Abstract

To the Editor
Given the cognitive impairment associated with schizophrenia, it is concerning that this may be worsened by the anticholinergic burden associated with some psychotropic medications (Joshi et al., 2021). Clozapine, the most effective medication for positive symptoms in treatment-refractory schizophrenia (Siskind et al., 2016), is a cholinergic receptor antagonist, while its active metabolite, norclozapine, is a partial agonist (McArdle et al., 2019). A higher clozapine/norclozapine ratio, with a higher cholinergic burden, has been found to be associated with greater cognitive impairment (McArdle et al., 2019; Rajji et al., 2015). Most of these studies have used small samples collected as part of routine clinical practice, rather than generating evidence from a larger sample collected within a formal clinical trial of people with schizophrenia who show persisting symptoms despite treatment with clozapine.
We used baseline data (n = 86) from a randomised controlled trial of participants with schizophrenia, prescribed clozapine, who had persisting negative symptoms (Rossell et al., 2016). We explored the relationship between the clozapine/norclozapine ratio and cognitive performance. Clinical symptoms were assessed using the Positive and Negative Syndrome Scale (PANSS) and IQ was estimated with Wechsler Abbreviated Scale of Intelligence (WASI) and cognition with the MATRICS consensus cognitive battery (see Table 2). For each participant, we calculated a clozapine/norclozapine ratio and anticholinergic cognitive burden (ACB) of co-prescribed medications using the ACB Scale (Joshi et al., 2021). Each cognitive (T-score) and PANSS raw score was correlated with the clozapine/norclozapine ratio and ACB score using Spearman’s rank correlation.
The demographic and clinical characteristics of participants are presented in Table 1. Overall, total psychosis symptoms were high (mean PANSS 73.9, SD = 14.1) and cognitive performance was characteristically poor (mean T-scores < 41, thus for most domains <1 standard deviation below standardised population means). The analysis found that clozapine/norclozapine ratio was not associated with any of the cognitive measures, nor with PANSS total or positive scales (Table 2). The correlation with the PANSS negative score (r = −0.288, p = 0.011) did not reach significance after applying Bonferroni correction for multiple comparisons (p < 0.003), nor did the association with Hopkins Verbal Learning Task–Revised (HVLT-R) delayed recall (r = −0.221, p = 0.054). There were no significant correlations between the ACB and cognitive tests.
Demographic and clinical characteristics of participants (N = 86).
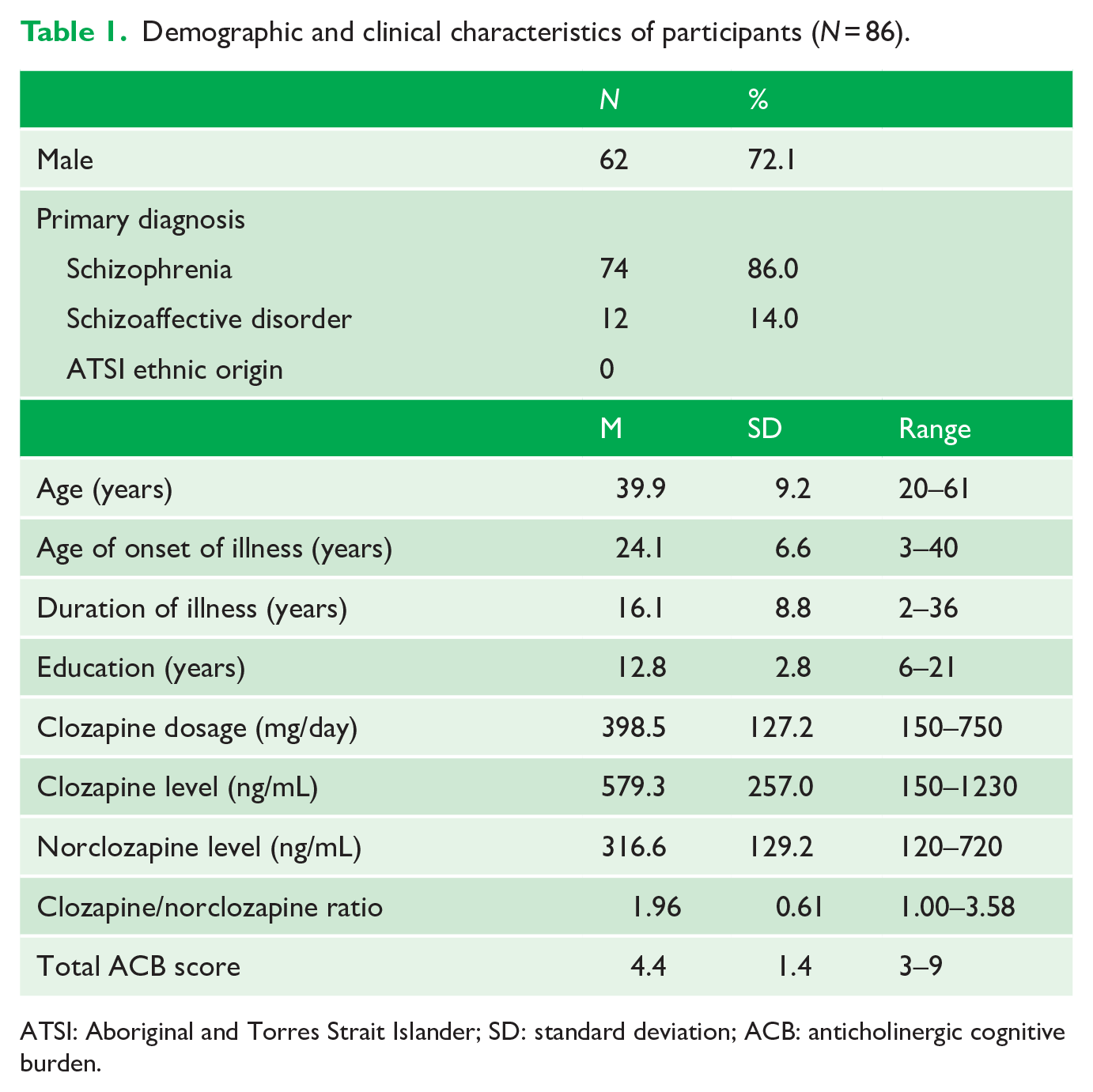
ATSI: Aboriginal and Torres Strait Islander; SD: standard deviation; ACB: anticholinergic cognitive burden.
Correlations between psychosis and cognitive scores, clozapine/norclozapine ratio and anticholinergic burden.
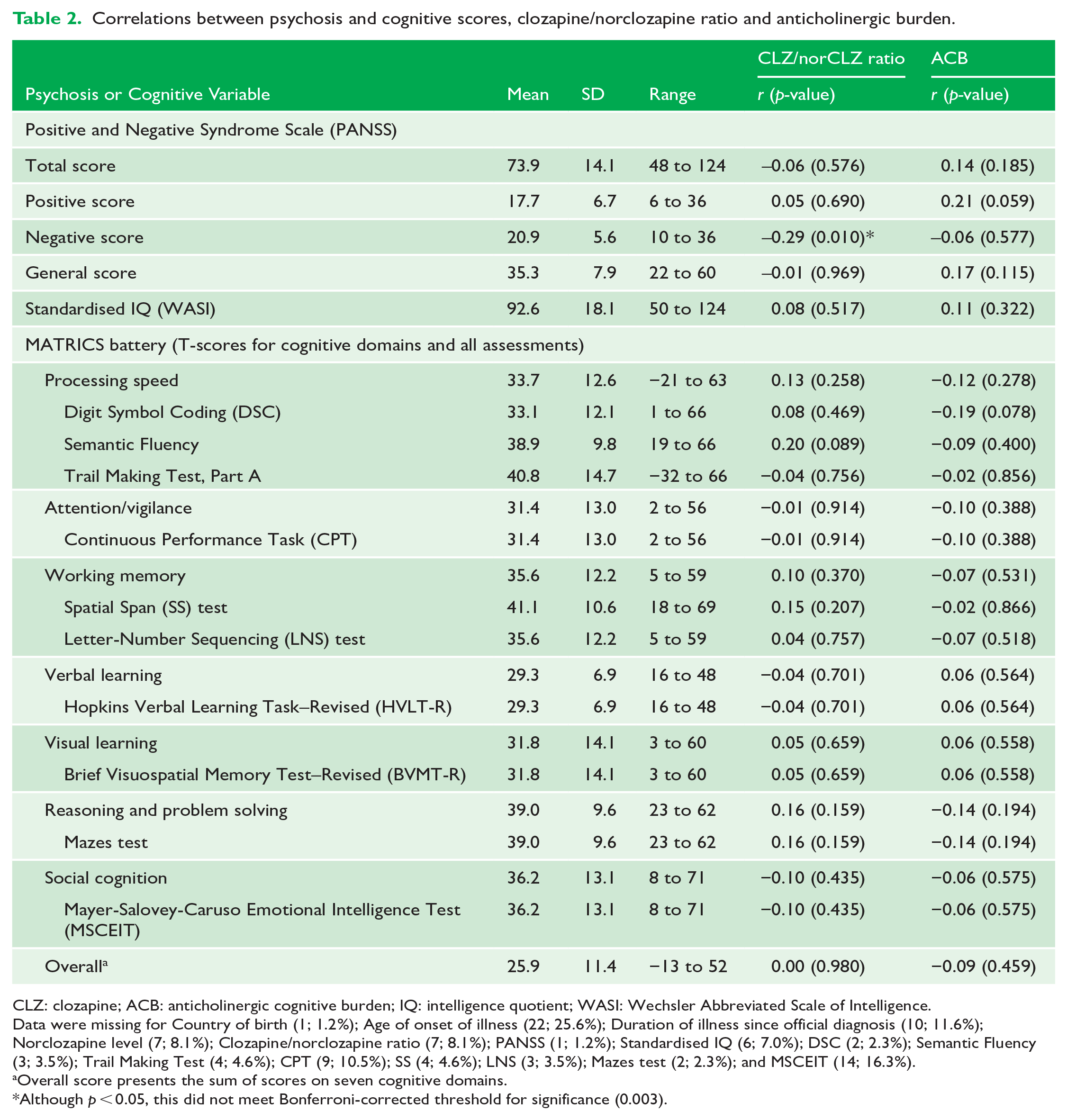
CLZ: clozapine; ACB: anticholinergic cognitive burden; IQ: intelligence quotient; WASI: Wechsler Abbreviated Scale of Intelligence.
Data were missing for Country of birth (1; 1.2%); Age of onset of illness (22; 25.6%); Duration of illness since official diagnosis (10; 11.6%); Norclozapine level (7; 8.1%); Clozapine/norclozapine ratio (7; 8.1%); PANSS (1; 1.2%); Standardised IQ (6; 7.0%); DSC (2; 2.3%); Semantic Fluency (3; 3.5%); Trail Making Test (4; 4.6%); CPT (9; 10.5%); SS (4; 4.6%); LNS (3; 3.5%); Mazes test (2; 2.3%); and MSCEIT (14; 16.3%).
Overall score presents the sum of scores on seven cognitive domains.
Although p < 0.05, this did not meet Bonferroni-corrected threshold for significance (0.003).
Despite previous studies finding that higher clozapine/norclozapine ratios were associated with greater cognitive impairment, specifically with deficits in processing speed (McArdle et al., 2019) and working memory (Rajji et al., 2015), our findings did not replicate these results. This discrepancy may be due to the specific symptom profile of individuals who are poorly responsive to antipsychotic drugs and, therefore, are considered treatment resistant. An inspection of the baseline psychosis and cognition levels of our cohort compared to previous similar studies (Joshi et al., 2021; Rajji et al., 2015) revealed higher overall cholinergic burden in this cohort (clozapine/norclozapine ratio of 1.96 compared to 1.64, Rajji et al., 2015, and ACB score of 4.4 compared to 3.8, Joshi et al., 2021), but also greater psychosis symptom severity (as measured with the PANSS). While mean cognitive scores show slightly lower cognitive performance compared to the patients included in the study by Rajji et al. (2015), the range of scores in our cohort tends to be wider. We speculate that the variation of scores, obtained in our larger sample, may contribute to the lack of observed association between clozapine/norclozapine ratio and cognition in this cohort.
Future research should continue examining the cholinergic burden of clozapine and co-medications in longitudinal studies. Moreover, changes in cognition at the individual level rather than the aggregate level should be explored. This could guide techniques to minimise cognitive effects in clinical practice. There is also a need to evaluate the cognitive learning potential among people with treatment-resistant schizophrenia, as they may present with poorer overall cognition, which may be only minimally affected by changes in cholinergic transmission.
Footnotes
Author Contributions
The letter was conceived by all authors. U.A. wrote the first draft which was finalised with contributions of all authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: D.S. and U.A. are funded in part by an NHMRC Investigator Grant GNT1194635. The ENHANCE RCT was funded by an NHMRC Project Grant GNT1098442.
