Abstract
Objective:
Concerns about intentional and unintentional poisoning present a barrier to wider use of clozapine in treatment-resistant schizophrenia. The objective of this study was to investigate decedent demographics and trends in fatal poisonings in Australia involving clozapine.
Methods:
This was a retrospective case series of all fatal drug toxicity reported to an Australian coroner between 1 May 2000 and 31 December 2016 where toxicological analysis detected clozapine. Cases were identified using the National Coronial Information System. Demographics extracted included age and gender of the decedent, year and location of death, cause and manner of death and drugs detected in post-mortem samples.
Results:
There were 278 poisoning deaths where clozapine was detected in toxicological analyses. Three-quarters of all cases (n = 207) involved men and the median age at death was 38.5 years (interquartile range: 16 years). Three-quarters of the deaths occurred in the home. Overall, 15.8% of deaths were deemed intentional, 57.5% unintentional and 24.5% of unknown intent. While the annual number of intentional self-poisonings remained constant with <5 per year, the overall number of fatalities increased due to an increase in unintentional poisonings. Multiple drug toxicity was reported in 55.0% of cases and clozapine alone in 45.0% of cases. The most common co-reported medications were antidepressants, benzodiazepines and opioids detected in 47.1%, 44.4% and 41.2% of multiple drug toxicities, respectively.
Conclusion:
This was the first Australia-wide review of all fatal drug poisonings reported to a coroner involving clozapine. Fatalities were most common in men and occurred at home. Multiple drug toxicity generally involved psychotropic, sedative or opioid analgesic medications. Despite increasing clozapine use, rates of intentional poisoning have remained constant and low. Developing a better knowledge of unintentional fatalities presents an opportunity to minimise harm.
Introduction
Up to one-third of individuals with schizophrenia will not respond to adequate trials of two first-line antipsychotics. For these individuals, clozapine is considered the most effective treatment, with up to 60% responding within 6 months (Lally and MacCabe, 2015). Clozapine significantly reduces suicidality and all-cause mortality compared to other antipsychotics (Meltzer et al., 2003; Taipale et al., 2020; Tiihonen et al., 2009). A nationwide cohort study in Finland between 1972 and 2014 reported clozapine reduced suicide (adjusted hazard ratio [aHR] = 0.21, 95% confidence interval [CI] = [0.15, 0.29]) and all-cause mortality (aHR = 0.39, 95% CI = [0.36, 0.43]) compared to non-use of antipsychotics (Taipale et al., 2020). Clozapine was superior to other antipsychotics, a result consistent with other similar studies (Tiihonen et al., 2009).
While clozapine use has increased over the last decade, it is still considered underutilised (Bachmann et al., 2017). Bachmann et al. (2017) examined the prevalence of clozapine use in 17 countries, and identified the highest rates in Finland and New Zealand (189.2/100,000 and 116.3/100,000, respectively) and lowest rates in Japan and the United States (privately insured population: 0.6/100,000 and 14/100,000, respectively). In Australia, Forrester et al. (2015) estimated that 33% of people with treatment-resistant schizophrenia living in Queensland were dispensed clozapine in 2013. Similarly, an Australian representative national survey of individuals with contact with mental health services in the previous 12 months (2010 Australian Survey of High Impact Psychosis) estimated one-third of individuals with chronic course schizophrenia were treated with clozapine, suggesting clozapine remains underutilised in Australia (Siskind et al., 2017).
Major barriers to use include concern regarding serious and potentially life-threatening adverse drug events (ADEs) and the requirement for routine blood monitoring (Cetin and Kose, 2016; Farooq et al., 2019). Neutropenia and agranulocytosis are reported to occur in 2.7% and 0.7% of individuals treated with clozapine respectively (Munro et al., 1999). However, a recent meta-analysis of controlled studies suggested clozapine is not associated with a higher risk of neutropenia compared to other individual antipsychotics and therefore challenged the need for routine monitoring (Myles et al., 2019). Another serious ADE is myocarditis which has been reported in over 5% of initiations in some Australian settings. However, the high rate may be explained by reporting bias, protocolised monitoring and higher overall awareness in Australia (Dawson et al., 2018; Nachmani Major et al., 2020; Siskind et al., 2020).
Clozapine has relatively high toxicity compared to other antipsychotics and is implicated in intentional and unintentional poisonings (Fountain et al., 2019; Handley et al., 2016; Pfeifer et al., 2020). Acute toxicity with clozapine can lead to central nervous system (CNS) depression resulting in coma and respiratory depression and may cause profound hypotension, tachycardia, cardiac arrest and seizure among other effects (Ciranni et al., 2009; Kramer et al., 2010). Advancing age and dose are factors associated with severity of toxicity (Kramer et al., 2010). A single dose of clozapine within the licenced range can be fatal in a clozapine-naïve individual (Stanworth et al., 2012), with doses as low as 100 mg being associated with moderate and severe toxicity (Kramer et al., 2010). A series of 12 suspected clozapine poisonings by overdose in the United Kingdom and Ireland between 1992 and 2003, of which 7 were fatal, identified that these patients were non-adherent or had taken clozapine prescribed for another patient (Flanagan et al., 2005). The investigators identified appreciable overlap in post-mortem blood plasma levels in those who died from clozapine self-poisoning and a further 54 deaths in individuals prescribed clozapine where clozapine self-poisoning was not suspected, suggesting a finding of clozapine self-poisoning cannot be made based on post-mortem blood levels alone.
A study of 1544 antipsychotic-related fatal poisonings in England and Wales between 1993 and 2013 identified 193 clozapine-related fatalities (Handley et al., 2016). Antipsychotic-related fatalities increased from 1.0 per million to 1.5 per million of the population over this period. There were 0–7 clozapine-related deaths annually between 1993 and 2002, increasing to 10–19 per year between 2003 and 2013 (Handley et al., 2016). In this study, increases in antipsychotic-related deaths reported from 2001 onwards were largely attributable to unintentional deaths associated with clozapine or concurrent opiate exposure. Pharmaceutical substances, including clozapine, accounted for 52% of the 1402 poisoning-related deaths recorded in the New Zealand cohort from the National Coronial Information System (NCIS) database from 2008 to 2013 (Fountain et al., 2019). Clozapine was the equal sixth most common pharmaceutical substance involved in deaths and accounted for the greatest proportion of antipsychotic-related deaths. This is despite restrictions on clozapine prescribing and relatively low utilisation compared to other antipsychotics. A 16-year study of relative toxicities of pharmaceutical substances in the greater Newcastle Hunter Area in New South Wales identified that fatal toxicity index and case fatalities for antipsychotics as a class were 7.9 (5.5–10.4) deaths/defined daily dose (DDD)/100 years and 1.72% (1.2–2.24), while clozapine had the highest toxicity estimates of all individual antipsychotics (50.10 deaths/DDD/100 years and 15.51%) (Brett et al., 2019). This is consistent with other studies comparing toxicity of individual antipsychotics in overdose (Ferrey et al., 2018; Pfeifer et al., 2020).
To date, no studies have investigated demographics of clozapine poisonings reported to a coroner outside of the United Kingdom and New Zealand (Flanagan et al., 2005; Fountain et al., 2019; Handley et al., 2016). Given increasing clozapine use in Australia and new reports of unintentional deaths (Dawson et al., 2018; Forrester et al., 2015; Nachmani Major et al., 2020; Ronaldson et al., 2011a, 2011b), a review of Australian clozapine-related poisoning deaths was warranted. Knowledge of the causes may provide information to improve safe use of clozapine, particularly as concerns about safety present a potential barrier to use. The objective of this study was to investigate decedent demographics and trends in fatal poisonings involving clozapine.
Methods
Study design and context
This was a retrospective case series of all fatal poisonings reported to an Australian coroner between 1 May 2000 and 31 December 2016 where clozapine was detected by toxicological analysis. Data were extracted from the NCIS database that contains records of all deaths investigated by Australian and New Zealand coroners since 2000. In Australia, a coronial investigation is conducted for all reportable deaths to identify a decedent, the medical cause and manner of death and to make recommendations to prevent avoidable deaths. What constitutes a reportable death varies between Australian jurisdictions; however, these are generally unexpected, violent or unnatural deaths such as those related to accidents or suicides, or deaths which have occurred suddenly or unexpectedly while the decedent was receiving care or held in custody (NCIS, 2020). Poisoning deaths are considered reportable because these are unnatural.
NCIS data
The NCIS database contains cases from May 2000. Data are coded by staff from each jurisdiction from the coronial brief produced during the investigation conducted by the coroner. Data include demographic information, date of death, mechanism of death, intent of death and non-coded reports including coronial finding, autopsy, toxicology and police reports. The NCIS have coding manuals and a quality assurance review process to maintain reliability.
Intent
The intent behind a death is coded from findings of the coroner at the conclusion of the investigation. Intentional self-harm is classified when the deceased intentionally self-inflicted harm regardless of the anticipated outcome. Other intent types include unintentional, assault or unknown intent.
Involvement of a drug in death
The involvement of a drug in a death is determined by a forensic pathologist by analysis of post-mortem blood samples and other contextual information. Establishing the involvement of drugs is challenging due to post-mortem redistribution whereby a drug diffuses from tissues and alters post-mortem concentration in samples (Drummer and Gerostamoulos, 2002). This process is more pronounced with lipophilic drugs such as clozapine that have a high volume of distribution (Saar et al., 2012). This means there is no standard concentration of clozapine that can be used to assign cause of death (COD). In the NCIS dataset, the presence of a drug in a post-mortem sample does not indicate whether it was obtained by prescription, illicitly or otherwise obtained, or the relative contribution that the drug made in the death.
Medical COD
Medical COD is coded based on the forensic pathologist’s report. If more than one drug is deemed to have contributed to the death, the term multiple drug toxicity is often used to describe the COD. In some cases, the specific drugs thought to have caused the death are stated while in other cases this is unspecified. Often it is not possible to determine the relative contributions of different drugs to each specific death.
Procedures for case inclusion
The NCIS database was examined for closed Australian cases of clozapine poisonings from May 2000 to 31 December 2016 at the time of data collection in April 2019. A search for ‘clozapine’ as a medical COD was used. To detect any additional cases, a search using the mechanism of death with subcategory of ‘poisoning by chemical or substance’ and selecting ‘clozapine’ as the substance was run. A key word search for ‘clozapine’ or the brand names ‘Clopine’ or ‘Clozaril’ was conducted through police reports and coroners’ findings function. The results from each search were combined and duplicate cases were removed.
Data synthesis and extraction
All cases where the primary mechanism for death included exposure to chemical or other substances were manually reviewed by a member of the research team (J.L.D.). Cases without coding for mechanism for death were also manually reviewed to identify any coded with a COD of clozapine toxicity. Of the resultant reports, cases were then removed if there was clear indication of a non-pharmaceutical drug-related fatality listed under the medical COD (e.g. carbon monoxide poisoning or hanging) or clozapine was not coded in the list of parent drugs detected in the case or where the medical COD was related to chronic use of clozapine (e.g. obesity). Extracted data included age and gender of the deceased, year and location of death (state and whether at home or elsewhere), intent of the death at completion of the coronial investigation, other medications detected and documented medical COD.
Data analysis
Descriptive statistics were used to describe the characteristics of the dataset. Intent was categorised as intentional, unintentional or unknown. Unknown included intent coded as ‘undetermined’, ‘unlikely to be known’ or where the coded field was blank. Where the medical COD was clozapine toxicity, it was considered ‘single drug toxicity’, regardless of other drugs detected. Where the term ‘multiple’ or ‘combined’ drug toxicity was the medical COD, all drugs or alcohol detected in post-mortem sample were considered involved in the toxicity.
Ethics
Ethics approval was granted by the Department of Justice Human Research Ethics Committee (CF/17/23257) and the Victorian Institute of Forensic Medicine Research Advisory Committee (RAC 020-17).
Results
There were 278 cases where the primary mechanism of death was poisoning by a chemical or substance, and where clozapine was detected in the toxicological analysis (Table 1). Three-quarters of all cases (n = 207) were men and the median age at death was 38.5 years (interquartile range: 16 years). Three-quarters of these deaths occurred in the home. There was a peak in poisoning deaths between 30 and 39 years of age. There were no deaths reported among children or adolescents below the age of 18 years. The annual average number of poisoning deaths reported to a coroner involving clozapine was 16.35 (standard deviation [SD] = 5.39).
Demographics of poisonings reported to an Australian coroner where clozapine was detected.
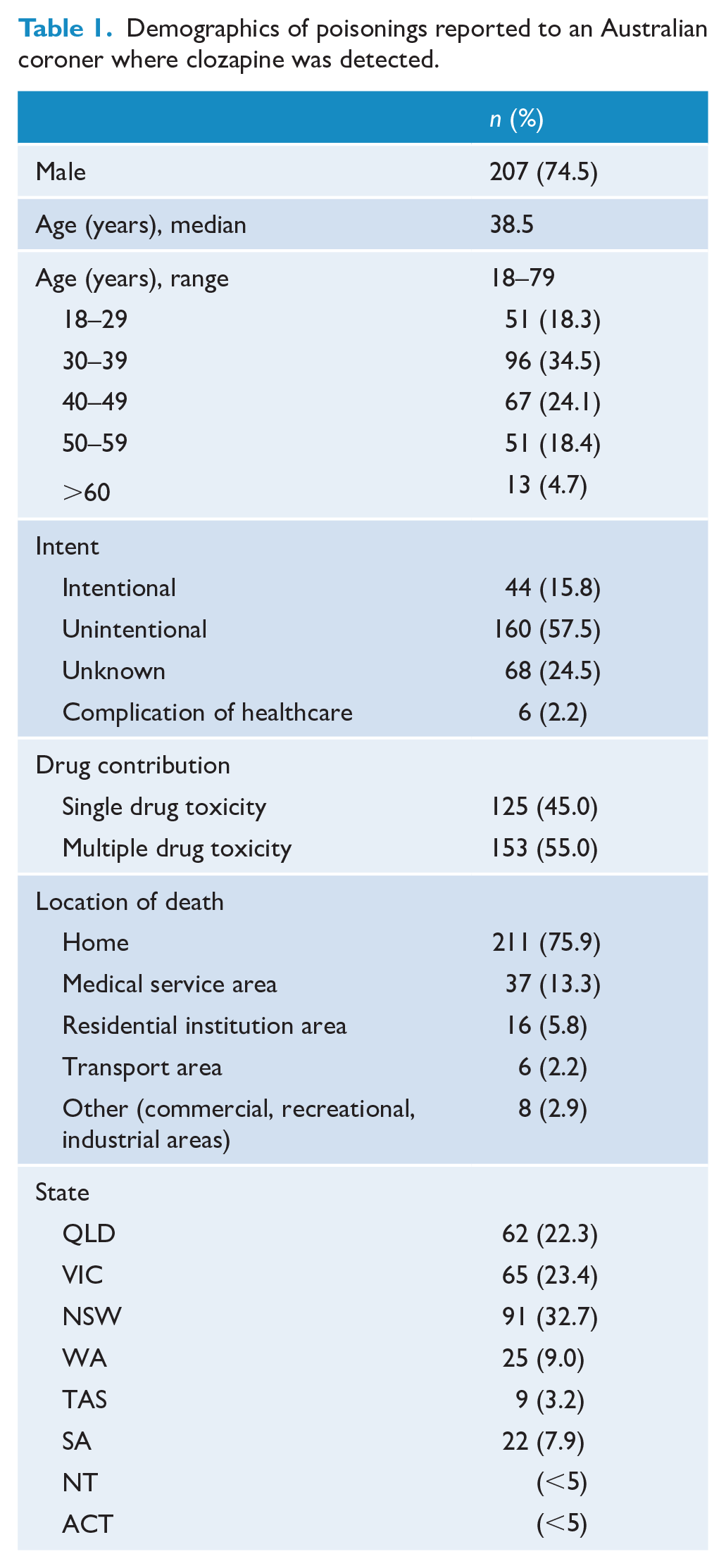
Figure 1 shows the number of poisoning deaths investigated by the coroner by year where clozapine has been detected. While the annual number of deaths has increased over the 16-year period, the number of intentional deaths has remained consistent and low with an average of 2.6 per year. The increase is primarily related to the number of unintentional deaths.
Drug poisonings reported to an Australian coroner where clozapine was detected, by year and intent at completion of investigation.
At conclusion of the coronial review, 44 deaths (15.8%) were determined to be intentional, while 160 (57.5%) were determined to be unintentional, and 68 (24.5%) were listed as unknown. In six cases the intent was determined to be a complication of medical care. The median age was similar for intentional and unintentional cases (37.5 vs 39.0 years); however, there were more multiple drug toxicities with unintentional compared to intentional cases (65.6.2% vs 52.3%, respectively). Males accounted for 65.9% of intentional deaths and 74.4% of unintentional deaths.
In 125 cases (45.0%), clozapine was reported as the medical COD, while multiple drug toxicity was reported in 153 cases (55.0%). The median age was similar between the groups, 38 and 40 years, respectively. Of cases where multiple drug toxicity was reported, these were generally other psychotropic, sedative or analgesic medications (Table 2). Alcohol was detected in 39 cases (25.5%), and marijuana, amphetamines or heroin were present in 25 cases (20.0%) of mixed drug toxicities. The median age of those with one or more known illicit drug detected was 35 years. Antidepressants were detected 75 times in 72 decedents, and were involved in 47.1% of multiple drug toxicity cases. The most common class of antidepressants involved in poisonings was selective serotonin reuptake inhibitors (SSRIs) with 50 reports. Citalopram was the most common co-detected SSRI, present in 30 cases (19.6% of multiple drug toxicity cases). Opioids were present in 63 decedents, contributing in 41.2% of the multiple drug toxicities. Codeine was the most detected opioid, followed by methadone. Morphine and metabolites were detected in 14.4% of mixed drug toxicities; however, it was likely detected as a metabolite of codeine and heroin in most cases. Multiple antipsychotics were present in 57 decedents, detected in 37.3% of mixed drug toxicities.
Co-administered substances implicated in cases of multiple drug toxicity with clozapine.
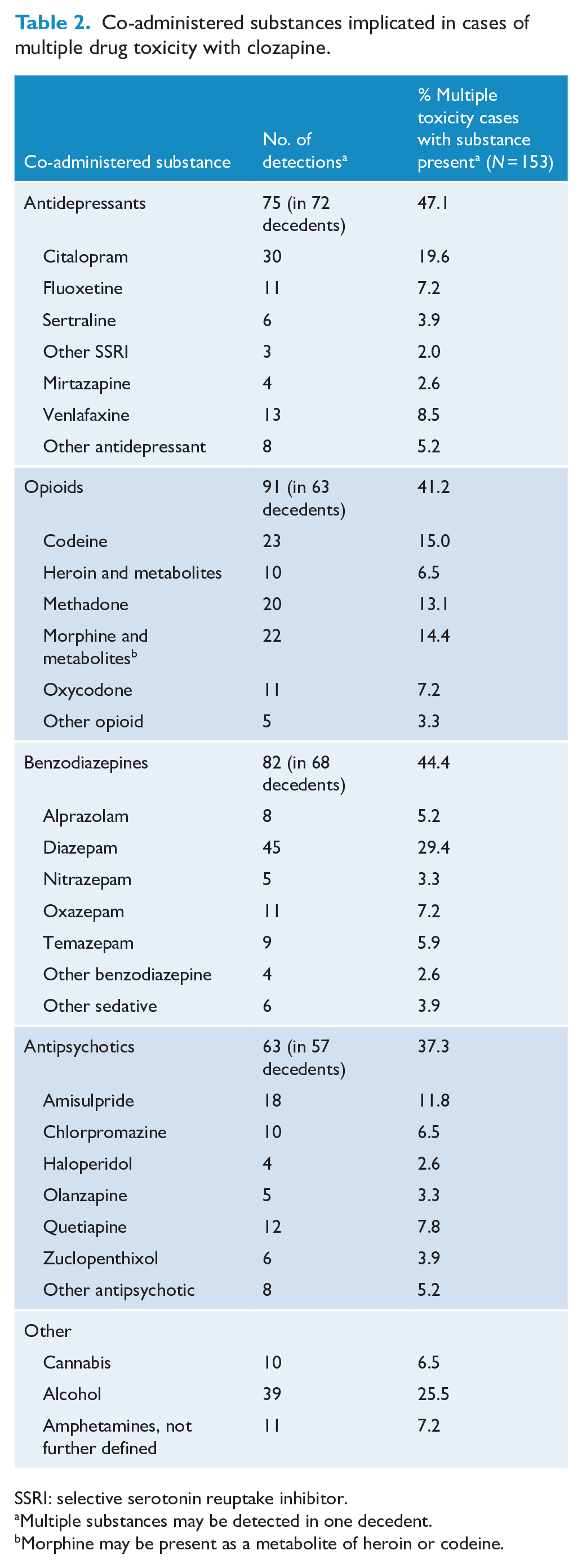
SSRI: selective serotonin reuptake inhibitor.
Multiple substances may be detected in one decedent.
Morphine may be present as a metabolite of heroin or codeine.
There were 24 cases which reported COD as drug toxicity along with infection. Of these infections, the majority were respiratory with 18 cases with bronchopneumonia, 4 cases of aspiration pneumonia, 1 reported of chronic bronchitis and 1 reported as sepsis.
Discussion
This 16-year national review of Australian coronial investigations identified 278 cases of fatal poisoning involving clozapine, corresponding to an average of 16 deaths per year. The median age of decedents was 38.5 years, three-quarters of deaths were among men and three-quarters occurred in the home. Multiple drug toxicity was present in 55% of cases. Psychotropic, sedative or analgesic medications were most frequently present in multiple drug toxicity.
Demographics of decedents in our study broadly matched those who are most likely to be prescribed clozapine. Bachmann et al. (2017) reported clozapine prevalence was highest in the 40–59 years age group in the 17 countries studied, with a male-to-female ratio ranging from 1.3 to 4.2 (median ratio: 1.5). Decedents in our study were younger with the largest represented age group of clozapine poisoning cases aged 30–39 years. A possible explanation may be high rates of illicit drug use among people aged in their 20s and 30s (Australian Institute of Health and Welfare (AIHW), 2017). The median age for a mixed drug toxicity death with the presence of a known illicit agent (marijuana cannabis, heroin and metabolites, and amphetamines) was 35 years which was lower than the median age of decedents overall. However, it was not possible to determine whether a prescription medication detected in toxicological analyses was obtained illicitly. Siskind et al. (2017) reported that the mean age of clozapine users in Australian mental health services was 37.4 years (SD = 9.51) and 73% were male. This is consistent with the median age of the decedents in our study. However, Siskind used data from clients of mental health services aged from 18 to 64 years.
Brett et al. (2019) investigated all drug-poisoning deaths occurring in the Greater Newcastle Hunter Area over a 16-year period and identified similar decedent demographics to our clozapine study, with decedents primarily male (62.8%), with a median age of 42 years and the majority of deaths occurring in the home (74.8%). Fountain et al. (2019) broadly investigated poisoning deaths by drugs and substances in New Zealand reported to a coroner over a 6-year period. There was similarly a higher proportion of males (63.5%), with a median decedent age of 46 years, and 67.4% occurring in the home (Fountain et al., 2019).
We identified that the overall number of poisoning deaths involving clozapine per year increased from the year 2000 and peaked in 2013. This increase was mostly attributable to an increase in cases with unintentional intent. Data on clozapine use are only available for the Australian state of Queensland. The rate of clozapine prescribing increased in Queensland from 44 to 60 per 100,000 people between 2004 and 2013, a 36% relative increase over the study period (Forrester et al., 2015). During the same period, we observed a two-fold increase in fatal poisonings relating to clozapine nationally, the majority of which were unintentional or of unknown intent. Overall, 15% of fatalities were the result of intentional-self harm and 58.9% were considered unintentional. The absolute annual number of intentional poisonings remained low (⩽5 cases per year) throughout the study period. This is reassuring given the increase in use of clozapine over the same period (Forrester et al., 2015). The rate of intentional deaths in this study was much lower than that identified in the New Zealand cohort of all substance related poisonings, where 50.6% were considered intentional, 39.2% were unintentional and 10.3% were unknown (Fountain et al., 2019). Handley et al. (2016) reviewed all antipsychotic-related fatal poisonings in England and Wales over a 20-year period and identified 36% to be unintentional and 29% to be intentional. However, this pattern in their study was substantially different for clozapine-related poisonings where intentional poisoning was reported in 17% of cases, and unintentional in 51% of clozapine poisoning fatalities. Our current study was markedly similar in rates of intentional and unintentional fatalities where clozapine was detected. It is likely that relative frequency of unintentional fatalities were partly attributable to the propensity of clozapine to cause CNS depression and cardiac toxicity.
In our study, decedent characteristics were similar for both intentional and unintentional poisonings; however, there was a higher proportion of toxicity from clozapine alone compared to mixed drug toxicity in the intentional group. In Australia, the median age of suicide in 2018 was 44.3 years for males and 44.7 years for females; males accounted for 76.2% of these cases (Australian Bureau of Statistics (ABS), 2019b). This study had a lower median age for intentional poisoning compared to overall decedents at 37.5 years and males accounted a relatively smaller proportion (65.9%). This contrasts to the median age of substance-related poisonings of 46 years in New Zealand (Fountain et al., 2019). This difference may reflect the relatively younger demographic that is prescribed clozapine as well as differences in suicide demographics in those with schizophrenia (Bachmann et al., 2017).
Mixed drug toxicities involving clozapine were most common with antidepressants, benzodiazepines, opioids and other antipsychotics in this study. This is consistent with the study by Fountain et al. (2019) who demonstrated opioids, antidepressants, antipsychotics and hypnosedatives were the most common medication classes contributing to poisonings in New Zealand. In our study, opioids and benzodiazepines were involved in 41.2% and 44.4% of multiple drug toxicity cases, respectively. Of opioid cases, codeine was detected as the parent drug most frequently, followed by methadone. In Australia, in 2018, almost two-thirds of drug related deaths involved opioids, more frequently detected than in this study (ABS, 2019a). Benzodiazepines, primarily diazepam, were present in 44.4% of multiple drug toxicity cases, and 24.5% of all cases. A cross-sectional study of all clozapine-treated individuals across three hospital clinics in New South Wales explored co-prescribed medications (Lappin et al., 2018). Lappin reported 5.9% of these individuals were co-prescribed benzodiazepines which is lower compared to their presence in decedents in this current study. The risk of fatal toxicity from clozapine may be greater in those co-prescribed opioids and benzodiazepines due to enhanced risk of CNS depression. Clinicians should regularly review the need for co-prescription of these medications with clozapine. Particularly, given unintentional poisonings were more frequent in the multiple drug toxicity group compared to clozapine alone (65.6.2% vs 52.3%, respectively).
Citalopram was detected in 30 cases. A case report suggested clozapine levels can be increased by citalopram (Borba and Henderson, 2000); however, to our knowledge, this finding has not been reproduced (Avenoso et al., 1998). Citalopram may be more likely to cause corrected QT (QTc) prolongation relative to other SSRIs (Beach et al., 2014; Isbister et al., 2004). In New Zealand, citalopram was the sixth most common substance identified in poisonings with multiple drug toxicities, and the second most common antidepressant after amitriptyline (Fountain et al., 2019). Acute clozapine toxicity may cause QTc prolongation (Kramer et al., 2010), and concomitant use of QTc prolonging drugs as well as drug overdose can cause additive toxicity and is a risk factor for fatal cardiac arrhythmia such as torsade de pointes (Haddad and Anderson, 2002). Furthermore, the most frequently co-ingested antipsychotic was amisulpride, present in 11.8% of multiple drug toxicities. Amisulpride has a relatively high potential to cause QTc prolongation in monotherapy and with other agents (Huhn et al., 2019). In our study, there were 12 cases involving fluoxetine and several reporting fluvoxamine. Fluoxetine is a strong inhibitor of CYP2D6, and fluvoxamine is a strong inhibitor of CYP1A2, which are both involved in clearance of clozapine. Fluoxetine increases clozapine levels by 30–75%, while fluvoxamine has been reported to increase levels by several fold (Preston, 2020) leading to toxicity. There were very few cases of mixed drug toxicities involving tricyclic antidepressants (TCAs) despite these having greater toxicity in overdose than SSRIs (Ojanpera et al., 2016; Pfeifer et al., 2020). This finding of high rates of SSRI prescribing may be due to awareness of the elevated chronic risk of suicide in individuals with schizophrenia compared to the general population, and the relatively safe profile of SSRIs compared to other antidepressants (Buckley and McManus, 2002; Hawton et al., 2010).
The 24 cases with infection co-reported as COD almost entirely involved respiratory infections. This finding is consistent with the increasing recognition of a connection between infection and inflammation, and the presence of clozapine toxicity. A recent systematic review identified 40 cases of clozapine toxicity during infection (Clark et al., 2018). Most infections in their review were respiratory (57.5%) and urinary (32.5%) with a mean clozapine level during infection of 1811 mcg/L (ranging from 744 to 4740 mcg/L). These levels are above the consensus therapeutic reference range for clozapine is 350–600 mcg/L indicating the individuals are at risk of toxicity (Hiemke et al., 2018).
Most of the poisoning deaths in this study were considered unintentional which may be due to the relatively high potential for toxicity and misadventure with clozapine compared to other antipsychotics. Handley et al. (2016) found that there were more fatalities involving clozapine alone or in combination with alcohol compared to any other antipsychotic indicating the potential of clozapine for toxicity. In New Zealand, clozapine accounted for the greatest proportion of antipsychotic-related deaths, despite restrictions and relatively low utilisation compared to other antipsychotics (Fountain et al., 2019). Clinician should be aware of the risks of concurrent infection and inflammation leading to clozapine toxicity, particularly pneumonia, as this may warrant earlier monitoring for signs of clozapine toxicity and interventions.
Strengths and limitations
NCIS data are well suited for investigating drug-related poisoning because poisoning deaths are considered unnatural and are generally investigated by an Australian coroner. To ensure as complete as possible data, we did not extend the study period beyond 2016 so that cases were closed. The case closure rate inclusive of 2016 was 87.6%. This study also has limitations related to the variability and completeness of data recorded in the NCIS database. There was variation in the level of detail recorded across the data fields making it difficult to determine the context of some poisoning deaths. The medical COD was not coded based on standardised terminologies or standardised blood concentrations. The pathology and police reports were not accessed to obtain possible further context to fatalities. Clozapine levels were not extracted as levels alone cannot determine role in a death (Flanagan et al., 2005). There was variation in whether ‘mixed drug toxicity’ was reported with specific culprit medications or not. We used the COD defined by the pathologist as indicator of the contribution of clozapine to the death, rather than trying to discern further based on examination of the toxicology results, and where mixed drug toxicity was reported we reported all co-prescribed medication as possible contributing medications. There is no consensus on how this is managed between studies. Fountain et al. (2019) assigned causality to a single drug deemed most likely to have caused the death based on review of the toxicology and coronial reports. Brett et al. (2019) treated each medication in a multiple drug toxicity as having contributed to death equally. This study likely underestimates clozapine-related deaths as those considered as having occurred naturally where clozapine was detected are not captured.
Conclusion
This study was the first Australia-wide review of all fatal drug poisonings involving clozapine reported to a coroner. These fatalities most often occurred in men and in the home. Multiple drug toxicity generally involved psychotropic, sedative or opioid analgesic medications. Clinicians should regularly review the need for co-prescribing these medications with clozapine due to additive risk for CNS depression or cardiac events. Although use of clozapine is increasing, rates of intentional poisoning have remained low. The increasing number of deaths involving clozapine is linked to an increase in unintentional poisonings. Further analysis of circumstances surrounding unintentional fatalities involving clozapine will present an opportunity to minimise harm.
Footnotes
Acknowledgements
The authors would like to acknowledge the National Coronial Information System for providing access to coronial data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
