Abstract
The Royal Australian and New Zealand College of Psychiatrists’ (RANZCP) 2018 position statement supports increased, regulated availability of e-cigarettes (ECs) as a harm-reduction measure and recommends further research into their use. Aligned with this recommendation, we aimed to critically evaluate the RANZCP’s stance on this issue through a literature review focused on the areas identified in the position statement as requiring further investigation: (1) the adverse health effects attributable to ECs; (2) use of ECs for smoking cessation (particularly for people living with severe mental illness); and (3) EC-associated risks for nicotine naïve young people. We identified and summarised evidence of harm attributable to ECs that is particularly relevant to young people through direct adverse health sequelae, onset of nicotine dependence and increased risk of combustible cigarette (CC) use. A small number of studies suggest ECs can be used for harm-reduction purposes in people diagnosed with nicotine dependence and severe mental illness. However, these results must be considered alongside robust evidence supporting the effectiveness of existing pharmacological interventions for smoking cessation in people with severe mental illness. The position statement is in urgent need of review in line with the available evidence.
Introduction
In 2018, the Royal Australian and New Zealand College of Psychiatrists (RANZCP, 2018) released Position Statement 97 supporting the legalisation and regulation of nicotine-containing electronic cigarettes (ECs), also known as vaporisers (‘vaping’). The underlying sentiment was to promote equitable access to a potential harm-reduction smoking cessation intervention for Australians living with mental illness and tobacco dependence. However, concerns were raised regarding the tenability of this position, given weak supporting evidence available at the time, and the risk of creating a new generation of smokers (Scott et al., 2019). Several years have passed, and in 2022 the RANZCP’s EC position statement remains unchanged.
The position statement recommended further research into three areas: (1) the long-term health effects of vaping; (2) the effectiveness of ECs as cessation tools; and (3) monitoring demographic patterns of use, including youth initiation rates. This paper aims to summarise recent findings relevant to these domains. Furthermore, the acceptability of the position statement will be evaluated in light of available evidence.
The long-term health effects of electronic cigarettes
It is widely accepted that ECs are safer than cigarettes (McNeill et al., 2020; Nutt et al., 2014) although the long-term toxicity of vaping remains uncertain (World Health Organization [WHO], 2019). Monitoring EC safety has been challenging in the context of a wide range of electronic nicotine delivery systems containing different flavours, emissions and nicotine dosages of variable accuracy (Ghebreyesus, 2019; WHO, 2019). ECs have only been commercially available since 2003. Malignancy risk increases with combustible cigarette (CC) smoking duration (Remen et al., 2018), and the population consequences of EC use in those who were previous CC smokers may not become apparent for many years. In 2014, Nutt et al. (2014) reported the consensus of an expert panel convened to consider relative harms of nicotine-containing products. They scored products for harm to users and others, utilising weighted criteria to derive a score between 0 and 100. Unsurprisingly, cigarettes were graded as the most noxious with an overall score of 99.6 while ECs were assigned a score of four. This led to widespread publication that ECs are ‘95% less harmful’ than cigarettes (Polosa, 2015). The original authors and others (Eisenberg et al., 2020; Polosa, 2015) acknowledged the absence of data regarding the harms associated with many of the products rated. Reviews by Public Health England (PHE; McNeill et al., 2015) and the Royal College of Physicians (RCP, 2016) supported ‘95% less harmful’ as a reasonable estimate based on studies of toxicants within EC aerosol and cigarette smoke. These studies demonstrate that EC vapour contains harmful constituents, but at substantially lower levels than cigarette smoke. Relative to sustained tobacco consumption, the studies concluded that toxin levels inhaled from EC vapour over many years would be unlikely to result in the same magnitude of health sequelae, with the caveat that longer-term EC risks remain unknown. No well-controlled longitudinal studies exist that can determine the relative safety of ECs vs CCs (Gotts et al., 2019) leading to ongoing uncertainty. As noted, available evidence concludes that vaping is significantly safer than smoking with respect to comparisons of carcinogenic chemical profiles (Goniewicz et al., 2014; Stephens, 2017). This does not mean that ECs should be considered harmless. The WHO advocates for stricter EC regulation in keeping with inconclusive but clear risks associated with these products, including cardiovascular and respiratory disease (Ghebreyesus, 2019; WHO, 2019).
Regarding the cardiovascular risks associated with ECs, Peruzzi et al. (2020) recently completed an umbrella review of seven systematic reviews totaling 183 studies. Generally, these reviews concluded that ECs are safer than CCs with respect to adverse cardiovascular sequelae (Peruzzi et al., 2020). However, acute effects of EC use identified at a cellular level included evidence of oxidative stress, cytotoxicity and cardiomyocyte dysfunction. Physiologically, ECs increased heart rate, blood pressure and arterial stiffness. Clinically, ECs were associated with increased risk of atrial fibrillation and myocardial infarction, although an underlying causal link has not been established. In a nationally representative, observational, longitudinal analysis of EC use and cardiovascular disease, Berlowitz et al. (2022) reported that the risk of incident myocardial infarction or bypass surgery, heart failure or stroke did not differ significantly between exclusive EC users and non-users. Relative to smoking, exclusive EC use provided a 34% risk reduction for the development of cardiovascular disease (hazard ratio [HR]: 0.66, 95% confidence interval [CI]: [0.46, 0.94]). However, the cardiovascular risk of dual EC and CC consumption was equivalent to those exclusively smoking CCs. Similarly, in a randomised study of the cardiovascular effects of switching from CCs to ECs, George et al. (2019) found significantly improved endothelial function within 1 month of transitioning, with the greatest benefits identified for those with the least dual-use. Together, these studies suggest that smokers who transition to exclusive EC use experience health benefits compared with those who continue using CCs.
Recently published results of a nationally representative US survey of 16,855 participants found EC users had significantly higher odds of having a myocardial infarction compared with non-smokers (odds ratio [OR]: 4.09, 95% CI: [1.29–12.98]; Vindhyal et al., 2020). Dual EC/CC users had significantly increased risk of myocardial infarction (OR: 5.44, 95% CI: [2.90–10.22]), stroke (OR: 2.32, 95% CI: [1.44–3.74]), and coronary heart disease (OR: 2.27, 95% CI: [1.37–2.44]) relative to non-smokers (Vindhyal et al., 2020). Causality cannot be determined in this cross-sectional study and the degree of cardiovascular morbidity present prior to commencing ECs is unclear. However, collectively these results provide evidence that ECs do not mitigate the risk of adverse cardiovascular events when CCs continue to be smoked.
Adverse respiratory sequelae have also been linked with ECs (Gotts et al., 2019). After controlling for CC smoking, EC use has been associated with increased risk of asthma (adjusted OR [aOR]: 1.39, 95% CI: [1.28–1.51]) and chronic obstructive pulmonary disease (COPD; aOR: 1.49, 95% CI: [1.36–1.65]; Wills et al., 2021). In laboratory studies, EC liquids affect biological processes relevant to respiratory disease pathogenesis, namely through cytotoxic effects and oxidative stress, and less consistently by increasing inflammatory indices (Wills et al., 2021). However, the impact of EC vapour upon lungs at a cellular level is substantially lower relative to cigarette smoke (Caruso et al., 2021; Iskander et al., 2019). In a recent systematic review, former smokers who switched to ECs showed ~40% lower odds of adverse respiratory outcomes relative to exclusive smokers (Goniewicz et al., 2020). This was based on a small number of mostly cross-sectional epidemiological studies.
Cases of EC or vaping-associated lung injuries (EVALI) have been well-publicised. In 2019, the Centre for Disease Control and Prevention (CDC) formed a lung injury task force following a nationwide outbreak of EVALI. As of February 2020, 2807 EVALI cases had been reported to the CDC, including 68 deaths (CDC, 2020). The syndrome has been strongly associated with an additive (vitamin E acetate) that was included in THC vaping products, although the evidence is not sufficient to rule out other causative agents including chemicals in non-THC products (CDC, 2020; Kligerman et al., 2020). EVALI cases have declined, likely due to removal of vitamin E acetate from regulated products, and law enforcement responses (CDC, 2020). Ongoing uncertainties continue regarding the exact causes of EVALI.
Adverse health sequelae of ECs cannot be considered in isolation from tobacco as most EC users also smoke CCs or are former smokers, placing them at risk for consequences associated with both products (Ghebreyesus, 2019). Combined smoking and vaping may be transitional for some. However, longitudinal evidence indicates that those with higher tobacco dependence are more likely to become or remain dual users than to switch to exclusive EC use (Morgan Snell et al., 2020). In a large, nationally representative US cross-sectional survey, dual CC/EC use was associated with 36% higher odds (95% CI: [1.18–1.56]) of cardiovascular disease compared with CCs alone (Osei et al., 2019). Similarly, in a nationally representative longitudinal analysis, the incidence of respiratory disease at follow-up was significantly associated with baseline reporting of former (aOR: 1.31, 95% CI: [1.07, 1.60]) and current (aOR: 1.29, 95% CI: [1.03, 1.61]) EC use (Bhatta and Glantz, 2020). ECs and smoking were independent risk factors for the development of adverse respiratory outcomes. Respiratory ailments were likely developing over years before diagnosis, and some smokers with symptoms probably tried ECs for therapeutic, harm reduction, purposes. Authors calculated that switching from smoking to exclusive EC use would lower the risk of developing respiratory disease by a factor of 0.58 (42%). However, dual-use was the predominant pattern, reported by >90% of EC users at follow-up. For dual users, the odds of developing respiratory disease were estimated at 3.30 compared with never smokers/vapers.
An emerging indirect harm relates to evidence that ECs may increase the risk of CC use in non-smokers and former smokers. In a systematic review, umbrella review and meta-analysis, data from 25 studies of EC use and CC-uptake among non-smokers were synthesised (Baenziger et al., 2021). Non-smokers who used ECs were on average three times more likely to start smoking CCs and become current tobacco smokers, and former smokers using ECs were twice as likely to relapse compared with non-EC users (Baenziger et al., 2021). ECs are not benign, and ongoing monitoring of associated direct and indirect health risks is needed.
The effectiveness of ECs as smoking cessation tools
The most recent smoking cessation clinical guidelines from the Royal Australian College of General Practitioners (RACGP, 2019) explicitly recommends cautious EC prescribing, stipulating that they are not first-line treatment. Concerns raised by the RACGP include (1) the lack of tested products; (2) the absence of high-level evidence that ECs are effective for smoking cessation; (3) limited data characterising long-term toxicity; (4) unease regarding potential for dual-use; and (5) re-normalisation and promotion of nicotine-consumption, particularly among younger populations.
Eleven recent systematic reviews and meta-analyses (see Table 1; Chan et al., 2021; Grabovac et al., 2021; Hanewinkel et al., 2022; Hartmann-Boyce et al., 2021; Hedman et al., 2021; Ibrahim et al., 2021; Pound et al., 2021; Thomas et al., 2021; Wang et al., 2021; Zhang et al., 2021) have examined the efficacy and safety of ECs for smoking cessation, including one that was prepared by the Australian National University for the Australian Government Department of Health (Yazidjoglou et al., 2021). Two reviews (Ibrahim et al., 2021; Pound et al., 2021) found no significant differences in 12-month abstinence rates or CC reductions between EC and comparison groups. The majority (9/11) showed that in randomised controlled trials (RCTs), smokers assigned to nicotine ECs were about twice as likely to be abstinent from CCs at follow-up relative to those assigned to placebo or treatment as usual. These findings are tempered by many of the trials being assessed as moderate-to-high risk of bias, and broad confidence intervals representing uncertainty in the efficacy of the EC intervention. Moreover, where complete nicotine abstinence was investigated, CC cessation rates were higher for ECs vs NRT, but those assigned to vaping were less likely to be abstinent from nicotine at follow-up (Hanewinkel et al., 2022). Therefore, ECs as an intervention may be more likely than other smoking cessation tools to perpetuate nicotine dependence. Of course, there is likely to be a group of smokers for whom other therapies have been ineffective where switching to ECs would reduce harm.
Summary of recent systematic-review level evidence for the efficacy of e-cigarettes as aids to cessation of combustible tobacco smoking.
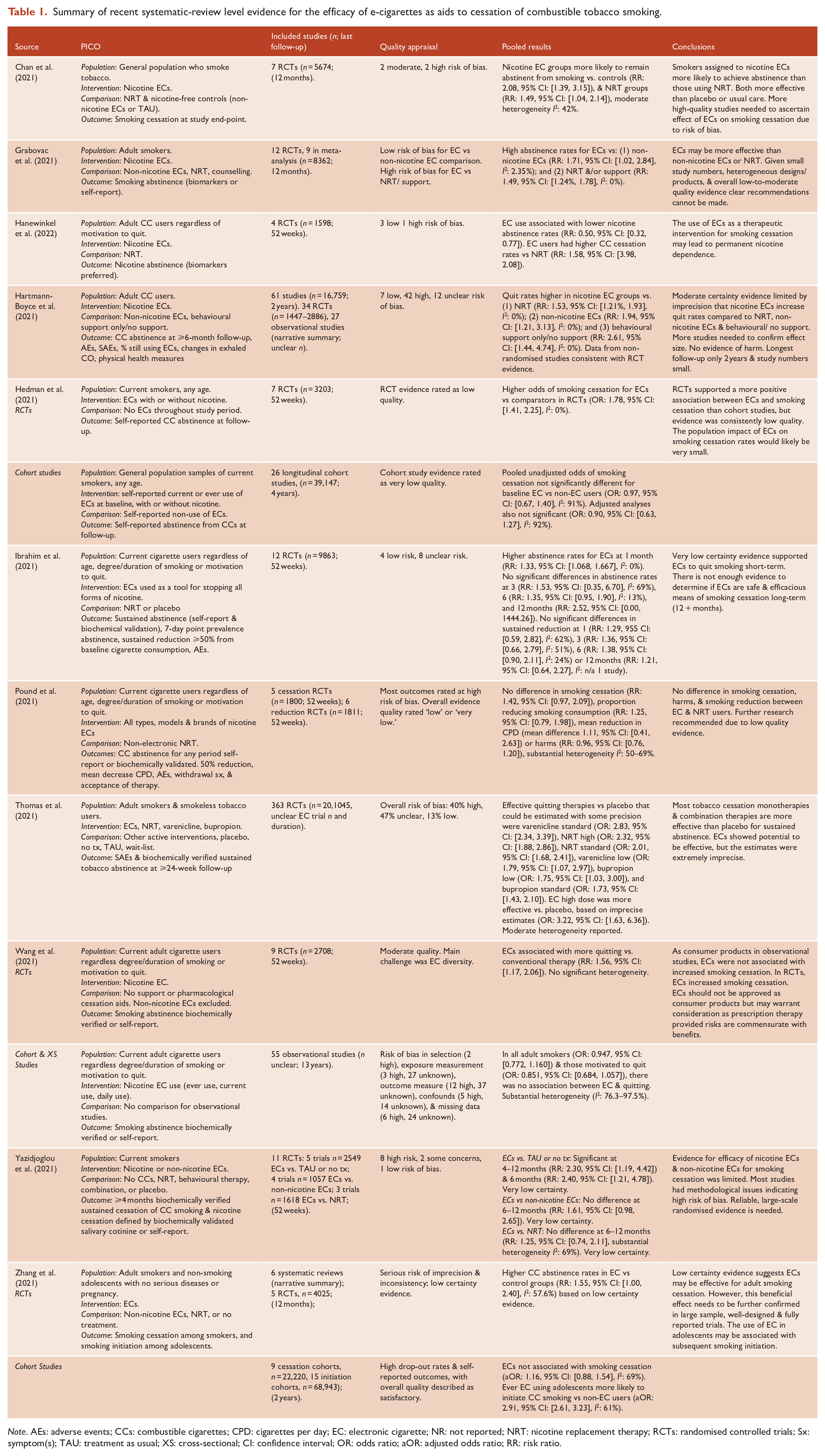
Note. AEs: adverse events; CCs: combustible cigarettes; CPD: cigarettes per day; EC: electronic cigarette; NR: not reported; NRT: nicotine replacement therapy; RCTs: randomised controlled trials; Sx: symptom(s); TAU: treatment as usual; XS: cross-sectional; CI: confidence interval; OR: odds ratio; aOR: adjusted odds ratio; RR: risk ratio.
Three reviews also examined non-randomised study designs. The latest Cochrane Review collated by Hartmann-Boyce et al. (2021) reported that data from non-randomised studies were ‘consistent with RCT data’ in a narrative synthesis. Wang et al. (2021) sub-divided observational study findings by motivation to quit in an attempt to limit heterogeneity. Substantial diversity remained in the context of ECs representing a broad product group. They found, however, that as consumer products ECs did not significantly increase smoking cessation rates (Wang et al., 2021). Other syntheses of observational cohort study findings were consistent with these conclusions (Hedman et al., 2021; Zhang et al., 2021). In addition to not finding a relationship between ECs and smoking cessation across cohort studies, Zhang et al. (2021) also identified increased risk of later CC initiation in adolescents who had used ECs.
In summary, the current evidence-base does not favour the inclusion of ECs as a population health measure. One recent cross-sectional Australian study identified that a small number of people who reported a smoking cessation attempt via ECs (n = 189) were more likely to achieve self-reported smoking abstinence (aOR: 1.68, 95% CI: [1.09, 2.60]; Chambers, 2022). This is promising, but it remains uncertain how increasing the availability of these products will affect population health through sustained CC cessation. The strongest evidence-base and first-line treatment for smoking cessation is approved pharmacological therapies (nicotine replacement therapy, varenicline and sustained-release bupropion hydrochloride) accompanied by behavioural support (RACGP, 2019).
The RANZCP’s (2018) position statement was premised on the ‘. . . disproportionately high smoking prevalence, and low quit rates, among people living with mental illness’. Widespread availability of effective smoking cessation interventions is particularly critical for people diagnosed with severe mental illnesses such as schizophrenia, where rates of CC use are substantially higher than the general population and life expectancy is reduced on average by 14.5 years (95% CI: [11.2, 17.8]; Hjorthoj et al., 2017), partially due to increased health risks secondary to smoking (Tam et al., 2016). The evidence suggesting ECs may be a helpful harm-reduction measure in people with psychotic disorders who smoke CCs is very weak, with six identified relevant studies summarised in Table 2. Across these studies, smoking abstinence was achieved by 7–35% of participants over 4-week to 12-month follow-up periods, and those who vaped significantly reduced CC intake. Methodologically, none of these were RCTs, and the generalisability of results to all people with severe mental illness is questionable. Most studies excluded people with physical health co-morbidities, recent exacerbation of mental illness, or a history of other substance abuse/ dependence. Moreover, the study with the largest sample (n = 86) defined mental illness via a proxy measure of self-reported psychotropic medication use. Of further concern is that nationally representative studies suggests that dual-use of ECs and CCs is common among people with mental health conditions, with estimates ranging from 8.6% to 40.8% (McNeill et al., 2020). There is robust evidence derived from RCTs that currently licenced pharmacological smoking cessation interventions accessible through the Pharmaceutical Benefits Scheme are effective for people diagnosed with schizophrenia (Siskind et al., 2020). To determine the effectiveness of ECs for smoking reduction and cessation in this population, more randomised and methodologically high-quality studies are needed.
Summary of studies investigating ECs for smoking cessation in people diagnosed with severe mental illnesses.
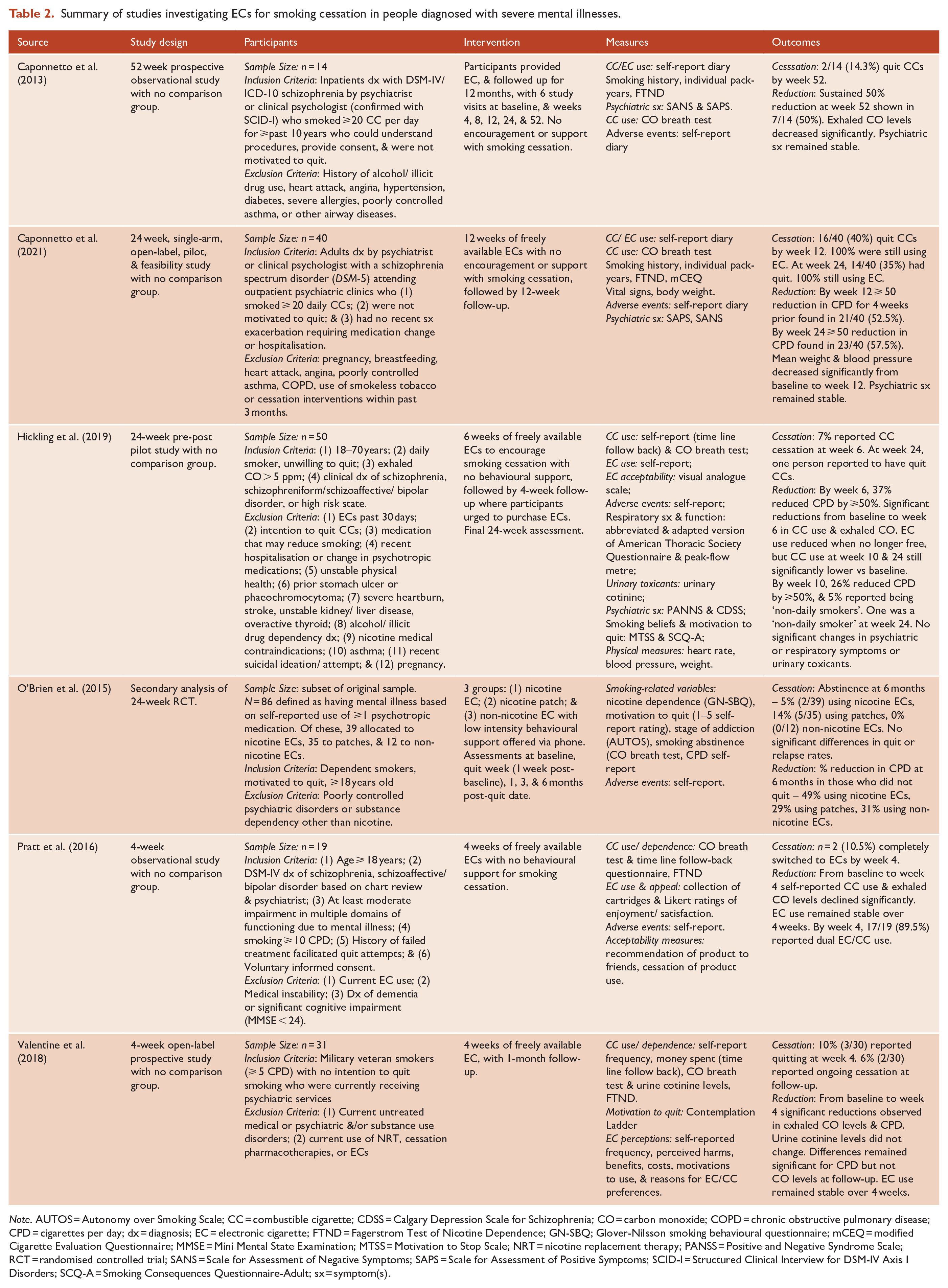
Note. AUTOS = Autonomy over Smoking Scale; CC = combustible cigarette; CDSS = Calgary Depression Scale for Schizophrenia; CO = carbon monoxide; COPD = chronic obstructive pulmonary disease; CPD = cigarettes per day; dx = diagnosis; EC = electronic cigarette; FTND = Fagerstrom Test of Nicotine Dependence; GN-SBQ; Glover-Nilsson smoking behavioural questionnaire; mCEQ = modified Cigarette Evaluation Questionnaire; MMSE = Mini Mental State Examination; MTSS = Motivation to Stop Scale; NRT = nicotine replacement therapy; PANSS = Positive and Negative Syndrome Scale; RCT = randomised controlled trial; SANS = Scale for Assessment of Negative Symptoms; SAPS = Scale for Assessment of Positive Symptoms; SCID-I = Structured Clinical Interview for DSM-IV Axis I Disorders; SCQ-A = Smoking Consequences Questionnaire-Adult; sx = symptom(s).
The use of electronic cigarettes by youth
The prevalence of youth EC use varies globally, with evidence of rates increasing in many countries over time (Yoong et al., 2018). In the United States, youth smoking rates have declined in association with increasing vaping popularity (Levy et al., 2019). It remains unclear, however, how the increased use of ECs by young people will affect public health over time. The population impact of reduced CC use in younger populations may not be offset by the larger increases in ECs in this group (Levy et al., 2019). In Australia, ECs pose a significant threat to the reductions in smoking rates achieved by public health interventions. The most recent National Drug Strategy Household Survey from the Australian Institute of Health and Welfare (AIHW, 2020) identified that daily smoking rates have decreased to 11.0% in 2019, from 24% in 1991. From 2016 to 2019, rates of current and lifetime EC use increased among Australian smokers (4.4–9.7%) and non-smokers (0.6–1.4%), with young adults being most attracted to these products (AIHW, 2020). In 2019, younger current EC users were more likely than older EC users to report that they had never smoked when they started vaping (65% of 14- to 17-year-olds and 39% of 18- to 24-years-olds vs <10% of people aged 40 years and older; AIHW, 2020). More than 70% of young people (14–24 years) used ECs ‘out of curiosity’ (AIHW, 2020). The novelty of vaping devices and availability of flavours likely appeals to adolescents and young adults during a period of developmental vulnerability associated with risk-taking, experimentation and susceptibility to the onset of substance use problems.
Evidence suggests that increased EC availability will potentially cultivate more CC smokers. Six meta-analyses summarised in Table 3 indicate that young non-smokers exposed to ECs had between two and five times the odds of commencing smoking CCs compared with those not exposed to ECs (Chan et al., 2020; Khouja et al., 2020; O’Brien et al., 2021; Soneji et al., 2017; Yoong et al., 2021; Zhong et al., 2016). These conclusions were mostly based on heterogeneous prospective observational studies, many of which were assessed as having a moderate to high risk of bias. The reasons for the association between exposure to ECs and subsequent CC use are uncertain. One theory is that vaping acts as a ‘gateway’ to more potent, accessible and toxic forms of nicotine (Chan et al., 2020). The association between EC use and later CC smoking has also been explained through the lens of ‘common liability’, that is, that there are shared susceptibilities for EC and CC use (Chan et al., 2020; Khouja et al., 2020). Regardless of the underlying drivers, there is a clear imperative for policy-makers to ensure that young people are adequately protected from the harms of nicotine dependence and smoking through careful EC regulation.
Summary of systematic-review level evidence regarding the implications of EC exposure for young people.
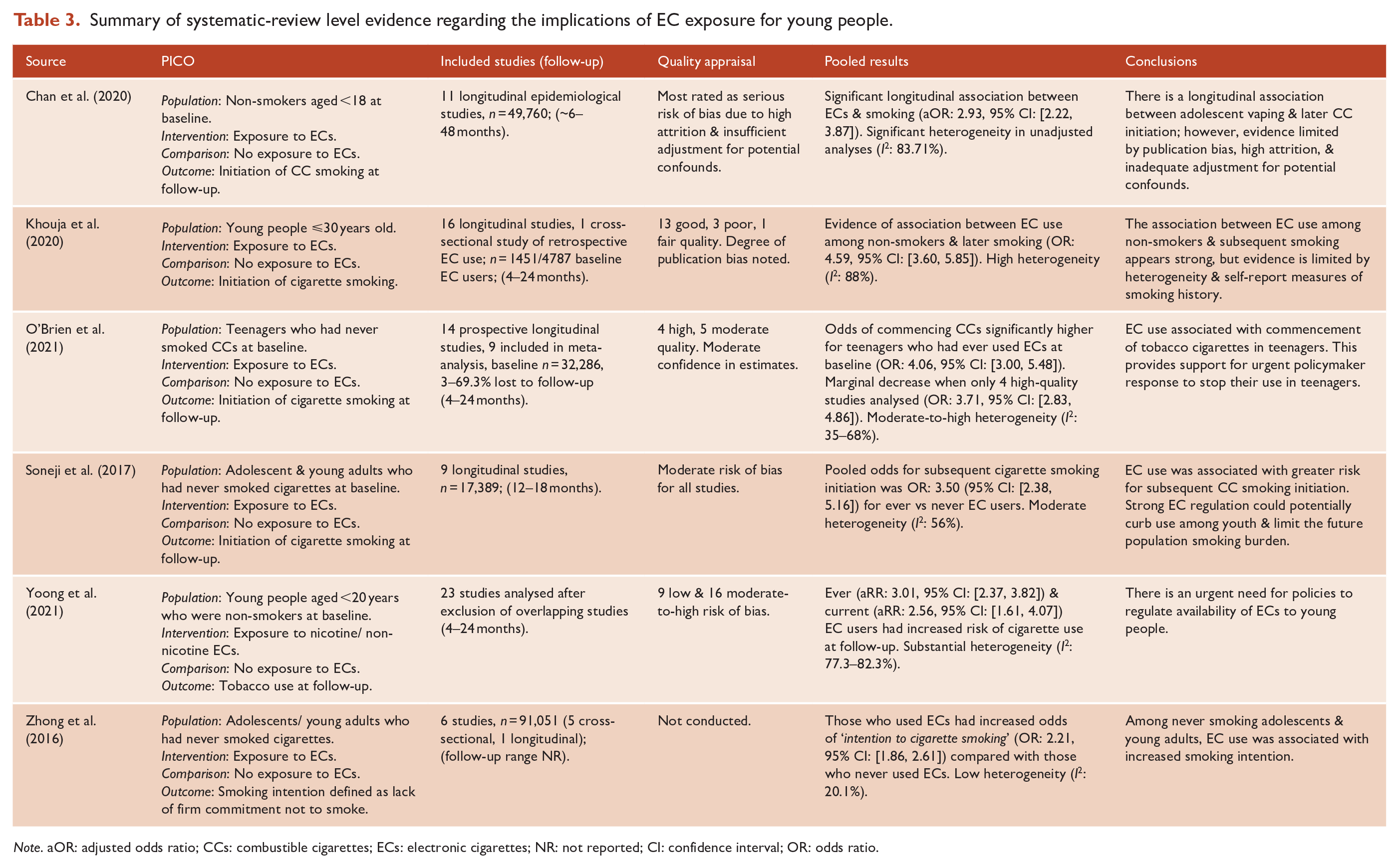
Note. aOR: adjusted odds ratio; CCs: combustible cigarettes; ECs: electronic cigarettes; NR: not reported; CI: confidence interval; OR: odds ratio.
Summary and conclusions
There is robust evidence that currently available pharmacological agents are the most effective interventions for smoking cessation in people with serious mental illness. The evidence supporting the use of ECs in this population for smoking cessation is very weak. ECs may assist people who are dependent on CCs with smoking cessation. However, many will combine the use of CCs with ECs, which will not mitigate health risks. Non-smokers and in particular young people who are exposed to ECs are at significantly increased risk of commencing CC smoking. It is recommended that the RANZCP (2018) reconsider Position Statement 97. We advocate for the RANZCP to either withdraw or substantially revise their position statement on ECs, which may in time become a serious public health risk. We also recommend the RANZCP develops a position statement on tobacco smoking in people with serious mental illness where high-quality evidence of effective interventions exists.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
