Abstract
The demand for natural infant care products, including diapers, has increased. However, few disposable diapers have been able to provide the performance caregivers desire while also incorporating ingredients consistent with the “natural” category. In an examiner-blinded clinical study, the performance of a new cotton-enhanced diaper with high-performance materials was compared with an existing natural diaper offering. A total of 131 infants wore 1 of the 2 diapers for a 4-week period. Diaper performance was assessed based on skin marking assessments, scored by a trained grader, and incidence of diaper dermatitis. Skin grading for diaper dermatitis was assessed at 4 sites in the diaper area. The new diaper offering was associated with less skin marking and significantly less diaper rash at the genitals and intertriginous regions versus the comparator. These data suggest that the new diaper provided significant improvement in both skin marking and prevalence of diaper rash.
Introduction
Diapers are an important and necessary product used by the incontinent child. At birth, the diaper is one of the first products worn by the newborn. In the mid-20th century, the predominant diaper was a cotton-containing cloth diaper. Cloth diapers have relatively poor absorbency, especially under high urine and stool loads, leading to diaper leakage. Furthermore, these products were inferior at sequestering urine and feces away from the infant’s skin, resulting in skin overhydration and enzymatic attack of the skin by fecal proteases and lipases, as evidenced by severe diaper dermatitis (DD) rates of ~60%. 1 The availability of disposable diapers containing cellulose reduced severe DD rates to ~39%, which was further reduced by superabsorbent diapers (29%), with even lower severe diaper rash observed in present-day diapers with superabsorbent polymers and breathable materials. 2 As such, diaper design plays a crucial role in infant health.
Infant skin is especially susceptible to irritation, particularly in the diapered area where it is in close contact with urine and feces. Irritant DD, also known as diaper rash, is a common skin condition among infants, which is caused by a mix of exposure to friction, excess moisture, and increased pH from urine and feces, and triggers an inflammatory response that is erythematous, papular, scaling, and sometimes painful.2-5 Published data indicate that DD prevalence is estimated to be between 25% and 50%, depending on age and the methods applied in specific studies. 6 Other estimates indicate that approximately 50% to 65% of babies will suffer from diaper rash at some time in their life. 7 The peak incidence of DD is generally believed to occur between the ages of 9 and 12 months. 8 DD is not inconsequential from a health care utilization standpoint, accounting for almost 20% of childhood dermatology visits. 9 The emergence of ultra-absorbent disposable diapers, which better protect against prolonged contact between skin, urine, and feces, has helped improve the care of diapered skin, particularly in reducing the severity and incidence of irritant DD.2,10,11 It is estimated that after the introduction of ultra-absorbent components into the diaper core in the late 1980s, the incidence of moderate to severe DD decreased by 50%; evolving designs continue to improve on the absorption properties of diapers today.1,2
In recent years, diaper design has been evolving alongside increasing demand from consumers for products that are natural or naturally derived as these are perceived by some to be better for infant skin. The natural category of personal care products has seen dramatic growth. For instance, the global organic and natural beauty market has an estimated value of $13.2 billion and is rapidly growing. 12 Growth in natural cosmetics has increased 7% in the United States, as compared with a 2% rise in the overall beauty market in 2015. 13 These trends in cosmetics and other personal care products are being mirrored in the diaper category as well. A recent survey indicated that mothers regularly choose products from the natural and organic category at 4 times the rate they did 10 years ago. 14 Furthermore, 40% of mothers believe that natural products are safer for their infant. 15 Concerns around safety are more considerable drivers of purchasing behavior than concerns about product performance. However, many parents also perceive that the natural diapers on the market today do not provide the same effective absorption as conventional diapers. As such, 1 in 3 mothers report using a mix of conventional products and natural products in order to care for their infants. Most commonly, mothers will diaper an infant in a natural diaper throughout the day but switch to a conventional diaper for overnight, as the conventional diaper provides better product performance as indicated by less leakage. 16
There are limited offerings of diapers in the natural category (those diapers positioned as having more natural/eco-friendly ingredients or manufactured without certain materials/processes, such as chlorine bleaching) that offer the same high-performance design as conventional diapers. A new diaper was designed to meet the needs of this emerging segment of consumers, while providing better performance than existing options in the “natural” category. The following clinical study compared the new diaper offering with similar performance-driving technologies as found in conventional diapers, such as an absorbent core and high-performing topsheet, with the goal of evaluating product performance and infant skin condition against the leading totally chlorine-free diaper.
Methods
Study Design
Diaper performance was evaluated in a parallel, randomized, examiner-blinded clinical study. The study was reviewed and approved by an ethics committee and all parents/caregivers provided written informed consent for their child. The study randomized 131 infants with 116 completing the study. Participants were stratified by baseline genital diaper rash score, current diaper product used, and sex. Following screening and enrollment, all infants wore a third-party store brand diaper (Parent’s Choice) for a 7-day “washout” period to acclimate the skin to the same conditions and were provided a standard, marketed baby wipe (Pampers Sensitive Wipes) to use throughout the study.
Following the washout period, subjects were randomized to 1 of 2 diaper products (Diaper A, Honest; Diaper B, Pampers Pure Protection), which were used exclusively for the 4-week study duration. Diaper A was chosen because at the time of the study it was the market leader in the “natural” diaper segment in North America according to sales. 17 Diaper B was a new diaper enhanced with cotton and containing other thoughtfully selected materials.
In-Use Diaper Questionnaire
An at-home diary was used to assess leakage, wear time, and topical product usage. Parents/caregivers were asked to record all diaper changes throughout the study, along with the type of soiling (urine, feces, or both), if there was leakage, and the time between diaper changes. Parents/caregivers were also asked to record and track usage of any topical products (cream, lotion, or powder) throughout the study.
Skin Marking
Skin marking in the diapered area was defined as an indentation (mark) on the skin caused directly by contact of the diaper with the skin. Skin marking, while primarily a cosmetic endpoint, can be indicative of the pressure applied by the product to the skin, and is therefore a useful endpoint in determining fit of the product as a variable in product performance. Pressure marking is apparent as an indentation in the skin; red marking is the redness within a pressure mark or redness in a defined pattern where there is no a pressure mark. Both types of marking are transient in nature. Skin marking assessments were conducted by a trained skin grader immediately at diaper removal. The scale for pressure marking was a 5-point, 0 to 4 scale, with categories of 0 = no indentation; 1 = slight indentation; 2 = moderate indentation; 3 = deep indentation; and 4 = very deep indentation. The scale for red marking was a 5-point, 0 to 4 scale, with categories of 0 = no redness; 1 = definite pink; 2 = definite red; 3 = intense red; and 4 = very intense red. Skin marking was evaluated at day 3 and day 28 of the study at the following 5 grading sites: left leg cuff, right leg cuff, front waist, back waist, and buttocks. The proportion of grading visits at which any pressure marking or red marking was observed, as well as the proportion of subjects with any pressure marking or red marking on any day post-baseline, was summarized by test product. The entire distribution of data was also summarized by test product. The data were also summarized separately by grading site to better understand the anatomical distribution of pressure marking and red marking.
Assessment of Diaper Dermatitis
DD, or diaper rash, severity was assessed at 4 diaper grading sites: perianal (anal groove), intertriginous (leg folds), genitals, and buttocks. Diaper rash was graded on post-baseline days 0, 3, 7, 10, 14, 17, 21, 24, and 28. Diaper rash assessment was scored using the P&G Diaper Rash Grading Scale, developed and used by Procter & Gamble, which ranks skin condition on a 7-point scale (0-3.0).2,3,18 Scores are based on the presence and intensity of erythema, erythema area of coverage, and presence of papules and pustules.
Rash assessed at a severity of “mild-to-moderate” (equivalent to a score of ≥1.5 on the Diaper Rash Grading Scale) was chosen based on the size of the study and rash incidence and severity observed in this population. Rash was analyzed based on 3 endpoints: (1) the percent of subjects with at least one score ≥1.5; (2) the percent of assessments with scores ≥1.5; and (3) the percent of subject with consecutive scores ≥1.5 at the same site.
Data Analysis and Statistical Methods
The intent-to-treat (ITT) population was defined as all subjects who were randomized and experienced at least one application of the assigned study product. The per protocol (PP) population included subjects in the ITT population who had no serious inclusion/exclusion violations, no serious continuance criteria violations, no serious concomitant medication violations, and high study product compliance. The PP population was used for the primary analysis and for all diaper rash comparisons. Analyses of the ITT population was done to confirm the PP results and as the main data source for adverse event (AE) evaluations and summaries.
Descriptive statistics were used to summarize the study data. Endpoints having to do with the proportion of subjects were analyzed with a χ2 test. Endpoints having to do with the proportion of grading sites were analyzed with analysis of variance, where the response is the proportion of graded sites with skin marking score ≥2.0 or rash score ≥1.5.
Results
Demographics
There were 131 infants randomized into the study with 116 completing the study. Infants were well balanced between the 2 diaper products with regard to sex, age, weight, race, ethnicity, and Fitzpatrick skin type (Table 1).
Subject Participant Demographics.
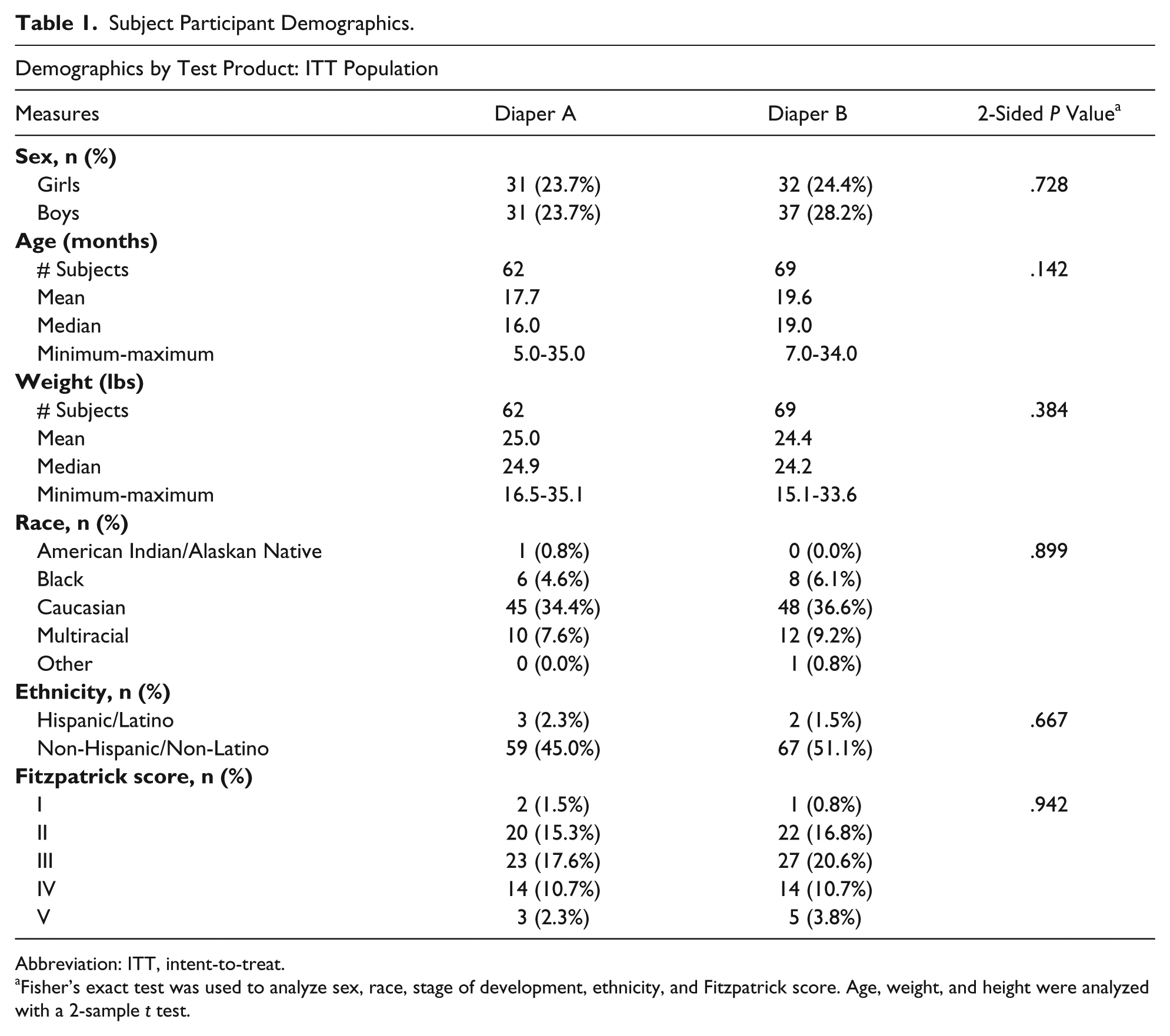
Abbreviation: ITT, intent-to-treat.
Fisher’s exact test was used to analyze sex, race, stage of development, ethnicity, and Fitzpatrick score. Age, weight, and height were analyzed with a 2-sample t test.
At-Home Diary Results
To assess the performance of the 2 diapers, data were collected at each diaper change for the presence of leakage for changes that contained urine only or a bowel movement (Figure 1). Infants wearing Diaper B experienced significantly lower urine leakage rate than Diaper A when comparing all changes (3.0% vs 6.9%; P < .05) and for urine-only changes (3.4% vs 8.1%; P < .05). There was less leakage for Diaper B for changes containing a bowel movement (1.6% vs 3.9%), but the results were not statistically significant (Figure 1).
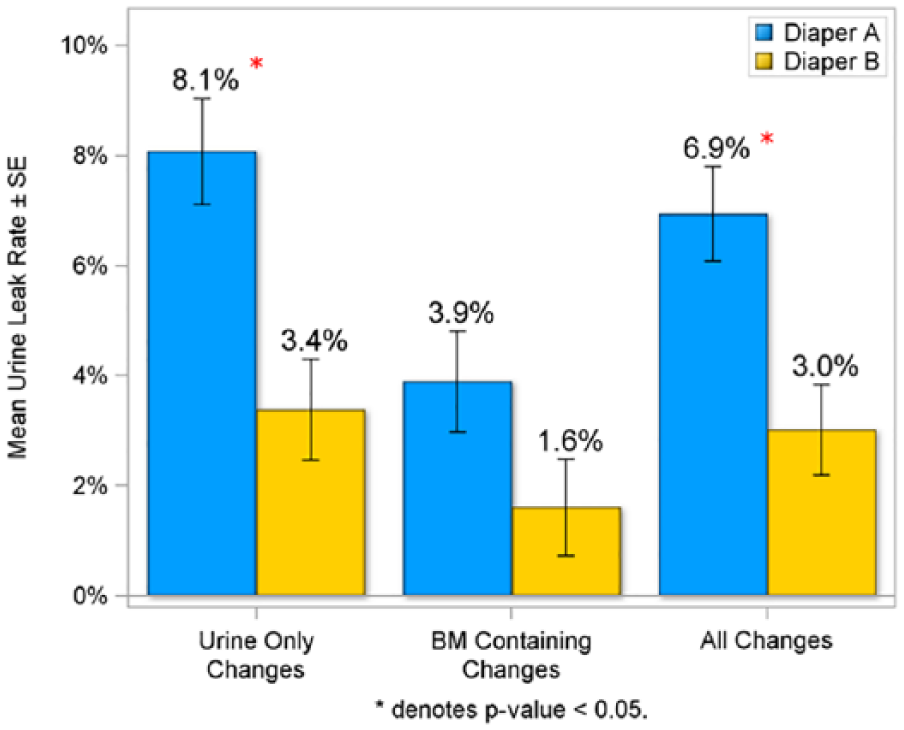
Urine leak rate by type of change in intent-to-treat population.
Parents were also asked to report on diaper habits, including diaper wear times and the use of topical skin products in the diapered area. Average wear times (in hours) revealed no differences between the 2 diaper products regardless of soiling type (Figure 2), suggesting similar diaper changing habits between groups.
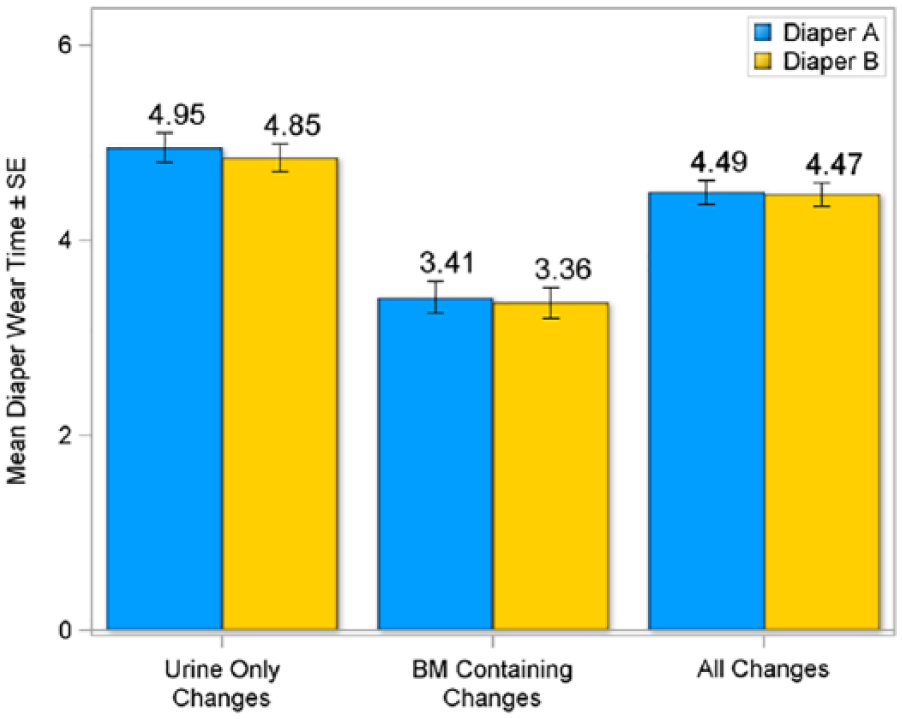
Mean diaper wear time by type of change in intent-to-treat population.
While parents were asked to refrain from using topical products for the duration of the study, the use of topical products was collected. The predominant form was cream, followed by powders and lotions. No statistically significant differences between the 2 diapers were noted with regard to topical product use (Figure 3).
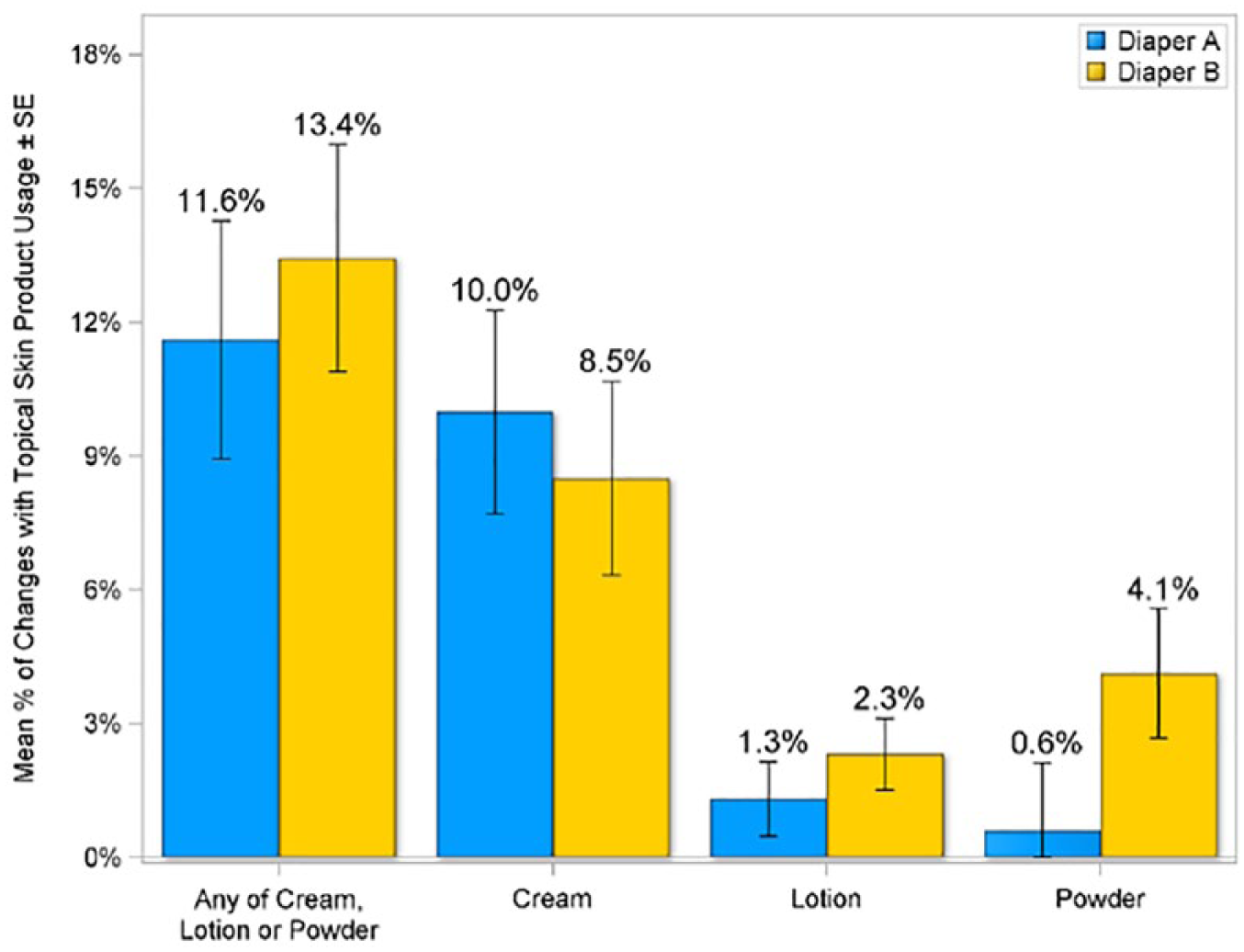
Topical skin product usage in intent-to-treat population.
Skin Marking Assessment
To compare the amount of skin marking between the 2 diapers, trained graders assessed the amount of transient pressure and red marking immediately on diaper removal on the right and left leg cuffs, front and back waist, and buttocks (Figures 4 and 5). In general, both pressure and red marking were highest at the leg cuffs, followed by the waist and then buttocks. Infants wearing Diaper A experienced significantly more pressure and red marking at all anatomical sites compared with infants wearing Diaper B.
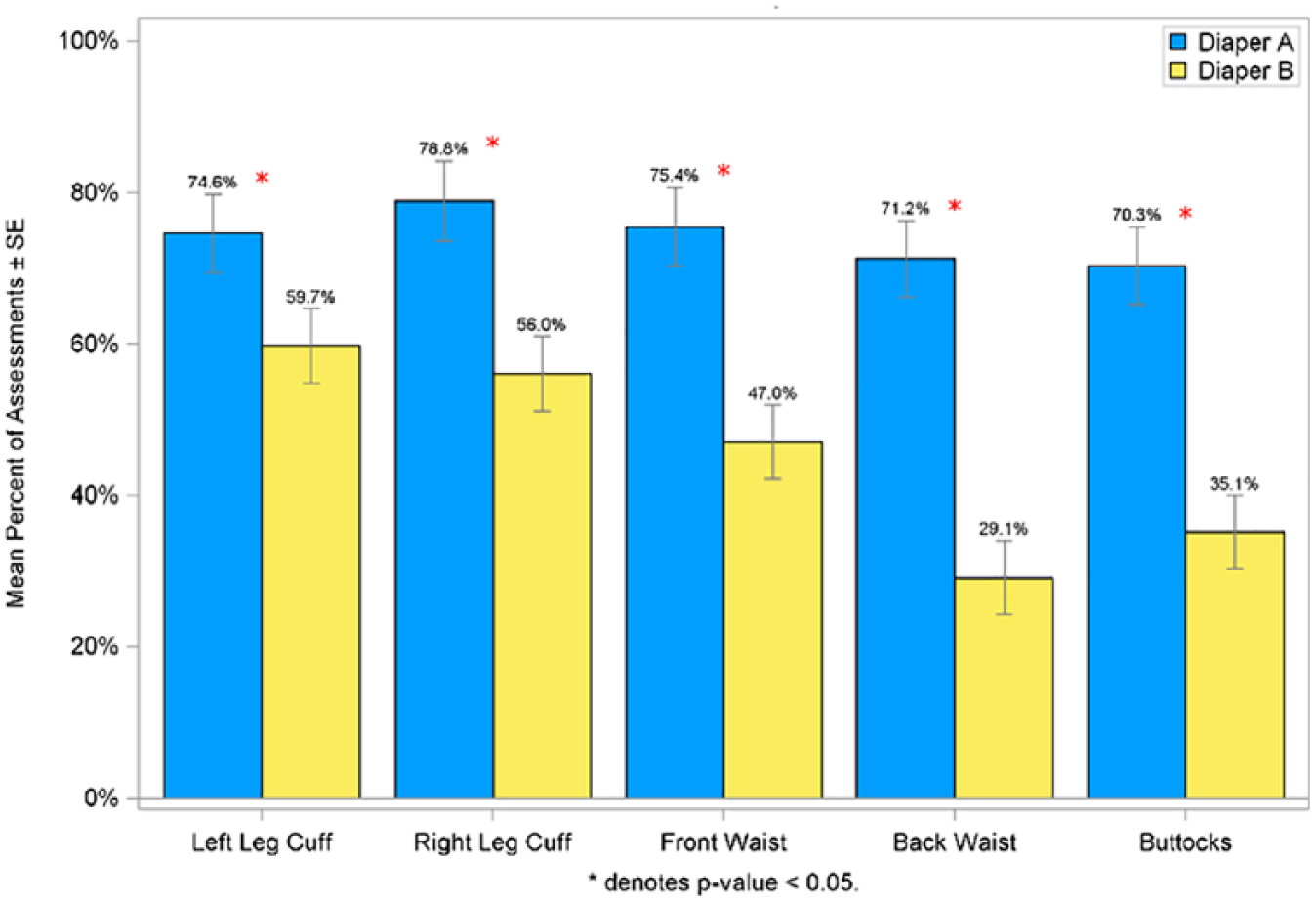
Mean percent of assessments with pressure marks scored at ≥2 in per protocol population.
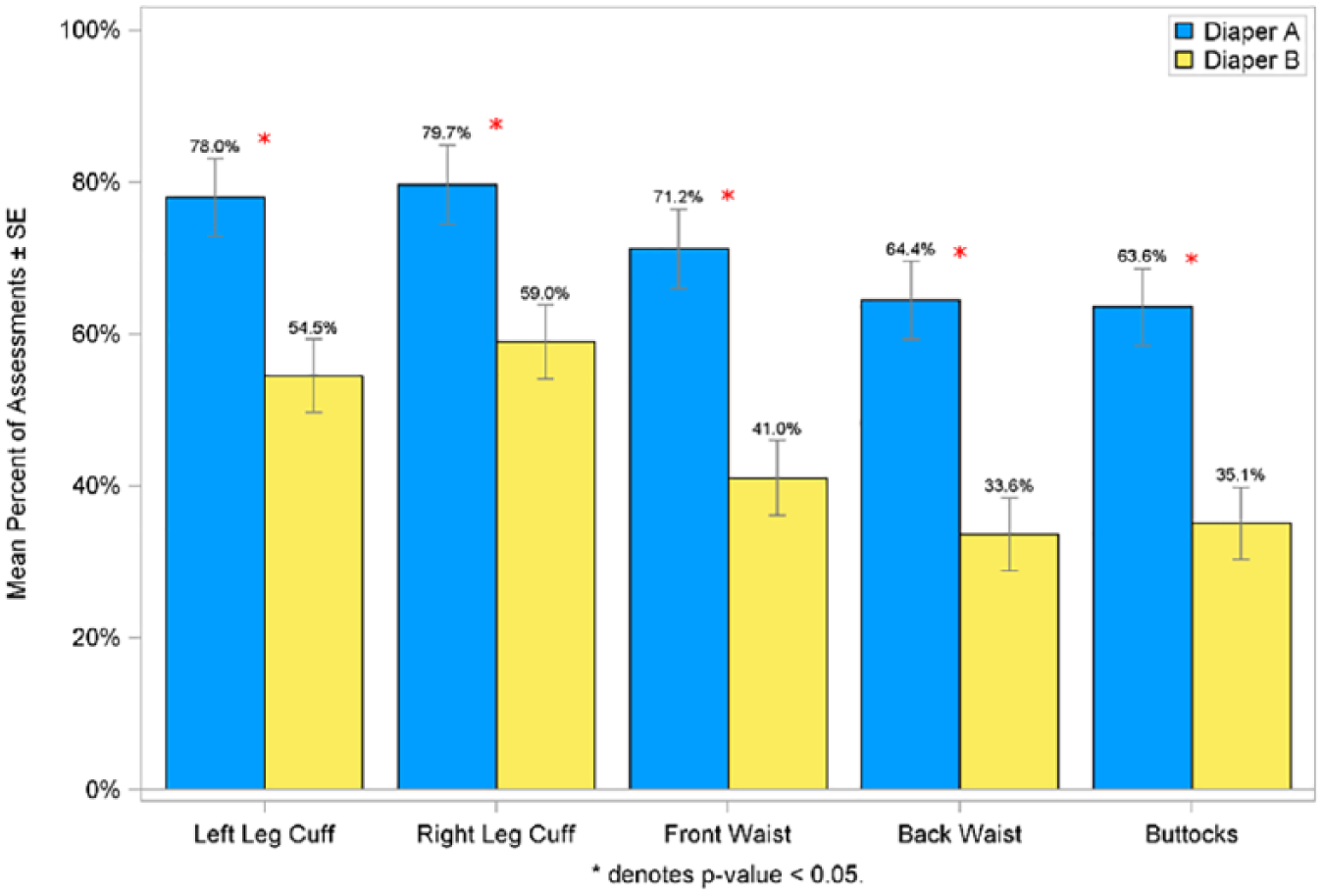
Mean percent of assessments with red marking scored at ≥2 in per protocol population.
The percent of assessments taken on day 3 and day 28 of the study showed that infants wearing Diaper B had significantly less (P < .05) pressure marks assessed as moderate or greater severity (score of ≥2.0) across each of the 5 grading sites compared with Diaper A. A similar finding was observed for red marking scored at ≥2.0 (definitely red) with significantly less (P < .05) red marking achieved at all grading sites for Diaper B compared with Diaper A.
Diaper Rash Scores
Diaper rash presence and severity was evaluated using the P&G Global Diaper Rash Grading Scale, a 7-point scale ranging from 0 to 3 with half point increments.2,3,18 Overall, diaper rash incidence and severity were lower for infants wearing Diaper B compared with Diaper A. For ease of comparing the 2 products in this study, rash is reported for “mild-to-moderate” or greater (score ≥1.5), which is the midpoint of the P&G Diaper Rash Grading Score. Rash at ≥1.5 was assessed based on the percent of subjects who experienced even one such rash event during the study, the percent of assessments occurring during the study, and the percent of subjects who experienced rash on consecutive visits.
For each of the rash measures, the perianal site experienced the greatest amount of rash, followed by the genitals, intertriginous areas, and the buttocks. The percent of subjects experiencing at least one episode of rash during the study was significantly lower on Diaper B in the genital (46.3% vs 63.8%) and intertriginous (29.9% vs 51.7%) regions compared with Diaper A (P < .05; Figure 6). Similar to these findings, the mean percent of post-baseline rash assessments was significantly lower in infants wearing Diaper B at both the genital (15.4% vs 24.1%) and intertriginous (8.4% vs 16.6%) regions compared with Diaper A (P < .05; Figure 7). The percent of infants having a rash score of ≥1.5 on consecutive visits was lower for infants wearing Diaper B in the genital (13.4% vs 31.0%) and intertriginous (7.5% vs 24.1%) regions than for Diaper A (P < .05; Figure 8).
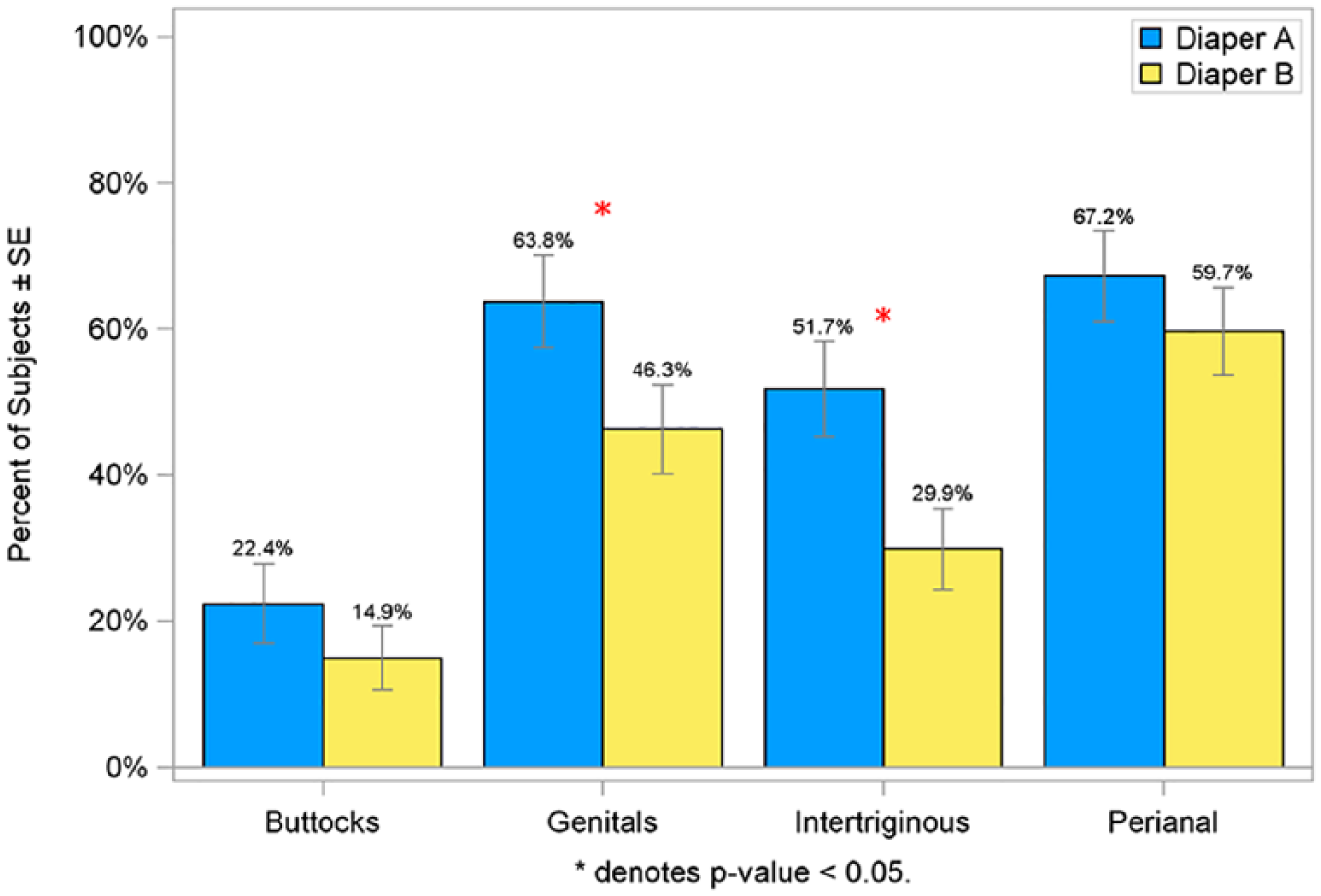
Percent of subjects with at least 1 post-baseline rash assessment ≥1.5 in per protocol population.
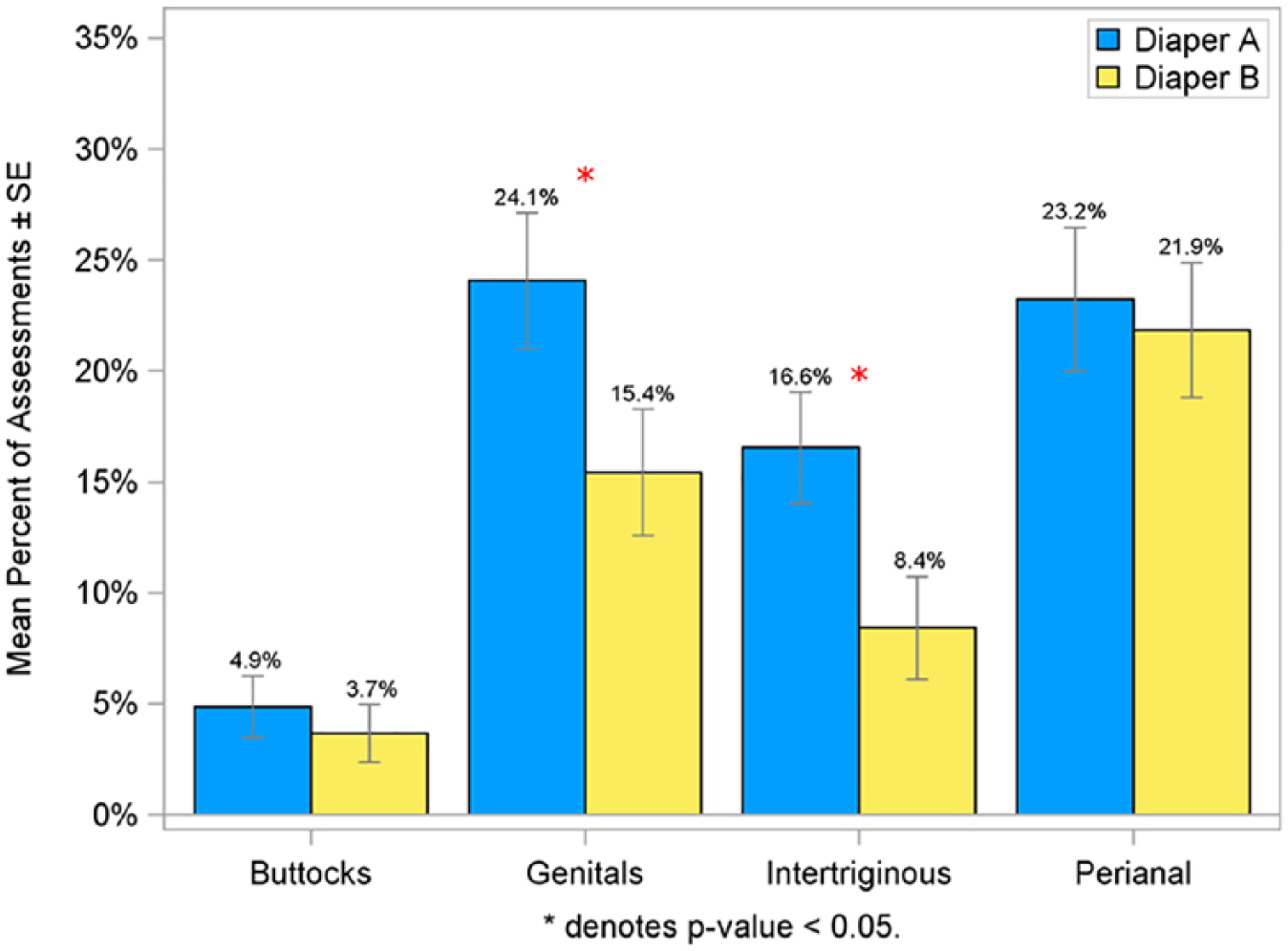
Mean percent of post-baseline rash assessments with score ≥1.5 in per protocol population.
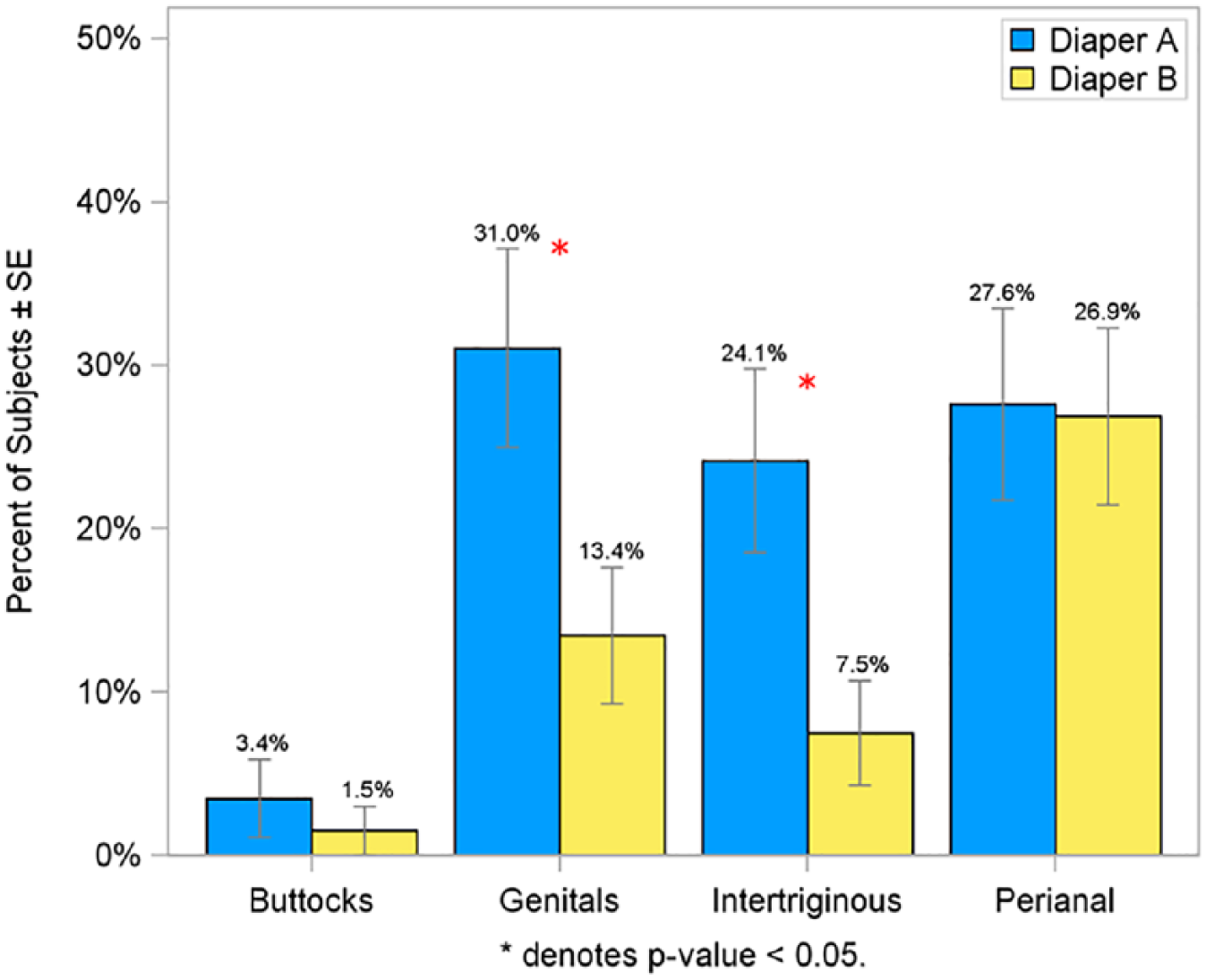
Percent of subjects having consecutive visits with rash score ≥1.5 in per protocol population.
Adverse Events
There were 135 treatment emergent AEs in 76 babies in this study. None of those events were considered serious AEs. Of the nonserious AEs, 101 were mild and 34 were classified as moderate in severity. Of these events, 124 AEs were classified as not related to test product (eg, ear infections, upper respiratory infections, and insect bites). Eleven AEs were classified as doubtful or possible for relatedness, included yeast infection, diaper rash of at least a 2.5, or other marks in the diapered area not classified as diaper rash (6 were on Diaper A and 5 were on Diaper B).
Discussion
In this clinical investigation, the performance of 2 natural diaper products was significantly differentiated based on leakage rates, the skin attributes of skin marking, and the incidence and severity of diaper rash. These findings suggest that key design differences between the 2 products exist, which are likely to be meaningful to parents who are searching for a natural product that meets their performance expectations typical of premium, traditional diaper offerings. These results may create new opportunities to meet the demands of parents and caregivers who are increasingly choosing baby care products that they perceive to be more “natural,” while providing the proper and necessary product performance to care for infants’ skin. 19
When a diaper leaks, it can be a source of frustration for parents and discomfort for infants, often requiring a clothing change. This is particularly problematic when it occurs at night, which can interrupt the sleep of the infant and parent. In this study, leakage rates were significantly reduced with Diaper B. Increased leak rate with Diaper A may be due to reduced diaper absorbency or poor diaper fit. A lower capacity diaper is unlikely to adequately sequester urine and/or feces, which are key components in provoking skin breakdown and are consistent with the increased DD in infants wearing Diaper A. Infants wearing Diaper A experienced significantly more skin marking compared with those wearing Diaper B at each of the 5 anatomical sites examined. While skin marking is transient and predominantly cosmetic in nature, more skin marking is suggestive of parents applying the diaper more tightly, which may reflect efforts by the caregiver to prevent diaper leakage with Diaper A. In this case, Diaper A may fit closer to the skin and be less capable of allowing air exchange, which could contribute to the worsening skin conditions seen in this study.
The lower diaper rash incidence and severity observed at the genital and intertriginous sites in infants wearing Diaper B versus those wearing Diaper A can be considered robust as these reductions were observed in all 3 unique analyses of rash. For a rash severity of ≥1.5, the percent of subjects experiencing rash was reduced, as was the percent of rash assessments over the wear period. There was also a reduction in the percent of subjects presenting with rash at consecutive assessments, suggesting that when rash was observed in infants with Diaper B, these events were of limited duration (<7 days). The reduced DD incidence and severity observed in infants wearing Diaper B cannot be attributed to caregiver habits related to diaper change frequency or application of topical product use, as the frequency of these behaviors was nearly identical between the 2 products. The reduced leakage observed in urine-only diaper changes suggests that the superior liquid/moisture handling properties of Diaper B are a reasonable explanation for the improved skin condition of these infants. Improvements in skin health are not unprecedented in the design of disposable diapers as analyses over the past several decades show progressive decreases in rash incidence and severity with improvements in disposable diaper technology.10,20
The natural category is growing, and many caregivers are choosing to use natural personal care products for their infants, regardless of product performance in comparison to conventional products. 21 Therefore, for the increasing number of parents who are choosing diapers containing more naturally derived materials over conventional diapers, it is crucial that they have a product available to them that incorporates high-performance technology, such as an absorbent core, high-performing topsheet, and materials that allow dehumidification of the diaper environment, all of which can provide superior skin outcomes.
From birth, many parents and caregivers receive mixed messages, sometimes even from health care professionals, that natural products are preferable to conventional products, particularly for skincare.21-23 This leads to tough choices, confusion, and anxiety for caregivers over whether they are choosing the right products for their infants, particularly if they are weighing perceived efficacy of a product against perceived risk or harm. This new diaper provides a viable option alongside conventional offerings for caregivers who want products that provide the ingredients and manufacturing techniques they desire, such as totally chlorine-free pulp, cotton, and other quality materials, without sacrificing product performance.
Conclusions
This clinical study comparing 2 diapers in the natural category showed that the new, cotton-enhanced diaper with high-performance materials for absorption outperformed the comparator diaper. As parents and caregivers are increasingly choosing more natural products for their infants, a diaper that combines the ingredients and manufacturing processes they desire, along with outstanding absorption and performance, can meet consumer preferences without sacrificing quality or infant health outcomes.
Author Contributions
All authors contributed to the design, analysis, and interpretation of the clinical study and results. All authors revised and reviewed the final manuscript.
Footnotes
Acknowledgements
Product support was provided by Sara Giovanni of the Procter & Gamble Company. Writing support was provided by Eric Moorhead, MS, of Spectrum Science Communications Inc, and supported by Procter & Gamble.
Declarations of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Robert J. O’Connor, Andrew N. Carr, Y. Wang, Roger Gibb, and Veronica Sanchez are full-time employees at Procter & Gamble. Donald L. Nofziger and Mary Bailey have received financial support from Procter & Gamble.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Procter & Gamble Company.
