Abstract
Owing to coronavirus disease 2019 (COVID-19), lung damage is seen as an important problem in patients after recovery. In this study, evaluation of respiratory symptoms and lung ultrasonography (LUS) findings of those who have had symptomatic and asymptomatic COVID-19 disease in children was aimed. A total of 81 patients with positive and 18 healthy children with negative COVID-19 antibodies were included to the study. The most common late presentation symptoms were cough (85.2%), shortness of breath (77.8%), and chest pain (60.5%). In LUS, 2 or less B lines, 3 or more B lines, and Z line were seen in 66.7%, 33.3%, and 9.9% of patients, respectively. There was no significant difference between control and patients in terms of these parameters (P > .05). Pleural effusion was detected in 2 patients in the late period. Respiratory system findings may develop in the late period in patients infected with COVID-19. Therefore, patients should be followed closely.
Introduction
Coronavirus disease 2019 affects people of all ages.1-3 Recently, the clinical and radiological findings among infected children have been described.4-6 Although COVID-19 is thought to be more mild in children, COVID-19 pneumonia has also been described in asymptomatic or symptomatic children.1-3,5 In addition, pneumonia requires hospitalization with the need for intensive care unit (ICU) admission in some cases. 7 Impaired lung functions, as a common finding, are not expected to improve in a short period of time.3,8 Moreover, shortness of breath, chest pain, and cough may develop in the later period.2,6 For these patients, imaging techniques involving high levels of radiation such as computed tomography (CT) are used for diagnosis. Lung ultrasonography is another important tool that can be used in the diagnosis and follow-up of pneumonia in children. 9 In this study, evaluations of respiratory system symptoms and LUS findings of children with symptomatic and asymptomatic COVID-19 disease were performed.
Materials and Methods
The study was approved by the local ethics committee (identification number: E-46418926-050.01.04—37158). Follow-up was conducted between July 2021 and October 2021 in accordance with the ethical standards of the 1964 Declaration of Helsinki. All patients or their parents gave their informed consent to participate in the study.
Participants
All children under the age of 18 years7,10 with late-stage COVID-19 complaints who applied to the Pediatric Allergy and Immunology Polyclinics of a tertiary-level hospital were considered. Those who met the relevant inclusion criteria were enrolled in the study.
The study group (group 1) was divided into 3 subgroups with the following criteria:
Patients with a history of COVID-19 infection: those who had COVID-19 infection proven by polymerase chain reaction (PCR) test and positive antibodies.
Patients with asymptomatic disease: those who had contact with a confirmed case of COVID-19 infection, had no symptoms during the contact period, and had positive antibodies.
Patients with symptomatic disease: those who had contact with a confirmed case of COVID-19 infection and had mild symptoms but were not diagnosed with COVID-19 infection during the contact period and had positive antibodies.
The control group (group 2) included healthy children who did not have any complaints and came to the Pediatrics Polyclinic for sports reports. Children in the control group did not have a history of COVID-19 or exposure to COVID-19 infection and were negative for COVID-19 antibodies.
The definition of late COVID symptoms is not standardized, and there are various definitions in the literature. 11 However, its general definition is accepted as symptoms that occur 4 weeks or more from the first diagnosis of COVID-19 infection. 12 Similar to the studies,11,12 late-stage complaints were defined as symptoms that started at least 1 month after the history of COVID-19 infection or after the COVID-19 contact. Patients who had an infection or contact at least 1 month previously and whose complaints (cough, shortness of breath, or chest pain) had not improved at least 2 months later were included in the study.
In the American College of Chest Physicians, the Australian and New Zealand thoracic community, and in many studies, long-term cough is defined as a cough lasting longer than 4 weeks.13,14 In contrast, the British Thoracic Society defines a long-term cough as a cough that lasts longer than 8 weeks. Because of this confusion, this study consists of patients whose symptoms persisted for at least 2 months. At this study, demographic characteristics, medical histories, and complaints of the patients were recorded. They were carefully examined for specific cough pointers (recurrent pneumonia, cardiac abnormalities, chest wall deformity, digital clubbing, failure to thrive, feeding difficulties, hemoptysis, cyanosis, neurodevelopmental abnormality, choking episode, etc), which is an underlying disease finding.13,14 The patient’s contact with a person who has diagnosed as tuberculosis or has family history of tuberculosis was questioned. Furthermore, they were evaluated for gastroesophageal reflux symptoms. Those with specific cough pointers were not included in the study because they could be signs of underlying long-term diseases such as parenchymal lung disease, suppurative lung disease, serious systemic pulmonary illness such as cystic fibrosis, aspiration, vascular abnormalities, cardiac disease, immunodeficiency, and atypical infections. In addition, those with tuberculosis contact or family history and those with gastroesophageal reflux symptoms were not included in the study. In addition, patients with known underlying long-term lung, cardiac, rheumatological, or neoplastic diseases; long-term renal failure; immunodeficiency; congenital or anatomical defects of the airway; or a history of asthma treatment in the last 6 months were not included in the study.
Polymerase chain reaction results of the patients and history of hospitalization due to COVID-19 infection were recorded. Participants were also asked about the presence of stomachache, muscle pain, headache, joint pain, rash, fever, constipation, hair loss, or forgetfulness.
Total severe acute respiratory syndrome coronavirus 2 antibodies
Total severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) antibodies (total IgM and IgG antibodies) against SARS-CoV-2 in plasma samples were tested using enzyme-linked immunosorbent assay kits for SARS-CoV-2 (Roche Diagnostics, Germany) in our laboratory with the routine protocol after ensuring 2-level quality control. Results of the evaluated samples are given as reactive or nonreactive with cutoff values (cutoff index: COI). Cutoff values were interpreted as follows: COI <1, nonreactive and negative for SARS-CoV-2 antibodies; COI ≥1.0, and reactive and positive for SARS-CoV-2 antibodies.
Lung ultrasonography examination
Lung ultrasonography examinations were performed in an average of 8 minutes in a warm and quiet room with heated gel using an 8-12 MHz transducer (Toshiba Xario XG, Japan). The examination method was explained to the children, who were taken to the ultrasonography (US) examination room while accompanied by their parents. It was performed for the posterior part of the thorax on both sides, from basal to apex, and at the level of the costophrenic sinuses while the child was sitting. The movement and integrity of the pleura were evaluated while the child inhaled and exhaled. Reflection artifacts caused by air in the lung tissue and therefore the presence of A lines indicating aerated normal lung tissue were recorded. The mean number of B lines in each intercostal space perpendicular to the pleural surface and indicating that the interstitial space was filled with a material other than air was determined. It is physiologically normal to have 2 or fewer B lines in each intercostal space. This finding was also recorded. Z lines that were longer than B lines, crossed the entire US screen, and appeared perpendicular to the pleural surface were interpreted as artifacts. The presence and the number of Z lines in the intercostal space were recorded for each participant. Pleural effusion evaluation was performed by examining both costophrenic sinuses.
Chest X-rays (CXRs) of all participants were reviewed at the time of the diagnosis.
Statistical analysis
Descriptive statistics for categorical variables in this study were expressed as numbers (n) and percentages (%). Chi-square tests were used to determine the relationships between categorical variables. The statistical significance level (α) was taken as 5% in the calculations, and IBM SPSS Statistics 25 for Windows (IBM Corp., USA) was used for analysis. Values of P < .05 were considered statistically significant.
Results
Of the 110 patients who presented with late-stage complaints, 81 patients with positive COVID-19 antibodies were enrolled in group 1 while patients with negative results were excluded from the study. A total of 18 healthy children found to be negative for COVID-19 antibodies formed the control group (group 2). The mean age was 10.2 ± 4.6 (range: 1-17) and 10.3 ± 4.8 (range: 1-16) years in group 1 and group 2, respectively. Considering age and sex, no significant difference was found between the groups (P > .05). Patients’ most common late-stage complaints were cough (85.2%), shortness of breath (77.8%), and chest pain (60.5%). Muscle pain (14.8%), rash (14.8%), joint pain (8.6%), headache (6.2%), stomachache (3.7%), fever (2.5%), hair loss (2.5%), and constipation (1.2%) were seen less frequently. There was no difference between symptomatic and asymptomatic COVID-19 disease in terms of late-stage complaints (P > .05) in group 1. Of the included patients, 39.5% reported no COVID-19 contact. Only 38.3% (31 of 81) of the patients had a history of COVID-19, and 27.2% of the patients (22 of 81) had the disease asymptomatically. The remaining 34.6% (28 of 81) were patients with mild symptoms who were not diagnosed with COVID-19 at the time of symptoms. Five of 81 patients were hospitalized due to COVID-19-related pneumonia.
Polymerase chain reaction test results were positive for all 31 patients with a history of COVID-19 and 21 of the asymptomatic patients. The remaining 29 patients in the study group did not undergo PCR testing, and the diagnosis was made by measuring COVID-19 antibodies. Polymerase chain reaction results of patients in group 1 are summarized in the diagram.
Postexposure symptoms thought to be associated with contact with a case of COVID-19 (49 of 81 patients) developed after 1 month for 13 (16.6%) patients, after 5 months for 16 (19.8%) patients, and after 1 year for 20 (24.7%) (Table 1).
Demographic Characteristics of Groups, Late Symptoms, and History of Illness of Group 1.
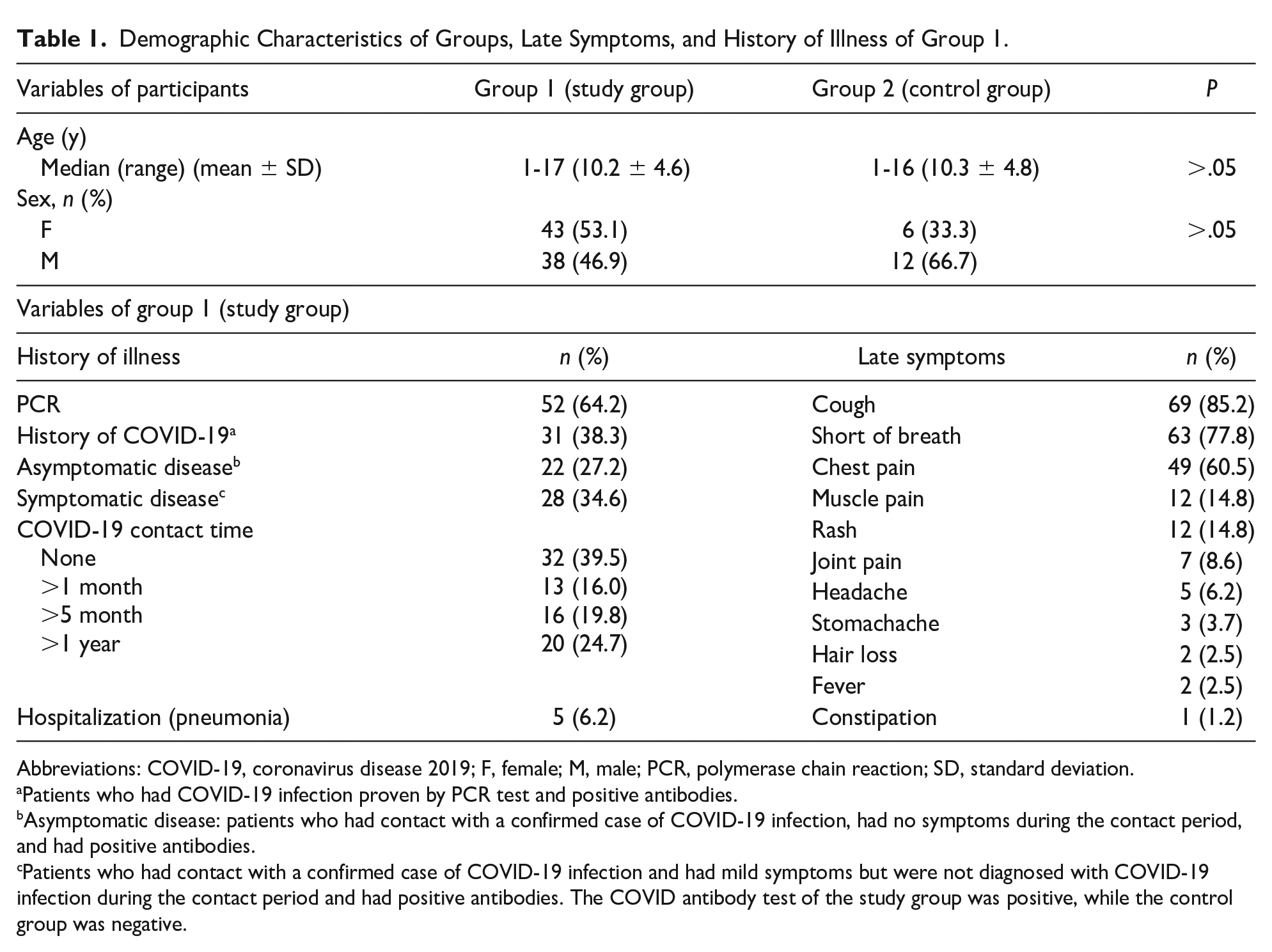
Abbreviations: COVID-19, coronavirus disease 2019; F, female; M, male; PCR, polymerase chain reaction; SD, standard deviation.
Patients who had COVID-19 infection proven by PCR test and positive antibodies.
Asymptomatic disease: patients who had contact with a confirmed case of COVID-19 infection, had no symptoms during the contact period, and had positive antibodies.
Patients who had contact with a confirmed case of COVID-19 infection and had mild symptoms but were not diagnosed with COVID-19 infection during the contact period and had positive antibodies. The COVID antibody test of the study group was positive, while the control group was negative.
Lung Ultrasonography Findings of Groups.
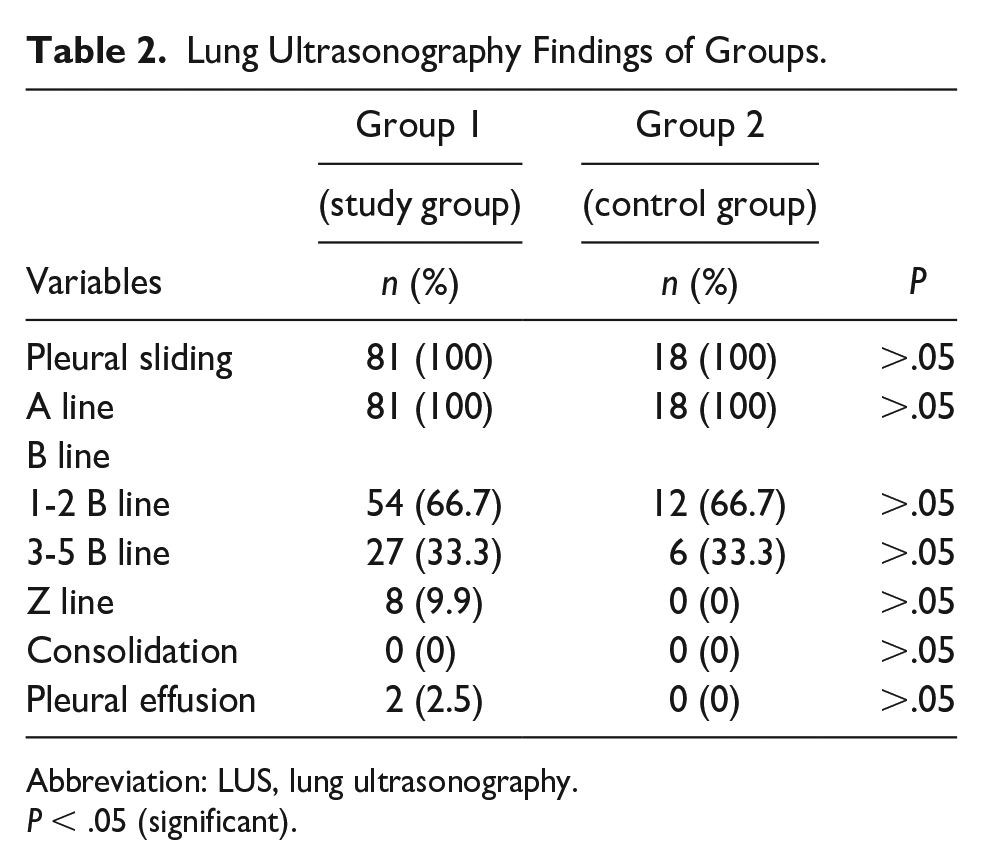
Abbreviation: LUS, lung ultrasonography.
P < .05 (significant).
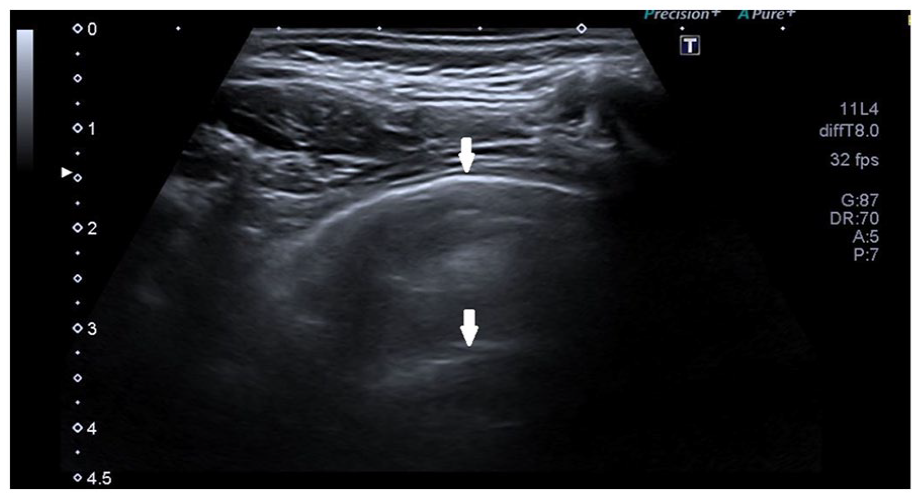
Normally ventilated lung tissue.
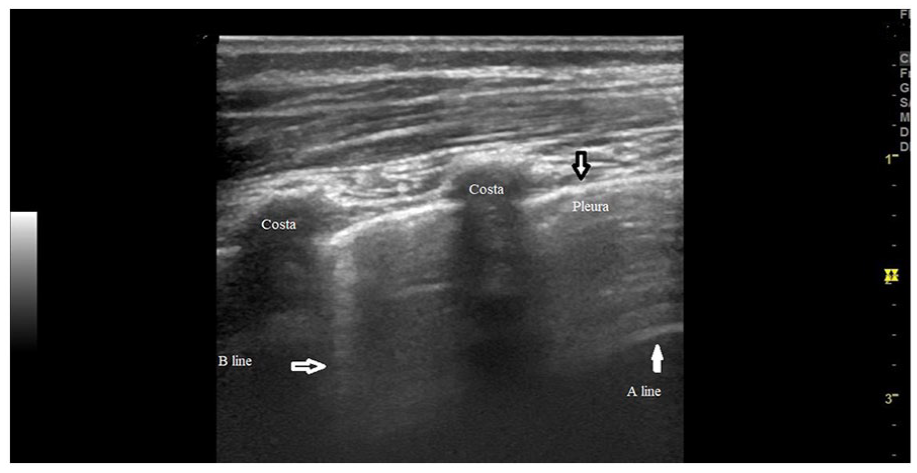
One B line in each intercostal space.
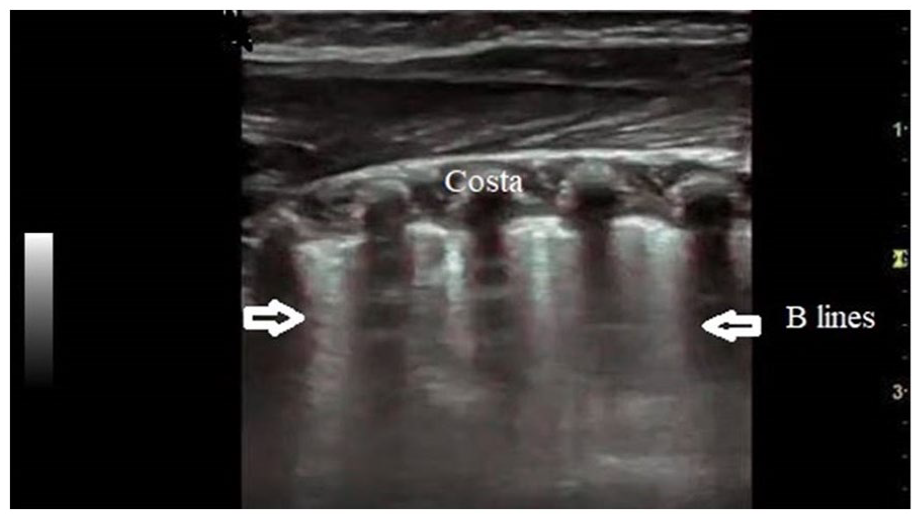
Multiple B lines in each intercostal space.
Control ultrasonography (USG) performed 1 month later in 2 patients with pleural effusion showed that the effusion improved and their symptoms regressed. In addition, 19 of 27 patients with B lines of 3 or more came to follow-up, while the others did not. It was shown that the symptoms of these patients regressed and the B lines on LUS decreased. Sample cases related to these results and change in LUS image with improvement in their symptoms are shown in Table 3.
Samples of Patients With 3 or More B Lines and Change in Lung Ultrasonography Image With Improvement in Their Symptoms.

Abbreviations: F, female; LUS, lung ultrasonography; M, male.
No specific finding was detected in the CXRs of any of the participants.
Discussion
How SARS-CoV-2 infection manifests in the late period is still unknown.15,16 Although there are many studies in adults,17,18 there are few reports of long-term COVID-19-related changes in respiratory function in children. 19 There is no consensus about the management and treatment of long-term sequelae, and studies are still ongoing.17,18 Among hospitalized adult patients with COVID-19 who did not require mechanical ventilation, one third reported dyspnea and cough and about half reported fatigue after discharge. 18 In 2 different studies with small numbers of children, no significant deterioration in diffusing capacity was found 2 months after COVID-19 infection.20,21 In another study conducted with a total of 73 children with a history of COVID-19 infection, either symptomatic or asymptomatic, it was shown that respiratory functions at an average of 2.6 months after infection were not different from those of children without a recent history of infection. 19
Severe acute respiratory syndrome coronavirus 2 basically enters the body through the respiratory tract and most commonly presents with COVID-19 pneumonia. 16 Acute lung injury may result in pulmonary fibrosis, leading to long-term deterioration of lung function.22,23 In adult studies, every additional comorbidity such as cardiomyopathy, long-term obstructive pulmonary disease, long-term renal failure, hypertension, obesity, organ cancer, and stroke was found to increase the risk of death.24,25 Furthermore, a higher prevalence of hypertension, diabetes mellitus, and cardiovascular disease has been reported in COVID-19 patients. 25 In this study, a comparison was not made in this regard because none of the patients had comorbid diseases and none required hospitalization in the late period.
Even if children have asymptomatic COVID-19 infections, they may have findings in their lungs at that time. Therefore, they may present later with respiratory system findings. Approximately one third of the patients in this study presenting with respiratory system findings in the late period had the disease asymptomatically. The occurrence of similar findings in the late period in children who had both symptomatic and asymptomatic COVID-19 suggests that COVID-19 caused similar damage to the lungs of these children. It is necessary to conduct a more comprehensive study on this subject.
It was previously reported that symptoms recurred in some patients (5 of 55 patients; 9%) who were discharged after having COVID-19 pneumonia. 26 Four of those 5 patients presented with fever, 1 had cough, 1 had sore throat, and 1 had constipation. All patients reported fatigue. 26 In our study, cough, shortness of breath, and chest pain were the most common symptoms while muscle pain, rash, joint pain, stomachache, fever, hair loss, and constipation were also seen to lesser extents. These findings suggest that patients should be followed in terms of the possible re-emergence of symptoms or the occurrence of new symptoms in the future.
One of the commonly used methods to diagnose COVID-19 is CT, but in follow-up, it may not be necessary for every patient. It is inappropriate to perform unnecessary diagnostic CT for children due to the harmful effects of radiation. Sedation may also be required for children while CT is performed. The harmful effects of CT can be prevented by performing LUS for children who have had a COVID-19 infection or who have been in close contact with a person infected with COVID-19. If there is no improvement despite the treatment given, CT can be used as a second choice for limited patients.
Although CXRs are frequently used for the diagnosis of respiratory diseases, including COVID-19 pneumonia, the findings are often nonspecific. 27 There are some studies of LUS findings in children during the period of COVID-19 infection, and LUS has been suggested as a promising diagnostic method by some pediatric studies.1,7 The efficacy of LUS in detecting pneumonia of any cause in children has been reported,10,28 and its accuracy is comparable to that of CXRs in detecting lung abnormalities in cases of COVID-19 infection. However, it was shown that US findings alone were not enough to predict COVID-19 infection in children.1,7 Vertical artifacts (70%) and pleural disorders (60%) have been reported to occur frequently, and consolidation and white lung appearance in the subpleural spaces during COVID-19 infection are also seen to a lesser extent in children.1,3,7 However, these various types of pulmonary involvement cannot be differentiated from the effects of other viral respiratory tract infections.1,29,30 In pediatric COVID-19 patients, and especially those with moderate disease rather than mild disease, pleural irregularities, subpleural consolidations, and B lines were observed in LUS, which may be compatible with viral bronchiolitis. 31 Most patients showed improvements in control LUS. 31
It has been reported that the findings of LUS performed for children with and without COVID-19 infection with similar symptoms were not different. 7 These LUS findings were bilateral, revealed consolidations, and included more than 3 B lines in both groups. Furthermore, pleural effusion was not detected in any patients. 7 It was stated that none of the findings were specific to COVID-19 infection. 7 The results of our study support these findings as no abnormalities identified COVID-19 infection. In our study, no differences were found between control subjects and symptomatic or asymptomatic patients in terms of LUS findings. However, in another study, it was shown that the LUS findings for patients with COVID-19 were mostly interstitial pulmonary edema and pulmonary consolidations occurring in the subpleural and peripheral pulmonary regions. 3
In studies conducted during the acute period of COVID-19, it has been reported that CXR findings are similar to those described in CT, but with less sensitivity. Therefore, children with COVID-19 may be referred to CT for diagnostic purposes. 32 CT has high sensitivity, so it has become an important diagnostic tool for COVID-19. In our study, we could not detect any significant findings in the CXR, since the patients were not in the acute phase of the infection. Other reasons were that they presented in the late period and their clinics were not severe. Although CXR findings were not specific in our study, CT is still planned due to the insensitivity of CXR and ongoing concern for serious COVID-19 pneumonia. The success of LUS in detecting the presence or absence of pneumonia is known. Moreover, the diagnostic efficiency of LUS is high, especially in severe cases but low in mild and moderate cases.3,27 At the same time, moderate agreement has been found between CT and LUS in detecting pulmonary manifestations of COVID-19. Some researchers have noted that LUS is useful in the diagnosis and monitoring of pediatric COVID-19 pneumonia.1,27 Thus, unnecessary radiation exposure and sedation among children can be avoided. 1 Because of this advantage, this concern can be alleviated by applying it instead of CT.10,28 In our study, we examined late-stage LUS findings among children. Pleural sliding and A lines were observed in all patients. Two or fewer B lines were seen in 66.7% of the cases, and 3 or more B lines were seen in 33.3% of the cases at each intercostal space. Z lines were seen in 9.9% of the cases. No differences were found between the patients and the control group in terms of these findings. Furthermore, no consolidation was observed, but pleural effusion was seen in 2 cases. In this study, we may not have detected different pathologies by LUS because none of the cases were severe and pneumonia was not observed. Nevertheless, we think that the routine use of LUS is useful in the diagnosis of COVID-19 pneumonia in children, and this is also valid for those with late-stage respiratory system findings, especially in severe cases.
Families are particularly worried about the risk of sequelae developing in the lungs due to COVID-19. Therefore, CT may be performed in the early period to make the diagnosis, but LUS is a good alternative for initial lung imaging. Computed tomography imaging can be performed for patients who do not improve during follow-up. Lung ultrasonography is a method that can be used easily for children, and it can reduce radiation exposure by reducing the number of radiological examinations. It may also be useful in confirming the diagnosis and following children who have had symptomatic or asymptomatic COVID-19 in the late period.
Limitations of the Study
An advantage of this study was that the person performing the LUS was an experienced pediatric radiologist. On the contrary, there are some limitations of this study.
During the pandemic period, hospital admissions of healthy children were low. Therefore, the number of control subjects enrolled in this study was relatively low. Asymptomatic patients were thought to have acquired the virus through contact with COVID-19 patients. It is unclear how soon after infection symptoms appear. Initial PCR testing could have shown when these children acquired the COVID-19 virus.
At the time of the study, the pandemic was ongoing, and the rate of COVID-19 was still very high. In addition, the viral panel could not be routinely performed under the current conditions. Since the children were at home during the pandemic, it was thought that symptoms were due to COVID-19 rather than other infections. In addition, COVID-19 infection was considered as a priority because the COVID-19 antibodies were positive even though the children were at home. If necessary, the next step was to investigate other viral and bacterial infections. However, the inability to definitively exclude other viral and bacterial infections is one of the limitations of the study.
Meanwhile, since the children did not have initial LUS images recorded during the active disease period, a comparison could not be made in this respect. We could not determine the sensitivity and specificity of LUS as our patients’ symptoms were not severe and CT could not be performed due to ethical constraints.
Patients with symptoms but with negative antibodies were excluded from the study. We would have had more comprehensive results if we had included such patients in the comparisons. Further studies with larger cohorts and longer durations are needed to obtain definitive findings.
Conclusion
It should be kept in mind that patients with COVID-19 may develop respiratory system findings in the late period, and they should be followed closely in this respect. This study is meaningful in terms of showing that late symptoms may also occur in patients who had the disease asymptomatically. In addition, it has been shown that unnecessary and invasive examinations can be avoided by expanding the use of LUS. Evaluation of the lungs by performing LUS for children is meaningful and useful. It also provides great convenience for pediatric patients, their families, and health care professionals by reducing the need for radiation and sedation in children. This study is important because of its emphasis on the need for longer clinical follow-up of patients with COVID-19 and the treatment of long-term sequelae. Larger randomized controlled studies with larger numbers of participants are needed in terms of follow-up and treatment management.
Authors’ Contributions
FD and AMK participated in the design, execution, and analysis of the paper equally. They read and approved the final version of the manuscript.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
