, of which only 6 (20.7%) were identified by culture. This discrepancy supports the need for alternative methods to detect S pneumoniae in CAP-Emp beyond culture alone. However, PCR is not routinely available in many hospitals, particularly in developing countries where S pneumoniae remains the most important cause of mortality in children less than 5 years old outside the neonatal period. 30 This is important not only for improving care of individual patients, but also to inform future vaccine development. Our data were generated 8-12 years after broad utilization of PCV-13, suggesting ongoing surveillance is necessary as newer vaccines with higher valency are introduced. In this respect, BINAX has the significant limitation of not providing the S pneumoniae serotype responsible for the infection; newer urinary pneumococcal antigen detection tests have been successfully evaluated for identification of S pneumoniae and pneumococcal serotype in adult patients with pneumonia. 31 Such studies are underway in children.
An important limitation of our study is that it was carried out at a single center in a county with a high prevalence of immunization compliance. Also, despite representing a diverse community with various socio-economic backgrounds and ethnicities, Orange County has a relatively low number of African American children, as reflected in our study’s patient demographics. Another limitation is that our study was affected by the SARS-CoV-2 pandemic, during which some authors have reported a noticeable decrease in S pneumoniae invasive disease including CAP.32,33 Some of the advantages of our study are the prospective nature of its design and the inclusion of 2 different PCR methodologies to identify S pneumoniae, strengthening our confidence that we were able to identify all bacterial pathogens responsible for CAP-Emp.
Conclusion
Here we present our experience with an immunochromatographic test (BINAX), a highly reliable, inexpensive, quick and easy to complete at laboratories in any country, performed in PF, which showed sensitivity and NPV of 100% to identify S pneumoniae as the pathogen responsible for CAP-Emp in children. The advantage of early identification of S pneumoniae as the responsible pathogen in these patients has been highlighted by others.7,19
We believe that BINAX in PF is a valuable tool in the care of these patients. Future studies should explore BINAX utilization in curbing use of broad-spectrum antibiotics and shortening length of hospital stay.
Author Contributions
Michael A. Crawford substantially contributed to the conception and design and acquisition of data this manuscript as well as drafting and critically revising the manuscript. Lindsay R. Grant contributed to the analysis and interpretation of data in this manuscript as well as critically revising the manuscript. Antonio C. Arrieta and Stephanie Osborne substantially contributed to the conception and design, acquisition, analysis, and interpretation of data in this manuscript as well as drafting and critically revising the manuscript. Alejandro Cane, Delma J. Nieves, Ashley Miller, and Tricia Morphew contributed to the analysis and interpretation of data in this manuscript as well as critically revising the manuscript. Raul Isturiz and Adriano Arguedas substantially contributed to the conception and design, analysis, and interpretation of data of this manuscript as well as critically revising the manuscript. Additional Author Contribution Note: All authors noted above gave final approval of the manuscript and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Footnotes
Ethical Considerations
This study was approved by the Children’s Hospital of Orange County Institutional Review Board. (IRB # 130996). Written Informed Consent (ICF) was obtained from parents/guardians of all participants; assent was obtained from participants aged 7 years and older.
Consent for Publication
N/A.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded through a research collaboration with Pfizer. Children’s Hospital of Orange County is the study sponsor.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Antonio C Arrieta received compensation from Pfizer Inc. for the oversight of and conduction of a comprehensive study on the etiology of community-acquired pneumonia and empyema, which included completion of the present report. LRG, AC, AM, and AA employees and shareholders of Pfizer.
Data Availability Statements
The data presented in this manuscript are kept at the Research Institute at the Children’s Hospital of Orange County.
