Abstract
Experts recommend screening for or presumptively treating strongyloidiasis in foreign-born immigrant children, though prevalence is unknown. Health care providers encounter barriers to following this recommendation. We aimed to describe Strongyloides seropositivity and risk factors among pediatric patients on a mobile clinic. Participants completed a written survey and provided blood for absolute eosinophil count (AEC) and Strongyloides IgG enzyme-linked immunosorbent assay (ELISA). From October 2022 to June 2023, 206 patients participated; 201 (97.6%) were born outside the United States; 97.1% (n = 200) were Hispanic. Most (68.9%) arrived less than 1 year before study enrollment. Four (1.9%) and 3 (1.5%) had positive and equivocal ELISA results, respectively. Peripheral eosinophilia was seen in 27 participants (13%). Previous antiparasitic use was reported by 106 (51.5%). We found no significant associations between Strongyloides seropositivity and AEC, exposures, symptoms, or medical history. Mobile clinics can feasibly conduct strongyloidiasis prevalence studies to create guidelines for providers who see foreign-born immigrant children.
Keywords
Background
Approximately 25% of children in the United States are from immigrant families (CIF), meaning they are foreign-born or at least one parent is foreign-born. 1 Many foreign-born immigrant children are at risk for unique medical conditions, including strongyloidiasis, 1 a soil-transmitted helminth (STH) infection caused by the nematode (roundworm) Strongyloides stercoralis. Soil-transmitted helminth infections are among the most common infections globally, with approximately 24% of the world’s population infected. 2 Moreover, strongyloidiasis is included in the 2030 World Health Organization (WHO) Roadmap for neglected tropical diseases elimination. 3 According to the Centers for Disease Control and Prevention (CDC) and the American Academy of Pediatrics (AAP), health care providers should screen for or presumptively treat strongyloidiasis in foreign-born immigrant children.4,5
Strongyloides stercoralis is endemic in temperate, subtropical, and tropical areas with moist soil and improper disposal of human feces, including areas of the southern United States.5,6 Although humans are definitive hosts, reservoirs include non-human primates, dogs, wild canids, and cats. 7 Transmission occurs via fecal-oral route or directly when S. stercoralis filariform larvae living in contaminated soil penetrate skin (eg, walking barefoot). Although most infections are asymptomatic, symptoms can include rashes (eg, larva currens), transient pneumonitis, abdominal pain, intestinal malabsorption, vomiting, diarrhea, and constipation. 5 The incubation period for strongyloidiasis is unknown, and S. stercoralis can persist in the human host for decades after exposure because it “autoinfects” via intestine or perianal skin. 5 Serious complications of strongyloidiasis include hyperinfection syndrome and disseminated disease, which are more common in immunocompromised hosts. Hyperinfection occurs with increased larval migration between gastrointestinal and pulmonary tracts. Dissemination occurs when larvae migrate to distal organs and cause pulmonary infiltrates, septicemia, or meningitis due to enteric gram-negative bacteria. Infection likely occurs when large larval load penetrates the intestinal wall and allows gram-negative bacteria to enter bloodstream. 5
Diagnosis is usually through stool ova and parasite microscopy. 5 However, obtaining stool is logistically challenging, and multiple stool specimens are necessary to improve test specificity. The more readily available Strongyloides IgG is the recommended blood test of choice.1,4 Because antibodies can remain positive after resolution of infection, a positive serology does not confirm active infection. 5 Although up to 50% of strongyloidiasis cases do not have eosinophilia, in an immunocompetent patient, the presence of peripheral eosinophilia may help diagnose strongyloidiasis. 5
In general, the treatment of choice for strongyloidiasis is ivermectin; this includes presumptive treatment with ivermectin when testing is not available.1,4,5 Albendazole is an alternative treatment, though it is associated with lower cure rates. 5 In 2024 the WHO began recommending annual mass drug administration with one dose of ivermectin for children age 5 years and older in endemic areas where the prevalence among school-aged children is ≥5% via stool test or ≥15% via serology. 3
For pediatric health care providers caring for foreign-born immigrant children, deciding to test for or presumptively treat strongyloidiasis can be complicated. Firstly, the global and US prevalence of strongyloidiasis is unknown. Country or region-specific prevalence studies have yielded varying results and often focus on adult participants. Among adult immigrants, asylees, and refugees, prevalence rates range from 1.9% to 4.2%.8 -10 Regional estimates of prevalence in adults in Latin America have a wide range from 5% to more than 15%. 11 Among children, 2.3% of children hospitalized in Italy were seropositive for S. stercoralis. 12 Providers may not know if testing for strongyloidiasis among asymptomatic foreign-born immigrant children is appropriate, and in areas with limited resources, testing and treating are both difficult. The average cost for Strongyloides IgG is $120 (LabCorp, email communication, June 27, 2024). Covering the cost of this test may be challenging for children without health insurance. Those prescribed ivermectin for presumptive treatment may also face barriers. Children in uninsured immigrant families are less likely to use prescription medications. 13 Without a known prevalence of strongyloidiasis among foreign-born immigrant children, true risk of infection and need for treatment is unclear.
Characterization of prevalence and risk factors for strongyloidiasis in the United States could assist health care providers and public health officials develop cost-effective comprehensive management plans in foreign-born immigrant children. Given gaps in literature, it is prudent to evaluate prevalence and risk factors for strongyloidiasis among foreign-born immigrant children in the United States. Our institution’s mobile clinic (MC) is an ideal site for such a pilot study. MC provides free medical services, including well-child examinations, to under-resourced children in an urban area in the United States. MC also provides care for a large proportion of newly arrived, uninsured Latino children from immigrant families. Characterizing the prevalence of strongyloidiasis among MC children could assist health care providers in providing effective, comprehensive care in resource-limited areas. The current study evaluates risk factors and describes the Strongyloides seropositivity rate among patients seen for well-child visits on MC.
Methods
We developed a survey (appendix) to capture participant demographics, environmental exposures, history of antiparasitic or antiretroviral medication use, history of medical conditions with increased strongyloidiasis risk (HIV infection, HTLV-1 infection, cancer, malnutrition, liver or kidney disease, receipt of organ or stem cell transplantation, immunodeficiency, or immunosuppression), and history of nonparasitic conditions associated with peripheral eosinophilia (such as environmental allergies and asthma). Certified bilingual members of the research team translated the survey into Spanish and reviewed the translation for accuracy. We used printed surveys in English and Spanish.
We offered enrollment to patients 0 to 18 years old who presented to the MC for a well-child visit and whose families spoke English or Spanish. MC providers informed caregivers about the study’s purpose and consent process, provided a written summary of the study and consent form, and obtained written consent. MC providers then assisted 18-year-old participants or caregivers of 0- to 17-year-old participants, as needed, to complete the printed survey.
On the MC, participants underwent venipuncture for (1) complete blood count (CBC) with differential and (2) Strongyloides IgG enzyme-linked immunosorbent assay (ELISA). Clinical staff performed venipuncture at the time of enrollment and attempted up to 3 times to obtain a sufficient blood sample. If unsuccessful, they requested participants return. If still unsuccessful after 3 attempts on return, the participant was removed from the study.
Study staff entered data from the written surveys and laboratory results into a REDCap instrument (REDCap version 12.4.8).14,15 A Strongyloides IgG ELISA result of index value (IV) < 1 was interpreted as negative, IV = 1 was equivocal, and IV > 1 was positive. We recorded AEC and considered an AEC >500 cells/µL to be indicative of peripheral eosinophilia.
We offered all participants with abnormal ELISA or CBC results medical management in accordance with clinic norms: ivermectin 200 μg/kg to participants with positive ELISA and retesting for participants with equivocal ELISA.
Our affiliated institutional review board approved this study.
Data Analysis
We report data as frequencies and proportions for categorical variables and as median and interquartile range (IQR) or mean and standard deviation (SD) for continuous variables. We determined differences between groups of ELISA results (negative, equivocal, and positive) by the Fisher exact test for categorical variables and by Kruskal-Wallis test or analysis of variance for continuous variables as appropriate. A P value of <.05 was considered significant.
Results
From October 2022 to June 2023, MC providers saw 309 patients for well-child visits (Figure 1), of which 228 consented to participate (74% enrollment). We excluded 22: 4 with incomplete study procedures and 18 with insufficient blood samples. We included 206 participants in our study. The median participant age was 8.72 years (IQR = 6.25-12.03) (Table 1). Gender distribution was nearly even, with 51.9% males and 48.1% females. The predominant ethnicity was Hispanic (n = 200, 97.1%). Participants self-reported race as 163 (79.1%) white, 9 (4.4%) black, 3 (1.5%) Asian, 1 (0.5%) Native American/Alaskan, and 11 (5.3%) other. Most (n = 201, 97.6%) were born outside of the United States. Countries of birth included Honduras (n = 46, 22.3%), Venezuela (n = 29, 14.1%), Cuba (n = 25, 12.1%), Mexico (n = 22, 10.7%), Nicaragua (n = 20, 9.7%), Guatemala (n = 17, 8.3%), El Salvador (n = 11, 5.3%), the United States (n = 5, 2.4%), and other countries (n = 31, 15.1%), namely Afghanistan, Bolivia, Chile, Colombia, Ecuador, Kazakhstan, and Spain. Regarding arrival to the United States, 142 (68.9%) participants reported arriving less than 1 year prior to study enrollment, 43 (20.9%) 1 to 4 years prior, and 16 (7.8%) 5 or more years prior. For participants who previously lived outside of the United States, more children were from urban (n = 111, 53.9%) than small town (n = 65, 31.5%) and rural areas (n = 15, 7.3%) (P = .03). Of note, some participants reported living in more than one country before moving to the United States. Only the first reported country was included in hometown area analysis.
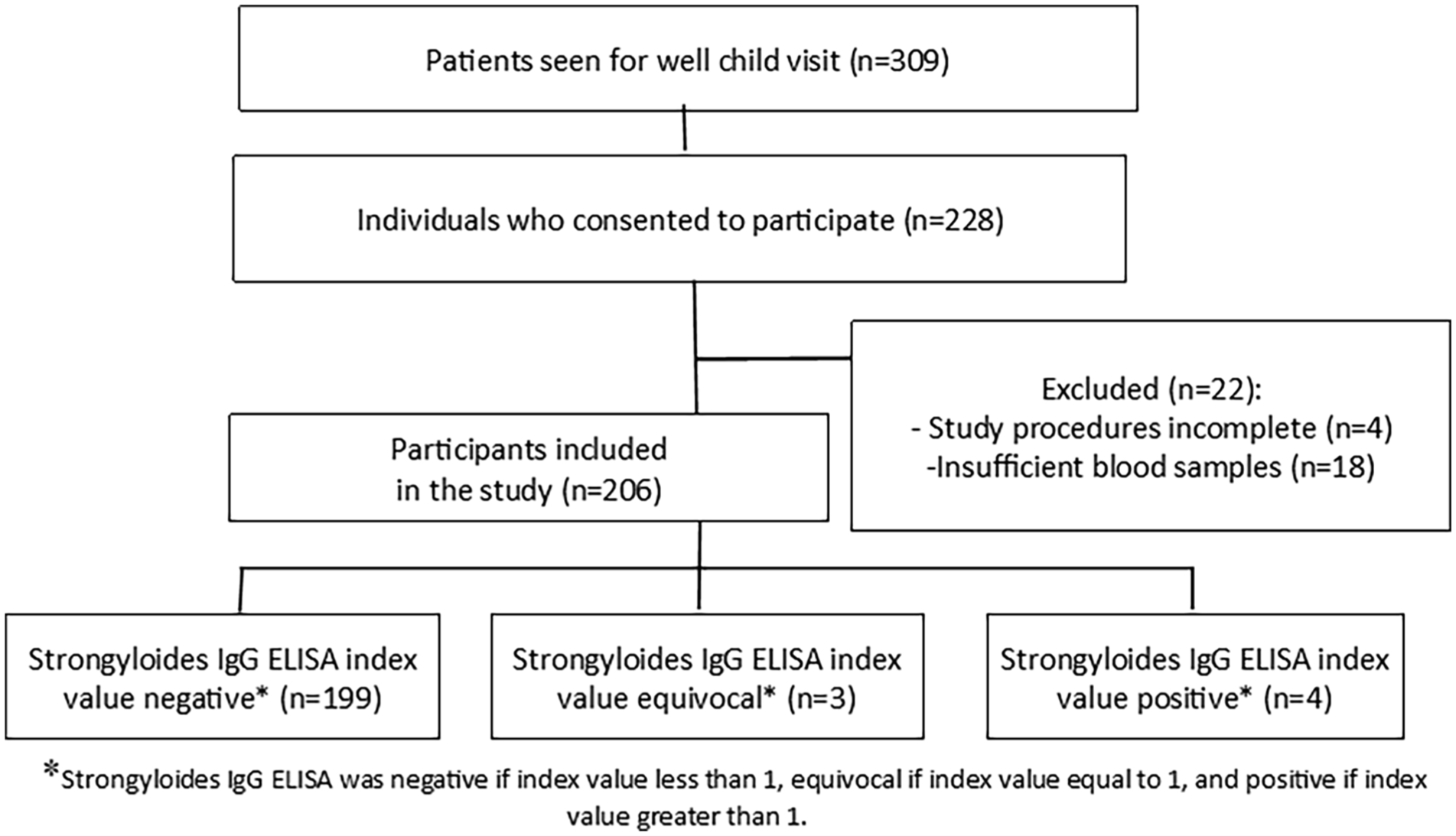
Flowchart of Study Participant Recruitment and Enrollment.
Demographics.
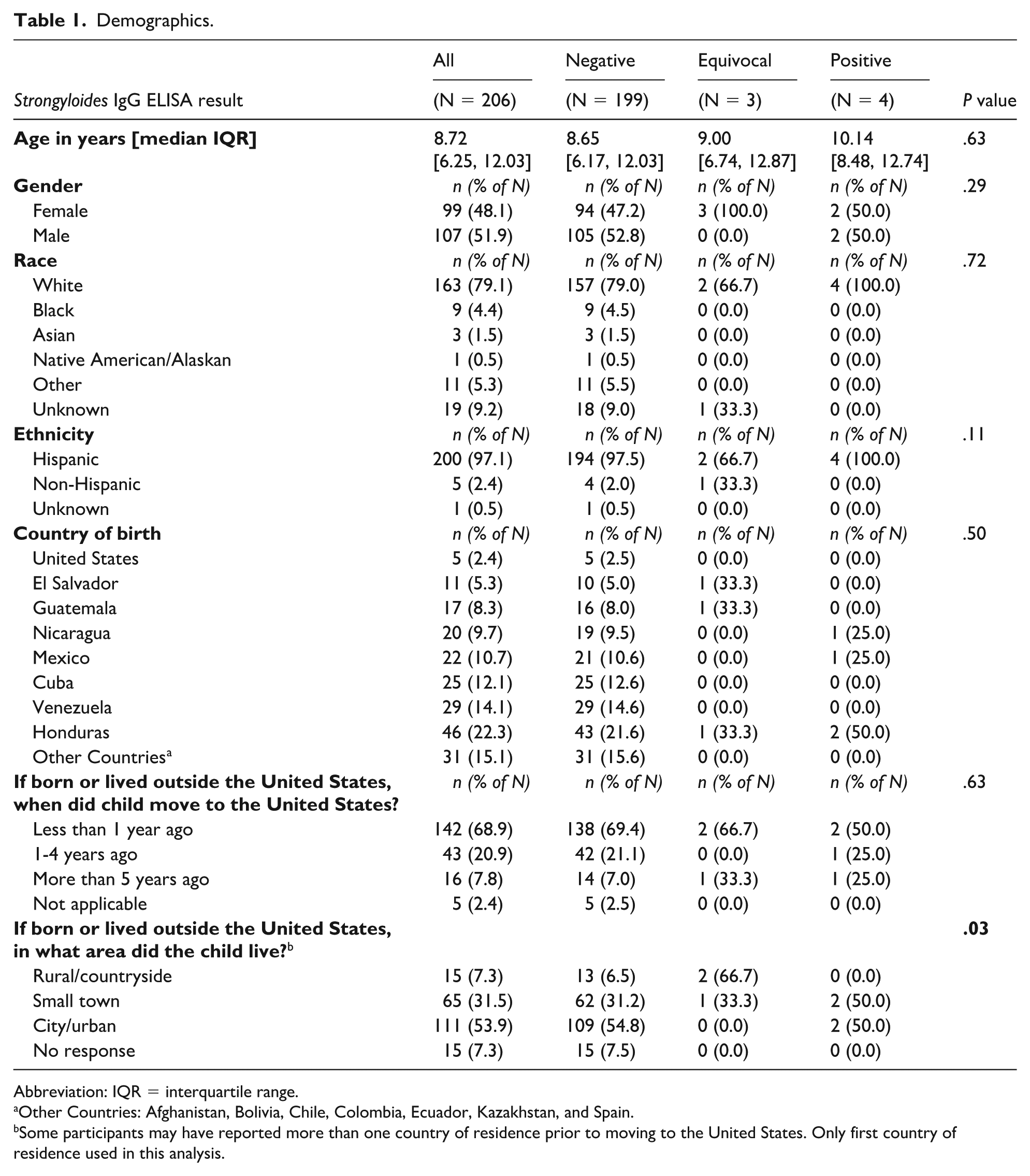
Abbreviation: IQR = interquartile range.
Other Countries: Afghanistan, Bolivia, Chile, Colombia, Ecuador, Kazakhstan, and Spain.
Some participants may have reported more than one country of residence prior to moving to the United States. Only first country of residence used in this analysis.
Of the 206 participants tested via Strongyloides IgG ELISA, 199 (96.6%) were negative, 3 (1.5%) equivocal, and 4 (1.9%) positive (Table 2). MC providers contacted the 3 participants with equivocal results, but none returned for retesting. The median AEC was 220 cells/μL; 27 participants (13.1%) had peripheral eosinophilia. We found no significant associations between ELISA result and country of birth (Table 1). We identified no significant associations between Strongyloides IgG result and the following variables: living in a home with or without potable water and/or a toilet or septic tank, going barefoot, regular exposure to animals (Table 3).
Laboratory Values/Absolute Eosinophil Count.
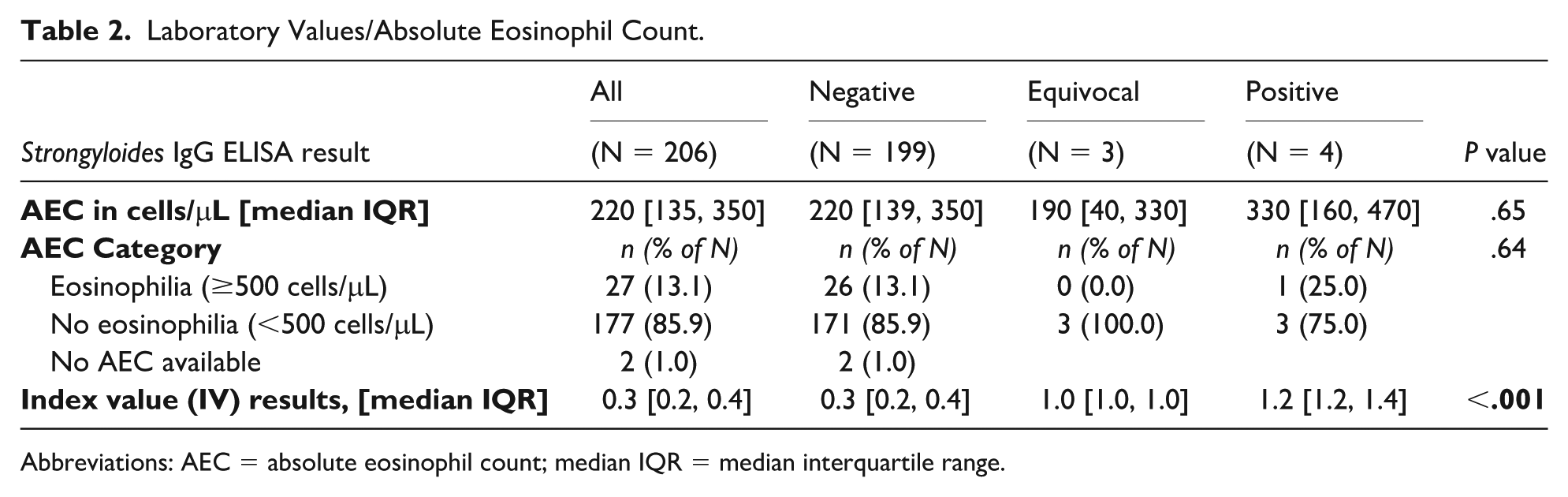
Abbreviations: AEC = absolute eosinophil count; median IQR = median interquartile range.
Home and Living Conditions.*
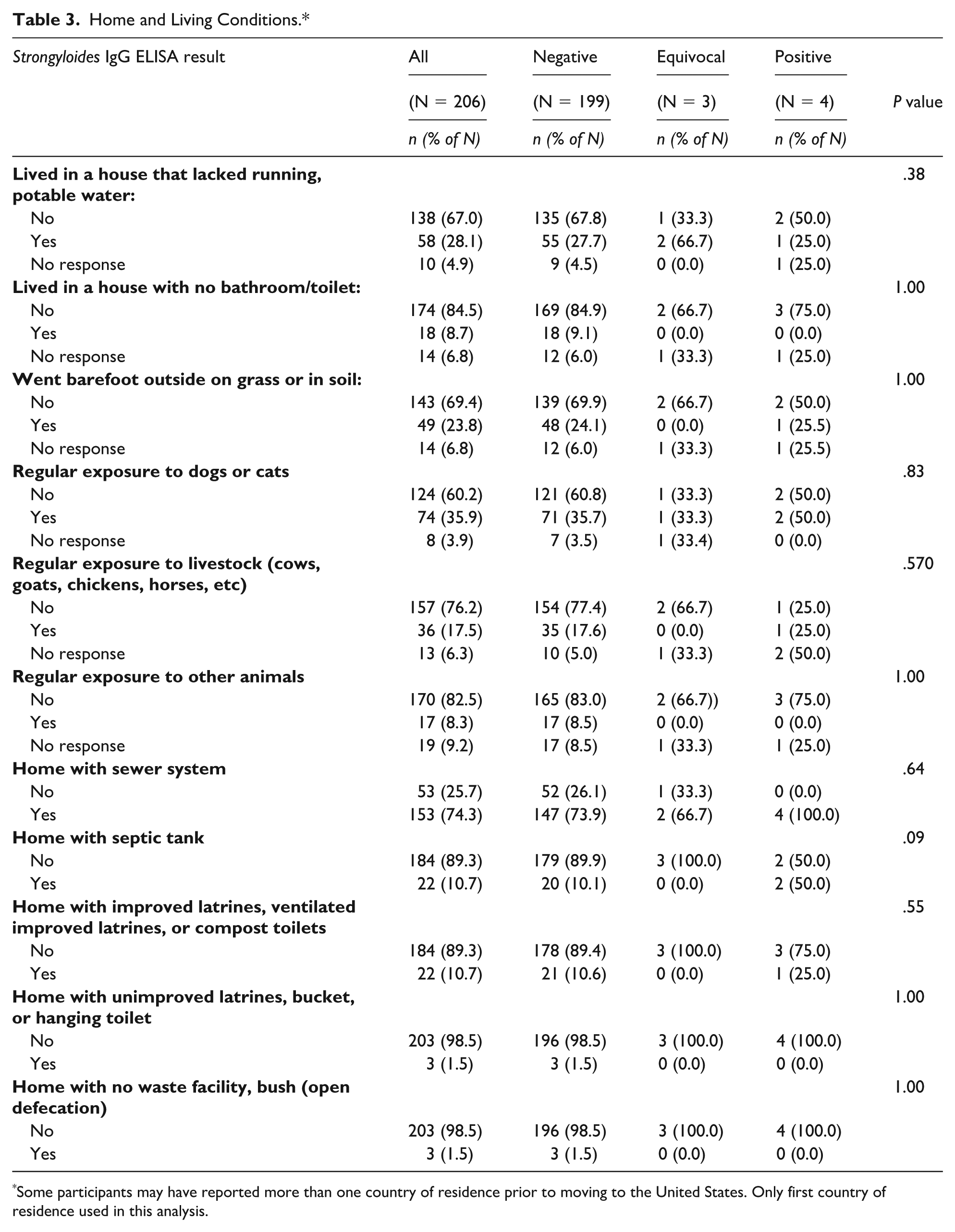
Some participants may have reported more than one country of residence prior to moving to the United States. Only first country of residence used in this analysis.
Fifty-four (26.2%) participants reported having symptoms in the month prior to enrollment, including abdominal pain, diarrhea, constipation, vomiting, rash, weight loss, and heartburn or acid reflux (Table 4). One hundred fifty-two (73.8%) participants reported no symptoms. The distribution of symptoms did not significantly differ between children with negative and positive ELISA results.
Symptoms and Medical Conditions.
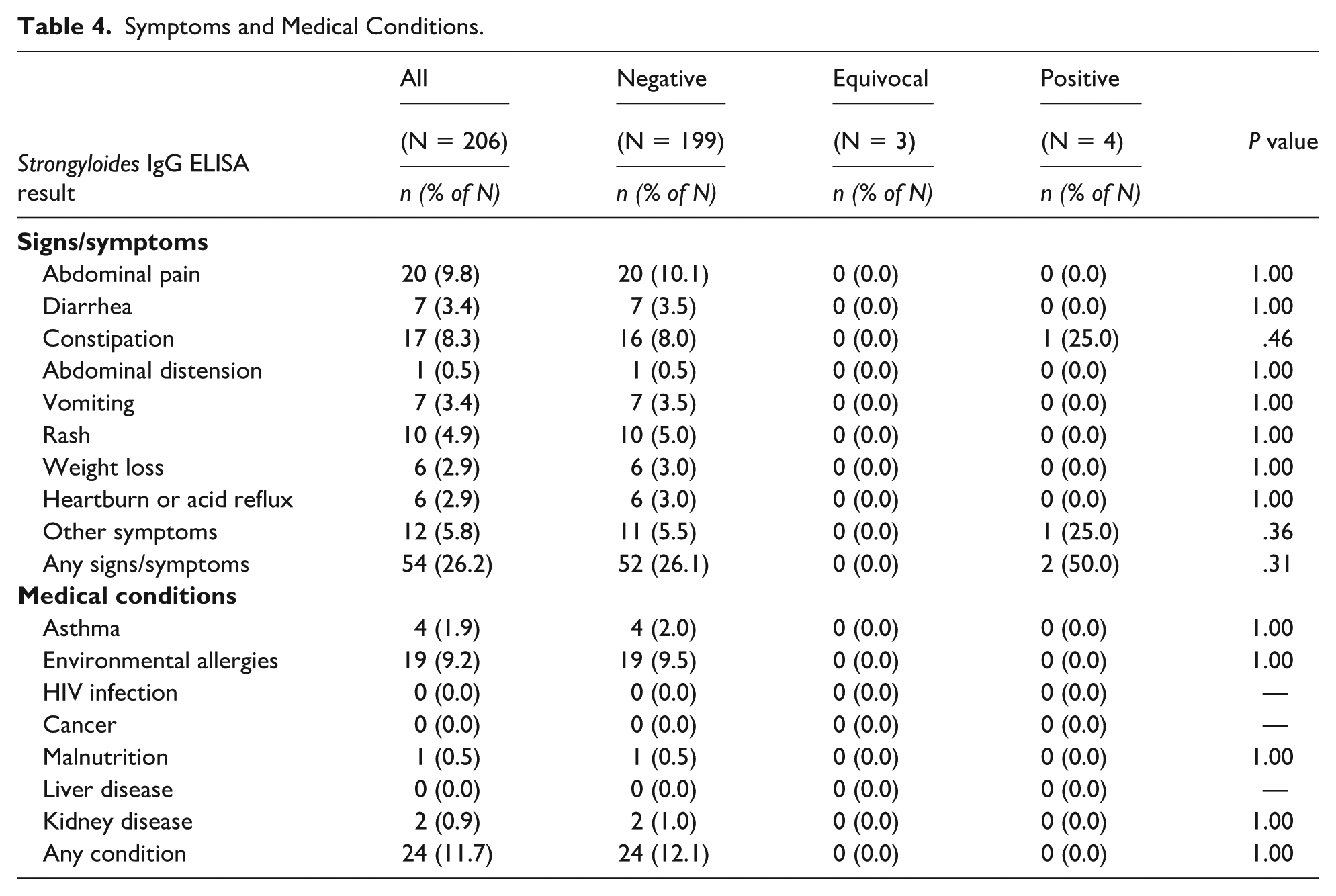
Only 24 (11.7%) participants reported a history of a medical condition, with the most common being environmental allergies (n = 19, 9.2%). We found no significant associations between history of a medical condition and ELISA result.
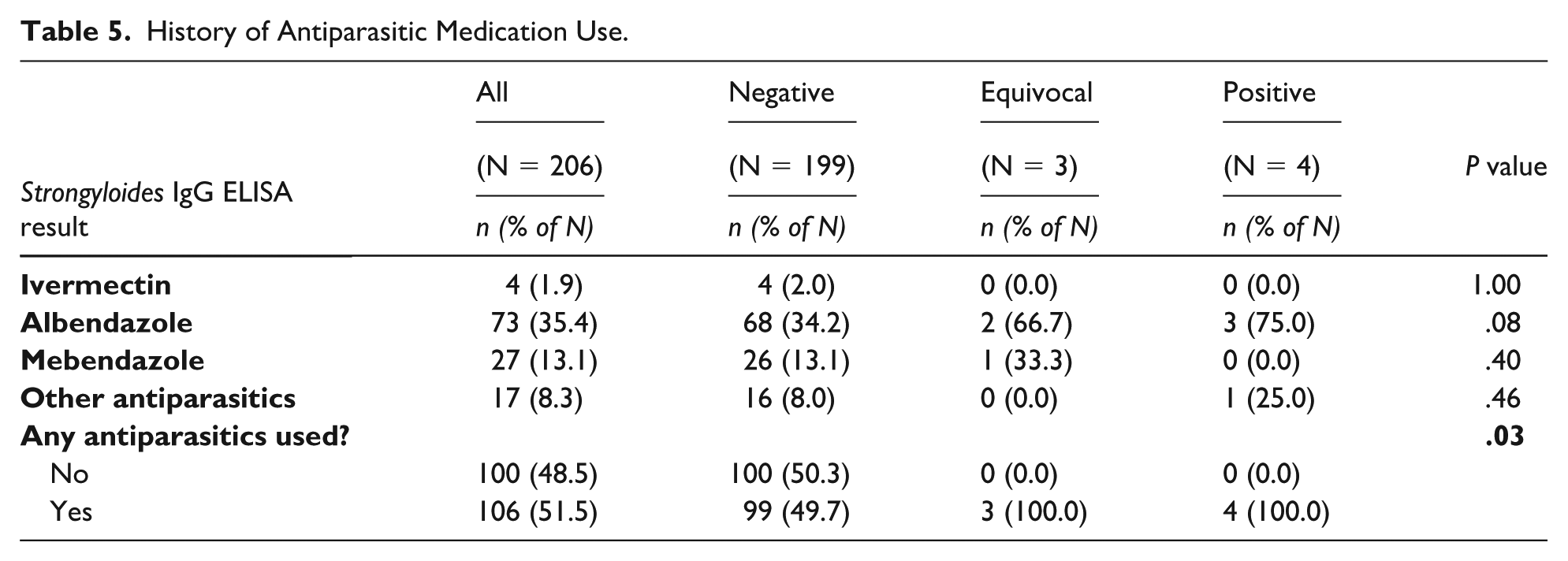
One hundred six (51.5%) participants reported use of any antiparasitic medication in their lifetime (Table 5). Albendazole was the most commonly used antiparasitic medication (n = 73, 35.4%), followed by mebendazole (n = 27, 13.1%), ivermectin (n = 4, 1.9%), and other antiparasitics (n = 17, 8.3%). We found no significant association between use of any antiparasitic and ELISA result.
History of Antiparasitic Medication Use.
All 4 participants with a positive ELISA result identified as white and Hispanic (Table 6). Their birth countries were Honduras (n = 2), Nicaragua (n = 1), and Mexico (n = 1). Two reported arriving in the United States less than 1 year prior, 1 arrived 1 to 4 years prior, and 1 arrived more than 5 years prior. Two reported no symptoms in the past month, whereas 1 reported constipation and 1 reported “other symptoms” without further explanation. All reported a history of antiparasitic medication use; 3 reported taking albendazole and 1 reported taking “other” antiparasitic. Only 1 reported a living condition known to increase their Strongyloides exposure risk, which was history of exposure to cats and dogs. Regarding AEC, only 1 participant had eosinophilia.
Characteristics of Participants with Positive Strongyloides IgG ELISA.

Participant numbers were randomly assigned and are not in chronological order of enrollment.
Discussion
Screening for and treating strongyloidiasis in resource-limited areas is challenging. To our knowledge, our study is the first to demonstrate that mobile clinics are a feasible venue for Strongyloides screening. Mobile clinics and other clinics in resource-limited areas often have the unique opportunity to care for foreign-born immigrant children. As this pilot study shows, these clinics can seek to describe the seropositivity of S. stercoralis among their patients, which would allow for an estimated prevalence of strongyloidiasis among this population.
We found a 1.9% S. stercoralis seropositivity rate among children presenting to MC for care, similar to estimates of 1.9% to 4.2% reported by studies of adult immigrants, asylees, and refugees in the United States8 -10 and to 2.3% seropositivity reported by a study of hospitalized children in Italy. 12 Unlike these studies, our study took place in a pediatric outpatient clinic serving an under-resourced, largely foreign-born immigrant population.
Our study aimed to describe risk factors for Strongyloides seropositivity in our mobile clinic patient population. However, our sample size did not yield enough seropositive participants to identify risk factors. We found no significant associations between Strongyloides seropositivity and presence of peripheral eosinophilia or AEC, birth country, living conditions/exposures, symptoms, or medical history. All seropositive participants were from Central America (Honduras and Nicaragua) or Mexico.
More than half of participants reported a lifetime history of antiparasitic medication use, most commonly albendazole and mebendazole. This likely reflects routine preventative chemotherapy (“deworming”) for STH in their countries of origin. The WHO recommends preventative chemotherapy with a single dose of albendazole or mebendazole either annually or biannually for all children above 12 months old, nonpregnant adolescent girls, and women of reproductive age where baseline prevalence of STH infection is at least 20%. 16 Fewer participants reported a lifetime history of ivermectin use, though strongyloidiasis is unlike other STH infections in that its treatment of choice is ivermectin instead of albendazole or mebendazole. 5 The WHO published their recommendation for preventative chemotherapy against strongyloidiasis in 2024, after our study concluded. 3 All participants with a positive ELISA reported prior use of at least one antiparasitic medication, suggesting that they had lived in higher-risk communities in their lifetime.
Our study had several limitations. We were unable to draw conclusions about active infection rates because we could not collect and evaluate stool specimens for ova and parasite microscopy. We did not pilot the survey; some participant families reported difficulty understanding several of our survey questions, including the questions about timeframe of sewage system exposure and presence of a septic tank at home. Such confusion may have affected our survey results. MC, like many other mobile clinics and clinics in resource-limited areas, cares for families facing multiple barriers to health care. Such barriers could have prevented more participants, potentially with positive ELISA results, from presenting for care and study inclusion. We included only English- and Spanish-speaking families, potentially leading us to miss seropositive children in families who speak other languages.
Conclusion
Strongyloidiasis is a potentially avoidable cause of morbidity in children globally, and children who immigrate to the United States from Strongyloides-endemic areas may unknowingly be infected with the parasite. While pediatric health care providers in the United States are advised to screen for or presumptively treat foreign-born immigrant children for strongyloidiasis, those practicing in under-resourced, outpatient areas recognize barriers to fulfilling this recommendation, including relatively high costs of testing and purchasing ivermectin, especially without health insurance. Health care providers need more information on prevalence and risk factors to develop appropriate management plans when resources are limited. This pilot serves as a foundation for future strongyloidiasis prevalence studies in foreign-born immigrant children who present to clinics in under-resourced areas such as mobile clinics.
Author Contributions
AL: contributed to conception and design; contributed to acquisition, analysis, and interpretation of data; drafted and critically revised manuscript; approved the final manuscript as submitted and agrees to be accountable for all aspects of the work
AG: contributed to design; contributed to acquisition of data; critically revised manuscript; approved the final manuscript as submitted and agrees to be accountable for all aspects of the work
NC: contributed to design; contributed to acquisition of data; drafted and critically revised manuscript; approved the final manuscript as submitted and agrees to be accountable for all aspects of the work
CD: contributed to design; contributed to acquisition of data; drafted and critically revised manuscript; approved the final manuscript as submitted and agrees to be accountable for all aspects of the work
EC: contributed to design; critically revised manuscript; approved the final manuscript as submitted and agrees to be accountable for all aspects of the work
AB: contributed to analysis of data; drafted and critically revised manuscript; approved the final manuscript as submitted and agrees to be accountable for all aspects of the work
SM: contributed to conception and design; contributed to acquisition of data; critically revised manuscript; approved the final manuscript as submitted and agrees to be accountable for all aspects of the work
Supplemental Material
sj-docx-1-cpj-10.1177_00099228251375274 – Supplemental material for Strongyloides Seropositivity Among Children Utilizing a Mobile Clinic
Supplemental material, sj-docx-1-cpj-10.1177_00099228251375274 for Strongyloides Seropositivity Among Children Utilizing a Mobile Clinic by Lynda Aririguzo, Aditi Gupta, Norma Castillo, Cassandra Duran, Eva H. Clark, Ankona Banerjee and Sanghamitra Misra in Clinical Pediatrics
Supplemental Material
sj-docx-2-cpj-10.1177_00099228251375274 – Supplemental material for Strongyloides Seropositivity Among Children Utilizing a Mobile Clinic
Supplemental material, sj-docx-2-cpj-10.1177_00099228251375274 for Strongyloides Seropositivity Among Children Utilizing a Mobile Clinic by Lynda Aririguzo, Aditi Gupta, Norma Castillo, Cassandra Duran, Eva H. Clark, Ankona Banerjee and Sanghamitra Misra in Clinical Pediatrics
Footnotes
Acknowledgements
The authors would like to thank Jill Weatherhead, MD, PhD, CTropMed from Baylor College of Medicine for her expertise and insight that were invaluable to this research.
ORCID iDs
Ethical Considerations
This study received ethical approval from the Baylor College of Medicine Institutional Review Board (protocol H-52050) on September 14, 2022.
Consent to Participate
All participants provided written informed consent prior to enrollment in the study.
Consent for Publication
Not applicable
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
At written request, an anonymized data set will be shared with other researchers.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
