Abstract
Background:
Bipolar disorder may undertake a progressive course in a subset of patients, and research efforts have been made to understand the biological basis underlying this process. This systematic review examined the literature available on biological markers associated with illness progression in bipolar disorder.
Methods:
Peer-reviewed articles were assessed using Embase, PsycINFO and PubMed, as well as from external sources. After initial screening, a total of 871 citations from databases and other sources were identified. Participants with a diagnosis of bipolar disorder were included in our systematic review; however, studies with participants younger than 15 or older than 65 were excluded. All studies were assessed using the Newcastle-Ottawa Scale assessment tool, and data pertaining to the results were extracted into tabular form using Google Sheets and Google Documents. The systematic review was registered on PROSPERO international prospective register of systematic reviews (ID Number: CRD42020154305).
Results:
A total of 35 studies were included in the systematic review. Increased ventricular size and reduction of grey matter volume were the most common brain changes associated with illness progression in bipolar disorder. Among the several biomarkers evaluated in this systematic review, findings also indicate a role of peripheral inflammatory markers in this process.
Discussion:
The studies evaluating the biological basis of the illness progression in bipolar disorder are still scarce and heterogeneous. However, current evidence supports the notion of neuroprogression, the pathophysiological process related to progressive brain changes associated with clinical progression in patients with bipolar disorder. The increase in peripheral inflammatory biomarkers and the neuroanatomical changes in bipolar disorder suggest progressive systemic and structural brain alterations, respectively.
Introduction
Bipolar disorder (BD) is a chronic and highly debilitating mental disorder that affects approximately 1% of the population worldwide (Merikangas et al., 2011). The recurrence of mood episodes is often associated with unfavourable clinical outcomes, including functional and cognitive impairments, higher rates of medical and psychiatric comorbidities and a lower response to treatment (Rosa et al., 2012; Van Rheenen et al., 2019; Wingo et al., 2009). A substantial proportion of patients with BD shows cognitive impairments that persist even following remission of mood symptoms (Bortolato et al., 2016), particularly in patients who experience multiple episodes (Van Rheenen et al., 2019). In addition, a recent study showed that functional deterioration may be progressive in nearly half of patients, characterized mainly by a higher number of relapses, greater neurocognitive impairment and greater severity of depressive symptoms (López-Villarreal et al., 2020).
BD is known to have a heterogeneous course of illness, where a subgroup of patients may present with progressive biological alterations in the brain and the periphery, including changes to neuroanatomical structure, neurotrophic factors, inflammatory markers and oxidative stress markers (Kapczinski et al., 2019; Kauer-Sant’Anna et al., 2009; Strakowski et al., 2002). Taking into consideration the hypothesis of neuroprogression, the pathological rewiring of the brain that takes place in parallel with clinical deterioration (Berk et al., 2011; Kupka et al., 2021; Rosa et al., 2014; Tatay-Manteiga et al., 2018), major efforts have been made to understand BD through clinical staging models (Berk et al., 2017; Cosci and Fava, 2013; Duffy, 2014; Kapczinski et al., 2014; McGorry et al., 2010; Reinares et al., 2013; Rosa et al., 2014). Furthermore, systemic biological changes observed in this illness may play a critical role in the neuroprogression of BD (Andreazza et al., 2009; Fries et al., 2012; Kapczinski et al., 2009; Tatay-Manteiga et al., 2017).
Although there has been a substantial increase in the understanding of illness progression, the pathophysiology of BD is highly complex, and its underlying neurobiological mechanisms remain largely unclear. Therefore, it is crucial to study the course of this illness further to identify potential biological pathways that may be involved in illness progression and provide insight on new approaches to diagnosis, prognosis and treatment.
The goal of this systematic review, therefore, was to synthesize the current research on biological markers (biomarkers) associated with illness progression in BD in (1) studies that assessed the levels of biomarkers in patients with BD according to illness progression and (2) studies that correlated biomarkers with relevant variables for illness progression.
Methods
In this study, we followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (Moher et al., 2015; Page et al., 2021). This study was registered on the PROSPERO international prospective register of systematic reviews (ID Number: CRD42020154305). A literature search was conducted using three databases (Embase, PsycINFO and PubMed) and included studies published until October 22, 2019.
Selection criteria
In this systematic review, cross-sectional studies, longitudinal studies, prospective studies, retrospective studies, non-randomized trials and case-control studies were included; the reference lists of the eligible studies were also assessed. Reviews, editorials, case reports, meta-analyses, randomized clinical trials and re-reports of raw data from a secondary source were excluded from this systematic review. No publication period, language or geographical restrictions were imposed in the current review. The inclusion criteria were participants between the ages of 15–65, with a diagnosis of BD. Studies consisting of paediatric populations were excluded from the systematic review. Geriatric populations were also excluded from the systematic review as older populations are considered more prone to medical comorbidities and neurodegenerative diseases, which may confound the data set (Lala and Sajatovic, 2012).
The following keywords were used in the systematic search strategy: (bipolar disorder OR bipolar disorders OR mania OR hypomania OR manic OR hypomanic OR manic-depressive disorder OR manic depressive disorder OR bipolar affective disorder) AND (neuroprogression OR illness progression OR early stage OR late stage OR early stage OR late stage OR staging OR progression) AND (biomarkers OR biological markers OR oxidative stress OR telomere length OR inflammation OR inflammatory markers OR cytokines OR neurotrophins OR neurotrophic factors OR imaging OR neuroimaging OR brain changes OR cellular changes OR cellular resilience OR blood biomarkers OR hormone changes OR protein changes).
Selection of eligible studies
The titles and abstracts were screened using Rayyan – a screening software for systematic reviews (Ouzzani et al., 2016). Duplicates were removed by the software and studies not fulfilling the inclusion criteria were excluded upon title/abstract screening. The full texts of the articles were then retrieved and assessed for eligibility. All of the review processes were performed independently by S.M. and S.G., and consensus regarding discrepancies between reviewers was achieved with other members of the review team, B.W.A. and B.P. The PRISMA flow diagram is shown in Figure 1.
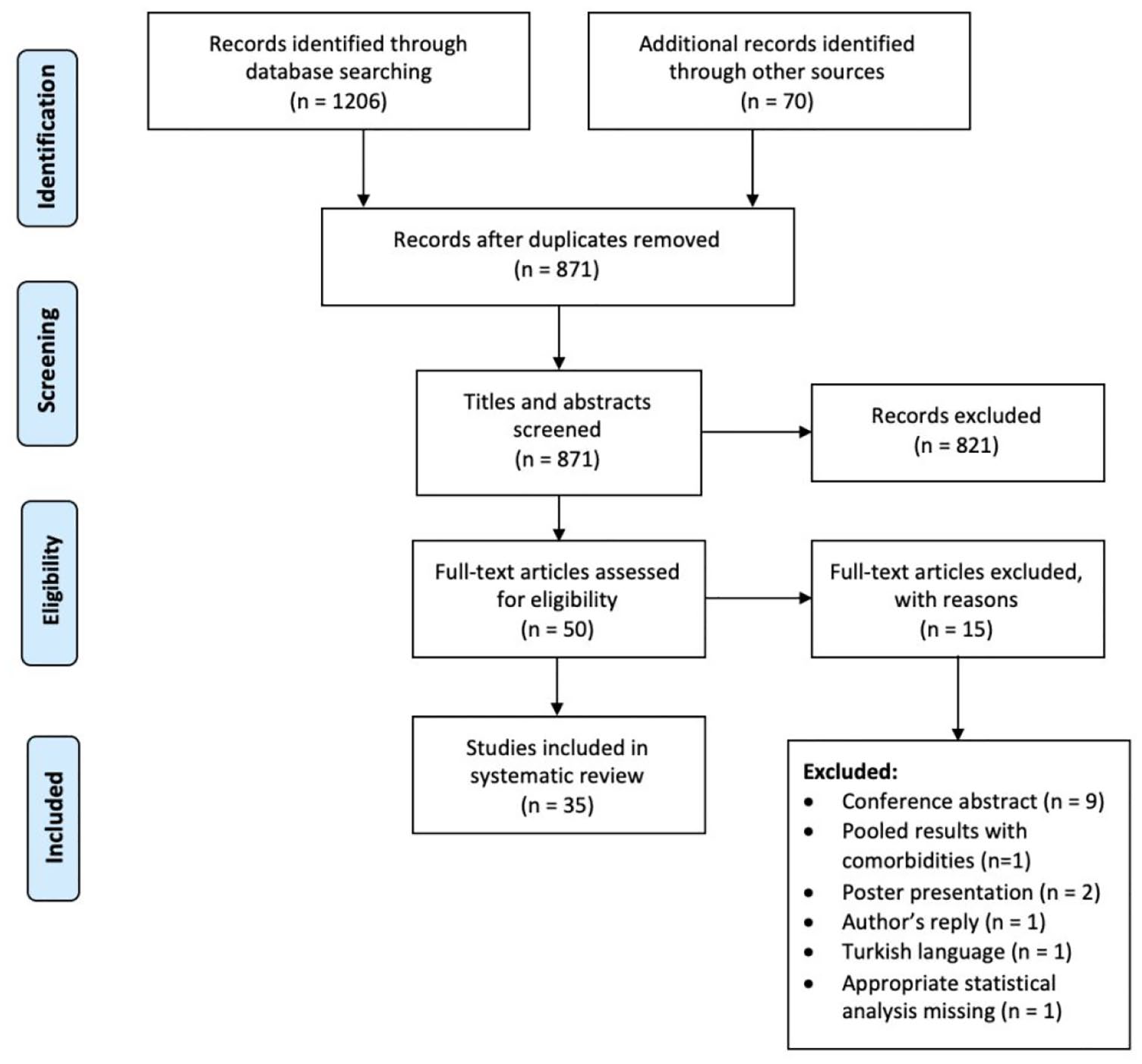
PRISMA Flow Diagram (Moher et al., 2015) of the literature search and selection.
Data extraction
Data from eligible studies were extracted and reported independently by two authors (S.M. and S.G.) using Google Sheets. Discrepancies were resolved through discussion with B.W.A. and B.P. The following information was retrieved: (1) population (sample size, sex and age of each group); (2) staging model (when applicable); (3) clinical variables used for staging (when applicable); (4) type of biological sample; and (5) biomarkers assessed regarding staging or associated with illness progression.
Quality assessment
A quality assessment using the Newcastle-Ottawa Quality Assessment Scale (Modesti et al., 2016) was also performed by S.M. and S.G., independently through a blind review. Conflicts were resolved with support from B.W.A. and B.P.
Results
Study characteristics
A total of 1206 studies were retrieved from three databases (581 from Embase, 211 from PsycINFO and 414 from PubMed), and 70 additional records were identified through other sources. Title/abstract screening was performed for 871 records, and 50 studies underwent full-text screening, for a total of 35 studies included in our systematic review.
Our systematic review included (1) studies that assessed biomarkers in patients with BD using staging models to classify patients into early and late stages, and (2) studies that presented correlations between biomarkers and clinical variables potentially associated with illness progression (such as duration of illness, number of hospitalizations, number of episodes, symptom severity and age at onset). The most common staging models for BD used in the publications were Kapczinski et al. (2009) and Rosa et al. (2014). The 35 studies included in our review reported changes in several peripheral biomarkers, including neurotrophic factors, inflammatory and oxidative stress markers, as well as brain changes. Data from the staging studies are in Table 1, and data from the studies investigating correlations between biomarkers and clinical variables associated with illness progression are in Table 2. An overview of the categorized peripheral biomarkers observed in more than one staging study is in Supplementary Table 1. The scoring for the Newcastle-Ottawa Quality Assessment is in Supplementary Tables 2 and 3.
Clinical and biological characteristics of the included staging studies.
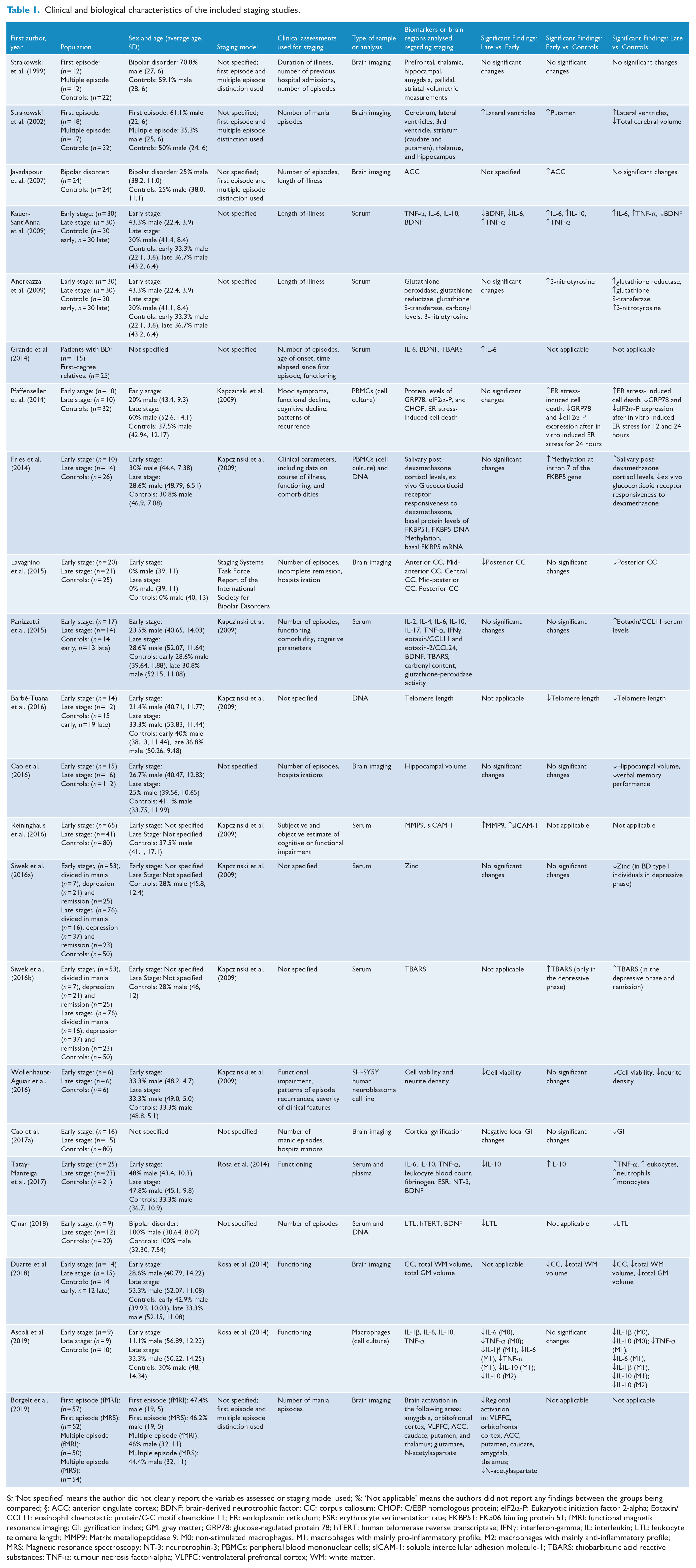
$: ‘Not specified’ means the author did not clearly report the variables assessed or staging model used; %: ‘Not applicable’ means the authors did not report any findings between the groups being compared; §: ACC: anterior cingulate cortex; BDNF: brain-derived neurotrophic factor; CC: corpus callosum; CHOP: C/EBP homologous protein; eIF2α-P: Eukaryotic initiation factor 2-alpha; Eotaxin/CCL11: eosinophil chemotactic protein/C-C motif chemokine 11; ER: endoplasmic reticulum; ESR: erythrocyte sedimentation rate; FKBP51: FK506 binding protein 51; fMRI: functional magnetic resonance imaging; GI: gyrification index; GM: grey matter; GRP78: glucose-regulated protein 78; hTERT: human telomerase reverse transcriptase; IFNγ: interferon-gamma; IL: interleukin; LTL: leukocyte telomere length; MMP9: Matrix metallopeptidase 9; M0: non-stimulated macrophages; M1: macrophages with mainly pro-inflammatory profile; M2: macrophages with mainly anti-inflammatory profile; MRS: Magnetic resonance spectroscopy; NT-3: neurotrophin-3; PBMCs: peripheral blood mononuclear cells; sICAM-1: soluble intercellular adhesion molecule-1; TBARS: thiobarbituric acid reactive substances; TNF-α: tumour necrosis factor-alpha; VLPFC: ventrolateral prefrontal cortex; WM: white matter.
Clinical and biological characteristics of the included studies correlating biomarkers to clinical variables.
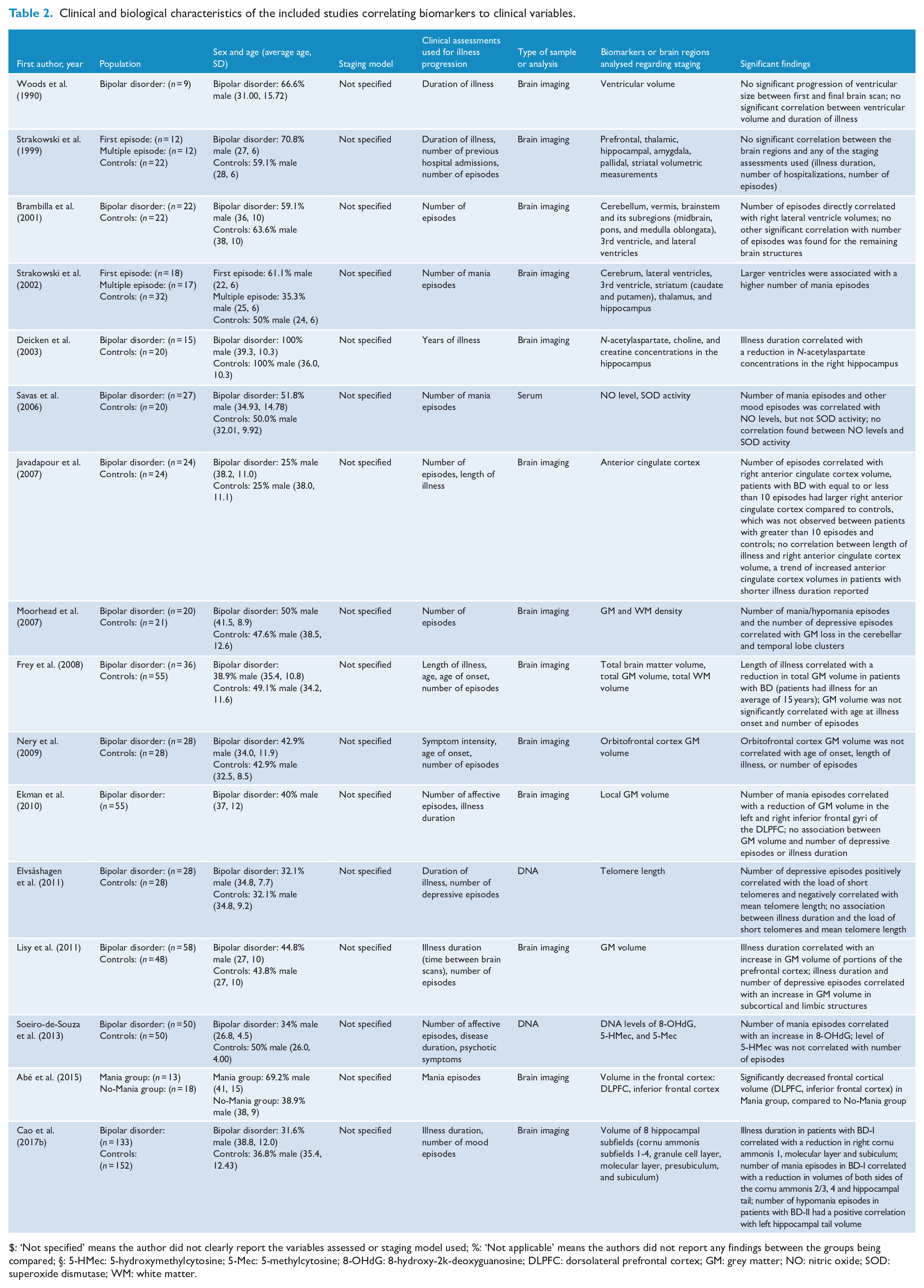
$: ‘Not specified’ means the author did not clearly report the variables assessed or staging model used; %: ‘Not applicable’ means the authors did not report any findings between the groups being compared; §: 5-HMec: 5-hydroxymethylcytosine; 5-Mec: 5-methylcytosine; 8-OHdG: 8-hydroxy-2k-deoxyguanosine; DLPFC: dorsolateral prefrontal cortex; GM: grey matter; NO: nitric oxide; SOD: superoxide dismutase; WM: white matter.
Neurotrophic factors
Five studies that investigated neurotrophic factors in regard to illness progression in BD were included in our review, all of which evaluated brain-derived neurotrophic factor (BDNF) peripheral levels. Çinar (2018), Tatay-Manteiga et al. (2017), Panizzutti et al. (2015) and Grande et al. (2014) found no statistically significant changes in BDNF levels between patients at the early and late stages of this illness. One study in particular, conducted by Kauer-Sant’Anna et al. (2009), found similar BDNF levels in patients with BD at the early stage compared with the control group; however, there was a decrease in BDNF levels in patients at the late stage of this illness when compared with controls and individuals at the early stage. Overall, the authors also found a negative correlation of length of illness with BDNF levels (Kauer-Sant’Anna et al., 2009).
Inflammatory markers
Interleukin-6
There were five studies that analysed inflammatory markers included in our review, all of which measured interleukin-6 (IL-6) levels. Although Tatay-Manteiga et al. (2017) and Panizzutti et al. (2015) found no significant changes in peripheral IL-6 levels between groups, Kauer-Sant’Anna et al. (2009) found an increase in IL-6 levels in patients at the late stage and at the early stage of the illness when compared to their respective control groups. However, these levels were lower in patients at the late stage when compared to patients at the early stage. Also, the authors found a negative correlation between length of illness and IL-6 levels (Kauer-Sant’Anna et al., 2009). However, Grande et al. (2014) identified an increase in IL-6 in patients at the late stage as compared to patients at the early stage. Another interesting study conducted by Ascoli et al. (2019) investigated the inflammatory response of monocyte-derived macrophages from patients with BD in regard to illness progression. In this study, M0 referred to non-stimulated macrophages, and M1 and M2 referred to macrophages polarized towards the mainly pro-inflammatory and mainly anti-inflammatory profiles, respectively. A decrease in the secretion of IL-6 was found in M0 and M1 from patients at the late stage compared to those at the early stage, and in M1 from patients at the late stage as compared to controls. No significant changes were found for any macrophage phenotype when comparing early stage patients to the control group.
Interleukin-10
The levels of interleukin-10 (IL-10), an anti-inflammatory cytokine, were examined in regard to staging for four of the five studies investigating inflammatory markers. Panizzutti et al. (2015) found no significant changes in serum IL-10 levels between groups. Although Kauer-Sant’Anna et al. (2009) and Tatay-Manteiga et al. (2017) found no differences in IL-10 levels between late stage patients and the control group, both studies noted a significant increase in IL-10 levels in patients at an early stage compared to controls. In addition, Kauer-Sant’Anna et al. (2009) found no change in serum IL-10 levels when comparing the early and late stage groups, and Tatay-Manteiga et al. (2017) noted a significant decrease in IL-10 levels in patients at the late stage compared to patients at the early stage. Furthermore, although Ascoli et al. (2019) did not find any changes in IL-10 when comparing early stage patients and the control group, there was a significant decrease in the secretion of IL-10 by all macrophage phenotypes from patients at the late stage compared to controls. There was also a decrease in IL-10 secretion by M1 and M2 in patients at the late stage as compared to patients in the early stage.
Tumour necrosis factor-α
While Panizzutti et al. (2015) found no significant changes in serum tumour necrosis factor-α (TNF-α) levels between groups, Tatay-Manteiga et al. (2017) found increased levels in patients at the late stage compared to controls, and Kauer-Sant’Anna et al. (2009) found an increase in TNF-α levels at the late stage group compared to the control group and also to the early stage, as well as an increase in patients at an early stage compared to controls. Ascoli et al. (2019) found a significant decrease in secretion of TNF-α in M0 and M1 from late stage patients compared to early stage patients. Furthermore, there were no significant changes in TNF-α levels in patients at the early stage compared to controls, but there was a significant decrease in TNF-α secretion in M1 from patients at the late stage compared to controls (Ascoli et al., 2019).
Other inflammatory markers
Other inflammatory markers have been explored in single studies. Panizzutti et al. (2015) found a notable increase in eosinophil chemotactic protein/C-C motif chemokine 11 (eotaxin/CCL11) serum levels in late stage patients compared to controls. In addition, Tatay-Manteiga et al. (2017) found a significant increase in leukocytes, neutrophils and monocytes count in patients at late stage compared to the control group. Furthermore, Ascoli et al. (2019) found that IL-1β secretion was decreased in M0 and M1 in patients at the late stage compared to controls, as well as compared to patients at the early stage in the M1 phenotype.
Oxidative stress
Our review included four studies that analysed oxidative stress in patients at early and late stages of BD. Thiobarbituric acid reactive substances (TBARS) levels were measured in three studies; two of them did not find differences between patients at early and late stages (Grande et al., 2014; Panizzutti et al., 2015), while one study conducted by Siwek et al. (2016b) found an increase in TBARS levels in patients at the early stage (in the depressive phase) and in patients at the late stage of BD (in the depressive phase and remission) compared to the control group. Carbonyl content and glutathione peroxidase levels were also analysed by Andreazza et al. (2009) and Panizzutti et al. (2015); however, no significant changes were found between groups.
Andreazza et al. (2009) also found increased levels of glutathione reductase, glutathione S-transferase and 3-nitrotyrosine in patients at late stage BD compared to controls. In addition, 3-nitrotyrosine levels were found to be increased in early stage patients when compared to the control group.
Furthermore, the number of manic episodes was positively correlated with nitric oxide levels (Savas et al., 2006) and 8-hydroxy-2′-deoxyguanosine levels (Soeiro-de-Souza et al., 2013), but was not correlated with superoxide dismutase activity (Savas et al., 2006) and 5-hydroxymethylcytosine levels (Soeiro-de-Souza et al., 2013).
Brain changes
Our review included 18 studies analysing brain changes and illness progression in BD. Lavagnino et al. (2015) identified significant decreases in the volume of the posterior corpus callosum in late stage patients compared to individuals in the early stage and the control group. In Duarte et al. (2018), the authors found a decrease in corpus callosum volume in early and late stage patients relative to the control group.
Duarte et al. (2018) also found a significant decrease in total white matter (WM) volume and total grey matter (GM) volume in late stage patients compared to the control group. While the authors identified a decrease in total WM volume in patients at early stages compared to the control group, no significant change in total GM volume between early stage patients and the control group were detected.
Cao et al. (2016) found a significant decrease in hippocampal volume as well as a decrease in verbal memory performance in patients at the late stage compared to the control group. Using the average of local gyrification index (GI) at each surface vertex on the cortex, Cao et al. (2017a) identified a significant association between BD staging and the GI. While there were no significant changes between early stage patients and the control group in GI, late stage patients did demonstrate an overall decrease in GI compared to the control group.
In a former study, no significant differences were found in specific brain structures (prefrontal, thalamic, hippocampal, amygdala, pallidal, striatal) between patients experiencing a single episode as compared to patients experiencing multiple episodes (Strakowski et al., 1999). In a subsequent study, Strakowski et al. (2002) found that multiple episode patients with BD presented larger lateral ventricles compared to first episode patients and controls, and a smaller total cerebral volume compared to controls, while the putamen was found to be significantly larger in first episode patients compared to the control group. Strakowski et al. (2002) also found a positive correlation between the number of manic episodes and ventricle size, while Brambilla et al. (2001) found a positive correlation between the total number of episodes and right lateral ventricle volume, and Woods et al. (1990) found no significant correlation between ventricular volume and illness duration.
Borgelt et al. (2019) found decreased regional activation in the bilateral ventrolateral prefrontal cortex, orbitofrontal cortex, anterior cingulate cortex (ACC), putamen, caudate, amygdala and thalamus in multiple episode patients compared to first episode patients. Furthermore, they identified a lower level of glutamate and N-acetylaspartate in the ACC of multiple episode patients (Borgelt et al., 2019), while Deicken et al. (2003) found a negative correlation between illness duration and N-acetylaspartate concentrations in the right hippocampus. In addition, Javadapour et al. (2007) found that individuals who experienced equal to or less than 10 previous mood episodes presented significantly larger right ACC compared to the control group, and a positive correlation was found between the number of episodes and right ACC volume.
Although a negative correlation was found between illness duration and total GM volume, number of episodes and age at illness onset was not found to be associated with total GM volume (Frey et al., 2008). Moreover, Nery et al. (2009) found no significant correlation between orbitofrontal cortex GM volume and illness duration, number of episodes and age at illness onset. A decrease in GM volume in the left and right inferior frontal gyri of the dorsolateral prefrontal cortex (DLPFC) was also found to be associated with the number of manic episodes, but not the number of depressive episodes or illness duration (Ekman et al., 2010). Although Lisy et al. (2011) found a positive correlation between GM volume in subcortical and limbic structures and illness duration and number of depressive episodes, Moorhead et al. (2007) found a negative correlation between GM volume in cerebellar and temporal lobe clusters and number of manic and depressive episodes. Lisy et al. (2011) also found that illness duration correlated with an increase in GM volume in portions of the prefrontal cortex, while Abé et al. (2015) found significantly decreased frontal cortical volume (DLPFC and inferior frontal cortex) in individuals who experienced manic episodes within a 6-year period compared to those who did not.
Finally, Cao et al. (2017b) found that the number of manic episodes in patients with BD-I was negatively associated with the size of cornu ammonis (CA) subfields 2/3, and 4, as well as the hippocampal tail. The number of hypomanic episodes in populations with BD-II was also linked with an increase in left hippocampal tail volume, while illness duration correlated with reductions in hippocampal subfields CA1, the molecular layer and the subiculum (Cao et al., 2017b).
Telomere length
Three studies investigating the telomere length according to illness progression of BD were included. Çinar (2018) and Barbé-Tuana et al. (2016) both found a significant decrease in telomere length in patients at a late stage of BD compared to the control group. Furthermore, a decrease in telomere length in late stage patients compared to patients at the early stage was shown in Çinar (2018), while Barbé-Tuana et al. (2016) showed a decrease in telomere length in patients at the early stage compared to the control group. In addition, the number of depressive episodes, but not the illness duration, was found to be correlated with the load of short telomeres and mean telomere length, in a positive and negative direction, respectively (Elvsåshagen et al., 2011).
Other biomarkers
Several biomarkers were investigated in regard to illness progression in only one study. Notably, Siwek et al. (2016a) found a decrease in serum zinc concentration in depressive patients at the late stage of BD compared to the control group or patients in remission; Reininghaus et al. (2016) found a significant increase in matrix metallopeptidase 9 and soluble intercellular adhesion molecule-1 (sICAM-1) in patients at the late stage compared to patients at the early stage of the illness; and Fries et al. (2014) noted patients at the late stage of BD showed increased salivary post-dexamethasone cortisol levels compared to the control group. Furthermore, two in vitro studies were included in this systematic review: Pfaffenseller et al. (2014) found an impaired endoplasmic reticulum (ER) stress response in late stage patients and increased ER stress-induced cell death in early and late stage patients compared to controls (where cell death was more pronounced in late stage patients with BD), and Wollenhaupt-Aguiar et al. (2016) found a significant decrease in cell viability and neurite density in cells treated with serum from late stage patients compared to the control group.
Discussion
In this systematic review, we found that increase in ventricular size and reduction of GM volume were the most pronounced brain changes associated with illness progression in BD. Overall, a decrease in GM volume in various regions of the brain was also found to be correlated to relevant variables of illness progression, further supporting the notion of neuroprogression, the pathophysiological process related to progressive structural, functional and neurochemical brain changes associated with clinical progression in BD (Ekman et al., 2010; Frey et al., 2008; Moorhead et al., 2007; Nery et al., 2009). A comprehensive summary of the findings is shown in Figure 2.
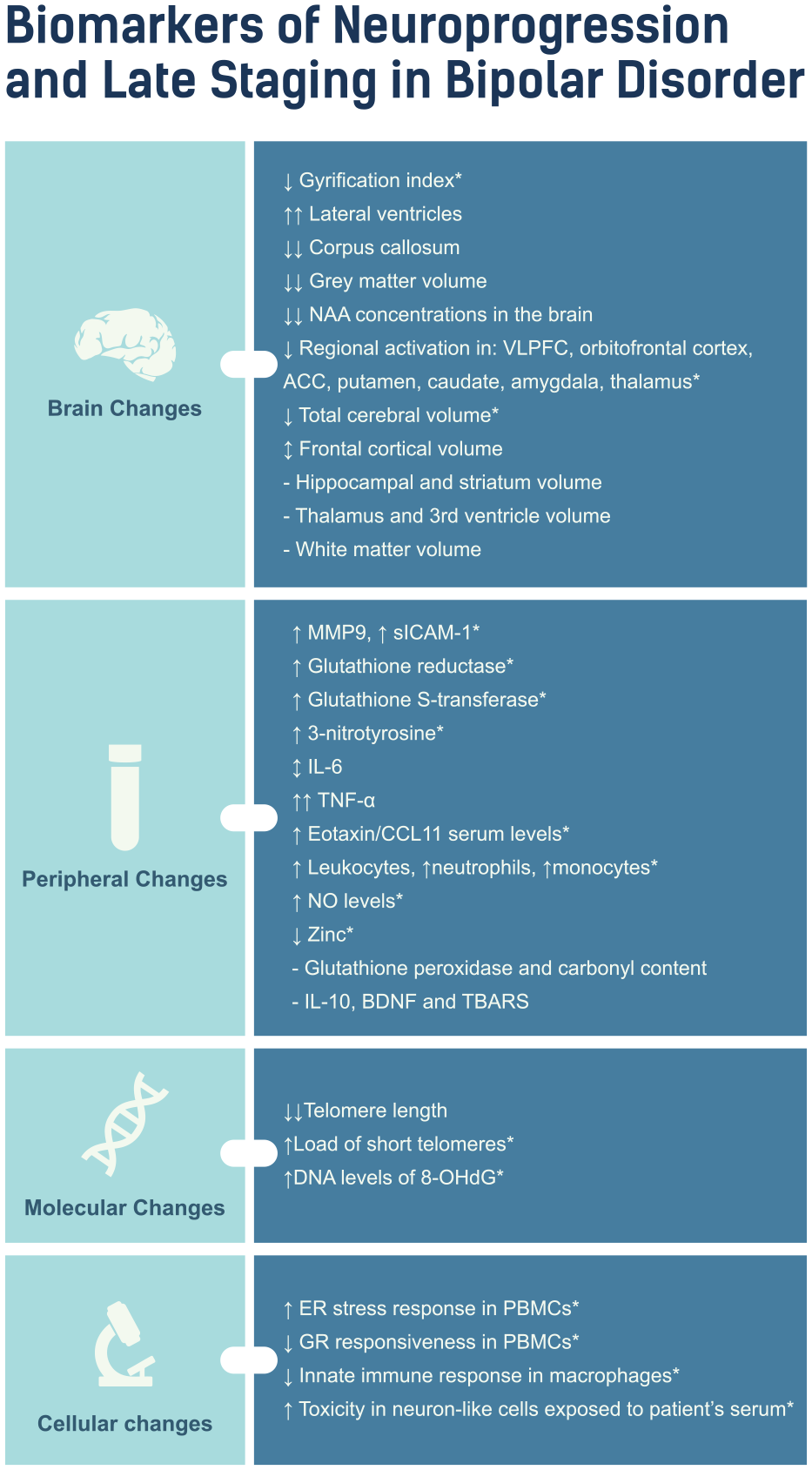
Summary of findings in the late stage patients with BD.
It is known that inflammation may play a critical role in the pathophysiology of BD, potentially underlying the biological basis of neuroprogression. Among the several biomarkers evaluated in this systematic review, the inflammatory markers were the most assessed among the included studies. Although there are conflicting findings, it seems some inflammatory markers have been altered since the early stages which would suggest that inflammation may be a trait marker of BD. Noteworthy, studies showed increased levels of the pro-inflammatory cytokine TNF-α at late stages of BD compared to their respective control groups (Kauer-Sant’Anna et al., 2009; Tatay-Manteiga et al., 2017). This indicates that inflammation may worsen in the later stages of the illness and that the cumulative effects of inflammation over time may contribute to the progressive biological alterations such as the brain changes and an impaired immune system response. It is possible that this may be due to the cumulative effects of mood episodes over time (Kapczinski et al., 2008) since previous research has reported an increase in pro-inflammatory cytokines during the mood episodes of BD (Brietzke et al., 2009; Kim et al., 2007). In addition, previous research has suggested that patients with BD may have increased permeability of the blood–brain barrier, which may facilitate increased migration of inflammatory markers into the brain (Patel and Frey, 2015). Reininghaus et al. (2016) found that patients at the later stages of BD had higher circulating levels of sICAM-1, which has been implicated in inflammation. The serum level of sICAM-1 acts as a proxy for the immune system, and it is hypothesized that they increase the permeability of the blood–brain barrier, allowing cytokines and immune cells from the periphery to enter the central nervous system (CNS) to a greater degree (Muller, 2019). It is also known that cytokines such as IL-6, IL-10, TNF-α, IFNγ and chemokines can activate microglia in the brain which participate in inflammatory processes in response to damage to the CNS (Stertz et al., 2013). These cells help manage neuronal injury in the acute phase. However, their chronic activation can cause an imbalance in homeostasis and induce a constant inflammatory environment which can lead to pruning and remodelling synaptic plasticity, causing damage to neurons; this could be the case in patients with BD due to the recurrence of mood episodes (Watkins et al., 2014), which may contribute to the aforementioned neuroanatomical changes that have been associated with illness progression, and ultimately, the cognitive and functional impairment outcomes seen in BD (Brietzke et al., 2011). In a recent meta-analysis by Velosa et al. (2020), BD was found to be a significant risk factor for the development of dementia, and the number of mood episodes was found to be a relevant predictor for this process. In conjunction with our findings, these results suggest that progressive inflammatory processes and brain changes associated with the number of mood episodes may be positively associated with cognitive impairment, and ultimately, increased risk of dementia in patients with BD. Overall, our findings support the hypothesis that BD may follow a progressive course, which may ultimately lead to deteriorating cognitive and functional performance (Figure 3).
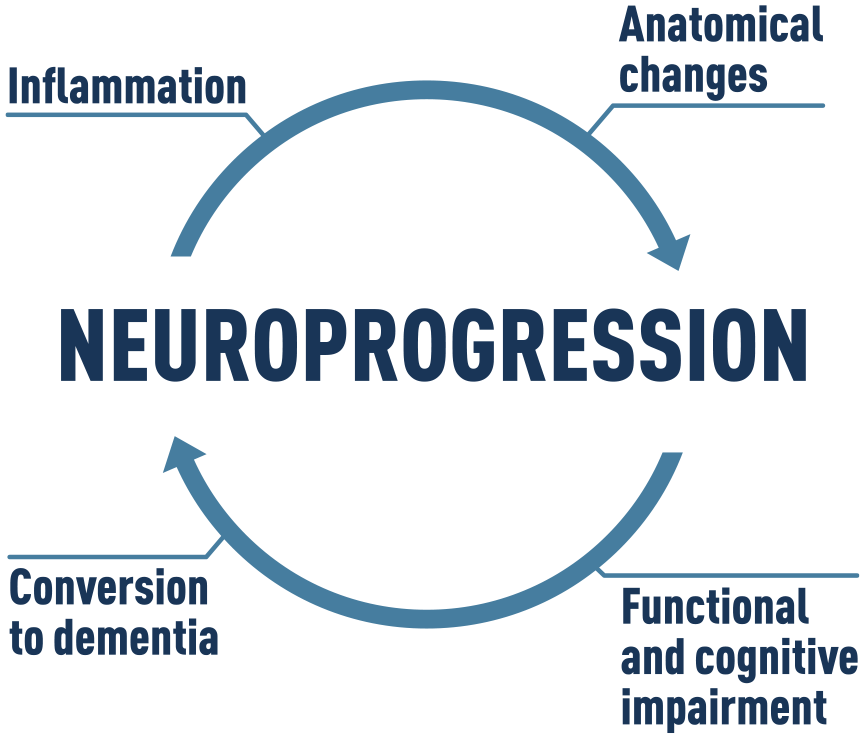
Positive feedback-loop hypothesis for neuroprogression in BD.
It is important to mention that although our focus has been on biomarkers, comorbidities may also be associated with illness progression. It is known that that the incidence of cardiovascular disease, diabetes, anxiety disorders and other comorbidities are high in BD (Aguglia et al., 2022; Krishnan, 2005; McIntyre et al., 2005; Spoorthy et al., 2019), and that these comorbidities have been associated with inflammation (Golia et al., 2014; Salim et al., 2012; Tsalamandris et al., 2019; Van Rheenen et al., 2019). In this sense, a potential relationship between inflammatory processes and comorbidities could contribute to the illness progression in BD. In a recent study by Aguglia et al. (2022), for instance, researchers reported a positive association between number of mood episodes and length of illness in patients with BD-I, and prospective cardiovascular complications. Therefore, future investigation should consider the influence of comorbidities in illness progression in patients with BD.
Although previous meta-analyses have indicated that oxidative stress markers (Brown et al., 2014; Rowland et al., 2018) and BDNF levels (Fernandes et al., 2015; Frey et al., 2013; Munkholm et al., 2016) are altered in patients with BD, more research is required to determine whether alterations also undertake a progressive course.
Another important biomarker that was evaluated in the included studies was the length of telomeres, repetitive sequences at the end of chromosomes that shorten naturally during cellular division. It has been suggested that BD may accelerate the body’s natural aging process, and the role of telomeres has been investigated (Fries et al., 2020; Powell et al., 2018; Squassina et al., 2019). Previous studies have suggested that, whenever the body experiences chronic stress, telomere shortening may be induced and even accelerated, resulting in apoptosis and genome instability (Muneer and Minhas, 2019; Simon et al., 2006). This process has therefore been proposed to be a potential biomarker in BD, alongside other markers such as inflammation and oxidative stress (Barbé-Tuana et al., 2016). However, more studies evaluating telomere shortening in patients at different stages of the illness are needed to clarify the potential association of illness progression and telomere shortening.
Noteworthy, across the included studies in this systematic review, contradictory results were shown and there were a few biomarkers that were not investigated by more than one study. It is also important to mention the heterogeneity of patients with BD, the small sample sizes in a few of the included studies, and the use of medications, which may have potentially confounded the findings. In addition, the majority of the included studies had a cross-sectional design; in a recent report by Vieta and Angst (2021), longitudinal cohort studies were proposed to help clarify the biological underpinnings of BD and inform prevention through timely therapeutic intervention. Through the implementation of a global BD cohort, the authors suggest long-term studies that better investigate progressive changes in biomarkers associated with illness progression will be possible.
The present systematic review also highlights the need for more consistency in the use of staging models in BD research to standardize the results and understand the variability of biomarkers across different studies. In this context, an updated task force was recently published proposing a standardized nomenclature about the clinical stages of BD (Kupka et al., 2021). This effort would be crucial for the consistency in future studies and will assist in the further understanding of the biological basis of illness progression in BD.
Staging models can be useful clinical tools, and therefore have also been proposed as a measure of illness progression in other psychiatric disorders, including schizophrenia and major depressive disorder (MDD) (Agius et al., 2010; Fuente-Tomas et al., 2019; Meisenzahl et al., 2008; Moylan et al., 2013; Pantelis et al., 2005). Progressive changes in biomarkers have also been identified in schizophrenia and MDD, indicating that the progressive course observed in a subset of patients with BD may not be unique to this illness. Future research into biomarkers and progression is encouraged to translate staging models for psychiatric disorders into practice.
In conclusion, the findings of this systematic review support the notion of neuroprogression in patients with BD, particularly changes in brain structures such as the ventricles and GM. The present systematic review further solidifies the importance of investigating biomarkers and their role in illness progression to obtain consistent findings that could highlight potential treatment targets and inform future clinical care.
Supplemental Material
sj-docx-1-anp-10.1177_00048674221091731 – Supplemental material for Biomarkers of neuroprogression and late staging in bipolar disorder: A systematic review
Supplemental material, sj-docx-1-anp-10.1177_00048674221091731 for Biomarkers of neuroprogression and late staging in bipolar disorder: A systematic review by Sonya Grewal, Stuart McKinlay, Flávio Kapczinski, Bianca Pfaffenseller and Bianca Wollenhaupt-Aguiar in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
The authors acknowledge Cezar Farias for the contributions in designing the Figures of this manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: F.K. reports personal fees as a speaker/consultant from Janssen–Johnson & Johnson, as well as grants from INCT–CNPq and the Canada Foundation for Innovation, outside the submitted work. S.G., S.M., B.P. and B.W.A. declare no conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
