Abstract
Objective:
Ketamine and related compounds are emerging as rapidly acting therapies for treatment-resistant depression. Ketamine differs from standard antidepressants in its speed of action, specific acute and cumulative side effects, risk of dependence and regulatory requirements. However, there is currently little guidance offering translation from research studies into clinical practice. We therefore detail a comprehensive model of care for ketamine treatment of depression.
Method:
We formulated a set of policies and procedures for a ‘compassionate use’ ketamine programme that developed out of our clinical research in ketamine. These policies and procedures were formulated into a detailed model of care.
Results:
The current Australian and New Zealand regulatory frameworks and professional bodies’ recommendations regarding ketamine are detailed along with clinical governance and infrastructure considerations. We next describe a four-step model comprising initial assessment, pre-treatment, treatment and post-treatment phases. The model comprises thorough psychiatric and medical assessments examining patient suitability, a rigorous consenting process and structured safety monitoring across an acute treatment course or maintenance therapy. Our ketamine dose-titration method is detailed allowing flexible dosing of patients across a treatment course enabling individualised treatment.
Conclusion:
The model of care aims to bridge the gap between efficacy studies and clinical care outside of research settings as ketamine and related compounds become increasingly important therapies for treatment-resistant depression.
Introduction
Ketamine therapy for depression has upended the dominant monoamine theory of depression. As an N-methyl-
A recent non-randomised study found ketamine equally as effective as electroconvulsive therapy (ECT) (Basso et al., 2020), introducing the possibility that it may be an alternative to ECT, especially in patients at increased risk for ECT-related cognitive side effects. Efficacy has also been reported in depression refractory to ECT (Ibrahim et al., 2011). Additionally, ketamine reduces suicidal ideation, with meta-analyses demonstrating reduction as early as 24 hours after treatment (Xu et al., 2016) and with effects lasting up to 72 hours (Witt et al., 2020). More recently, studies have examined repeated dosing, with esketamine found to be more effective than a placebo control, added on to existing AD in a treatment refractory sample (Popova et al., 2019), and with reduced relapse rates when added to conventional AD therapy (Daly et al., 2019). A meta-analysis of 20 studies (Kryst et al., 2020) found a significant reduction of depression severity scores at 2–3 weeks with repeated dosing ketamine treatment, with medium to large effect sizes (SMD = 0.70–0.81), depending on dosing regimen.
As evidence from clinical trials accumulates, and newer ketamine preparations are approved (e.g. intranasal esketamine has recently been approved by the US Food and Drug Administration and the European Medicines Agency for TRD as well as for major depressive disorder (MDD) and depressive symptoms with suicidal ideation or behaviour), there is a need for treatment guidelines to translate research findings into a model of care. The literature is sparse with regard to models of care for ketamine therapy in depression. Aside from efficacy studies and meta-analyses, the majority of recent ketamine literature comprises commentaries or opinion pieces on legal and ethical considerations (Loo, 2018; Malhi et al., 2016; Ryan and Loo, 2017; Sanacora et al., 2017b; Schatzberg, 2014; Singh et al., 2017, 2019; Zorumski and Conway, 2017). The American Psychiatric Association (APA) released a consensus statement on use of ketamine in the treatment of mood disorders (Sanacora et al., 2017a), which detailed expert clinical opinion and considerations for off-label use of ketamine. Locally, a clinical memorandum issued by the Royal Australian and New Zealand College of Psychiatrists (RANZCP, 2019) offered indicative guidance and the recent RANZCP Clinical Practice Guidelines on Mood Disorders (Malhi et al., 2021) discusses both ketamine and esketamine though without detail as to clinical translation. The above guidelines do not address the emerging interest in ketamine combined with an adjunctive psychological therapy (e.g. psychedelic-assisted psychotherapy), with studies required to test the efficacy of such approaches (Greenway et al., 2020).
From our experience of using ketamine to treat depression in clinical trials (Galvez et al., 2018; George et al., 2017; Lai et al., 2014; Loo et al., 2016; Ketamine for Adult Depression Study [ACTRN12616001096448]), we established a ‘compassionate use’ ketamine programme for participants who had completed the research trials, benefitted from treatment and requested further treatment. Clinical treatment policies and procedures were developed for this programme. The aims of this paper are twofold: first, to detail the current regulatory and professional guidelines surrounding ketamine treatment in Australia and New Zealand and second, to detail insights and important considerations derived from our ketamine programme with reference to the literature. Our overall key recommendations are listed in Box 1.
Key recommendations for ketamine treatment for depression.

KSET: Ketamine Side Effect Tool.
Regulatory aspects of ketamine in Australia and New Zealand
In Australia, ketamine is bound by regulations pertaining to its Schedule 8 (S8) drug status, meaning it is considered to have a high potential for abuse and addiction. In New Zealand, ketamine is classified as a ‘Class C’ or Schedule 3 drug considered to pose only a moderate risk of harm (New Zealand Legislation: Acts. Misuse of Drugs Act, 1975). In comparison to Australia and New Zealand, ketamine is listed in the United States as a Schedule III drug (defined as having moderate to low potential for dependence) and the United Kingdom as a Class B drug (or as Class A if it is injectable, which is deemed most harmful). Prescription of ketamine is bound by regulations which differ across Australian states and territories as well as New Zealand (see Bayes et al., 2019). The regulations broadly relate to the maximum time period of prescribing permitted without seeking an authority (most frequently a 2-month period), with an authority typically necessary for prescribing to drug-dependent patients. For example, in NSW, for any injectable or intranasal S8 drug, if the treatment period is less than two continuous months – no permit is required; if the treatment period is greater than two continuous months – an authority is required from the NSW Ministry of Health. For drug-dependent individuals in an outpatient setting, a permit is required prior to initiating treatment regardless of treatment period; for inpatients, no permit is needed if the treatment period does not exceed 14 consecutive days. The intranasal preparation of esketamine has recently been approved by the Therapeutic Goods Administration (TGA) in Australia, though it is unclear if any specific regulations outside those existing for S8 drugs will be involved. Appropriate storage facilities such as a lockable cabinet or refrigerator are required as well as an S8 drug register (e.g. in NSW, this is governed by the Poisons and Therapeutic Goods Regulation, 2008) and practitioners should ensure they comply with appropriate ketamine storage, disposal and recordkeeping policies. Familiarity with local regulations is of critical importance for clinicians, as failure to comply has led to disciplinary action (e.g. by the NSW Health Care Complaints Commission; Bayes et al., 2019; HCCC, 2018). Use of real-time prescription monitoring systems in certain jurisdictions (e.g. SafeScript in Victoria) should be adhered to as required, with such programmes reducing potential for doctor shopping (Boyles, 2019).
‘Off-label’ prescribing of ketamine for TRD
In Australia, ketamine is currently approved as an anaesthetic drug by the TGA, but is not currently licensed for treating depression, and as such its use for mood disorders is considered ‘off-label’. In New Zealand, the New Zealand Medicines and Medical Devices Safety Authority (Medsafe) affords ketamine similar status. In contrast, intranasal esketamine has been approved for TRD in both Australia and New Zealand with practice parameters clearly set out in the licence e.g. esketamine safety monitoring is mandated via the Risk Evaluation and Mitigation Strategy (REMS). Whereas off-label ketamine has no such licence for depression and instead requires parameters informed by models of care and guidelines. The RANZCP (2018) professional practice guideline ‘Off-label prescribing in psychiatry’ details a number of considerations relevant to ketamine prescription for depression. These include that off-label prescribing should occur only when therapy outside of non-standard treatment is considered necessary and appropriate; that this should be explained and documented in the medical record; relevant monitoring of clinical progress should be undertaken of both mental and physical status (e.g. vital signs); informed consent should be obtained and documented with the patient understanding the ‘purpose, nature, benefits, side effects, risks and potential out-of-pocket cost’ of the medication as well as information on alternative treatments. The guideline notes that some jurisdictions may require a second opinion e.g. in Tasmania a second opinion from a psychiatrist is required if ketamine is being prescribed outside of the framework of a clinical trial or the prescriber does not have access to a Medicines Advisory Committee/Medicines Assessment Advisory Committee (Department of Health, Tasmanian Government, 2020; RANZCP, 2018). Additionally, clinical indicators for a response to the proposed treatment (e.g. improved mood) should be discussed with the patient as well as monitoring of use, outcome and any side effects; an end-point to overall treatment should be decided as part of a management plan; explicit communication to general practitioners (GPs) that the treatment is off-label; and finally, that the psychiatrist has appropriate indemnity insurance prior to prescribing an off-label medication (RANZCP, 2018). It behoves practitioners to follow these guidelines if considering prescribing ketamine in an ‘off-label’ context for depression.
Professional bodies’ consensus statements on ketamine therapy for TRD
Currently, the RANZCP (2019) in their clinical memorandum on ‘Use of ketamine in treatment-resistant depression’ does not recommend use of ketamine in clinical practice, citing gaps in knowledge about dosing, treatment protocols and effectiveness and safety of long-term use. However, they do provide limited recommendations for psychiatrists considering prescribing ketamine outside of a research setting, suggesting first ‘to proceed with caution’ and recommends ketamine therapy be discussed with peers, preferably including a formal second opinion, and/or seek institutional review by a medicines advisory committee and/or seek consideration by an institutional research ethics committee (RANZCP, 2019). The APA consensus statement on the use of ketamine in the treatment of mood disorders (Sanacora et al., 2017a) also warns of risks, but goes on to provide guidance to shape clinical practice that is already occurring – a pragmatic approach based on a ‘harm minimisation’ model. An expert panel has recently provided ketamine and esketamine treatment guidance in clinical practice, highlighting practice parameters at point of care (McIntyre et al., 2021).
Infrastructure considerations
Infrastructure requirements typically include a clinic room in which to administer ketamine (especially if given by injection) and then to monitor a patient post drug administration – particularly relevant if outside of a hospital setting. The setting should be comfortable for the patient (e.g. a recliner chair), with a low stimulus environment to reduce the likelihood of unwanted psychotomimetic effects. Depending on the route of administration, specific supplies may be required, e.g. for a ketamine injection or infusion, cannulation equipment, infusion pump, syringes, needles, sharps disposal bin and gloves. Relevant monitoring equipment at a minimum should include a sphygmomanometer and pulse oximeter (see below, Safety monitoring section). A supply of pro re nata (PRN) medications should be available in the event that management of side effects arising from ketamine therapy is necessary. In practice, our experience is that this is not commonly required, but it is advisable to have available antiemetics, antihypertensives, analgesia (e.g. for headache), as well as benzodiazepines and antipsychotics for anxiety or agitation emerging during treatment.
Staff roles
A psychiatrist with expertise in prescription of ketamine and management of mood disorders should prescribe and oversee treatment. Depending on the treatment setting, the psychiatrist may provide supervision to other staff (e.g. psychiatry trainees, medical officers, nurses) who may be involved in treatment delivery, monitoring and preparation for discharge. A medical officer trained in basic and advanced life support should be available to attend at the time of treatment in the case that any medical complications arise, e.g. cardiovascular events or acute behavioural disturbances. In non-hospital healthcare settings, there should be a protocol for transfer of patients to a hospital emergency department should the patient experience an acute cardiovascular, or other, event.
Credentialling
The service should establish a credentialing system, with clinic health care workers credentialled according to the requirements of their role and responsibilities in the treatment process. This includes having a relevant knowledge base (which may be covered by attendance at a training course) and assessment of practical skills, including direct observation to establish competence where relevant (e.g. in use of instruments to measure efficacy and safety).
Clinical governance
Local policies and procedures should be established by any group planning to administer ketamine for depression. Any treatment programme should have a committee that convenes regularly to ensure appropriate credentialing of staff; compliance with local protocols for ketamine delivery; collection of clinical indicator data for auditing and benchmarking; as well as review of side effects, safety issues and any serious adverse events.
Phases of treatment
Our model involves the following four phases: (1) initial assessment, (2) pre-treatment, (3) treatment and (4) follow-up, as detailed below.
Initial assessment phase
Psychiatric diagnostic assessment
An appropriate clinical framework should include robust screening processes to determine the suitability of individuals to receive ketamine therapy. Ketamine and esketamine are not considered first-line treatments, with individuals required to have ‘treatment-resistant’ depression. Treatment resistance has been variably defined across ketamine efficacy studies, but typically involves failure of two therapeutic AD medication trials.
There is currently no consensus on where ketamine fits in treatment algorithms for TRD e.g. at what point in a patient’s treatment journey should ketamine be considered, with options ranging from failure of a single AD up to failure of response to ECT. Assessment of treatment resistance should involve consideration of the nature of the depression; aetiological and maintaining factors; the number of previous AD trials, including whether ADs from different classes have been used (e.g. selective serotonin reuptake inhibitors vs tricyclic ADs); whether the trials were of a sufficient duration and at an adequate dosage; whether there was a response; and if medications were ceased was this owing to a lack of efficacy or tolerability issues, the latter of which may also be an indication for ketamine. Augmentation strategies (e.g. with lithium carbonate, atypical antipsychotics) should also be detailed. Response to any trialled neurostimulation therapies, in particular ECT, should be documented. A standardised staging instrument (e.g. Maudsley Staging Method) may assist in quantifying the degree of treatment resistance (Fekadu et al., 2018). Current or past psychological therapies trialled should also be listed.
In addition to establishing TRD, a careful diagnostic assessment should examine whether the patient has a unipolar or bipolar condition as well as any comorbid diagnoses. Efficacy of ketamine in bipolar depression has been previously reported, though only a minority of the overall studies have been conducted in this patient population (Diazgranados et al., 2010). The onset of ketamine’s AD effect in bipolar depression is more rapid (vs unipolar depression) and patients should be monitored for switching (Diazgranados et al., 2010). Those with severe suicidality in the context of MDD may benefit from ketamine treatment, with a rapid reduction in ideation found in this patient group (Xu et al., 2016) with a similar result seen with esketamine (Canuso et al., 2018). Careful assessment for presence of psychotic features is important, as ketamine is not generally considered as indicated in such patients because of concern of exacerbating symptoms (Sanacora et al., 2017a). However, a recent case series of four patients with psychotic depression treated with low-dose esketamine found positive results without exacerbation of psychosis (Ajub and Lacerda, 2018).
It remains unclear which patient groups are more likely to respond to ketamine treatment. There has been a suggestion that patients with melancholic depression may have a superior response to ketamine based on a number of very small studies and case reports (Atigari and Healy, 2013; Galvez et al., 2018; Lai et al., 2014; Paslakis et al., 2010), with one such patient deemed a ‘super-responder’ who went into remission for 5 months after a single intravenous dose of ketamine (Galvez et al., 2014). Other studies have noted female gender, older age (>45 years), greater treatment resistance and higher levels of disability were factors increasing the probability of response to esketamine commenced with AD medication versus placebo (Popova et al., 2019).
Those with depression and comorbid personality disorder (in particular borderline personality disorder) warrant careful evaluation, as ketamine has rapid-onset and rapid-offset AD effects, which may have complex interactions with personality-based emotional dysregulation. In such patients, the role of ketamine in treating suicidal ideation remains to be elucidated, as mood can rapidly decline after an initial improvement, which may theoretically increase suicidality (Ryan and Loo, 2017). There may be additional concerns with potential misuse in this clinical population. Patients with major depression and comorbid substance abuse also warrant careful consideration, given the abuse potential of ketamine and its known use as a recreational drug. In Australian states and territories, there are regulatory restrictions around prescribing S8 drugs to substance-dependent patients (see Bayes et al., 2019). Prior or current recreational use or abuse of ketamine or phencyclidine (PCP) should be considered an absolute contraindication to ketamine treatment. Prior or current use/abuse of other substances should be considered a probable contraindication. Prior treatment or use of ketamine should be enquired about (e.g. for chronic pain), noting any adverse reactions that may have occurred or features of dependence.
Physical health assessment
We recommend patients undergo a routine medical assessment to determine suitability for ketamine therapy. A recent systematic review (Short et al., 2018) examined side effects of ketamine treatment in depression, noting a number of potential physical, psychiatric and cognitive side effects. The potential of ketamine to induce hepatotoxicity, acute hypertension (Wan et al., 2015; Zarate et al., 2006), cystitis and lower urinary tract dysfunction warrant screening for pre-existing conditions affecting these organ systems. Hepatic and urological comorbidities as well as hypertension should be screened for, to determine whether these preclude treatment or require management prior to treatment (e.g. involvement of the GP or treating specialist to stabilise hypertension). Screening for cognitive impairment may additionally be required, as frequent and long-term ketamine use has been associated with cognitive dysfunction (Morgan et al., 2010, 2014); and these patients could be at increased risk. For those patients with explicit cognitive deficits, ketamine should only be considered when benefits (e.g. control of severe suicidal behaviours) far outweigh risks and with consideration given to capacity to consent to treatment. A structured safety assessment framework, the Ketamine Side Effect Tool (KSET), includes a screening questionnaire to identify medical risks relevant to treatment with ketamine (Short et al., 2020).
Pre-treatment phase
Work-up
In addition to initial screening, it is important to seek background information (e.g. discharge summaries). We recommend a general physical examination be undertaken including weight and vital signs – blood pressure (BP), heart rate (HR), respiratory rate (RR) and oxygen saturation (SaO2). In keeping with active safety monitoring, investigations should include baseline liver function tests (LFTs), a urine dipstick test if there is concern about urological conditions, and an electrocardiogram (ECG) if indicated (KSET screening module, Short et al., 2020). For women of childbearing age, a pregnancy test should be undertaken and the patient advised of potential risks of ketamine during pregnancy (as per product information; category B3). Baseline cognitive testing using standardised tests (e.g. to assess memory and attention: see KSET Baseline form, Short et al., 2020) should be documented to allow for comparison to repeat testing if clinical concerns arise about treatment. Documentation of other prescribed medications and checking for possible interactions with ketamine should be undertaken.
Consent
Appropriate consenting of patients is essential before commencing treatment. This should contain at least the following elements: detail of the current evidence base for use of ketamine in depression (including response and remission data); alternative treatments available; description of the treatment programme (assessment, dosing-related information, frequency of treatments, observations during treatment, duration of treatment period [noting regulatory limits], follow-up post-treatment); safety of ketamine (including acute, cumulative and long-term side effects, noting evidence for the latter two not yet clearly established) and financial consent. Women should agree they will utilise measures to prevent pregnancy and refrain from breastfeeding during the treatment period. We recommend involving a family member in the consenting process. If cognitive impairment is present, capacity to consent needs to be carefully evaluated and involvement of a family member is essential in this instance. All patients should agree as part of the consenting process to desist from working, driving, operating heavy machinery or making important decisions on the same day as ketamine treatment. An appropriate consent form should detail all relevant information and be signed and retained in the patient’s medical record. For long-term or maintenance treatment (if permitted by regulations), re-consenting the patient periodically is required.
Patient expectations
It is important to manage expectations of a patient embarking on a course of ketamine. Popular news reports have hailed ketamine as a ‘wonder drug’ – however, the complexity of many patients’ presentations, often with established treatment resistance and comorbidities, means that measures apart from ketamine will likely be required for overall recovery. Additionally, patients should be informed of the rapid onset of AD action, but that relapse may also occur within days, and recurrence of symptoms may be rapid if therapy is ceased. As the majority of studies of repeated dosing esketamine have been undertaken in individuals where their pre-existing AD was continued or they were managed with a concomitant AD commenced during the trial (Daly et al., 2019; Popova et al., 2019), we advise patients that ketamine should be used as add-on therapy.
Treatment phase
A psychiatrist or a medical practitioner (under the supervision of a psychiatrist) familiar with the evidence on AD effects of ketamine should prescribe ketamine, including specifics of dosage and route of administration, be responsible for the ketamine treatment process and for review in the case of any medical complication.
Route of administration
Many of the original ketamine studies used an intravenous (IV) route, typically an infusion (Berman et al., 2000; Zarate et al., 2006), though ketamine can also be administered by intramuscular (IM) injection or subcutaneously (SC) (infusion or injection), per oral (de Gioannis and de Leo, 2014), intranasal (Galvez et al., 2018; Popova et al., 2019), sublingual or per rectal (Andrade, 2019). The various routes of administration lead to differences in bioavailability and pharmacokinetic profile, though the relationship between drug pharmacokinetics and AD response is not well understood. The comparable efficacy of these various routes of administration has not been established. From our prior studies utilising the IV, IM, SC and intranasal routes, we decided to use the SC route in a multicentre study (ACTRN12616001096448) and compassionate treatment programme, based on prior findings of relatively few side effects and good efficacy (Galvez et al., 2018; George et al., 2017; Lai et al., 2014; Loo et al., 2016). We note that in NSW, injections of S8 drugs should be given by a medical practitioner or registered nurse.
Dose-titration method
In a series of double-blind studies examining an individualised dose-titration approach, each participant was initially treated at 0.1 mg/kg IV with dosage increased in subsequent sessions (by 0.1 mg/kg each step) until response and remission were achieved (George et al., 2017; Lai et al., 2014; Loo et al., 2016). These studies demonstrated the important principle that the mg/kg dose required for response and remission differed between individuals, suggesting an individual dose-titration approach is necessary. The same principle is evident in the large studies of intranasal esketamine. Fixed-dose schedules examining repeated dose esketamine (in conjunction with standard AD medication) did not yield statistically significant results when compared to standard AD with active placebo (Fedgchin et al., 2019; Ochs-Ross et al., 2020). However, the same drug formulation, when used with a flexible dosing approach with patients able to receive an increased dose depending on response (Popova et al., 2019) – resulted in a statistically significant reduction in depression scores compared to placebo, again given in conjunction with newly commenced AD medication.
Based on our prior studies, we developed a dosing approach for SC ketamine. The first aspect of our dose-titration method is to determine the optimal dose for an individual patient. This is undertaken by assessing the patient clinically and using a standardised depression rating scale (e.g. Montgomery–Asberg Depression Rating Scale [MADRS]; Montgomery and Åsberg, 1979) before each treatment session. Based on the change in scores between sessions, and tolerability of ketamine at the previous session, we determine the dose at subsequent sessions based on an ascending dose titration schedule with five levels (see Table 1), starting at the standard dose of 0.5 mg/kg and with subsequent levels, i.e. level 2 (0.6 mg/kg), level 3 (0.75 mg/kg), level 4 (0.9 mg/kg) and level 5 (1.0 mg/kg). If the MADRS score has decreased between sessions, then the dose typically continues at the same level. If the MADRS score has not improved or it has increased, consideration is given to increasing the dose to the next level. Such a decision also takes into account safety and tolerability, e.g. if ketamine led to medically significant adverse effects (e.g. rise in BP) or was poorly tolerated in the previous session, continuing at the same dose or reducing dose may be advisable.
Prescribing guide.
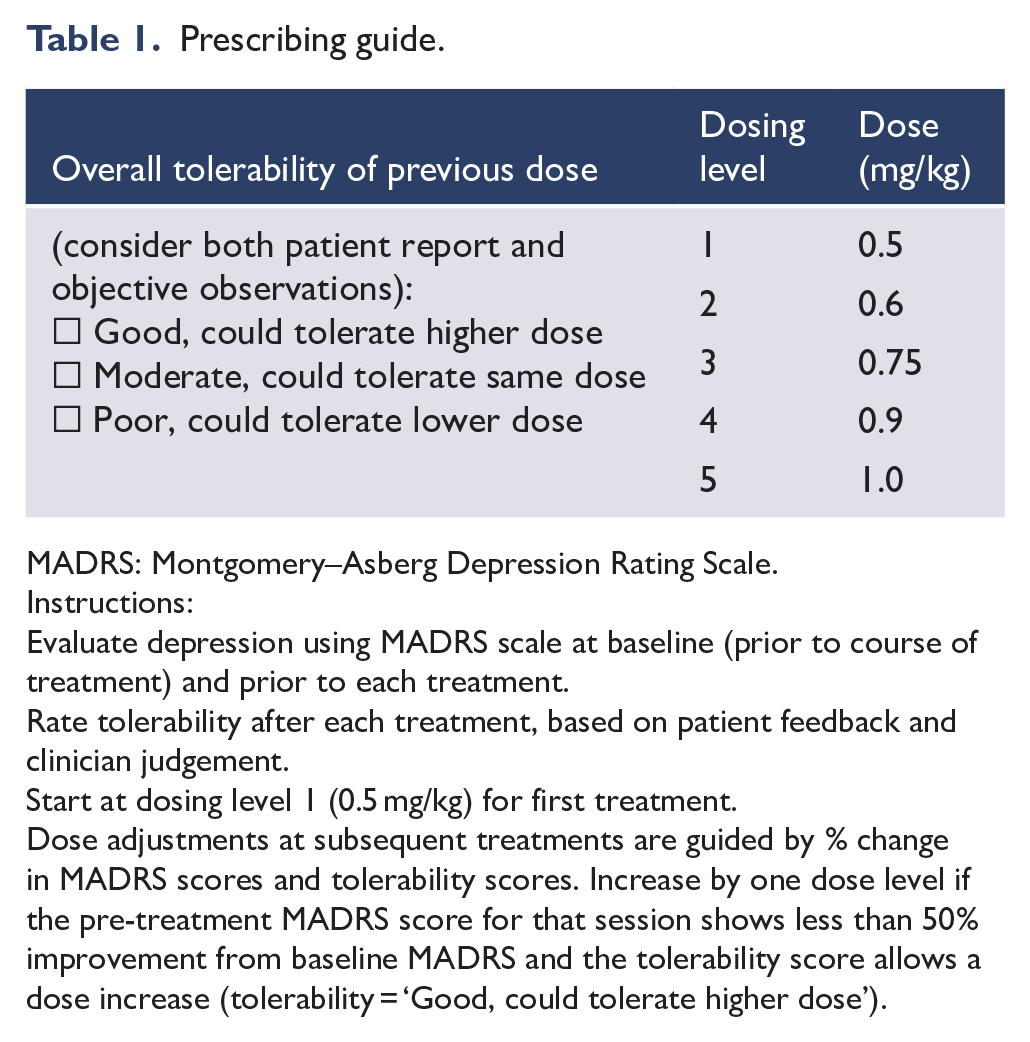
MADRS: Montgomery–Asberg Depression Rating Scale.
Instructions:
Evaluate depression using MADRS scale at baseline (prior to course of treatment) and prior to each treatment.
Rate tolerability after each treatment, based on patient feedback and clinician judgement.
Start at dosing level 1 (0.5 mg/kg) for first treatment.
Dose adjustments at subsequent treatments are guided by % change in MADRS scores and tolerability scores. Increase by one dose level if the pre-treatment MADRS score for that session shows less than 50% improvement from baseline MADRS and the tolerability score allows a dose increase (tolerability = ‘Good, could tolerate higher dose’).
Singh et al. (2016) in a repeated dosing study found thrice weekly ketamine dosing at 0.5 mg/kg IV no more effective than twice weekly dosing. Twice weekly dosing is therefore recommended for the acute treatment phase, ideally spaced throughout the week. This twice weekly frequency is continued, with dosing based on improvement in depression as well as tolerability, and continued ideally until the patient enters remission (e.g. MADRS < 10 on several occasions).
Once the optimal dose is found for a patient, the second aspect of our dose-titration method is to alter dosing frequency. Dose is held constant while the frequency of dosing is adjusted, in the first instance reducing to once weekly. If remission persists, then consideration can be given to a further reduction to fortnightly dosing, with this continued for ongoing maintenance treatment. Should the patient relapse, frequency of dosing should revert to the prior regimen, indicating a shift back to an acute course of treatment.
An acute treatment course may need to be ceased if there is a lack of response. As ketamine has an individualised dose–response profile, an adequate trial would require treatment sessions at a range of ascending doses. Treatment should be aborted if there is a failure of an adequate response or if patients cannot tolerate therapeutic doses.
Side effects and safety monitoring
Ketamine has potential to cause acute physical, psychiatric, psychotomimetic and cognitive side effects following a single dose and cumulative side effects resulting from repeated dosing. As per the systematic review by Short et al. (2018), the majority of studies of ketamine in depression have examined side effects only acutely and after a single dose, with limited studies examining safety in repeated doses. Acute physical side effects may include sedation, light-headedness, dizziness, poor coordination, headache, nausea and vomiting – all of which tend to be self-limiting. Cardiovascular changes, including transient elevation in HR and BP, may also occur acutely post-dose (Wan et al., 2015; Zarate et al., 2006). The most common psychiatric SEs include anxiety, agitation, irritability and mood elevation, which tend to occur acutely. Psychotomimetic symptoms, which may include dissociation, disorganised thought, altered perceptions (e.g. hallucinations or illusions), emotional withdrawal and suspiciousness, tend to be transient and with severity dependent on dosage and route of administration. Cognitive changes may include working memory deficits, poor concentration, confusion and diminished mental capacity (Krystal et al., 1999; Larkin and Beautrais, 2011; Short et al., 2018).
Across a course of treatment, there may be side effects arising from cumulative exposure to ketamine, including hepatotoxicity and bladder dysfunction (Katalinic et al., 2013; Short et al., 2018). Transient elevations in LFTs may not preclude ongoing treatment, but should be monitored. If LFTs are severely abnormal, treatment may need to be discontinued. Urinary changes may include increased frequency, dysuria or incontinence. Repeated use of ketamine at doses 5–30 times higher than used in our treatment programme have been found to lead to lower urinary tract pathology, including cystitis (Shahani et al., 2007; Short et al., 2018). Additionally, symptoms of substance dependence may arise and should be enquired about. It remains unclear whether there may be cumulative negative effects on cognition, with a lack of quality data in controlled medical settings (Short et al., 2018), though with such adverse effects observed in chronic recreational users of ketamine (Morgan et al., 2010). Given this uncertainty, it would be prudent for clinicians treating patients with ketamine to assess cognition. Limited studies have assessed side effects across a course of maintenance therapy. Wajs et al. (2020) in a large open label maintenance study of esketamine with oral AD (with a median exposure of 23 weeks) found no cases of interstitial cystitis. There were reported cases of cystitis which resolved despite continuing esketamine treatment, a case of frequent urination requiring dose reduction and urinary incontinence requiring discontinuation (Wajs et al., 2020). Cognitive performance was found to be either improved or stabilised and there were no cases of drug-seeking or abuse (Wajs et al., 2020).
A systematic approach should be implemented to monitor the above symptoms (e.g. KSET; Short et al., 2020), which measures both acute and cumulative SEs. During a treatment session, we screen for potential acute symptoms pre-dose and post-dose (e.g. at 1 hour) to check for emergent side effects (e.g. dissociation), and then at 2 hours to assess whether these have resolved prior to discharge. We recommend cardiovascular monitoring including BP and HR at pre-treatment and at regular intervals during treatment. A medical officer should be informed if BP or HR is elevated for prolonged periods of time (e.g. systolic > 180 mmHg and/or diastolic > 110 mmHg over 30 minutes) or if emergent symptoms are clinically concerning. As there are no significant changes in oxygen saturation in patients administered ketamine (Wan et al., 2015), we do not routinely monitor oxygen saturation across a treatment episode unless there are reasons for concern (e.g. patient is very sedated, has sleep apnoea or a comorbid pulmonary condition – which may lead to desaturation). Overall tolerability of a ketamine session should be noted which can inform dose adjustment at the next session. Following a treatment session, it is important to assess the patient is safe for discharge. Using the KSET structured measure, this occurs by examining whether orientation, BP, HR and any acute emergent symptoms have returned to safe and acceptable levels. In a ketamine naïve patient receiving their first treatment session, we recommend monitoring for up to 4 hours post-dose. For subsequent sessions, we recommend monitoring for up to 2 hours post-dose, though this may have to be extended if the discharge criteria are not met.
We monitor for cumulative side effects across a course of treatment, by reviewing the patient prior to administering each treatment (KSET Acute Treatment form) as well as periodic evaluations of liver function, cognition and urinalysis (KSET Follow Up form). The KSET Acute Treatment form enquires about symptoms prior to subsequent treatments, checking for emerging cumulative side effects which may warrant further enquiry and investigation before the treatment course continues – urinary symptoms, features of dependence, psychiatric changes (Short et al., 2020). Repeat cognitive testing to compare with the pre-treatment assessment may be undertaken if clinically indicated.
We have detailed the KSET measure, though additional rating scales may also be used, including the Bladder Pain and Interstitial Cystitis Symptom Score (BPICSS) (Humphrey et al., 2012), Clinician Administered Dissociative States Scale (CADSS) (Bremner et al., 1998) and the psychotic symptoms subscale of the Brief Psychiatric Rating Scale (BPRS) (Overall and Gorham, 1962), noting that the latter two were not designed to detect psychotomimetic effects of ketamine and may not be sensitive to these.
Duration of treatment
More recently, studies have examined a course of esketamine treatment (e.g. Daly et al., 2019). After initial improvement during the acute treatment course, ongoing maintenance treatment with esketamine reduced relapse rates compared to maintenance placebo treatment, both given in conjunction with conventional ADs (Daly et al., 2019). An open label study of esketamine commenced concurrently with an oral AD (Wajs et al., 2020) found at 48 weeks, 76.5% were responders and 58.2% were remitters. Prescribing needs to adhere to local regulatory guidelines (see Bayes et al., 2019) though in light of emerging safety and efficacy data, it is possible state regulations may be revised. Duration of treatment is also affected by factors such as efficacy, tolerability and cost to the patient. Some patients may be able to withdraw from maintenance treatment if they achieve a sustained response with gradual reduction in the frequency of ketamine treatments; however, there is no data on this. The limited data on the safety of long-term treatment (Short et al., 2018) means that careful monitoring for cumulative side effects is essential if repeated dosing occurs.
Post-treatment phase
Following a course of treatment, it is advised that follow-up of the patient continues. As relapse may occur following cessation of an acute course of ketamine, we recommend maintenance treatment with an AD to reduce the probability of relapse (similar to recommendations for ECT). Patients should be monitored for a worsening of their mood and suicidality upon cessation of ketamine, with the suggestion the latter may be the result of withdrawal symptoms (Schatzberg, 2019). Our model provides follow-up at 2 weeks and 1, 3, 6 and 12 months (in addition to the patient’s regular clinical care) where we undertake a clinical assessment, MADRS, global assessment of functioning (GAF) (APA, 2002), safety monitoring, repeat cognitive testing if indicated, as well as LFTs and urine dipstick if previously abnormal or symptoms suggest their use (KSET Follow Up form). Following a period of remission after an acute course of ketamine, patients may relapse. In this instance, they could be reassessed for potential to have a further acute course of ketamine.
Conclusion
Ketamine’s profile as a rapidly acting AD with specific acute and cumulative SEs, unique dosing requirements, risk of dependence and specific restrictions around its prescription in Australia and New Zealand, mean that a model of care for ketamine therapy in depression is timely. We detail our model based on a ‘compassionate use’ ketamine programme which developed out of our clinical research programme. We detail the current regulatory framework and professional bodies’ recommendations regarding ketamine and discuss clinical governance and infrastructure considerations. We then describe our four-step model comprising the initial assessment, pre-treatment, treatment and post-treatment phases. Critical to the model are thorough initial psychiatric and medical assessments to assess patient suitability, a rigorous consenting process and structured safety monitoring across an acute treatment course or maintenance therapy. We also detail our dose-titration method which allows for a logical and sequential method to flexibly dose patients across a treatment course so that they receive individualised therapy. Such a model of care aims to bridge the gap between efficacy studies and clinical care outside of research settings as ketamine and related compounds become increasingly important therapies for TRD.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
