Abstract
In May 2021, Qatar launched the BNT162b2 COVID-19 vaccine campaign for adolescents aged 12 to 15 years across all 27 health centers. Our study assessed the safety and efficacy of the vaccine among vaccinated and nonvaccinated adolescents in Qatar. Using a retrospective observational study, we analyzed the medical records of 1956 adolescents who were severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) positive from June 17 to December 17, 2021. The mean age for the vaccinated group was 13.89 ± 0.93 years, and for the nonvaccinated group, it was 12.99 ± 0.93 years. In the vaccinated group, 46% were male (n = 185) compared with 53% in the nonvaccinated group (n = 827) and 54% were female in the vaccinated group (n = 217) versus 47% in the nonvaccinated group (n = 727). Our findings demonstrate satisfactory protection provided by the Pfizer-BioNTech COVID-19 vaccine, with only one fifth of the study population contracting SARS-CoV-2 infections after the double-dose regimen. These results highlight the importance of maximizing vaccination coverage and considering booster doses for adolescents to enhance protection.
Keywords
Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection was declared a pandemic by the World Health Organization on March 11, 2020. 1 Worldwide as of October 17, 2023, 696 637 148 people are affected by SARS-CoV-2 infection, with mortality of 6 926 610 and recovered cases of 668 573 986. 2 The data from Qatar as on October 17, 2023, declare 514 524 positive cases with a mortality rate of 690, and recovered cases of 513 687. 3
Coronavirus disease 2019 (COVID-19) has affected the health and lives of the whole world with high morbidity and mortality. Compared with the adult population, the incidence of SARS-CoV-2 infection was lower among children and adolescents. National statistics from countries in Asia, Europe, and North America showed that pediatric cases account for 2.1% to 20.2% of confirmed SARS-CoV-2 infection cases.4,5 However, the impact in terms of severity, comorbidity, and mortality has been significant among children and adolescents. In the United States, SARS-CoV-2 infection ranks as 1 of the top 10 causes of death for children and adolescents. 6 In addition, children who contracted SARS-CoV-2 infection also develop serious complications, such as multisystem inflammatory syndrome in children (MIS-C), a condition where multiple organ systems including the cardiovascular, respiratory, renal, nervous, integumentary, or gastrointestinal systems undergo inflammatory responses. 6 Since the pandemic began, more than 2300 cases of MIS-C have been reported in children aged 5 to 11 years. 7 In addition to the physical harm, their intellectual and emotional development also had been adversely affected by the pandemic. It was estimated that approximately 1.5 billion young people worldwide had been obliged to stay at home, negatively influencing their health and social functioning. 8 Therefore, an urgent need for combating the deadly virus through mass vaccination has been adopted worldwide as a preventive and protective measure. On December 11, 2020, the US Food and Drug Administration (FDA) under an emergency use authorization (EUA) has authorized the emergency use of the Pfizer-BioNTech (BNT162b2) COVID-19 vaccine for individuals 16 years of age and above. Two doses are delivered, 3 weeks apart estimating the vaccine is safe and effective to prevent SARS-CoV-2 infection in individuals 16 years of age and older. 9 Since then, the BNT162b2 COVID-19 vaccine campaign has started worldwide among the adolescent population. In Qatar, the BNT162b2 COVID-19 vaccination campaign started on December 23, 2020, across primary and secondary care, allowing nationwide vaccination of people 16 years and above age group. 10 On May 10, 2021, the FDA expanded the EUA for the BNT162b2 COVID-19 vaccine in adolescents, that is, 12 to 15 years of age, for the prevention of SARS-CoV-2 infection. The FDA’s expansion of the EUA for the BNT162b2 COVID-19 vaccine to include adolescents 12 through 15 years of age is a significant step in the fight against the SARS-CoV-2 pandemic. 11 European and US agencies later recommended the use of the BNT162b2 COVID-19 vaccine as a 2-dose regimen in children aged 5 to 11 years and as a booster dose in adolescents aged 12 to 15 years. 12
Two doses of the BNT162b2 vaccine are highly effective across all age groups (≥16 years, including older adults aged ≥85 years) in preventing symptomatic and asymptomatic SARS-CoV-2 infections and associated hospitalizations, severe disease, and death. There were marked and sustained declines in SARS-CoV-2 infection incidence corresponding to increasing vaccine coverage suggesting the vaccine effective against SARS-CoV-2 infection. 13
On May 17, 2021, following the order of the Ministry of Public Health, the Primary Health Care Corporation (PHCC) Qatar started the BNT162b2 COVID-19 vaccine campaign among adolescents, that is, 12 to 15 years of age across its all 27 health centers. 14 This study focused on the efficacy of the BNT162b2 COVID-19 vaccine against the prevention of SARS-CoV-2 infection and its influence on the rate of hospitalization, morbidity, and mortality in Qatar. Although the clinical trials done by the BNT162b2 COVID-19 vaccine have shown 100% efficacy in 2260 enrolled adolescents from 12 to 15 years of age in the United States, 15 no similar studies on the effectiveness of vaccine on children are conducted in Qatar. In addition, to our knowledge, no high-quality randomized control trials are conducted to explain the safety and efficacy profile of the BNT162b2 COVID-19 vaccine among the 12 to 15 age group. Consequently, this article will summarize and analyze the safety and protective efficacy of the BNT162b2 COVID-19 vaccine among the vaccinated and unvaccinated adolescent limited subpopulation in Qatar to provide reliable evidence for further clinical application.
Methods
A quantitative retrospective observational study assessed the medical records of SARS-CoV-2–positive adolescents (12-15 years of age) of all 27 health centers of PHCC. All data were obtained during the second wave of the SARS-CoV-2 pandemic from June 17 to December 17, 2021. The study included anonymized details of SARS-CoV-2–positive adolescents who sought treatment in the health centers of PHCC. Severe acute respiratory syndrome coronavirus 2-positive status confirmed using reverse transcription polymerase chain reaction (RT-PCR) was taken as samples. The data were extracted from Health Information Management software named CERNER of the organization.
Data with anonymized personal identifiers were initially categorized into 2 groups: the vaccinated group (who have completed both the doses of Pfizer-BioNTech [BNT162b2] COVID-19 vaccine, with the second dose taken after 2 weeks or more) versus nonvaccinated group (who have not received any vaccine dose). A predetermined template focusing on the following attributes was used to classify the data into 4 major categories among vaccinated and nonvaccinated SARS-CoV-2 infection patients:
Symptomatic and asymptomatic RT-PCR–positive cases of SARS-CoV-2 infection.
Comorbidities or preexisting conditions.
Number/rate of hospitalizations related to SARS-CoV-2 infection.
Number/rate of deaths related to SARS-CoV-2 infection.
The participants were later categorized based on their age, that is, 12 to 13 and 14 to 15 years; sex: male and female; and nationalities: Qatari nationals and non-Qatari residents of Qatar.
Study Setting and Population
Primary Health Care Corporation is the primary health care provider for the people of Qatar. At the time of the study, the PHCC was operating 27 primary health care centers distributed in 3 regions of Qatar, namely, Central, Western, and Northern. The health centers (HCs) provide appropriate and effective health care services focused on the needs of patients. Services delivered emphasize health promotion, prevention, and diagnosis, as well as treatment and provision of long-term and appropriate support to patients and their families.
As of the most recent update in September 2023, the population of Qatar was estimated to be approximately 3 057 507 individuals. It is important to note that the population of Qatar is notably diverse, primarily characterized by a significant expatriate (non-Qatari residents) workforce, constituting 89% of the total population, and a smaller portion of Qatari nationals, making up 21%. This expatriate population in Qatar comprises individuals from a wide range of countries who have come to contribute their skills and expertise across various sectors, including construction, health care, education, and finance.
Ethical Guidelines
This study was conducted in accordance with the Declaration of Helsinki (1964).
Outcome Measurement
Outcome measurement for the efficacy of the BNT162b2 COVID-19 vaccine was analyzed by comparing the postvaccination outcomes observed in the vaccinated group with the nonvaccinated group. Criteria for efficacy measurement emphasize on the following:
Incidence of the SARS-CoV-2 infection.
Rate and severity of the symptoms specified to SARS-CoV-2 infection.
Rate of hospitalization due to SARS-CoV-2 infection including intensive care unit (ICU) admission.
Impact of comorbidities and chronic conditions on the SARS-CoV-2 infection such as diabetes mellitus, hypertension, dyslipidemia, asthma, chronic obstructive pulmonary disease (COPD), obesity, rheumatic fever, kidney diseases, neurological diseases, hematological diseases, and so on.
Specification on postvaccination duration among participants categorized in the vaccinated group.
Statistical Methods
Data management and statistical analysis were performed by STATA 11.2 (College Station, Texas). The chi-square test for goodness of fit was used to find the significant difference between the age groups, gender, nationality, type of swabs test, symptoms, and comorbidities with groups (vaccinated and nonvaccinated, respectively) and it is expressed as frequency and percentage. P < .05 is considered statistically significant.
Results
The study data involved 1956 SARS-CoV-2 infection (RT-PCR positive) cases with an inclusion criterion of adolescents aged between 12 and 15 years reported in the primary health care centers of Qatar. Out of 1956 children, 20.55% were vaccinated (n = 402) and 79.45% were nonvaccinated (n = 1554). The mean age for the vaccinated group was 13.89 ± 0.93 years and that for the nonvaccinated group was 12.99 ± 0.93 years. Among the vaccinated group (total n = 402), 37% were aged 12 to 13 years (n = 150) and 63% were aged 14 to 15 years (n = 252). In the nonvaccinated group (total n = 1554), 68% were aged 12 to 13 years (n = 1060), while 32% were aged 14 to 15 years (n = 494). In the vaccinated group, 46% children were male (n = 185) versus 53% in the nonvaccinated group (n = 827) and 54% children were female in the vaccinated group (n = 217) versus 47% in the nonvaccinated group (n = 727). In terms of nationalities, the total percentage of vaccinated Qatari citizens was 33% (n = 132) versus nonvaccinated Qatari citizens 50% (n = 779). For non-Qatari residents all calculated as a single group, the vaccinated group included 67% (n = 270) versus the nonvaccinated group 50% (n = 775) (Table 1).
Demographic Details of Vaccinated and Nonvaccinated Children.
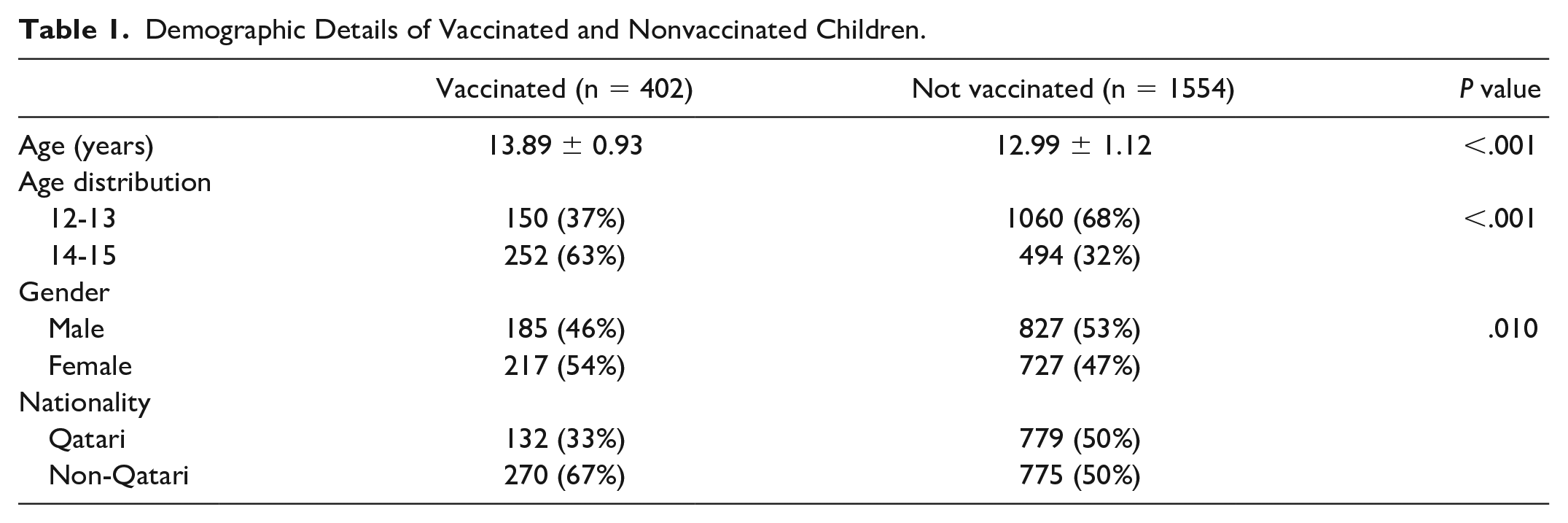
During the study period, the number of vaccinated participants who contracted SARS-CoV-2 infections was significantly less compared with the unvaccinated population. About 20.55% (n = 402) of the total sample size comprised the vaccinated group, while 79.45% (n = 1554) of the sample size comprised the unvaccinated group. Incidence of the SARS-CoV-2 infection was found to be statistically significant among the vaccinated group who received 2 doses of Pfizer-BioNTech (BNT162b2) COVID-19 vaccine (P < .001) rendering the vaccine effective against prevention of the SARS-CoV-2 infections (Table 1).
The association of the SARS-CoV-2 infections with hospital admission, existing comorbidities, and chronic illness analysis revealed relative similarity between the vaccinated and nonvaccinated groups. In the vaccinated group, 75% (n = 304) were asymptomatic (mild to moderate symptoms) and 23% (n = 96) were symptomatic (severe symptoms). Numbers seen in nonvaccinated group were 73% (n = 1142) asymptomatic (mild to moderate symptoms) and 24% (n = 378) symptomatic (severe symptoms). Among the vaccinated group, 4% (n = 19) were hospitalized with 0 ICU admissions, while for the nonvaccinated group, 5% (n = 85) were admitted to the hospital with 4 ICU (0.26%) admissions. In addition to asthma or COPD, children with other comorbidities including diabetes, hypertension, circulatory system disorders, hematological disorders, kidney diseases, neurological problems, and obesity were not found to be statistically insignificant in the development of complications with SARS-CoV-2 infection (Table 2). Samples contained a supportive medical history of asthma or COPD in both groups, that is, 8.7% (n = 35) in the vaccinated group and 11.6% (n = 181) in the nonvaccinated group.
Showing Types of Swabs, Presence of Symptoms, Admission, Comorbidities, and Chronic Patients Within Both Study Groups.
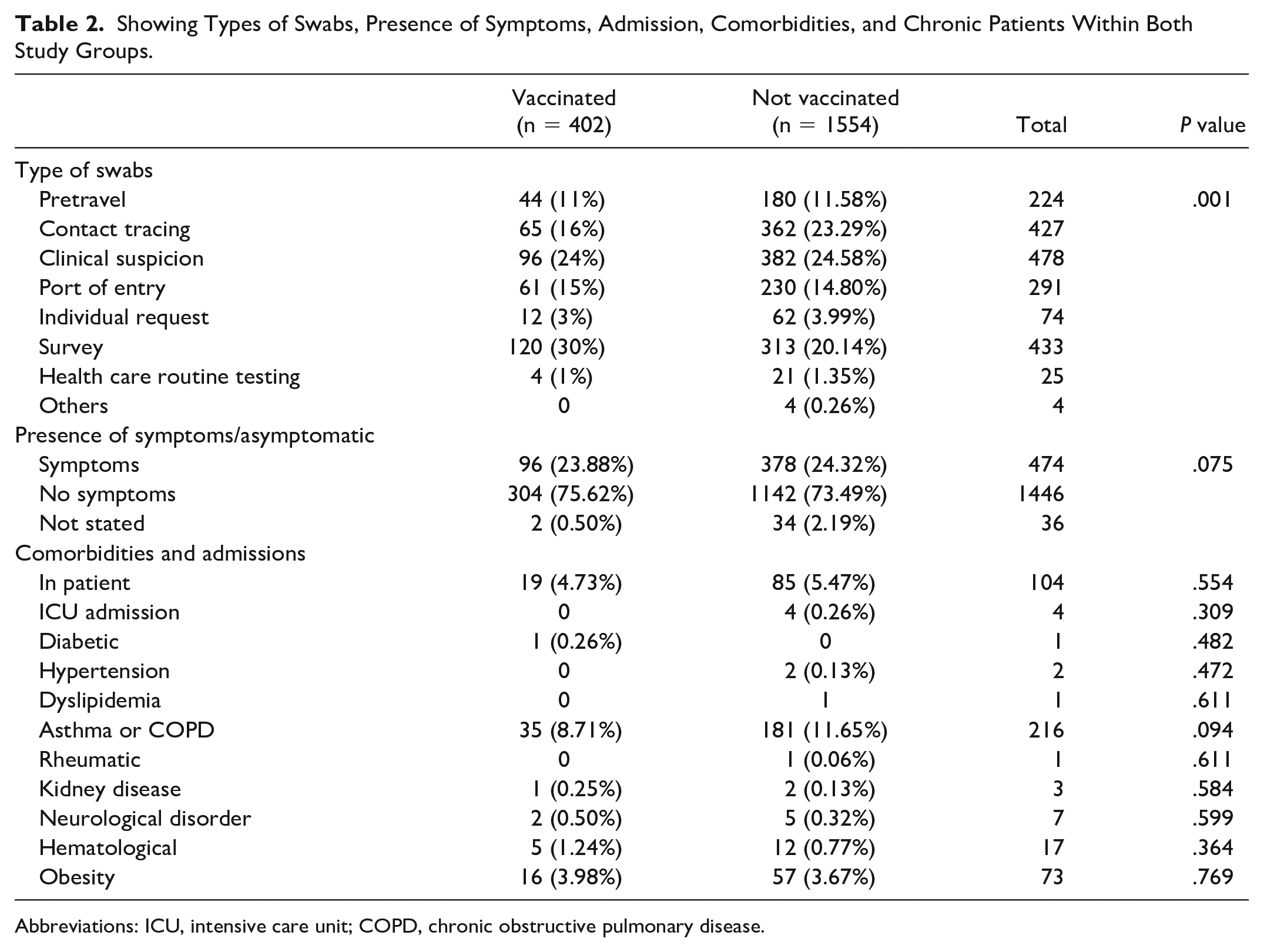
Abbreviations: ICU, intensive care unit; COPD, chronic obstructive pulmonary disease.
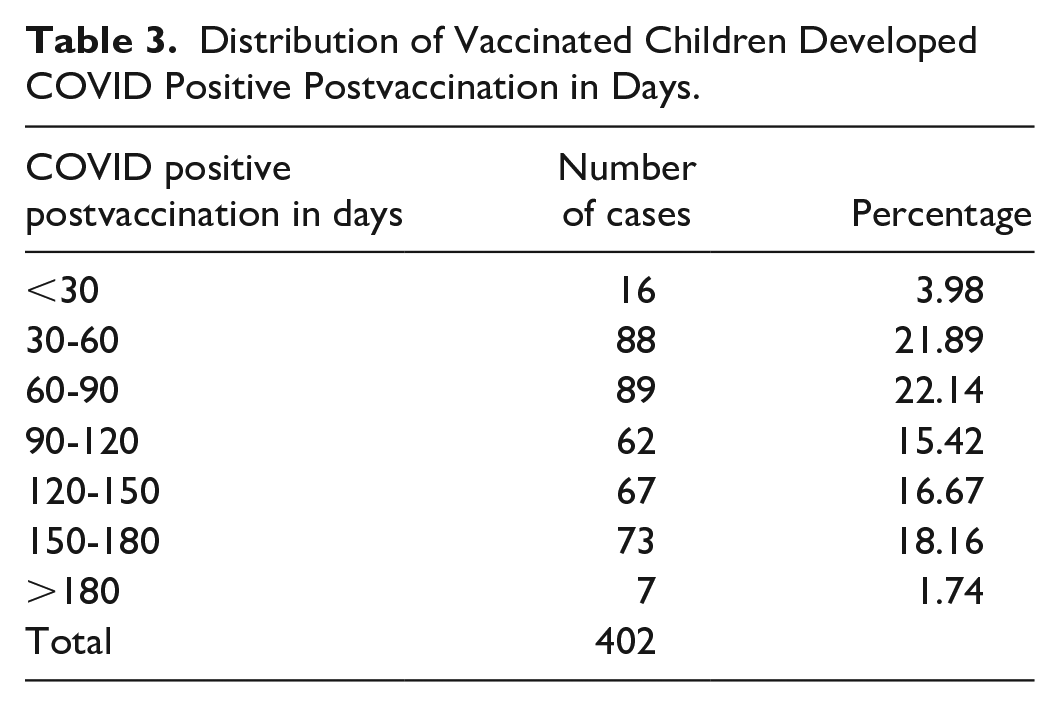
Further analysis of the association between the postvaccination period and the incidence of SARS-CoV-2 infection revealed that distribution of the positive cases after the BNT162b2 COVID-19 vaccine was found to be less during the initial 30-day postvaccination status. A total of 402 adolescents tested positive for SARS-CoV-2 infection as per postvaccination duration with the least during the initial month of vaccination and most of the cases between 30 and 90 days (Table 3). The maximum apparent effectiveness of the vaccines was noted during the initial 4 weeks of the vaccination with subsequent increased incidence in SARS-CoV-2 infections thereafter. See Table 3.
Distribution of Vaccinated Children Developed COVID Positive Postvaccination in Days.
An analysis of the reason for encounters among vaccinated and nonvaccinated individuals revealed different contexts. Among vaccinated individuals, encounter reason for the SARS-CoV-2 infection screening was majorly survey (30%) conducted as a part of the National Screening Campaign. Clinical suspicion from symptomatic infections was recorded next (24%) followed by contact tracing of the SARS-CoV-2–positive patients (16%). Port of entry after travel (15%) and pretravel screening (11%) were listed next. The remaining percentages were attributed to individual (3%) and health care routine (1%) testing. Similarly, an analysis of the nonvaccinated individual revealed a different trend: clinical suspicion (25.58%); contact tracing (23.29%); national survey (20.14%); post-travel screening (14.80%); pretravel screening (11.58%); and others (5.6%), respectively (Table 2).
Discussion
This retrospective observational study conducted in primary health care centers of Qatar showed that the incidence of SARS-CoV-2 infections among individuals who were fully vaccinated with Pfizer-BioNTech (BNT162b2) COVID-19 vaccine was significantly lower (20.55%) compared with the unvaccinated population (79.45%). The rate of infection was one fifth of the total samples denoting the decreased incidence among vaccinated adolescents. The findings are consistent with studies conducted in the United States, United Kingdom, and Israel.16,17 The vaccinated participants with a double-dose regimen showed increased efficacy in terms of humoral immunogenicity and prophylactic efficacy irrespective of the variants of the virus as the study was conducted during the omicron wave of the pandemic.18,19
The mechanism of development and maintenance of immunity following SARS-CoV-2 infections and BNT162b2 COVID-19 vaccination is still not well understood in children. Humoral and cellular antibody-mediated response to the actual virus or inactivated vector is depicted to play a major role in combating the infection and prevention of further onset developing antigen-specific adaptive immunity and memory against future infection.18,20 Combined IgM- and IgG-mediated immune response contributes to extended protection against the infection.21,22 An analysis of the association between the postvaccination period and the incidence of SARS-CoV-2 infection revealed that the distribution of positive cases after the COVID-19 vaccine was found to be less during the initial 30-day postvaccination status. Decreased incidence of SARS-CoV-2 infections among adolescents could be related to a stronger T-cell–mediated immune response of the BNT162b2 COVID-19 vaccine in preventing the infection. The observed drop in efficiency of the vaccine in preventing SARS-CoV-2 infections after 30 days and subsequently could be because of the waning of vaccine immunogenicity after immediate T-cell–mediated immunity and physiologic response to develop IgG-mediated immune response through memory mechanism.18,20 The subsequent reduction in the incidence of the infection after 90 days could be related to the memory-mediated immune mechanism. This difference may also be related to the fact that immune function is not yet well developed in young children.
The association of the SARS-CoV-2 infections with hospital admission, existing comorbidities including respiratory conditions, and chronic illness analysis revealed relative similarity between vaccinated and nonvaccinated in the vaccinated group. A similar trend was noticed in exhibiting symptoms associated with infections also. Fowlkes et al 19 cohort study among children and adolescents also revealed that irrespective of symptoms, 2 doses of the BNT162b2 COVID-19 vaccine were effective to prevent symptomatic and asymptomatic SARS-CoV-2 infection including delta and omicron variants (51% of omicron infections and 34% of delta infections were asymptomatic). The children who receive the COVID-19 vaccine have not only a lower risk of SARS-CoV-2 infection but also a significantly lower prevalence of various complications. 23 The findings of this study contradicted the causal link between SARS-CoV-2 infections with the rate of hospitalization; existing comorbidities; chronic illness; and need for critical care in developed nations and low- and middle-income countries.24-26
However, there are some limitations as well. First of all, the study was retrospective and there was no differentiation made among the variants of the SARS-CoV-2 strains. The data could be related/contradicted with the delta during late 2020 and omicron variants of SARS-CoV-2 virus during January 2022 where the efficacy of the BNT162b2 COVID-19 vaccine differed. The vaccine uptake was considerably low in the study population, which could have led to selection biases in the study groups. In addition, the increased incidence of SARS-CoV-2 infections during 30 to 60 and 60 to 90 days and further decrease after 90 days is contradictory and should be analyzed in detail seeking scientific justification. The lack of data on long-term follow-up on the efficacy of the vaccine and association with comorbidities and hospitalizations needs to be assessed further to understand the physiological and immunological variation of adolescents in the prevention, development, and surviving the infection. Further prospective studies will be needed to assess the duration of protection and immune prophylactic response and the need for booster doses.17,24 In a nutshell, the study revealed that the vaccine is effective in preventing SARS-CoV-2 infections and the immune efficacy of the vaccine decreased gradually over time demanding the need for booster dose.
Conclusion
In summary, our findings indicate that protection of Pfizer-BioNTech (BNT162b2) COVID-19 vaccine against SARS-CoV-2 infections is satisfactory as only one fifth of the total study population contracted SARS-CoV-2 infections after the double-dose regimen. However, it was found that protection against infection declined after 90 days of vaccination, making the population susceptible to the infection. The exact mechanism justifying the increased incidence of infection is not clear whether the drop in efficacy is due to memory-mediated humoral immune mechanism or other factors. Our findings support the importance of maximizing vaccination coverage for considering booster doses for adolescents to enhance protection. Further studies will be needed to assess the duration of protection and immune prophylactic response and the need for booster doses.
Author Contributions
MAA: Contributed to the conception, design, and data collection; drafted the manuscript; and revised it critically for final content.
MSH: Contributed to the conception of the proposal.
STV: Contributed to the design of the study; drafted the manuscript; and revised it critically for final content.
MSAM: Contributed to the conception of the proposal.
ACM: Contributed to the conception of the proposal; drafted the manuscript; and revised it critically for final content.
JA: Contributed to the data retrieval and analysis.
JIK: Contributed to the analysis and interpretation of data and results.
Footnotes
Acknowledgements
The authors would like to acknowledge the Primary Health Care Corporation Department of Clinical Research for the support and fund provided for the publication of this study. They take responsibility for all aspects of the reliability and freedom from bias of the data presented and their discussed interpretation.
Data Availability
The data sets generated and/or analyzed during this study are not publicly available due to the institutional rules and regulations but are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received funding approval for the article publication charge of 6500 QAR from the Primary Health Care Corporation Department of Clinical Research.
Ethical Guidelines
This project has been ethically approved by the Department of Clinical Research, Research Subcommittee in Primary Health Care Corporation, Doha, Qatar, with the Research Proposal Ref No: PHCC/DCR/2021/05/037. This study was conducted in accordance with the Declaration of Helsinki (1964)
