Abstract
The Vaccines for Children (VFC) program distributes one half of all vaccines administered to US children. Compared with commercial vaccine distribution, VFC distribution is more complex. This prospective observational study compares the delivery and administration of VFC versus non-VFC influenza vaccine in US pediatricians’ offices across 3 influenza seasons and its apparent impact on 2-dose compliance rates. Because of earlier shipping (mean = 29-42 days) of non-VFC vaccines, administration of VFC vaccines was delayed relative to non-VFC vaccines by approximately 1 month in 2007-2008 and 2008-2009 and 2 weeks in 2010-2011. Two-dose compliance rates for the VFC and non-VFC populations were 38.5% and 47.5% (P < .001) in 2007-2008, 45.9% and 55.1% (P < .001) in 2008-2009, and 50.0% and 52.9% (P < .001) in 2010-2011, respectively. Despite delays, earlier VFC shipment in 2010-2011 enabled greater equity in 2-dose compliance.
Introduction
The annual delivery of influenza vaccinations is challenging because vaccines must be distributed and administered during a limited time interval. In the United States, children 6 months through 8 years of age have been recommended to receive 2 doses of influenza vaccine at least 4 weeks apart if they have not been previously vaccinated or if they were vaccinated in the previous season for the first time and received only 1 dose of vaccine. 1 In 2010-2011, the need for 2 doses of influenza vaccine was based on prior receipt of trivalent seasonal influenza vaccine and the 2009 monovalent pandemic H1N1 vaccine. 1 Compliance with the recommended 2-dose regimen has been suboptimal in recent seasons and is frequently <25% among children 2 years of age and older. 2,3
The US Vaccines for Children (VFC) program was implemented in 1994 with the aim of providing all recommended childhood vaccines free of charge to eligible children. Under the program, the Centers for Disease Control and Prevention purchases vaccines and distributes them to state/local health departments, which then distribute vaccines at no charge to provider offices and clinics. Children eligible to receive VFC vaccines in provider offices include those who are <18 years old and eligible for Medicaid, do not have health insurance, or are American Indian or Alaska native. Since implementation, the VFC program has significantly improved vaccination rates among uninsured children. 4 -9 The VFC program provides approximately one half of all vaccines provided to children in the United States. For influenza vaccines specifically, the VFC program appears to provide approximately one-third of vaccines delivered in US pediatrician offices. 10,11 Compared with commercial distribution of vaccines, the VFC vaccine distribution system is more complex and has the additional burden of ensuring equity across jurisdictions and product types.
Few studies have simultaneously evaluated the delivery of influenza vaccines through the VFC system and privately purchased influenza vaccinations (non-VFC). A previous study of US pediatric offices in 2007-2008 and 2008-2009 demonstrated a delay in the delivery and administration of VFC influenza vaccine relative to non-VFC vaccine. 11 The delay was consistent across geographic regions and vaccine manufacturers and appeared to be responsible for lower 2-dose compliance among the VFC population; this was supported by another study in a large health maintenance organization, which also demonstrated lower 2-dose compliance among subjects with later first-dose receipt. 3 Using data acquired from a continuation of the same study of pediatric offices conducted during the 2010-2011 season, the present analysis describes the timing of delivery and administration of VFC versus non-VFC influenza vaccines in US pediatric offices in 2010-2011 relative to the previously described 2007-2008 and 2008-2009 influenza seasons.
Methods
Study Design
A prospective, observational, noninterventional study was conducted in US outpatient pediatric offices during the 2007-2008, 2008-2009, and 2010-2011 influenza seasons. Although the study was also conducted in 2009-2010, these data were not included in the analysis because of the anomalous nature of the 2009-2010 season, which was dominated by the 2009 H1N1 pandemic response. Study methods have been previously described and are also summarized below. 10,11 Each season, a sample of outpatient pediatric offices from the American Medical Association list of pediatricians was recruited to achieve a geographically balanced sample. To be eligible for inclusion, each practice had to provide influenza vaccines at their location and be able to calculate the total number of patients under their care by age group through an electronic database or chart count. Hospital-based offices were excluded. No individual participants were enrolled in this study, and no patient-identifying information was collected.
Offices were required to record each influenza vaccination that was given to their patients as it was administered in the office. Vaccinations were tracked by age group, first or second vaccination, whether the child required 1 or 2 doses of vaccine during the season, type of vaccine (multidose injectable, prefilled injectable, or intranasal spray), and VFC status (VFC vs non-VFC). The age groups studied were 6 to 23 months, 24 to 59 months, 5 to 8 years, and 9 to 18 years (in season 1, this age group was 9-17 years old). Vaccinations administered outside of the office were not tracked. Counts of vaccinations given to children <9 years of age who were recommended to receive 2 doses during the season were tracked separately from those who required only 1 dose. Each office submitted a cumulative record of the number of vaccinations for each half-month interval throughout the influenza season. Participating practices completed surveys at the beginning and end of each season on various parameters, including all influenza vaccine shipment quantities by vaccine type and their dates of arrival.
Data Analysis
Data on influenza vaccinations were aggregated across sites and analyzed as a function of time. Because first and second vaccinations given to children requiring 2 doses were tracked separately, aggregate compliance with the 2-dose regimen could be estimated by dividing total second vaccinations by total first vaccinations for children requiring 2 doses; compliance was calculated by age group and VFC status. Temporal patterns of VFC and non-VFC vaccinations given to children requiring 2 doses were analyzed, and cumulative compliance rates were calculated for each half-month interval. Offices that reported a large number of children receiving first vaccinations at other venues (eg, local school vaccination programs) were excluded from analyses related to 2-dose compliance because a child’s inclusion in the numerator (by receiving a second dose in the office) but not the denominator (by receiving a first vaccination elsewhere) would skew the compliance calculation; accordingly, 6 of 42, 9 of 84, and 1 of 105 offices were excluded from the 2-dose compliance calculation in seasons 1, 2, and 3, respectively.
Results
Vaccination in Pediatric Offices
Characteristics of the pediatric offices studied are presented in Table 1. In all seasons, initial shipments of privately purchased, non-VFC vaccines arrived on average 29 to 42 days earlier than initial VFC shipments (Table 1). The median first dates that any influenza vaccine was offered to patients were September 16, 2007, September 1, 2008, and August 17, 2010, respectively. In the first season, 42 433 first vaccinations and 5508 second vaccinations were administered, 27% (n = 12 981) of which were VFC vaccines. In the second season, 84 868 first vaccinations and 10 806 second vaccinations were administered, 32% (n = 30 835) of which were VFC vaccines. In the third season, 145 228 first vaccinations and 20 676 second vaccinations were administered, 27% (n = 45 401) of which were VFC vaccines. The proportion of influenza vaccinations that were supplied through the VFC system was lower among children 9 years of age and older compared with younger age groups.
Characteristics of Pediatric Offices and Vaccine Shipment Timing by Season
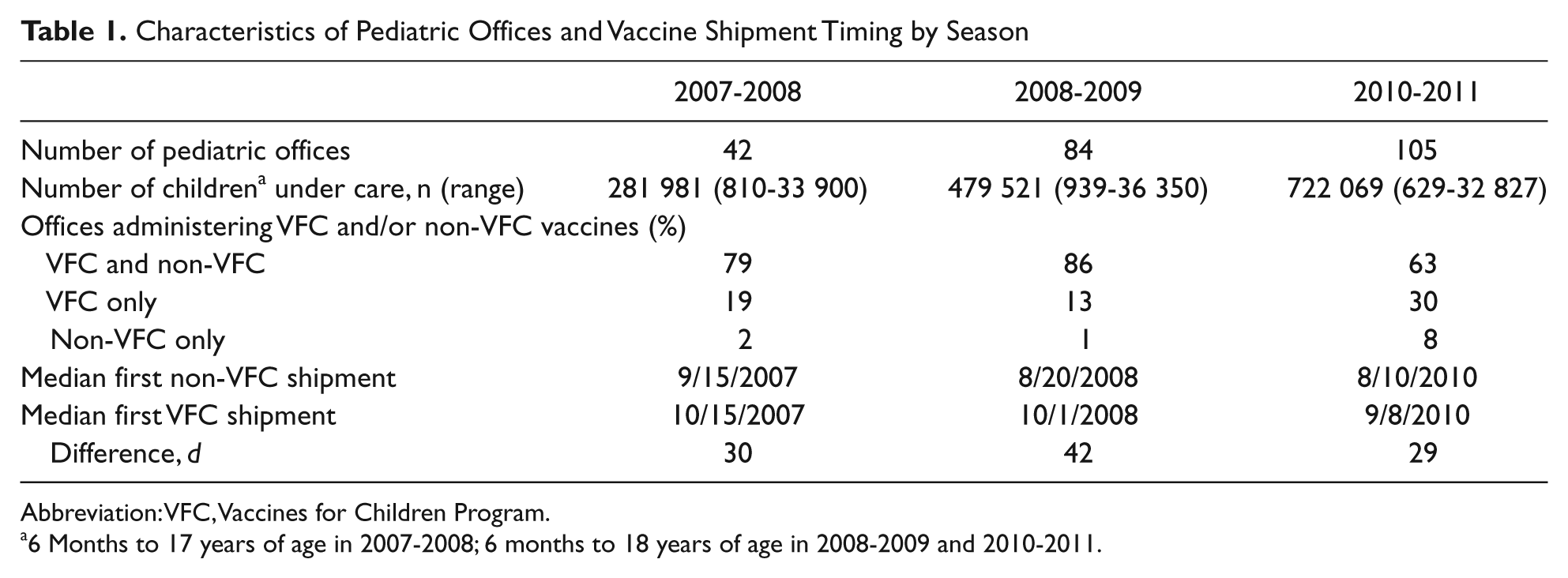
Abbreviation: VFC, Vaccines for Children Program.
6 Months to 17 years of age in 2007-2008; 6 months to 18 years of age in 2008-2009 and 2010-2011.
Receipt of Vaccine and Timing of Vaccination
Commensurate with the delay in shipment arrival, VFC vaccine administration was delayed compared with non-VFC vaccine by approximately 1 month in 2007-2008 and 2008-2009 and by approximately 2 weeks in 2010-2011. The delay in first-dose administration resulted in a subsequent delay in second-dose administration for children requiring 2 doses (Figure 1). A similar delay was seen for all vaccine types, including multidose vials of injectable vaccine, prefilled syringes of injectable vaccine, and intranasal vaccine; there was no discernible geographic regional pattern to the observed delay. In season 3, to better understand the effects of delayed VFC vaccine shipments, offices were asked if they ever delayed the administration of non-VFC vaccine because of a delay in VFC vaccine shipment. Only 8 of 105 offices (8%) responded affirmatively.
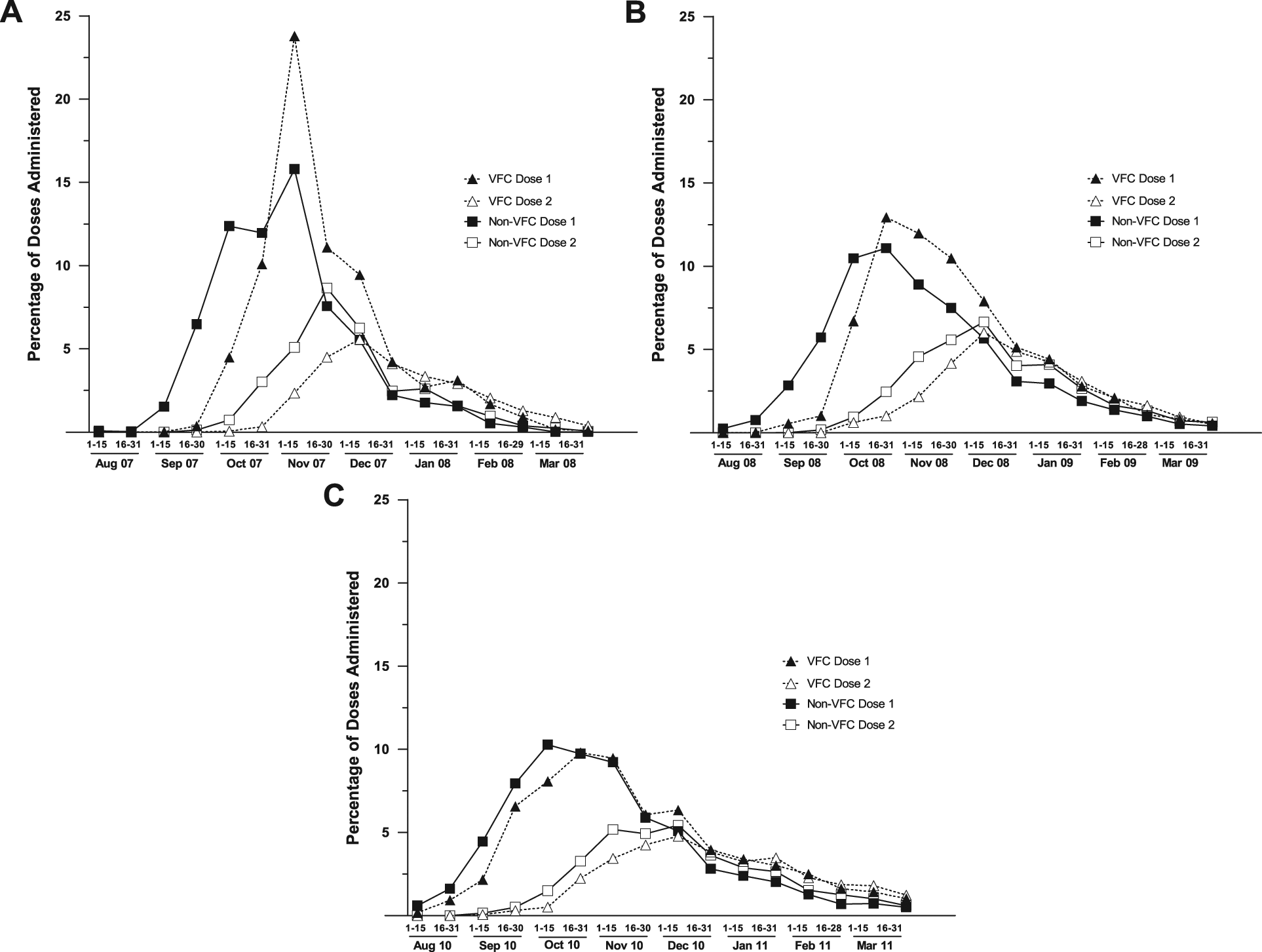
Timing of first and second influenza doses administered to children requiring 2 doses, by dose number and VFC status. To adjust for differences in the size of the VFC and non-VFC populations within and across seasons, the figures depict the percentage of all VFC and non-VFC vaccinations during each 2-week interval. The denominator for the VFC dose 1 and VFC dose 2 was the total number of VFC doses administered; similarly, the denominator for non-VFC dose 1 and non-VFC dose 2 was the total number of non-VFC doses administered. A. 2007-2008 influenza season. B. 2008-2009 influenza season. C. 2010-2011 influenza season
Two-Dose Compliance
In seasons 1 and 2, compliance rates among children receiving the VFC vaccine were 17% to 19% lower than among children receiving privately purchased, non-VFC vaccines; the absolute difference in 2-dose compliance was approximately 9% for both seasons (Table 2). In season 3, there was a 5% relative difference in compliance and a 3% absolute rate difference. Each season, the difference in compliance between the VFC and non-VFC populations was consistent across all age groups, with the 1 exception of similar rates among children 24 to 59 months of age in the 2008-2009 season.
Two-Dose Compliance Rates for VFC and Non-VFC Populations Among Children Younger Than 9 Years Identified as Needing 2 Doses
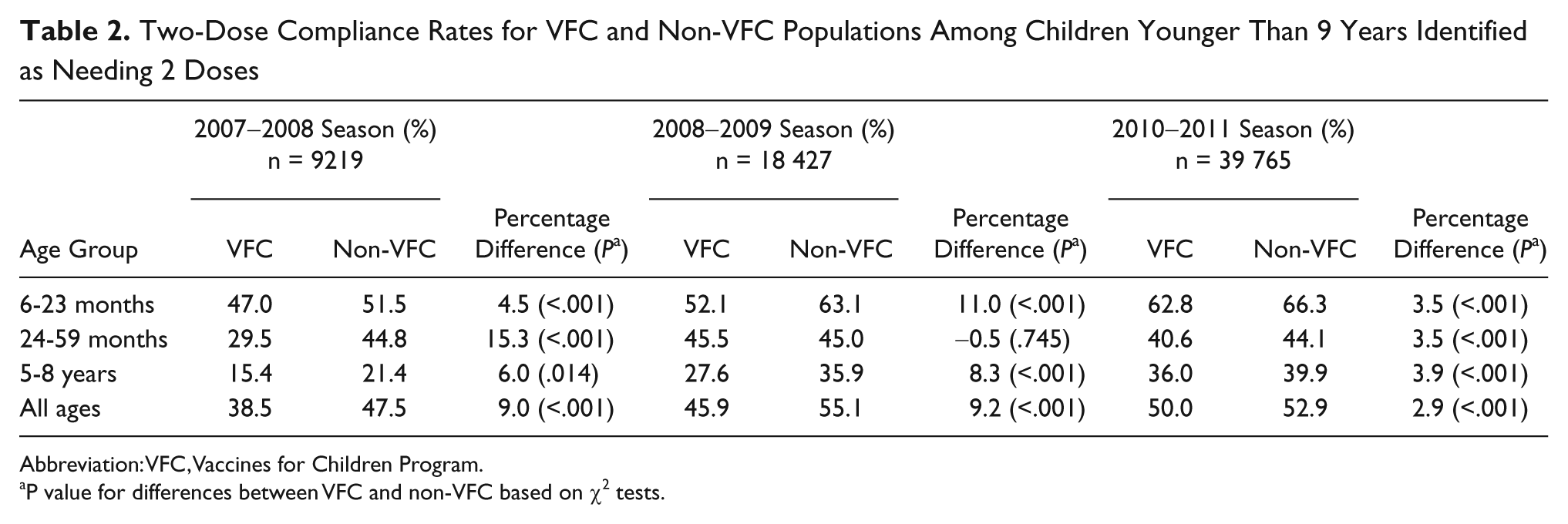
Abbreviation: VFC, Vaccines for Children Program.
P value for differences between VFC and non-VFC based on χ2 tests.
When plotted by the number of weeks after the first administration of VFC and non-VFC vaccines, the difference in cumulative 2-dose compliance between the VFC and non-VFC populations was not a constant, generalized phenomenon throughout the vaccination period. Instead, 2-dose compliance among VFC vaccine recipients fell below that of non-VFC vaccinees only in December and January, simultaneous with the overall decline in influenza vaccinations (Figure 2). This effect was pronounced in 2007-2008 and 2008-2009 but more subtle in 2010-2011.
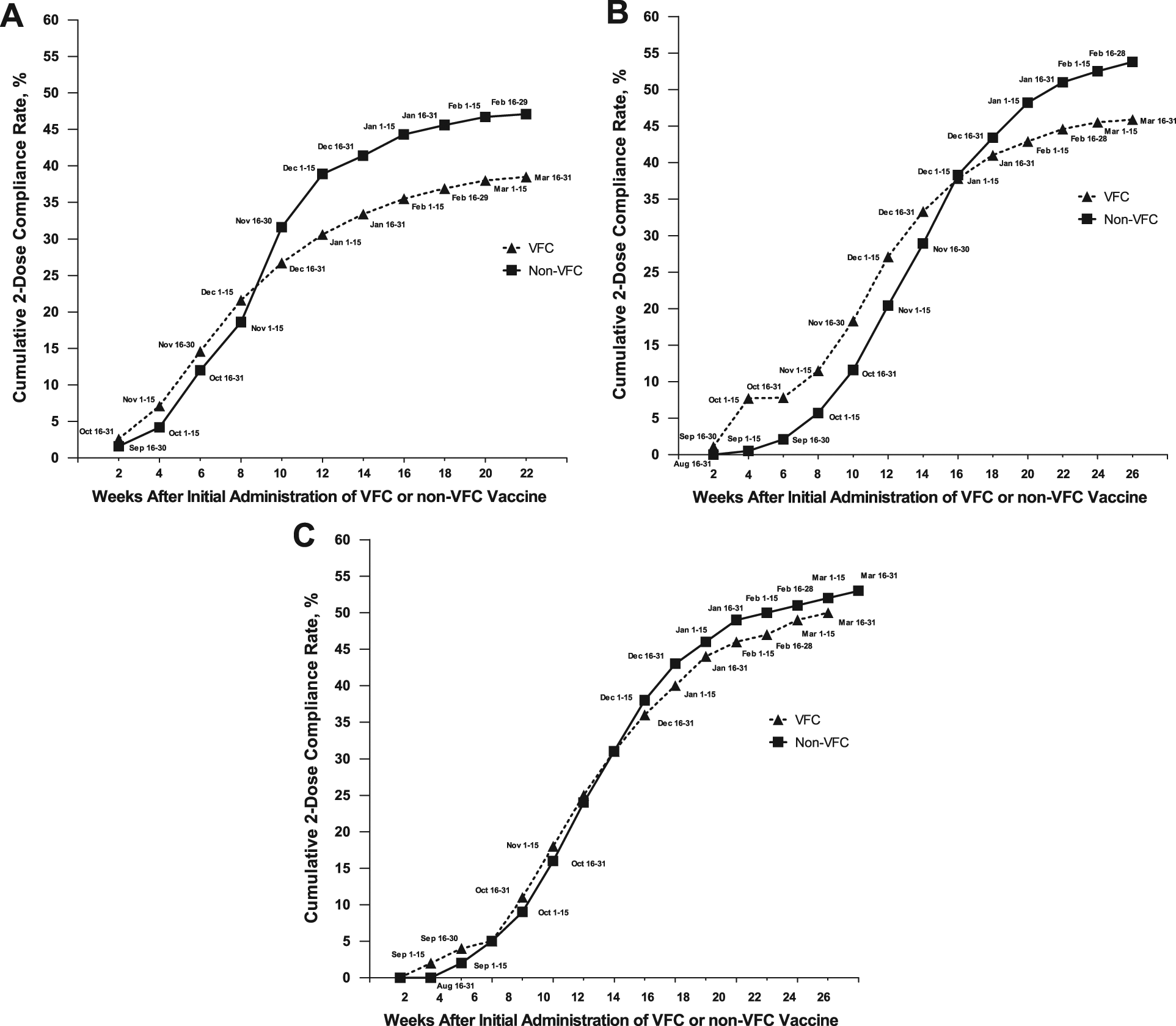
Cumulative 2-dose compliance by VFC status by week after initial vaccination. A. 2007-2008 influenza season. B. 2008-2009 influenza season. C. 2010-2011 influenza season
Discussion
The VFC program is critical to ensuring that financially vulnerable children in the United States have access to vaccination. The current study suggests that although delivery of the VFC influenza vaccine was delayed relative to the non-VFC influenza vaccine, arrival of the VFC vaccine by early September may mitigate the effects of delayed VFC vaccine shipment. VFC vaccine shipment was consistently delayed in all study seasons by approximately 1 month. In 2007-2008 and 2008-2009, this shipment arrival delay led to the median first VFC shipment occurring in October. Given the large number of non-VFC vaccinations administered in September, the limited availability of VFC vaccines in September led to a significant overall delay in VFC vaccine administration. This initial delay and the general end-of-season decline in vaccination effectively shortened the time in which the VFC population could receive a second dose of vaccine, and lower 2-dose compliance rates were observed for VFC vaccinees. These findings suggest that 2-dose compliance is significantly influenced by parental convenience, with larger time windows increasing the probability of second-dose receipt. In 2010-2011, a different pattern was observed. VFC shipments arrived on average by early September, and the general administration delay between VFC and non-VFC vaccines was minimal. This similar timing of administration appears to have enabled greater equity in 2-dose compliance rates between the VFC and non-VFC populations.
Because of the additional requirements and complexity of the VFC distribution system, it may not be possible to ensure simultaneous delivery of VFC and non-VFC influenza vaccine shipments to providers. Based on the current analysis, a more realistic and similarly effective target could be for VFC influenza vaccine shipments to arrive by early September. To accomplish this goal, manufacturers, distributors, and public health entities should prioritize early-season shipments of VFC influenza vaccine to pediatric providers. Additionally, pediatric health care providers should be aware that children in the VFC program may have less time to receive an influenza vaccine and should consider increasing their efforts to vaccinate this population in later months.
In the previous report that described reduced 2-dose compliance in the VFC population versus the non-VFC population in 2007-2008 and 2008-2009, trends in cumulative 2-dose compliance by weeks following initial availability suggested that time was the principal factor responsible for the difference. 11 However, socioeconomic or other baseline population differences could not be excluded as contributing factors. The reduced discrepancy in 2-dose compliance that accompanied improved VFC vaccine shipment timing in 2010-2011 supports the conclusion that the differences observed in 2007-2008 and 2008-2009 were primarily driven by the timing of VFC vaccine availability in provider offices.
The current data also highlight the recent increase in US pediatric influenza vaccinations administered in August and September. Each season, the median first administration date moved earlier, as early as August 17 in 2010-2011. A recent analysis of US electronic health care reimbursement claims for pediatric influenza vaccination demonstrated a consistent increase in early-season vaccination from 2006-2007 through 2008-2009. 12 The current data suggest that this trend has continued through 2010-2011.
The primary strength of this analysis is that the data regarding influenza vaccinations come from a large, prospectively captured sample from offices that actively recorded all vaccinations as they were administered. The primary limitation relates to the absolute point estimates of 2-dose compliance. Compliance rates in this study may overestimate true compliance because the estimates are based on the rate of second receipt in those children who were identified by the office at the time of vaccination as requiring 2 doses; any child not identified by the office as requiring 2 doses would not be included in the denominator. Accordingly, the calculated rates of compliance presented here should be interpreted as measures of compliance in children to whom providers intended to administer 2 doses. This potential overestimation should not affect comparisons between different populations such as the VFC and non-VFC populations because the effect should be similar in both. Additionally, the present study lacked the power to assess delay on the state level. The timeliness of VFC vaccine delivery may vary at the state grantee level, and data from a larger study may help identify best practices to minimize delay.
Conclusion
Because of administrative complexities, VFC influenza vaccines have typically been available later in the influenza season compared with non-VFC vaccines, resulting in a delay in vaccination among children receiving VFC vaccines. During 2010-2011, VFC vaccines were shipped earlier in the season, resulting in greater equity in 2-dose compliance rates between the VFC and non-VFC populations. VFC vaccines should be shipped to providers as soon as possible. Vaccine delivery by early September should help decrease disparities in access to influenza vaccination between the VFC and non-VFC populations.
Footnotes
Acknowledgements
Formatting the manuscript for submission was performed by Complete Healthcare Communications, Inc (Chadds Ford, PA) and was funded by MedImmune.
Authors’ Note
As sponsor of the study, MedImmune was involved in the study design, which included the collection, analysis, and interpretation of data as well as the writing of the manuscript.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs Ambrose and Toback were both employees of MedImmune, LLC, at the time the study was conducted.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was sponsored by MedImmune, LLC, Gaithersburg, MD, USA.
