Abstract
Introduction
Parkinson’s disease (PD) is a progressive neurodegenerative condition caused by the loss of dopamine cells located within the basal ganglia (Pavese et al., 2010). PD is associated with a variety of motor and non-motor symptoms (Davis et al., 2003) with fatigue being one of the most common non-motor symptoms. Fatigue impacts occupational performance, participation, and quality of life (Herlofson & Kluger, 2017). The exact etiology of fatigue is unknown, but it can arise at early or later stages of the disease (Dogan et al., 2015).
Fatigue can impact everyday function and occupational performance (Olsson et al., 2013). For example, fatigue is associated with reduced social participation, increased risk of social isolation, and psychological distress (Herlofson & Kluger, 2017). For people with PD, fatigue is also associated with early retirement and reduced working hours, sometimes resulting in financial distress and a diminished sense of productivity (Friedman et al., 2007; Zesiewicz et al., 2007).
Despite the impact of fatigue on health and daily life, it is under-recognized in many health care settings (Shulman et al., 2002). Even when recognized, currently available meta-analyses and systematic reviews in PD report no definitive pharmacological solution (Elbers et al., 2016; Lou, 2015; Nassif & Pereira, 2018), emphasizing the importance of developing and testing non-pharmacological interventions. Among non-pharmacological treatments, cognitive-behavioral therapy (CBT), exercise, and energy conservation interventions have been reported in the literature (Asano & Finlayson, 2014). However, the scarcity of high-level Randomized Controlled Trials (RCTs) hinders the adoption of non-pharmacological interventions, especially in people living with PD (Elbers et al., 2016; Lou, 2015; Nassif & Pereira, 2018).
One of the most frequently studied fatigue interventions is the “Managing Fatigue: A Six-Week Energy Conservation Course.” Managing Fatigue is a client-centered occupational therapy program which is aligned with the Person-Environment-Occupation (PEO) model (Law et al., 1996). The program teaches and supports energy management strategies such as activity simplification, task analysis, environmental modification, communicating about fatigue, planning, and prioritizing (Packer et al., 1995).
The Managing Fatigue intervention is internationally recognized and has been evaluated in various countries including the United States (Finlayson et al., 2011; Mathiowetz et al., 2005), Australia (Ghahari et al., 2010), and the Netherlands (Blikman et al., 2017). The Managing Fatigue intervention has been included in the Sweden clinical guidelines for people with multiple sclerosis (MS) due to the strong evidence base for the program (Socialstyrelsen, 2016). Studies in MS samples have shown effectiveness in decreasing the impact of fatigue on daily life activities (Finlayson et al., 2011; Liepold & Mathiowetz, 2005; Sauter et al., 2008), depression (Ghahari et al., 2010; Sauter et al., 2008), sleep problems (Sauter et al., 2008), and improving participation (Beckerman et al., 2013), self-efficacy (Ghahari et al., 2010; Liepold & Mathiowetz, 2005), and quality of life (Finlayson et al., 2011; Ghahari et al., 2010; Liepold & Mathiowetz, 2005).
The Managing Fatigue intervention was originally developed and evaluated in a face-to-face group format (Beckerman et al., 2013; Mathiowetz et al., 2005; Sauter et al., 2008) but has since been adapted for teleconference (Finlayson et al., 2011), internet (Ghahari et al., 2010), and one-to-one formats (Blikman et al., 2017; Van Heest et al., 2017).
The one-to-one format has only been tested in two studies. Blikman et al. (2017), using an RCT design, evaluated a twelve-session version of the program for people living with MS, while Van Heest et al. (2017), in a one-group pre-test, post-test design, evaluated a version with five out of the six modules of the program (the last module was excluded) in people with chronic conditions (MS, fibromyalgia, cancer, and stroke).
Blikman et al. (2017) found no significant difference in fatigue between the experimental and information-only control groups as measured with the Checklist Individual Strength (CIS20r), the Modified Fatigue Impact Scale (MFIS), and the Fatigue Severity Scale (FSS). Moreover, no significant changes were found for social participation, measured with the Impact on Participation and Autonomy questionnaire (IPA), the Medical Outcomes Study Short Form 36 (SF-36), or the Rehabilitation Activities Profile (RAP). However, Van Heest (2017) found significant improvements in post-test fatigue measured by the Functional Assessment of Chronic Illness Therapy–Fatigue Scale; quality of life, measured with the Functional Assessment of Cancer Therapy–General; and self-efficacy, measured with the Self-efficacy for Performing Energy Conservation Strategies Assessment (SEPECSA). The heterogeneity in the delivery of the program, as well as the differing outcome measures and populations in each of these studies makes the results difficult to interpret, and more studies are required to assess the one-to-one delivery approach of the Managing Fatigue intervention.
While the Managing Fatigue intervention has been tested extensively in people with MS, only one study has included people living with PD to date. Ghahari et al. (2010) evaluated fatigue due to MS (n = 74), post-polio (n = 13), and PD (n = 8) in an RCT design testing the internet format of the program. Results demonstrated marginal improvement in self-efficacy and significant reductions in stress compared to the non-intervention group. However, results were not reported separately for each condition.
In summary, prior studies have shown promising results for the Managing Fatigue intervention, however, there is limited evidence of effectiveness for people living with PD, and evidence for the one-to-one format is contradictory. The planned study will address these gaps by evaluating the feasibility of a one-to-one videoconference delivery format of the Managing Fatigue intervention, named “The Managing Fatigue: Individual Program (MFIP),” for people living with PD.
The MFIP consists of six weekly sessions adapted for one-to-one delivery via videoconferencing by our research team, which includes the original author of the Managing Fatigue intervention. Videoconference reduces geographic and transportation barriers and allows participants to benefit from the program while following public health restrictions imposed by the COVID-19 pandemic. Regulations enforced by the government in response to the pandemic have changed the landscape of healthcare practices. With many jurisdictions now adopting virtual care and telehealth interventions as usual care, evidence of effectiveness of video conference-based interventions is needed more than ever (Hassan et al., 2020).
Methods
Study Design and Ethics
A mixed-methods approach (Creswell, 2013) nested in a pilot RCT, assigning participants in a 1:1 ratio to either usual care or MFIP arms will be employed. Qualitative and quantitative data will be collected simultaneously to evaluate the feasibility and preliminary effectiveness of the MFIP. Consistent with the feasibility/RCT pilot study design (Eldridge et al., 2016), we will assess two main aims. Aim 1 is to evaluate the feasibility of the MFIP and the pilot protocol in people living with PD. For this aim, the following research questions will be addressed:
To what extent is the MFIP relevant, acceptable, and impactful from the perspective of people living with PD? How effective are recruitment strategies to include participants from a range of sociodemographic backgrounds? To what extent are the selected outcome measures acceptable to people living with PD? Are there any statistical differences among study outcome measures between the MFIP and usual care arms? What is the required minimum sample size for a future full-scale RCT based on differences between study arms?
Aim 2 is to assess the preliminary effectiveness of the MFIP, more specifically to answer:
As this is a feasibility study, there will be no primary versus secondary outcomes. The outcomes for evaluation of preliminary effectiveness will be occupational performance, occupational balance, fatigue impact, quality of life, sleep quality, and self-efficacy. Results will be used to inform the primary outcome of any future RCT. This protocol has been approved by the Nova Scotia Health Research Ethics Board (ref: 1027048).
Participants
Estimating an attrition rate of 20%, a convenience sample of 54 participants (27 in each arm) will be recruited in Canada. We conducted both a sample size calculation and consulted previous literature to inform our estimation. To calculate the sample size, the type-1 error was set at 5% and the type-2 error at 20% for a power of 80%. Consulting previous relevant studies on fatigue in PD, effect sizes were only available for two of the outcome measures to be used in this study: The Multiple Fatigue Inventory (MFI) with an effect size of 0.664 (Bivard et al., 2017) and the Canadian Occupational Performance Measure (COPM) with an effect size of 0.37 (Sturkenboom et al., 2013). Thus, using the minimum effect size reported for the COPM, a total sample size of 42 will be required. Considering a 20% attrition rate, 54 participants will be recruited. The calculated sample size also aligns with methodological reviews that recommend a sample size of 10–50 participants for pilot/feasibility studies (Hertzog, 2008; Sim & Lewis, 2012).
The inclusion criteria for this study will be the ability to provide informed consent; age 18 years or older; having been diagnosed with PD (self-report); a score of ≥ 4 on the FSS; ability to read and communicate in English; access to the internet and an electronic device (i.e., Smart cellphone, tablet, computer); and a private place for videoconferences. Exclusion criteria will be the previous completion of the Managing Fatigue intervention, or a co-morbidity that causes severe fatigue. Since participants are required to take an active role in the program, having severe cognitive impairment, demonstrated by a score of < 13 on the Mini-Mental Status Exam (MMSE), will also be an exclusion criterion.
This study will recruit participants in several different ways: (a) web-based advertisements, (b) posters, (c) social media, (d) community advertising, and (e) word of mouth. Individuals who are interested in participating will contact the research team by email. Potential participants will first complete an email screening procedure to confirm they meet the preliminary eligibility criteria (are 18 years or older, live with PD, experience fatigue, and have access to an electronic device). Once confirmed, they will receive the study information and the consent form by email. Final eligibility will be confirmed after completion of the FSS and the MMSE during a secure videoconference. Consent will be confirmed electronically before any data collection. Participants will be informed that they may withdraw from the study at any time and for any reason. Whenever possible, reasons for withdrawals will be recorded.
After the screening process, a research staff member, not involved in the screening or data collection, will randomly assign eligible participants to either the usual care or the MFIP arms using sealed envelopes. After randomization, the research staff will assign participants in the MFIP arm to a therapist and will respond to any queries from participants. The assessor who is responsible for screening and data collection will be masked to group assignment. Study participants will be instructed not to share any information on group allocation with the assessor. The masking of the assessor will be monitored by documenting any information received during pre, post, and follow-up testing. A full CONSORT flow diagram is presented in Figure 1.
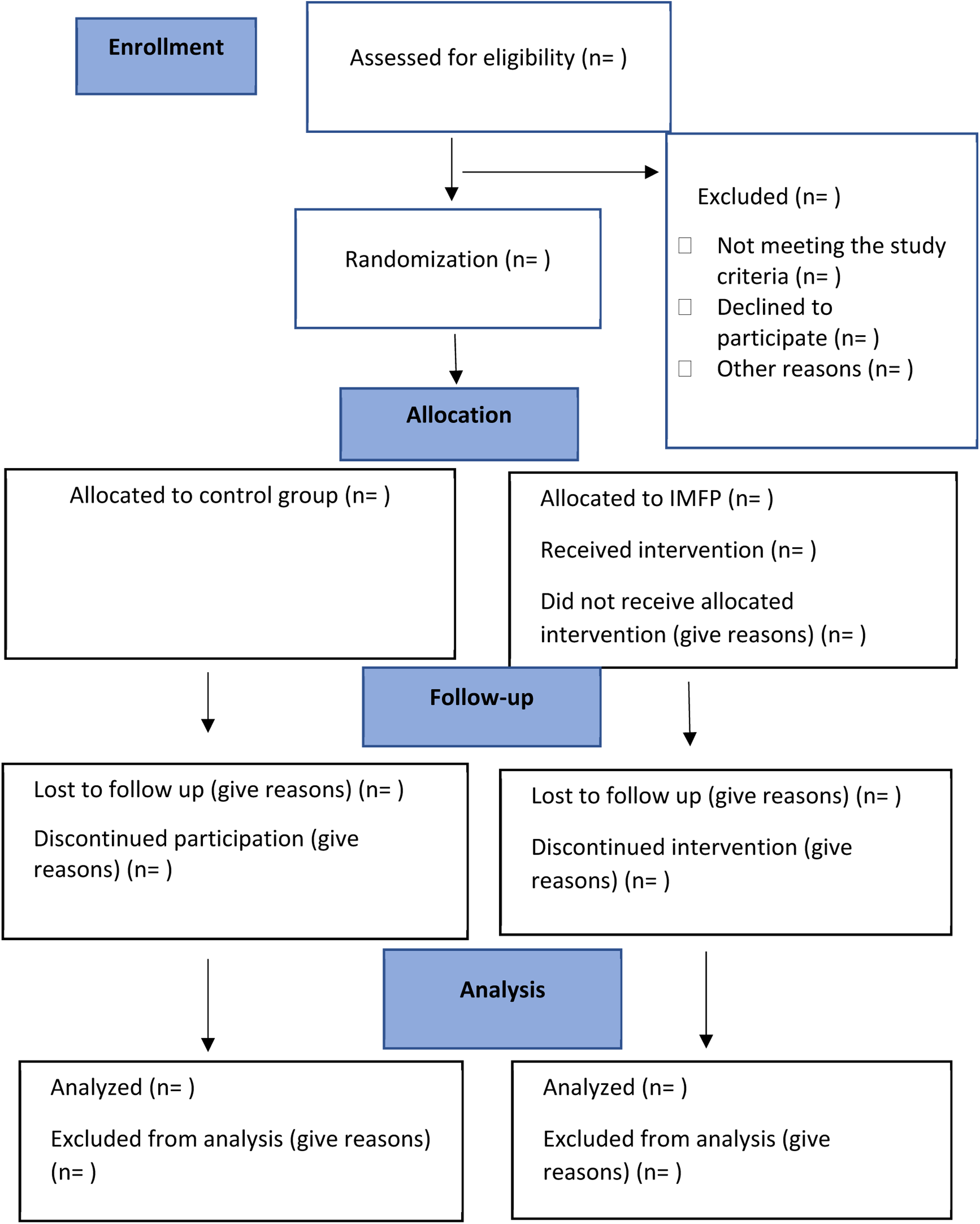
CONSORT flow diagram.
In this study, participants cannot be masked to their assigned group (MFIP versus usual care) as they will be actively involved in the sessions. Participants in the MFIP arm will receive the six-week MFIP in addition to their current healthcare services. Those in the usual care arm will not receive the MFIP and will continue receiving usual care. After completion of the study, participants in the usual care arm will have the option to receive the program's manual and attend an online, three-hour workshop on fatigue management held by occupational therapists at no cost. In this workshop, they will receive an abbreviated program that includes the program content and an introduction to the pre-session activities, in-session activities, and homework.
Intervention
The MFIP consists of weekly sessions of structured discussion between an individual participant and a qualified occupational therapist. The MFIP is described in two manuals, one for participants and one for therapists. The content is similar to that in the original program; however, two additional topics have been added: sleep hygiene and cognitive fatigue. The content is arranged in six sessions: (a) the importance of rest and sleep; (b) communication and body mechanics; (c) activity stations; (d) priorities and standards; (e) balancing your schedule; and (f) course review and future plans. Content related to cognitive fatigue is threaded through all sessions. Each session includes three main parts: (1) pre-session where participants are asked to complete activities prior to the session so they are prepared for discussion with their therapists; (2) in-session activities/information where participants discuss and learn about topics, based on their priorities; (3) homework where participants practice at home to ensure they are building the skills discussed in sessions. Participants will be supported to evaluate and tailor the strategies suggested in the program according to their prioritized occupational performance issues. Each weekly session will be approximately 90 min, although therapists may adjust the pace depending on participants’ needs and preferences. The goal is to support participants to complete the program within six to eight weeks.
Licensed occupational therapists, who complete an online training course designed for this study, will deliver the program. Therapists will be required to complete all modules and successfully answer all corresponding quiz questions. Therapists will have the opportunity to review content and re-take quizzes until all questions are correctly answered.
The therapist training covers: (a) history and development of the MFIP; (b) evidence of the effectiveness of the program; (c) fatigue; (d) an introduction to fatigue measurement; (e) energy management; (f) self-management and chronic disease management; (g) building self-efficacy; (h) motivational interviewing; (i) the transtheoretical model of behavioral change; (j) put it all together; (k) using secured videoconferencing; (l) PD; and (m) fatigue in PD.
Data Collection
Data for study outcome measures will be collected using Opinio Survey Software (ObjectPlanet. Inc, Copyright, 1998–2021) a secure, university supported platform. Participants will complete questionnaires online during a videoconference call with the assessor.
Demographic information will be collected after participants are enrolled and have consented. Participants will answer questions regarding age, gender, years since diagnosis, living status, employment status, and any treatments they are currently receiving to manage their fatigue. Data collected for age, gender, years since diagnosis and living status will be used to assess the effectiveness of recruitment strategies to include participants from a range of sociodemographic backgrounds.
Data collection for aim 1 will consist of feasibility questionnaires and focus groups to understand the perspectives of participants in the MFIP arm. Two feasibility questionnaires were developed by our research team to assess, from the perspective of participants, the relevance, acceptability, and perceived impact of the program. Individualized links to the feasibility questionnaires will be sent by a research assistant to participants via email. Feasibility Questionnaire #1 will be completed weekly to evaluate the relevance, acceptability, and perceived impact of the content of each session. Feasibility Questionnaire #2 will be completed at the end of the program and will assess relevance, acceptability, and perceived impact of the entire program. The response scale for both is a five-point Likert scale (Likert, 1932). Collected data will be exported to Stata: Software for Statistics and Data Science (StataCorp., 2019) following the completion of the data collection phase.
After completion of the program, 15 participants will be recruited to take part in one of three focus groups (n = 5 per group). Selection of participants will be based on maximum variation sampling accounting for disease duration, fatigue severity, and gender. Participants will be provided additional information about the focus groups and will be asked to provide a separate informed consent before participation. An experienced research assistant will lead the focus groups via a secure videoconference using a prepared interview guide. Participants will be encouraged to discuss the feasibility of the program including relevance, acceptability, barriers to completion of the program, acceptability of study measures, and perceived impact/changes.
To evaluate the effectiveness of recruitment strategies for future RCT designs, the following data will be collected and analyzed: how participants learned about the program; the proportion of participants who withdraw or are lost to follow-up, and, where possible reasons for withdrawal; and sociodemographic characteristics associated with each of the above.
To assess the preliminary effectiveness of the MFIP (Aim 2), data will be collected using standardized outcome measures administered at baseline, following completion of the program, and three months after completion. The COPM will be conducted using the standard interview-based protocol during a synchronous videoconference call. All other study measures are self-report surveys and will be completed online. To our knowledge, only the Occupational Balance Questionnaire (OBQ) and the Pittsburgh Sleep Quality Index (PSQI) have been used online. Formatting of the paper versions of all measures (COPM excluded) selected for this study, will be faithfully reproduced on the online platform. To maximize similarity to the original method of administration, the assessor will be available via videoconference during completion to respond to participants’ questions.
Study Outcome Measures
The outcome measures were selected based on the content and expected impact of the program. In addition, studies that employed the Managing Fatigue intervention or focused on fatigue in people living with PD were reviewed to identify potential outcome measures.
Respondents identify three to five self-selected occupational performance issues and then rate each on a 10-point Likert scale. The average performance and satisfaction scores will be calculated by summing individual occupational issue scores then dividing by the number of issues (Law et al., 1990). The COPM is responsive to change in individuals with chronic conditions (N = 150) with high sensitivity and has acceptable test-retest reliability for both performance and satisfaction scores (ICC = 0.63 and 0.84, respectively; Eyssen et al., 2011). The COPM was developed based on the Canadian Model of Occupational Performance (Law et al., 1990) and has previously been used as a primary outcome in an RCT evaluating an intervention for people with PD (Sturkenboom et al., 2014). In that study, the COPM was shown to be sensitive to change and able to detect significant differences between groups (Sturkenboom et al., 2014).
The OBQ measures satisfaction with the amount of time that a person takes to accomplish tasks. It uses a 4-level ordinal response scale for each item ranging from 0 “completely disagree” to 3 “completely agree.” The OBQ total score ranges from 0 (no occupational balance) to 35 (maximum occupational balance). The psychometric properties of the OBQ have not been explored in PD. However, in the general population, it has shown high internal consistency (Cronbach's alpha = 0.936) and test, re-test reliability (Spearman's Rho = 0.926) for its total score (N = 67). Neither ceiling nor floor effects were reported with this measure (Håkansson et al., 2020).
Two additional measures will be used to assess disease severity (measured by Estimated Hoehn and Yahr scale) and depression (measured by Geriatric Depression Scale). The GDS-15 is a short, yes/no self-report measure used to screen for depression in the elderly. Although not extensively tested in PD, it appears to have adequate discriminant validity for a diagnosis of major and minor depressive disorder in PD at a cut-off of 4/5 (Schrag et al., 2007). The Estimated Hoehn and Yahr scale (HY) is a widely used clinical rating scale that identifies the broad categories of motor function in PD (Mischley et al., 2017).
Data Analysis
Focus groups will be audiotaped and data transcribed verbatim. Any potentially identifying information will be removed prior to analysis. Data will be analyzed using the six-stage content analysis framework by Braun and Clarke (2013). The text will be coded verbatim without changing the meaning. Coded material will be categorized semantically until themes emerge. Codes and themes will be reviewed and refined until the final distinctive themes can be created.
Quantitative data will be downloaded from Opinio to Stata. After cleaning, data will be examined for skewness, outliers, and systematic missing data. Extreme outliers, defined as greater than ± 2SD from the mean, will be removed if they are less than 5% of all data (Allison, 2002).
Data from the Feasibility Questionnaires will be analyzed using descriptive analysis, including frequencies and proportions for categorical data, and means and standard deviations for continuous data. Baseline data on age, depression, and disease severity will be examined for equality of groups. In the case of significant differences between groups in any of these variables, a series of sensitivity analyses will be used to test the effect of the variables.
Because all measurements will be completed during a videoconference call and under the supervision of the assessor, we do not expect significant missing data at the item level. Any missing data, not at random, will be managed by following the protocol of each measurement tool or imputed using mean substitution. If the maximum number of missing items has not been included in the measurements’ protocol, it will be set at 20%.
A general linear mixed model will be used to assess the preliminary effectiveness of the outcome variables: fatigue impact, occupational balance, occupational performance, quality of life, sleep quality, and self-efficacy. The models will include the group assignment variables, time, and interaction of time and group. We will conduct analyses using both intention-to-treat and per-protocol principles. Similar to previous studies (Plow et al., 2020), for per protocol analysis, participants having complete data or received at least four intervention sessions will be included in the analysis.
Data from outcome measures will be used to calculate effect sizes to estimate the preliminary effectiveness and the required sample size for future RCTs and evaluate the sensitivity and responsiveness of the study outcome measures. Measurement tools with the highest effect size and the smallest significance level will be defined as the most sensitive measures (Corzillius et al., 1999). Measurement tools with higher levels of mean variability at baseline will have a smaller effect and, therefore, will be defined as less responsive measures (Husted et al., 2000). The sensitivity and responsiveness of measurement tools alongside their acceptability, obtained from the focus group data, will be used to identify the most suitable outcome measures for the future RCT.
Discussion
The effectiveness of fatigue interventions in people with PD has been investigated in only a few RCTs and fatigue has rarely been a primary outcome measure (Elbers et al., 2016; Lou, 2015; Nassif & Pereira, 2018). Moreover, the use of small sample sizes, variable definitions for fatigue, and a lack of consistency in accounting for confounding variables, such as depression and sleep problems (Ridder & Chou, 2016), makes it difficult to draw reliable conclusions from these studies. Therefore, there is a need for more robust studies that explore how best to manage fatigue, especially in people living with PD. In order to conduct a rigorous RCT, robust preliminary data on outcome measures, design, and feasibility of delivering a program are required. This preliminary pilot study is specifically designed to address these issues and provide answers needed for future larger and fully powered studies.
The Managing Fatigue intervention has been proven to be effective in reducing the impact of fatigue associated with neurological conditions, thus there is potential for a similar result for people with PD. Recommendation for clinical use, however, requires evidence of effectiveness from RCTs. Further, our planned study will put emphasis on occupation-related outcomes which have not been adequately measured and evaluated in previous studies that tested the Managing Fatigue program. To the best of our knowledge, only Veenhuizen et al. (2019) a more accurate evaluation of the impact of the program on everyday activities evaluated occupational performance as an outcome for this program. Our study will use occupational performance and occupational balance as study outcomes to better allow a more accurate evaluation of the impact of the program on everyday activities and occupational functioning of participants.
In our pilot RCT design, we are using a one-to-one delivery approach via videoconference. Although one-to-one delivery allows therapists to focus on tailoring the program to participants’ distinct priorities and situations, and improves access for people in remote areas, or where lower population density makes forming groups more difficult, there is still insufficient evidence to support its benefits in the PD population. This study will therefore contribute to the body of evidence examining the feasibility of using a one-to-one delivery approach.
This study will also contribute to the growing body of knowledge related to virtual care. Since the COVID-19 pandemic in 2020, the feasibility of using telehealth has been amplified. Therefore, developing and evaluating studies to assess the delivery of health programs using videoconference is becoming more relevant. However, using online communications and delivery approaches, when conducting studies, introduce additional challenges. For example, in the current study, there are no validated online versions for many of the study's self-report outcome measures. Thus, we had to adapt these tools for online administration. This is a potential limitation of our planned study, but we will implement measures to mitigate this limitation including consulting with the authors of the outcome measures and ensuring that assessors will be available via videoconferencing to help address any issues or questions that might arise for participants. Future studies to validate online versions of outcome measures will be needed to fully understand the effectiveness of virtual care.
Another possible limitation of using the videoconference delivery is participants’ unfamiliarity with the technology, potentially causing frustration and/or fatigue, especially in older individuals. Although these challenges exist, in this pilot study we will learn about the perspectives of people with PD regarding the acceptability of the online versions of the intervention and the study outcome measures. This will contribute to our understanding of the face validity of the online measures.
Conclusion
The proposed study will evaluate the feasibility of the MFIP in people living with PD. If the known beneficial effects of this program for other conditions extend to the PD population, this research will provide the preliminary evidence needed to support further studies that will help guide integration into the process of care for people living with PD.
Key Messages
Effective fatigue management interventions are not currently available for people with PD.
Testing the feasibility and preliminary effectiveness of the Managing Fatigue: Individual Program will provide occupational therapists with evidence to support their practice in addressing fatigue with individuals with PD.
Footnotes
Acknowledgments
We would like to thank our research team at the International Chronic and Complex Condition Research Group (IC3RG) at Dalhousie University, especially Sabrena Jaswal, for their assistance in running this trial. We would also like to thank the occupational therapists who will deliver the program and the participants who will give of their time to help improve the lives of others.
The authors received no financial support for the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
