Abstract
Objective:
Olfactory deficits have been reported in bipolar disorder, but this finding is controversial. This study investigated whether olfactory deficit can serve as a specific marker for bipolar disorder by comparing olfactory function in different mood episodes of bipolar disorder. We also compared olfactory function in bipolar disorder and other mental disorders – namely, major depressive disorder and schizophrenia.
Methods:
The study consisted of two experiments. Experiment 1 enrolled 175 bipolar disorder patients (70 depressed subgroup, 70 manic subgroup and 35 euthymic subgroup) and 47 controls. Experiment 2 enrolled the participants from Experiment 1, along with 85 major depressive disorder and 90 schizophrenia patients. The Sniffin’ Sticks test was used to evaluate odour identification ability and odour threshold (as a measure of odour sensitivity). The Hamilton Depression Rating Scale and Young Mania Rating Scale were used to assess depressive symptoms in all subjects and manic symptoms in bipolar disorder patients, respectively. We also used the Positive and Negative Syndrome Scale to assess clinical symptoms in schizophrenia patients.
Results:
All three bipolar disorder patient subgroups (depressed, manic and euthymic subgroup) showed reduced odour identification ability compared to controls; however, only patients in the acute phase of a mood episode (depressed, and manic subgroup) showed impaired odour sensitivity. Clinical symptoms were negatively correlated with odour sensitivity but not odour identification ability. Bipolar disorder and major depressive disorder patients showed less odour identification and sensitivity impairment than schizophrenia patients.
Conclusion:
Odour sensitivity is a potential dopaminergic marker for distinguishing between bipolar disorder patients in acute phase vs remission, while odour identification is a trait but a nonspecific marker of bipolar disorder.
Introduction
Bipolar disorder (BD) is an affective disorder characterised by recurrent emotional dysregulation that includes manic and depressive episodes (acute phrase) and periods of remission (American Psychological Association, 2015). According to Diagnostic and Statistical Manual of Mental Disorders, 5th Edition (DSM-5), patients in manic episode would show abnormally and persistently elevated, expansive or irritable mood disturbance and increased energy and goal-directed activity (either socially, at work or school, or sexually) for at least 1 week. Depressed episode means a period for at least 2 weeks that patients would experience depressed mood, loss of interest or pleasure, significant weight loss, insomnia or hypersomnia, suicidal ideation or even suicide attempt. All those mood disturbances have to cause marked social or occupational functioning impairment or make hospitalisation necessary to prevent injury (American Psychological Association, 2015).
Imaging studies have shown that BD patients exhibit functional and anatomic changes in the prefrontal cortex, temporal lobe, limbic regions (amygdala, hippocampus and striatum) and insula (Keener and Phillips, 2007; Kempton et al., 2008; Lu et al., 2019; McDonald et al., 2004; Zhang et al., 2020), which are also involved in olfactory information processing (Atanasova et al., 2008; Cohen et al., 2012; Farbman, 1991; Fjaeldstad et al., 2017; Oral et al., 2013). Dopaminergic dysfunction has been suggested as a critical role in BD (Ashok et al., 2017; Cousins et al., 2009). Ashok et al. (2017) indicated the unbalance of dopamine receptor and transporter in BD patients might partially be the cause of this disorder. Recent studies have revealed the association between striatal dopamine transporter and olfactory function, especially odour identification (OI) ability in patients with Parkinson’s disease and healthy controls (Liu et al., 2020; Pak et al., 2018). These findings combined together may reflect underlying dopaminergic olfactory mechanism in BD. A few studies have investigated olfactory function in BD; however, the findings are contradictory, with some reports showing significant deficits in OI ability in BD patients (Cumming et al., 2011; Kamath et al., 2018; Lahera et al., 2016) and others demonstrating a normal ability (Hardy et al., 2012; McLean et al., 2004; Swiecicki et al., 2009). Most studies have found comparable odour sensitivity (OS) between BD patients and controls (Hardy et al., 2012; Kamath et al., 2018; Swiecicki et al., 2009), but one found abnormalities in patients with BD (Krüger et al., 2006).
There are many possible reasons for these conflicting findings such as ambiguous mood episodes in BD and small sample sizes. Most studies on olfactory function in BD patients did not distinguish between subtypes or mood episodes (Cumming et al., 2011; Hardy et al., 2012; Lahera et al., 2016; McLean et al., 2004); only one study categorised patients into BD I and II subtypes (Kamath et al., 2018) and two classified patients based on clinical mood episodes, focusing on those in the euthymia phase (Krüger et al., 2006; Lahera et al., 2016). Lahera et al. (2016) found impaired OI ability in euthymic BD patients compared with controls. Kamath et al. (2018) found OI deficits widely existed in patients with BD and major depressive disorder (MDD), but significantly reduced OI ability was only showed in BD I patients with psychotic features, while no difference was found among BD patients in two subgroups (BD I and II). Emotional symptoms and olfactory function are closely related (Hardy et al., 2012). BD patients in the manic or depressive phase show clear behavioural changes, implying that olfactory deficits in BD patients vary according to the mood episode. In addition, with the exception of one study (BD I: n = 43; BD II: n = 48) (Kamath et al., 2018), the BD patient cohort in most investigations was relatively small, ranging from 12 to 39 patients (Cumming et al., 2011; Hardy et al., 2012; Krüger et al., 2006; McLean et al., 2004; Striebel et al., 1999; Swiecicki et al., 2009).
Olfactory deficits have been reported in mental disorders other than BD such as schizophrenia (SCZ) (Cohen et al., 2012; Jiang et al., 2018; Li et al., 2020; Moberg et al., 2014; Zou et al., 2015, 2018) and MDD (Chen et al., 2018a, 2018b; Croy and Hummel, 2017; Taalman et al., 2017; Zucco and Bollini, 2011), suggesting that olfactory deficit is a potential marker for SCZ spectrum disorders and MDD. It has been proposed that mental disorders are a continuum from unipolar depression to BD to typical SCZ (Crow, 1986). To date, studies found that MDD patients showed worse performance in OS (Chen et al., 2019), whereas OI was more severely impaired in patients with SCZ than in those with BD (Cumming et al., 2011). On the other hand, no differences in OI and OS were found between patients with unipolar and bipolar depression (Swiecicki et al., 2009). It was also demonstrated that among BD patients, only those with psychotic features showed poor olfactory performance relative to MDD patients (Kamath et al., 2018). However, there have been no direct comparisons among these three patient populations, and it remains unclear whether olfactory deficits differ between BD, MDD and SCZ.
We addressed the above question in this study, which consisted of two experiments with relatively large sample sizes. In Experiment 1, we investigated whether olfactory function can serve as a marker for BD by comparing OS and OI ability between patients experiencing different clinical mood episodes (manic, depressed and euthymic) and healthy control subjects. We hypothesised that OS and OI ability deficits would exist in BD patient groups instead of controls, with relatively mildest impairment in euthymic BD patients compared with manic and depressed BD patients. In Experiment 2, we examined whether the olfactory deficits observed in Experiment 1 are specific to BD or are common to other mental disorders (i.e. MDD and SCZ). We hypothesised that the OS and OI ability deficit would present as a continuum where SCZ and MDD patients would have the lowest and highest olfactory test scores, respectively, while BD patients would show an intermediate performance.
Experiment 1
Methods
Participants
A total of 175 BD patients were recruited from Zhujiang Hospital, Guangdong Provincial People’s Hospital, Guangdong 999 Brain Hospital and Wu Zhongpei Memorial Hospital in Guangdong, China. All patients were diagnosed by trained psychiatrists according to the DSM-5. Clinical symptoms were evaluated with the Hamilton Depression Rating Scale (HAMD) and Young Mania Rating Scale (YMRS). Patients who scored ⩽7 on both scales were included in the euthymic group; those who fulfilled the DSM-5 diagnostic criteria for mood episodes were considered depressed or manic. Among the BD patients, 70 were depressed inpatients, 70 were manic inpatients and 35 were euthymic outpatients (for sex distribution and age of each group, see Table 1). All patients were free of upper respiratory tract infections, nasal diseases, traumatic head injuries, other serious illness and drug abuse or alcohol addiction, and all had an IQ ⩾ 80; those in the control group – which consisted of 47 healthy subjects (mean age: 33.11 ± 10.60 years) – also had no family history of psychosis.
Sociodemographic and clinical characteristics in Experiment 1.
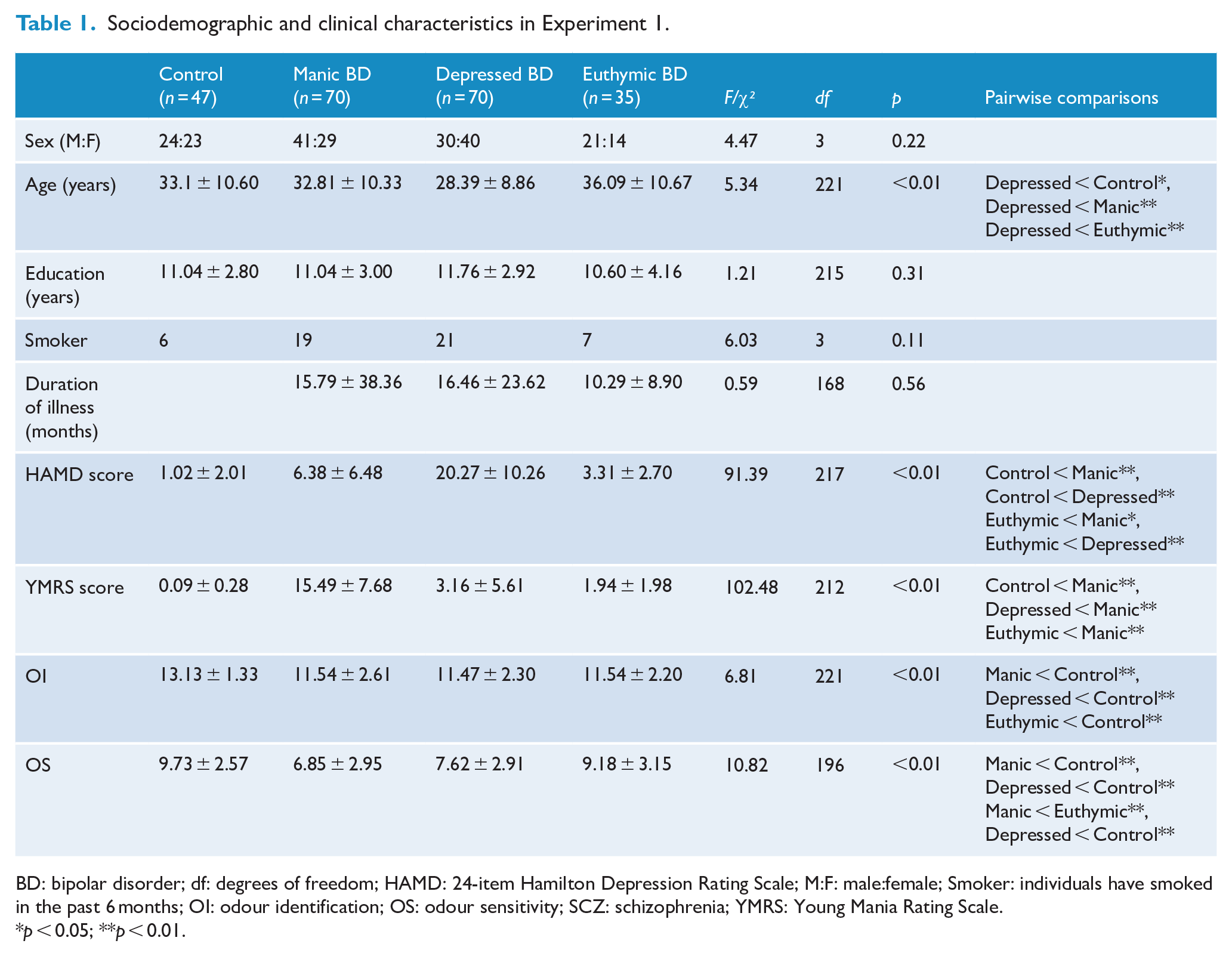
BD: bipolar disorder; df: degrees of freedom; HAMD: 24-item Hamilton Depression Rating Scale; M:F: male:female; Smoker: individuals have smoked in the past 6 months; OI: odour identification; OS: odour sensitivity; SCZ: schizophrenia; YMRS: Young Mania Rating Scale.
p < 0.05; **p < 0.01.
The study was approved by the Ethics Committee of Southern Medical University in Guangzhou, Guangdong, China. Written informed consent was obtained from all participants after they had been provided with a complete description of the study.
Measures
Sniffin’ Sticks test
The Sniffin’ Sticks test (SST) (Hummel et al., 1997), which is used to evaluate olfactory function, comprises three subtests: OI, odour threshold (OT) and discrimination. In this study, the OI test was used to identify odours and the OT test was used as a measure of OS. In the OI test, subjects were given felt-tip pens (length: 14 cm; diameter: 1.3 cm) containing a liquid odourant diluted with propylene glycol. The following 16 familiar odours were presented to each subject: orange, leather, chocolate, mint, banana, lemon, liquorice, ginger, garlic, coffee, apple, soy sauce, pineapple, rose, anise and fish. The experimenter opened each pen in turn and placed it ~2 cm below the nostrils of the subject for about 3 s, and the subject was required to select one of four possible answers to identify the odour of the pen. Each correct response was scored as 1, with total score ranging from 0 to 16 and higher scores indicating better OI ability.
To determine OS, 16 different dilutions of phenyl ethyl alcohol solution were used as the target odourant. In each trial, the experimenter presented three pens to the subject: one containing the target odourant and two containing odourless solutions. The subject was required to identify the pen containing the target odourant. OT was calculated using a single-staircase method that was reversed when the subject correctly identified the target odour pen 2 consecutive times or failed to detected the target odour once. The final score was calculated as the average of the last four rounds in seven reversals, and ranged from 1 (lowest sensitivity) to 16 (highest sensitivity).
HAMD
The HAMD rates depressive symptoms based on 24 items (Hamilton, 1960). Total scores range from 0 to 76, with a score between 20 and 35 indicating mild-to-moderate depression and a score ⩾35 indicating severe depression.
YMRS
The YMRS, which is used to assess symptoms of mania in BD (Young et al., 1978), consists of 11 items; 7 are scored on a 4-point severity scale and the other 4 are assigned a score ranging from 0 to 8.
Procedure
All tests were conducted by two well-trained psychology staff (one licenced doctoral-level psychiatrist and one master’s-level psychology student). The demographic data of participants were collected. OS test was conducted in a well-ventilated and uninterrupted room following steps introduced above. Fifteen minutes were provided as a break to reduce the confounding factors between two olfactory tests and clinical assessments (e.g. HAMD, YMRS) were conducted during this period of time. Finally, OI ability was tested. The whole test lasted for about an hour and all patients completed all test at once in the same day. The manic and depressed BD patients were all inpatients and all tests needed to be completed during hospitalisation. Patients in euthymic group were outpatients who completed all tests in the follow-up visit day.
Data analysis
Data are presented as mean ± standard deviation. Statistical analyses were performed using SPSS v22.0 software (SPSS Inc, Chicago, IL, USA) with the significance level set at p < 0.05. The chi-square test and one-way analysis of variance (ANOVA) were used to evaluate differences in sociodemographic and clinical characteristics and olfactory test results (OI and OS) between groups. The least significant difference post hoc test was used for multiple comparisons. Pearson’s correlation coefficient was used to analyse the relationship between olfactory performance and subject characteristics and partial correlations were performed to reanalyse those relationship after controlling for age.
Results and discussion
Table 1 summarised the demographic and clinical information of BD patients and control subjects. There were no differences between the four groups in terms of education level, depressed BD patients, n = 65; manic BD patients, n = 68; euthymic patients, n = 35; controls, n = 47; F(3, 212) = 1.21, p = 0.31; and duration of illness, depressed BD patients, n = 68; manic BD patients, n = 66; euthymic patients, n = 35; F(2, 166) = 0.59, p = 0.56. However, depressed BD patients were younger than patients in the other groups, F(3, 218) = 5.34, p < 0.01, and had higher HAMD scores, depressed BD patients, n = 67; manic BD patients, n = 69; euthymic BD patients, n = 35; controls, n = 47; F(3, 214) = 91.39, p < 0.01. Meanwhile, manic BD patients had higher YMRS scores than the other groups, depressed BD patients, n = 62; manic BD patients, n = 69; euthymic BD patients, n = 35; controls, n = 47; F(3, 209) = 102.48, p < 0.01.
There were significant differences between groups in OI, F(3, 218) = 6.81, p < 0.01; Figure 1(A), and OS, depressed BD patients, n = 57; manic BD patients, n = 60; euthymic BD patients, n = 33; controls, n = 47; F(3, 193) = 10.82, p < 0.01; Figure 1(B). Depressed and manic BD patients had lower OI and OS scores than controls (all p < 0.05), while euthymic BD patients showed a worse performance for OI (p < 0.05) but not OS (p > 0.05).
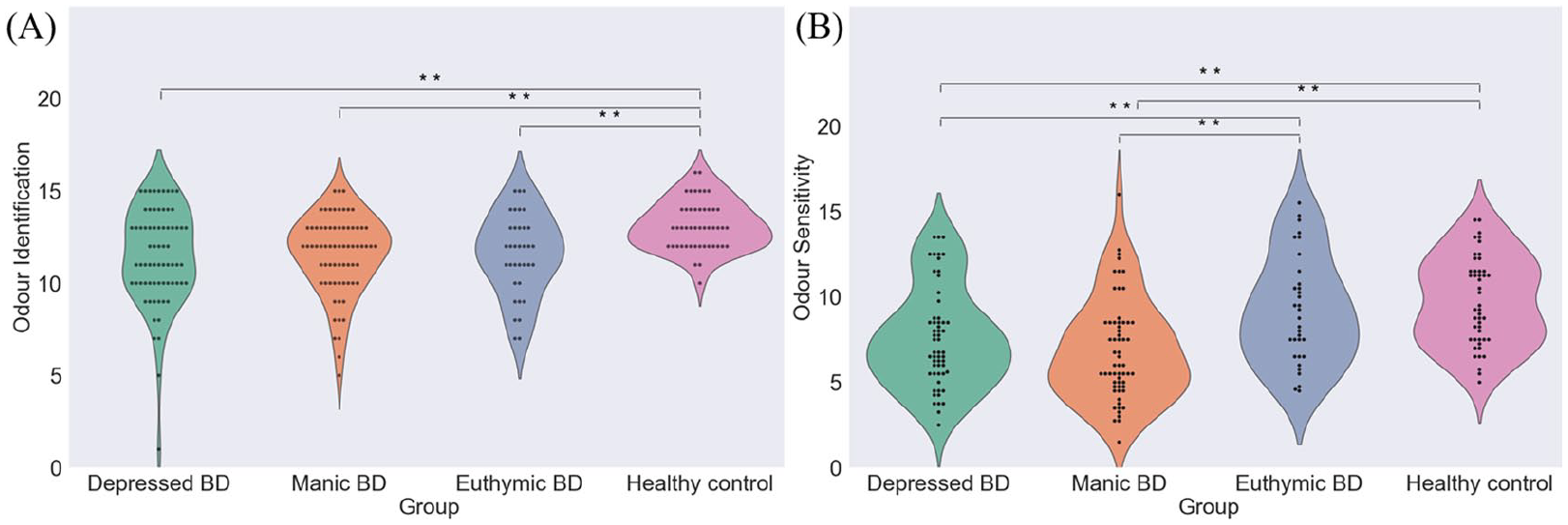
Multiple comparison of olfactory performance in Experiment 1: (A) and (B) olfactory functions in Experiment 1.
Correlations between clinical characteristics of BD patients and olfactory function are shown in Table 2. OS score was negatively correlated with HAMD score (r = −0.18, p < 0.05; Figure 2(A)) and YMRS score (r = −0.25, p < 0.01; Figure 2(B)), but showed no correlation with OI (all p > 0.05). Even after controlling for age, partial correlation analysis showed similar results (YMRS: OI, r = 0.06, p = 0.48, OS, r = −0.25, p < 0.01; HAMD: OI, r = −0.08, p = 0.35). Trend-level significance was observed for correlation between OS and HAMD (r = −0.16, p = 0.053).
Correlations between demographic characteristics and olfactory performance in Experiment 1.
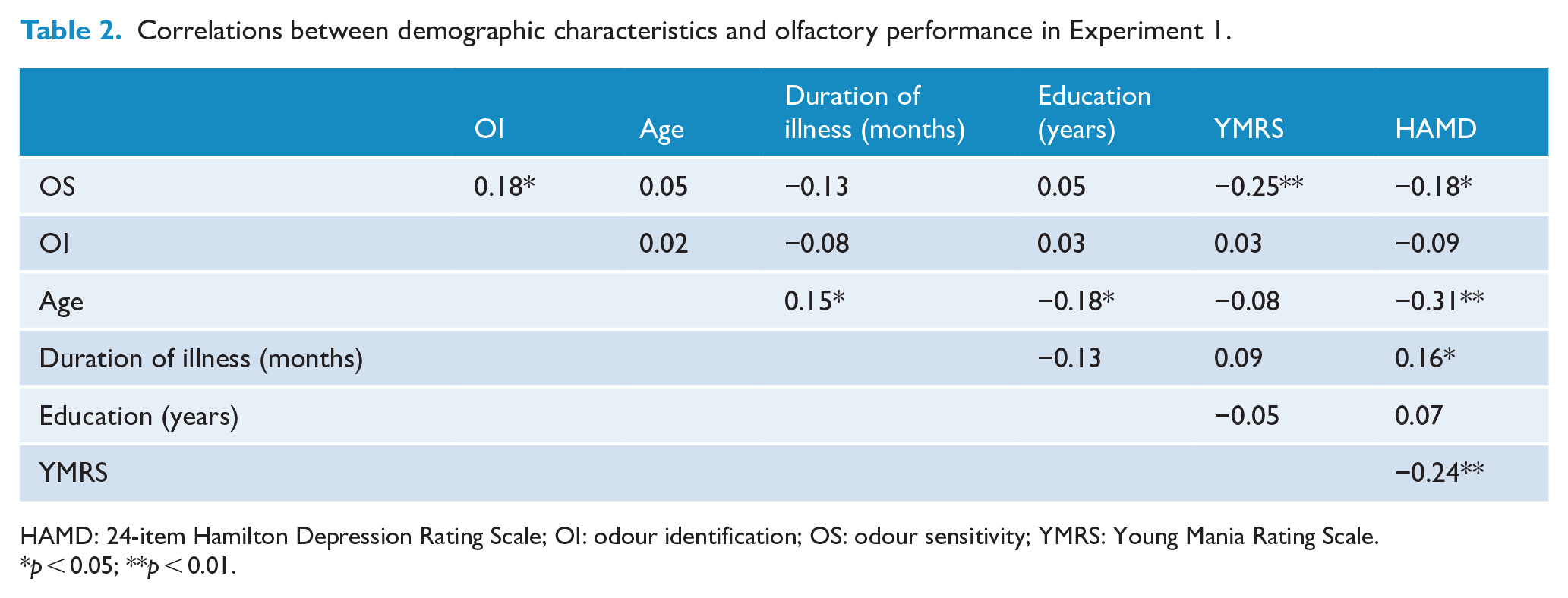
HAMD: 24-item Hamilton Depression Rating Scale; OI: odour identification; OS: odour sensitivity; YMRS: Young Mania Rating Scale.
p < 0.05; **p < 0.01.
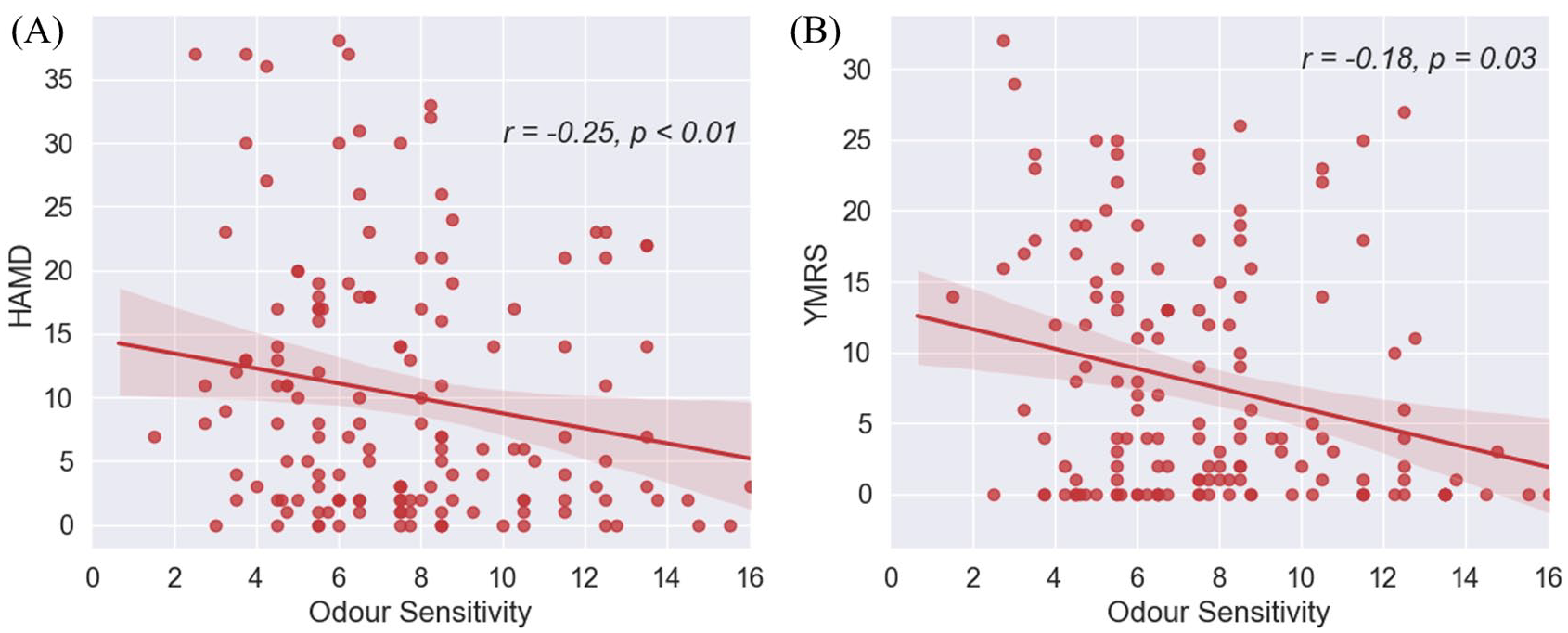
Correlations between odour sensitivity and clinical tests scores in BD patients in Experiment 1. (A) The relationship between odour sensitivity and HAMD and (B) the relationship between odour sensitivity and YMRS in Experiment 1.
In summary, OI deficits were observed in all BD patients; however, OS was impaired only in acute BD, including the depressed and manic BD patient subgroups. We also found that HAMD and YMRS scores were negatively correlated with OS but showed no correlation with OI. These results suggest that OI may indicate a trait marker for BD, whereas OS may be a state marker that can differentiate the acute phase of BD from remission. Deficits in olfaction have been proposed as a marker for MDD (Croy and Hummel, 2017; Croy et al., 2014) and SCZ (Zou et al., 2018). To determine whether the observed OI impairment was specific to BD or was associated with other mental disorders, we carried out Experiment 2 in which we compared olfactory function in BD, MDD and SCZ patients.
Experiment 2
Method
Participants
In addition to the subjects in Experiment 1, we recruited patients with MDD (n = 85; mean age: 22.12 ± 5.30 years) or SCZ (n = 90; mean age: 24.42 ± 7.04 years) from Zhujiang Hospital, Guangdong Provincial People’s Hospital, Guangdong 999 Brain Hospital and Wu Zhongpei Memorial Hospital in Guangdong, China. Patients were diagnosed by trained psychiatrists according to DSM-5. Clinical symptoms were evaluated with the Positive and Negative Syndrome Scale (PANSS) (Kay et al., 1987) in SCZ patients and with HAMD in SCZ and MDD patients. The subjects were free of upper respiratory infections, nasal diseases, traumatic head injuries, other serious illnesses and drug abuse or alcohol addiction, and all had an IQ ⩾ 80. The study was approved by the Ethics Committee of Southern Medical University in Guangzhou, Guangdong, China. Written informed consent was obtained from each participant after they had been provided with a complete description of the study.
Data analysis
Descriptive analysis was carried out for sociodemographic characteristics (sex, age, education level and duration of illness), clinical information (HAMD, YMRS and PANSS scores) and olfactory test scores (OI and OS). Differences in sex ratio between the five patient groups (three BD subgroups, MDD and SCZ) and the control group were compared with the chi-square test. One-way ANOVA and a post hoc test were used to compare sociodemographic characteristics (age, education level and duration of illness), HAMD scores and olfactory function (OI and OS) between the six groups. Analysis of covariance (ANCOVA) was performed to examine the difference of olfactory performance between three BD subgroups and other patient groups after controlling for age. Two-way ANOVA was used to determine whether olfactory function was influenced by sex in the six groups, and a correlation analysis was performed to evaluate whether age, education level and duration of illness influenced OI and OS.
Results and discussion
There were significant differences in sex, age, education level and duration of illness between patient groups (all p < 0.01, for details see Table 3). With the exception of euthymic BD group, other four patient groups had higher HAMD scores than control subjects, euthymic BD, n = 35; manic BD, n = 69; depressed BD, n = 67; MDD, n = 58; SCZ, n = 32; controls, n = 47; F(5, 302) = 51.77, p < 0.01. Manic BD patients had higher YMRS scores than depressed and euthymic BD patients and controls, manic BD, n = 69; depressed BD, n = 62; euthymic BD, n = 35; controls, n = 47; F(3, 209) = 102.48, p < 0.01.
Sociodemographic and clinical characteristics in Experiment 2.
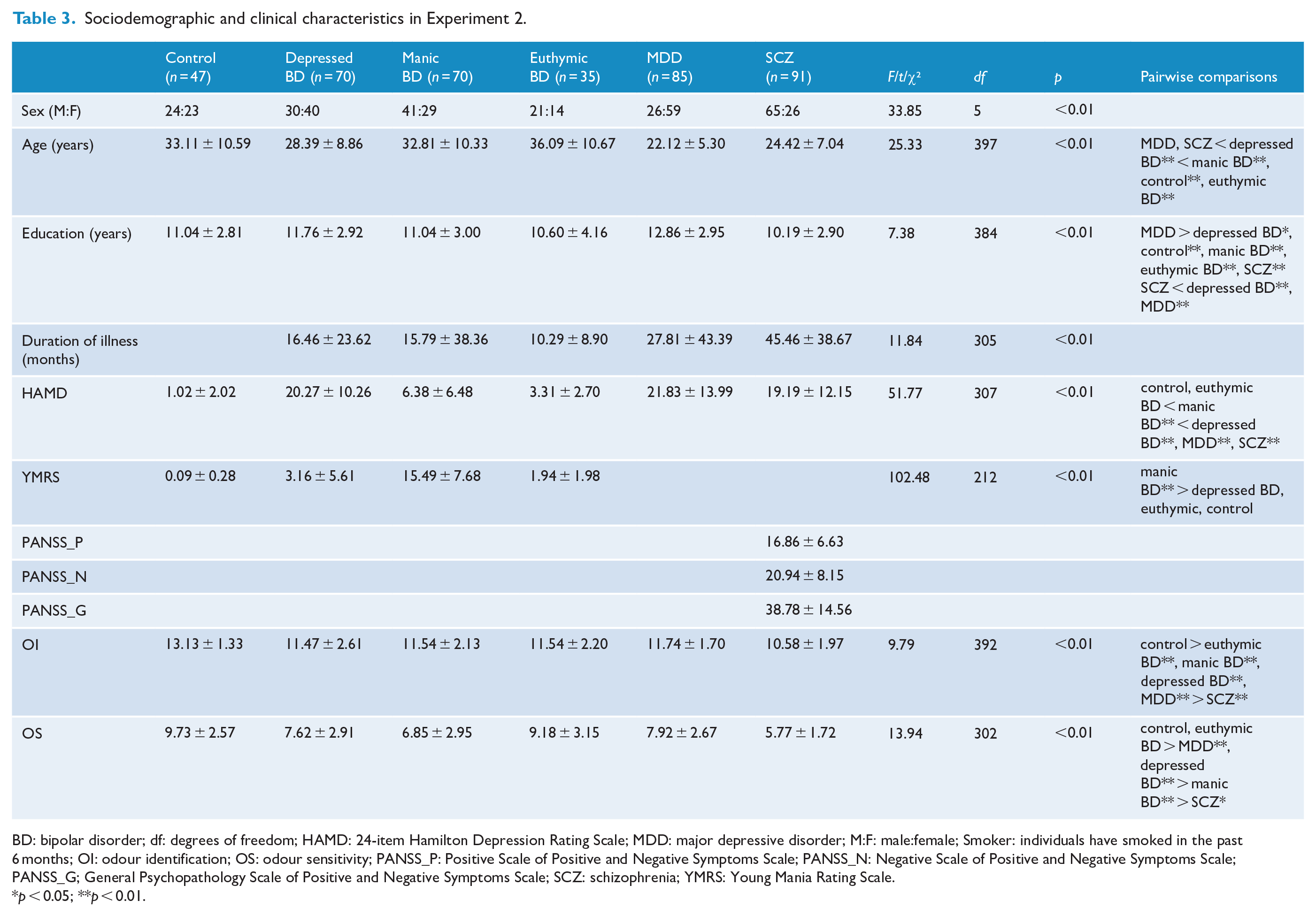
BD: bipolar disorder; df: degrees of freedom; HAMD: 24-item Hamilton Depression Rating Scale; MDD: major depressive disorder; M:F: male:female; Smoker: individuals have smoked in the past 6 months; OI: odour identification; OS: odour sensitivity; PANSS_P: Positive Scale of Positive and Negative Symptoms Scale; PANSS_N: Negative Scale of Positive and Negative Symptoms Scale; PANSS_G; General Psychopathology Scale of Positive and Negative Symptoms Scale; SCZ: schizophrenia; YMRS: Young Mania Rating Scale.
p < 0.05; **p < 0.01.
All five patient groups showed worse OI performance than control subjects, F(5, 387) = 9.79, p < 0.01; see Figure 3(A). There were no differences in OI ability between all three BD subgroups and MDD group (all p > 0.05); however, SCZ patients showed worse OI ability than BD and MDD patients (all p < 0.01). With the exception of euthymic BD group, other four patient groups showed impaired OS than controls, while the severity of OS impairment increased from unipolar and bipolar depressed group (MDD and depressed BD) to manic BD group, and finally, to SCZ group, F(5, 297) = 13.94, p < 0.01; see Figure 3(B). There were no significant correlations between olfactory function and demographic characteristics in SCZ patients (all p > 0.05), suggesting that age, education level and illness duration do not account for the greater olfactory deficits in this group relative to BD and MDD patients.
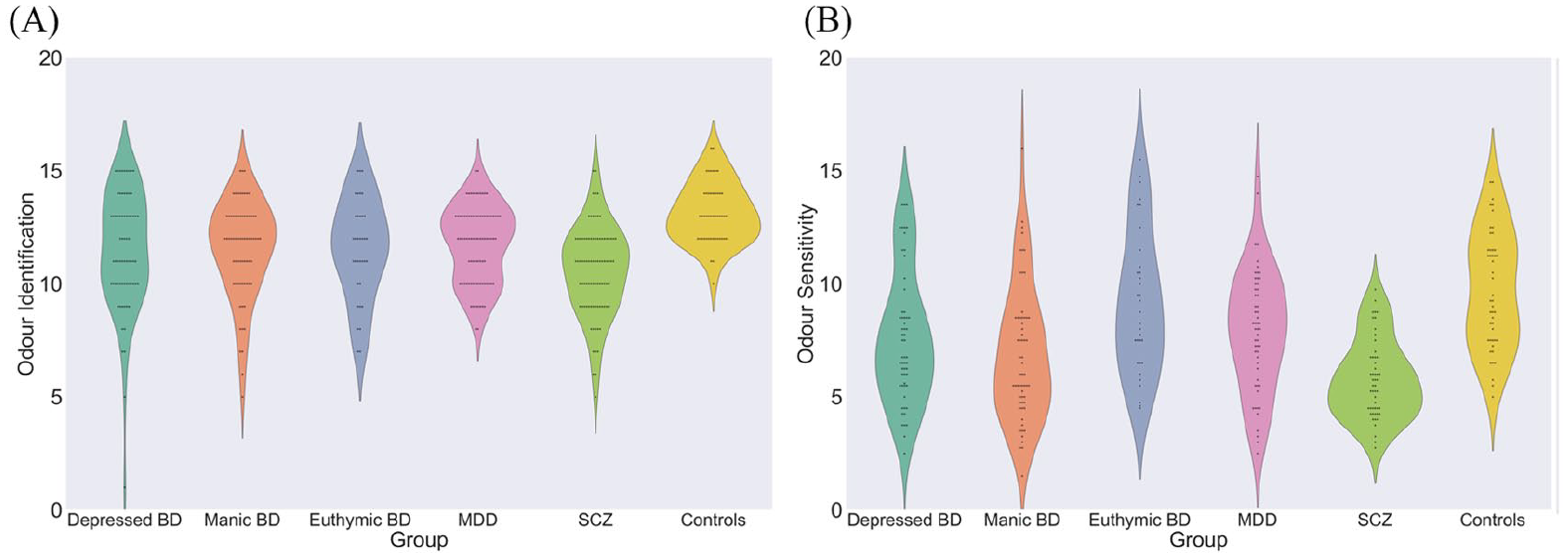
Multiple comparison of olfactory performance in Experiment 2: (A) odour identification ability and (B) odour sensitivity ability of all participants in Experiment 2.
After controlling for age, we conducted an ANCOVA of olfactory performance among six groups (three subgroups of BD, MDD, SCZ and controls) and we found similar results. All patient groups (three BD subgroups, MDD and SCZ) showed impaired OI than controls, F(5, 392) = 9.45, p < 0.01, and SCZ patients had the worst OI ability than other patient groups (MDD and three BD subgroups, all p < 0.05). Moreover, euthymic BD patients had similar OS with healthy control (euthymic BD, n = 33; control, n = 47; p = 0.46) and other patients showed more severe OS impairment than these two groups, depressed BD, n = 57; manic BD, n = 60; MDD, n = 55; SCZ, n = 51; F(5, 302) = 14.19, p < 0.01. In addition, SCZ group was more severe than depressed BD and MDD (all p < 0.05).
Two-way ANOVA was carried out in order to evaluate the effects of sex and type of illness on OI and OS. No main effect of sex or interaction between sex and type of illness was found for OI, sex: F(1, 300) = 0.10, p = 0.75; sex × type of illness: F(5, 300) = 1.59, p = 0.16; and OS, sex: F(1, 300) = 0.08, p = 0.78; sex × type of illness: F(5, 300) = 0.76, p = 0.58. Only type of illness showed a significant effect on olfactory function, OI: F(5, 300) = 7.10, p < 0.01; OS: F(5, 300) = 11.89, p < 0.01.
In summary, these results revealed that while deficits in OI and OS were present in patients with BD, MDD and SCZ, they were more severe in the latter disorder than in BD and MDD. The concept of a continuum of affective psychoses has been proposed (Crow, 1986), whereby the severity of symptoms increases from unipolar to bipolar affective disorders and, finally, to SCZ, suggesting a continuum of olfactory function that varies with the severity of psychotic symptoms.
General discussion
This is the first study comparing olfactory function in a large sample of BD patients with different mood episodes and between BD and other mental disorders – namely, MDD and SCZ. The results revealed impairment of OI in all subgroups of BD patients; however, only patients experiencing acute mood episodes (including depressed and manic episodes) had worse OS performance than control subjects. HAMD and YMRS scores were both negatively associated with OS, but they showed no correlations with OI. While OI and OS deficits were observed in all patients, they were more severe in SCZ than in BD or MDD.
Consistent with our hypothesis, OI impairment was observed in BD patients in different mood episodes (i.e. depressed, manic or euthymic), which is consistent with previous reports of impaired OI in BD patients (Cumming et al., 2011; Kamath et al., 2018; Lahera et al., 2016; Striebel et al., 1999). The proximity of brain structures involved in BD and olfaction (i.e. prefrontal cortex, temporal lobe, limbic regions and insula) may explain this observation (Atanasova et al., 2008; Fjaeldstad et al., 2017; Keener and Phillips, 2007; Kempton et al., 2008; Lu et al., 2019; Zhang et al., 2020). Deficits of OI caused by mood disturbance in manic episode could be explained by the reduced brain activation in orbitofrontal cortex, where is the most associated with OI ability (Altshuler et al., 2005). Our results also showed that OS was impaired in depressed and manic BD patients but not in those with euthymic BD, in agreement with previous studies that found no difference in OS between stable BD patients and healthy individuals (Hardy et al., 2012; Negoias et al., 2019). Depressive symptoms have been shown to be negatively related to olfactory bulb volume, which could influence the risk of emotion processing imbalance (Negoias et al., 2010). Colle et al. (2020) found that depression improvement mediated olfaction improvement. Thus, a reduced OS could reflect temporal changes in peripheral structures (i.e. nasal epithelium, olfactory receptors and olfactory bulbs) in patients with acute BD. Moreover, the negative correlations between HAMD and YMRS scores and OS but not OI indicate that OS can distinguish euthymic BD patients from those in acute phase, whereas OI may constitute a trait marker of BD.
In Experiment 2, we compared OI and OS between patients with BD, MDD and SCZ and found impaired olfactory function in all patient groups, with SCZ patients showing the worst performance in both olfactory function tests, except normal OS in euthymic BD patients. The lack of differences in olfactory function between depressed BD and MDD patients is in accordance with previous reports (Kamath et al., 2018; Kazour et al., 2020; Swiecicki et al., 2009). Recent imaging studies indicate that compared to BD, SCZ patients show a greater reduction in grey matter volume in the frontal-temporal cortex, hippocampus and amygdala (Maggioni et al., 2016; Nenadic et al., 2015), which are involved in olfactory processing. Our results are also consistent with previous work in which olfactory function was directly compared between these two patient groups; the authors proposed that OI deficits lie on a continuum, with limited dysfunction in BD and the greater impairment in SCZ (Cumming et al., 2011).
The education level was lower, and the SCZ group had a higher percentage of male patients than the BD and MDD groups in our study. However, the degree of olfactory impairment did not appear to be influenced by these two variables. We did not find any correlation between education level and olfactory function in the SCZ group, which is in line with the results of a meta-analysis demonstrating that education level had no influence on the effect size of olfactory performance in patients with SCZ (Moberg et al., 2014). Male and female SCZ patients did not differ in terms of olfactory function, and the two-way ANOVA showed no main effect of sex or the interaction between sex and type of illness. Thus, the more severe olfactory deficits in SCZ patients may be attributable to the disease itself.
The impaired OI in BD, MDD and SCZ could reflect shared clinical features or abnormalities in neuroanatomy or gene expression (Arango et al., 2013). Family and twin studies suggest a common genetic basis for BD and SCZ, which also show similarities in neurotransmitter dysfunction (Möller, 2003). The concept of a continuum of affective psychoses has been proposed (Crow, 1986), whereby the severity of symptoms increases from unipolar to bipolar affective disorders and, finally, to SCZ. This is supported by our observation that even after controlling for age, the lowest OS score existed in SCZ and manic BD patients, the better OS score in depressed BD and MDD patients and the highest OS score in euthymic BD patients and healthy controls. For OI ability, SCZ and MDD patients had the lowest and highest olfactory test scores, respectively, while BD patients showed an intermediate performance.
A limitation of this study was the cross-sectional design. Follow-up studies are needed to assess olfactory function by monitoring BD patients in different mood episodes. In addition, we did not investigate the mechanisms underlying the observed similarities and differences in olfactory function between BD, MDD and SCZ patients. Future studies using brain imaging technique among these patient groups are needed. Besides, we have only investigated participants’ OS and OI abilities. Investigations in other olfactory domains (e.g. odour discrimination, odour memory) may provide a new insight of these mental diseases. Nonetheless, our results suggest that OI impairment is a trait but not a disease-specific marker in BD, whereas OS impairment is a potential mood episode marker, which could have important implications in the diagnosis and management of BD.
Footnotes
Acknowledgements
The authors thank Prof. Thomas Hummel for his helpful comments on this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant no. 31700963); Natural Science Foundation of Guangdong Province, China (grant no. 2019A1515012135); and Medical Science and Technology Foundation of Guangdong Province, China (grant no. A2019192). These funding agencies had no other role in any aspect of the study or the writing of this paper.
