Abstract
Objective:
Heart rate variability, a quantitative measure of mainly parasympathetic activity, has been applied in evaluating many types of psychiatric and neurological disorders, including dementia (or neurocognitive disorders). However, although dementia patients often showed significantly lower heart rate variability (various indices) than healthy controls, and different types of dementia had distinct heart rate variability features, the results were not identical across studies. We designed a systematic review and meta-analysis for incorporating data from different studies.
Methods:
We gathered studies comparing heart rate variability in patients with dementia and in healthy controls. Heart rate variability was analysed in several ways: parasympathetic function in hierarchical order (main analysis), total variability, comparison of heart rate variability between different subtypes of dementia, specific indices of heart rate variability and heart rate variability reactivity.
Results:
In the initial search, we found 3425 relevant articles, from which 24 studies with a total of 1107 dementia patients and 1017 control participants finally entered the main meta-analysis. The dementia patients had a significantly lower resting heart rate variability for parasympathetic function (Hedges’ g = −0.3596, p = 0.0002) and total variability (Hedges’ g = −0.3059, p = 0.0002) than the controls. For diagnostic subgroup analysis relative to the controls, heart rate variability was significantly lower in patients with mild cognitive impairment (Hedges’ g = −0.3060) and in patients with dementia with Lewy bodies (Hedges’ g = −1.4154, p < 0.0001). Relative to patients with Alzheimer’s disease, heart rate variability in patients with dementia with Lewy bodies was significantly lower (Hedges’ g = −1.5465, p = 0.0381). Meta-regression revealed that gender proportion was significantly associated with effect size.
Conclusion:
Our results show that dementia patients (especially those with dementia with Lewy bodies and mild cognitive impairment) have lower parasympathetic activity than healthy people. The influence of gender on the results should be carefully interpreted.
Keywords
Introduction
Dementia or neurocognitive disorders (NCDs) form a group of disorders that present with cognitive impairment (American Psychiatric Association, 2000, 2013). The disease course of primary dementia is irreversible and has a huge impact on both personal functioning and global socioeconomic burden (Wimo et al., 2013). The symptoms of dementia originate from neurodegeneration or injury of the brain, and different types of dementia have distinct mechanisms of brain injury that involve various brain regions (American Psychiatric Association, 2013). Although cognitive deterioration is the major presentation for diagnosing dementia/NCDs, biomarkers such as neuroimaging (which detects regions of brain atrophy and vascular lesions) and cerebrospinal fluid examinations are important for differentiating distinct dementia subtypes (American Psychiatric Association, 2013). Among the various categories of psychiatric diagnoses, dementia/NCDs is one with abundant information on biomarkers (Maier, 2016). However, many of the biomarkers involved in neuroimaging are not equally accessible globally (Health Quality, 2014). Whether there is any convenient non-invasive index reflecting biological mechanisms for diagnosing dementia or differentiating its various subtypes is a clinically meaningful topic.
Heart rate variability (HRV) is an estimation of autonomic control of the heart and its association with dementia is worthy of discussion (Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology, 1996). HRV has been investigated in many mental disorders, such as depression, anxiety, schizophrenia, alcohol use disorder and somatic symptom disorder; low HRV is a common finding in these disorders (Alvares et al., 2016; Cheng et al., 2019, 2020). HRV is often viewed as the main indicator of parasympathetic activity, which in turn is considered to be associated with subjective well-being and psychological flexibility (Colzato et al., 2018). The root mean square of successive differences (RMSSD), high-frequency power (HF) and respiratory sinus arrhythmia (RSA) are often considered as parasympathetic-specific indices (Cheng et al., 2020). Furthermore, some components of HRV reflect both sympathetic and vagal modulations (such as low-frequency power, LF) or oscillatory breathing and cardiorespiratory coupling, vasomotor activity and thermoregulation (such as very-low-frequency power) (Penzel et al., 2016). Sympathetic modulation of the heart is especially important in some diseases, such as heart failure (Diaz et al., 2020). From a mental perspective, dementia can be thought of as similar to other psychiatric disorders with maladaptation and subjective torment; this is a potential pathway for connecting dementia with low HRV (Bar, 2015). From a physiological perspective, low HRV reflects reduced vagal activity, which can also be associated with dementia. For example, deficiency of the cholinergic system plays a major role in the cognitive decline of Alzheimer’s disease (AD). Acetylcholinesterase inhibitors can be used as cognitive enhancers for AD patients (Calabria et al., 2009) as acetylcholine is the main neurotransmitter of the parasympathetic nervous system (Burnstock, 2009). Furthermore, several types of dementia present with autonomic failure, such as multiple system atrophy (Lipp et al., 2009). Both multiple system atrophy and Parkinson’s disease belong to the α-synucleinopathy group and have shared pathophysiological features; Parkinson’s disease with dementia (PDD) and dementia with Lewy bodies (DLB) have also been reported to show dysautonomia (Stubendorff et al., 2012). The above information indicates that HRV could be helpful for diagnosing dementia and its subtypes.
HRV presentations in the dementia population have been investigated (Bar, 2015), but the methods of measurement and the study populations were heterogeneous across studies. Some studies analysed HRV according to pure heart rate signals and adopted HRV indicators such as the standard deviation of normal-to-normal RR intervals (SDNN), RMSSD, HF and total power (Kim et al., 2006; Zulli et al., 2005). Respiratory signals were considered as HRV indicators in other studies, with RSA being the most common (Jensen-Dahm et al., 2015). Lower HRV in dementia patients than in healthy individuals has been reported in association with most of the above HRV indices. Considering the situations of HRV measurement, resting-state (baseline) HRV and HRV reactivity can be distinguished; HRV reactivity means the HRV was measured under situations other than the resting state, such as change of posture, doing exercise, performing tasks or watching videos (Cheng et al., 2020). Regarding the study population, most studies compared HRV values in dementia patients and controls and the subtypes of dementia included AD, vascular dementia (VaD), frontotemporal dementia (FTD), DLB, PDD and mild cognitive impairment (MCI) with a lower level of cognitive decline; the comparisons of HRV in different subtypes of dementia were executed in several studies (Allan et al., 2007; Issac et al., 2017; Kim et al., 2018), but the results were not always identical. Therefore, we believe that incorporating these data using a meta-analytical approach may generate a clearer result regarding the autonomic features of dementia. A comparison of HRV among patients with different types of dementia and also between patients with different types of dementia and a healthy population can also be investigated in the meta-analysis.
This systematic review and meta-analysis was designed in light of the above information. The major aims of this study were to compare HRV for the following populations and situations: (1) resting HRV in dementia/NCD patients (including MCI/mild NCDs) and controls (main analysis); (2) resting HRV in dementia/NCD subtypes (such as AD, VaD, FTD, DLB, MCI) and controls; (3) resting HRV in different subtypes, such as DLB versus AD; (4) resting HRV in dementia/NCD patients and controls using specific HRV indices (such as only HF or only RMSSD); and (5) HRV reactivity in dementia/NCD patients and controls. Furthermore, the potential influence of age, gender and Mini-Mental State Examination (MMSE) score on the main analysis was analysed with meta-regression.
Methods
Data sources and search strategy
This systematic review and meta-analysis was prepared according to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) statement guidelines (Moher et al., 2009). We conducted an electronic literature search of PubMed, Medline, EMBASE, PsychINFO and PsycARTICLES from the earliest available date to February 2020. The literature search was conducted using the following search terms/keywords without any limits: (‘neurocognitive disorder’ OR ‘dementia’ OR ‘alzheimers’ OR ‘dementia with lewy bodies’ OR ‘dementia with Parkinson’s diseases’ OR ‘frontotemporal dementia’ OR ‘vascular dementia’ OR ‘mild cognitive impairment’ OR ‘cognitive impairment’ OR ‘memory impairment’) AND (‘heart rate variability’ OR ‘HRV’ OR ‘vagal nerve activity’ OR ‘autonomic nervous system’ OR ‘autonomic activity’ OR ‘vagal parasympathetic’ OR ‘respiratory sinus arrhythmia’). All titles meeting the inclusion criteria were retrieved and reviewed in full text. Original studies reporting on HRV in individuals with dementia/NCDs were eligible for review. HRV and other reported HRV measures, such as RSA, were included as the primary outcome for meta-analysis. Additional eligible studies were sought by searching the reference lists from primary articles and relevant reviews to identify any further studies that were not found with the electronic search. Figure 1 summarizes the review flowchart in accordance with the PRISMA statement (Moher et al., 2009).
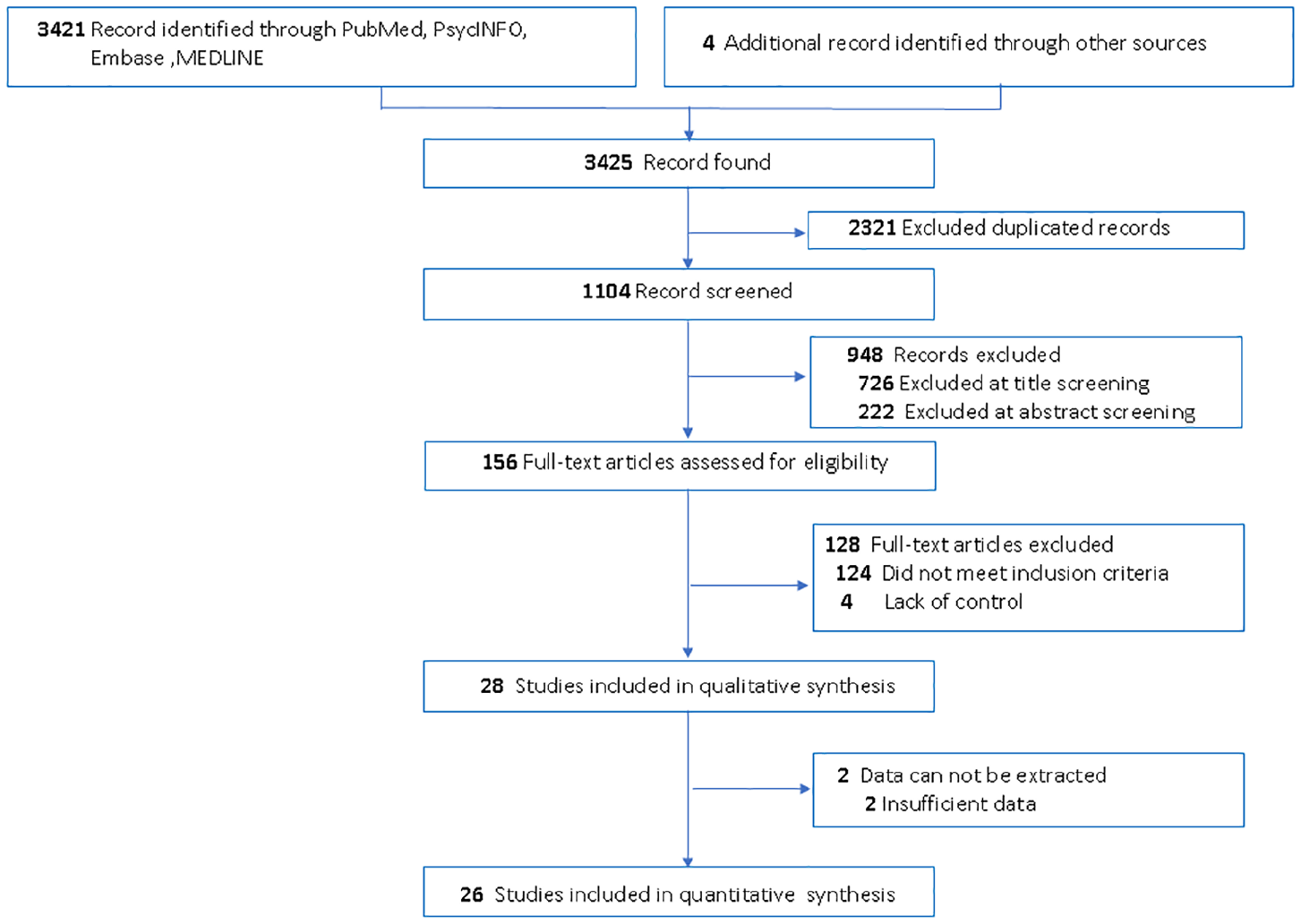
Flow diagram of the study selection process for the systematic review and meta-analysis. In the initial search 3425 relevant articles were found, from which 26 studies entered the quantitative meta-analysis (24 studies with a total of 1107 dementia patients and 1017 controls entered the main analysis; 2 studies were about the comparison of HRV in patients with different dementia subtypes).
Inclusion and exclusion criteria
The primary eligibility criterion was comparison of HRV in subjects with any type of dementia/NCD and in healthy controls, or comparison of HRV between different subtypes of dementia/NCD. The diagnosis of different types of dementia/NCD was defined via clinical evaluation and neuropsychological tests as: AD, defined by the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition; the International Statistical Classification of Diseases and Related Health Problems, Tenth Revision; and criteria of the National Institute of Neurological Communicative Diseases and Stroke along with the Alzheimer’s Disease and Related Disorders Association; VaD, defined by the National Institute of Neurological Disorders and Stroke along with the Association Internationale pour la Recherche et l’Enseignement en Neurosciences; PDD, defined by the United Kingdom Parkinson’s Disease Society Brain Bank; DLB, defined by DLB consensus criteria; FTD, defined by international consensus criteria for behavioural variant FTD; and MCI, defined by Mayo Clinic MCI criteria. In 18 included studies with MMSE data, the mean MMSE score is 21.62 (median = 22.7, range = 6.77–26); detailed data are shown in Supplementary Table 1. Furthermore, studies that reported the mean and standard deviation (SD) of at least one HRV index or provided data to estimate these values (e.g. confidence interval [CI], interquartile range) were also included. HRV data with recording length between 3 minutes and 24 hours and analyses from electrocardiogram or RR time series data and for different positions (supine, sitting or standing) were all included; detailed information is shown in Supplementary Table 1. Exclusion criteria were designs such as case series, case reports and animal studies and also cross-sectional studies without comparison groups.
Data extraction and quality assessment
We independently extracted the relevant information from the included studies and evaluated the methodological quality of eligible trials using the Newcastle-Ottawa Scale (Stang, 2010). Information on the studies included first author, publication year, country, study design, number of dementia/NCD subjects and controls, age, gender distribution, HRV indices and diagnostic criteria. Details on HRV were also obtained, including length and condition of HRV recording (e.g. baseline or reactivity). Authors who reported HRV but did not provide sufficient quantitative data (e.g. only a graphical display) were contacted to request the necessary information in order to derive effect size estimates and CIs for the selected indices. The Newcastle-Ottawa Scale is an eight-item scale with a version for assessing the quality of non-randomized studies in meta-analyses. This scale evaluates the domains of selection, comparability and outcome or exposure. Studies awarded seven or more stars were considered to be high-quality studies. All potentially relevant manuscripts were reviewed independently by two investigators (Y.-C.C. and Y.-C.H.) and areas of disagreement or uncertainty were adjudicated by a third investigator (W.-L.H.).
Outcome measures
We defined the resting HRV of any measure reflecting parasympathetic function as its primary outcomes in hierarchical order: RSA, HF (absolute, logarithmically transformed and normalized values), RMSSD, total power (absolute and logarithmically transformed values) and SDNN. We also searched articles adopting non-linear HRV indices (such as SD1, SD2, entropy) but did not find any relevant studies. The advantage of a hierarchical approach is the opportunity to include a large amount of data, as has been adopted in previous meta-analyses (Alvares et al., 2016; Cheng et al., 2019). Secondary outcomes were defined as indices reflecting the total variability (HRV indices in the full duration of measurement, not considering frequency; for example, total power, SDNN) of HRV (Cheng et al., 2019, 2020). Furthermore, distinct resting HRV indices (e.g. only HF, only RMSSD, only SDNN, only RSA) were analysed separately for examining the results with a lower sample size but higher homogeneity (Cheng et al., 2020; Koch et al., 2019; Koenig et al., 2016). For HRV reactivity outcomes, the post-challenge HRV indices reflecting parasympathetic functions were grouped to evaluate the differences between dementia/NCD patients and healthy subjects. Furthermore, for investigating the sympathetic activity of dementia/NCD patients, we also analysed LF, LF/HF and the sympathetic-specific measurement skin conductance (both level and rate) (Cheng et al., 2020; Kuo et al., 1999).
Data synthesis and statistical analysis
We performed a pooled estimate of HRV in dementia/NCD individuals compared to controls or compared to another dementia/NCD subtype. For indices reflecting different HRV categories, individual meta-analyses were performed. Where studies reported more than one index of HRV, hierarchical inclusion criteria were implemented to prevent conflation of effect size estimates (Alvares et al., 2016; Cheng et al., 2020). To estimate the true effect size across the different studies with variable reported units, standardized mean differences were calculated using Hedges’ g, along with the 95% CI. Hedges’ g is related to Cohen’s d and can be interpreted using the same conventions, with values of 0.2, 0.5 and 0.8 indicating small, medium and large effect sizes, respectively (Cohen, 1988). An added benefit of Hedges’ g is correction for biases found in small sample sizes. Both absolute and log-transformed values were included in the calculation of effect sizes. When only the standard error of the mean was reported, SD was calculated by multiplying this by the square root of sample size. In seven studies, no data were retrieved on SDs, only range and interquartile range, but according to the Cochrane guidelines SDs may be estimated from both of these (Higgins and Green, 2011; Hozo et al., 2005).
Possible sources of heterogeneity or inconsistency among trials in the magnitude or direction of effects were investigated. Heterogeneity was investigated using the I2 test (Higgins and Thompson, 2002). A random effect model was used to assume that the true effect size could vary among studies and thus offer more generalizable results than a fixed effect model. Publication bias was examined using a funnel plot, depicting the effect size against the standard error for asymmetry. Egger’s regression test was also used to assess publication bias (Egger et al., 1997).
We conducted planned subgroups and moderator analysis to identify the possible heterogeneity. Subgroup analysis was stratified based on different subtypes of dementia/NCD and individual HRV indices. To explore the potential effect of trial-level modifiers on the result of the main analysis, we considered several covariates in meta-regression approaches, conducted with one covariate at a time. The five covariates were mean age, MMSE score, male proportion, risk of study bias and duration of illness.
To investigate the potential confounding effects of any outliers within the recruited studies, a leave-one-study-out sensitivity analysis was performed by sequential exclusion of each trial at one time and calculating the pooled standardized mean difference for the remaining studies. If omitting one study at a time did not significantly alter the pooled results, this suggested good robustness of the study. All meta-analytic computations were performed using R software (using meta package version 3.5.2).
Results
Baseline characteristics of included studies
Of the 3425 records screened, 28 studies met the inclusion criteria. All included studies were case-control studies, with 26 articles fulfilling the eligibility criteria. Among them, 24 articles provided detailed data on resting HRV between NCD subjects (n = 1107) and age-matched controls (n = 1017); mean ages were 70.60 years (range = 62.0–79.6) for the NCD individuals and 70.87 years (range = 62.4–78.3) for the controls. Of the 26 studies with detailed data for analysis, the Newcastle-Ottawa Scale scores ranged from 6 to 9. A summary of included studies for meta-analysis and their results is presented in Supplementary Table 1.
Meta-analyses of resting HRV in individuals with dementia/NCD
The results of each meta-analysis are summarized in Table 1 (detailed results of each analysis are shown in Figures 2, 3 and Supplementary Figures 1–16). Twenty-four studies were extracted for synthesis of the main effect. In the main analysis, resting HRV was significantly lower in dementia/NCD patients as a whole compared to their control counterparts (24 studies: Hedges’ g = −0.3596, 95% CI = [−0.5520, −0.1672], p = 0.0002) (Figure 2). Visual inspection of the funnel plot and Egger’s regression test revealed no evidence of publication bias (p = 0.1028). Of the studies included for total variability, HRV was significantly lower in individuals with dementia/NCDs (13 studies: Hedges’ g = −0.3059, 95% CI = [−0.4643, −0.1475], p = 0.0002), but the funnel plot and Egger’s regression test indicated publication bias (p = 0.0433) (see Supplementary Table 2 and Supplementary Figure 1). For subgroup analysis of specific indices, dementia/NCD subjects exhibited significantly lower HF (18 studies: Hedges’ g = −0.3541, 95% CI = [−0.5837, −0.1245], p = 0.0025), RMSSD (11 studies: Hedges’ g = −0.2913, 95% CI = [−0.5624, −0.0201], p = 0.0353), SDNN (10 studies: Hedges’ g = −0.2681, 95% CI = [−0.4215, −0.1148], p = 0.0006) and RSA (5 studies: Hedges’ g = 0.3845, 95% CI = [−0.6659, −0.1031], p = 0.0074). The funnel plot and Egger’s regression test for HF (p = 0.0433) and RMSSD (p = 0.0318) indicated publication bias (Supplementary Table 3 and Supplementary Figures 12 and 14). For subgroup analysis of LF/HF, no significant differences were found between dementia/NCD patients and healthy controls (14 studies: Hedges’ g = 0.0608, 95% CI = [−0.2149, 0.3365], p = 0.6655).
Meta-analysis results of HRV in patients with NCDs.
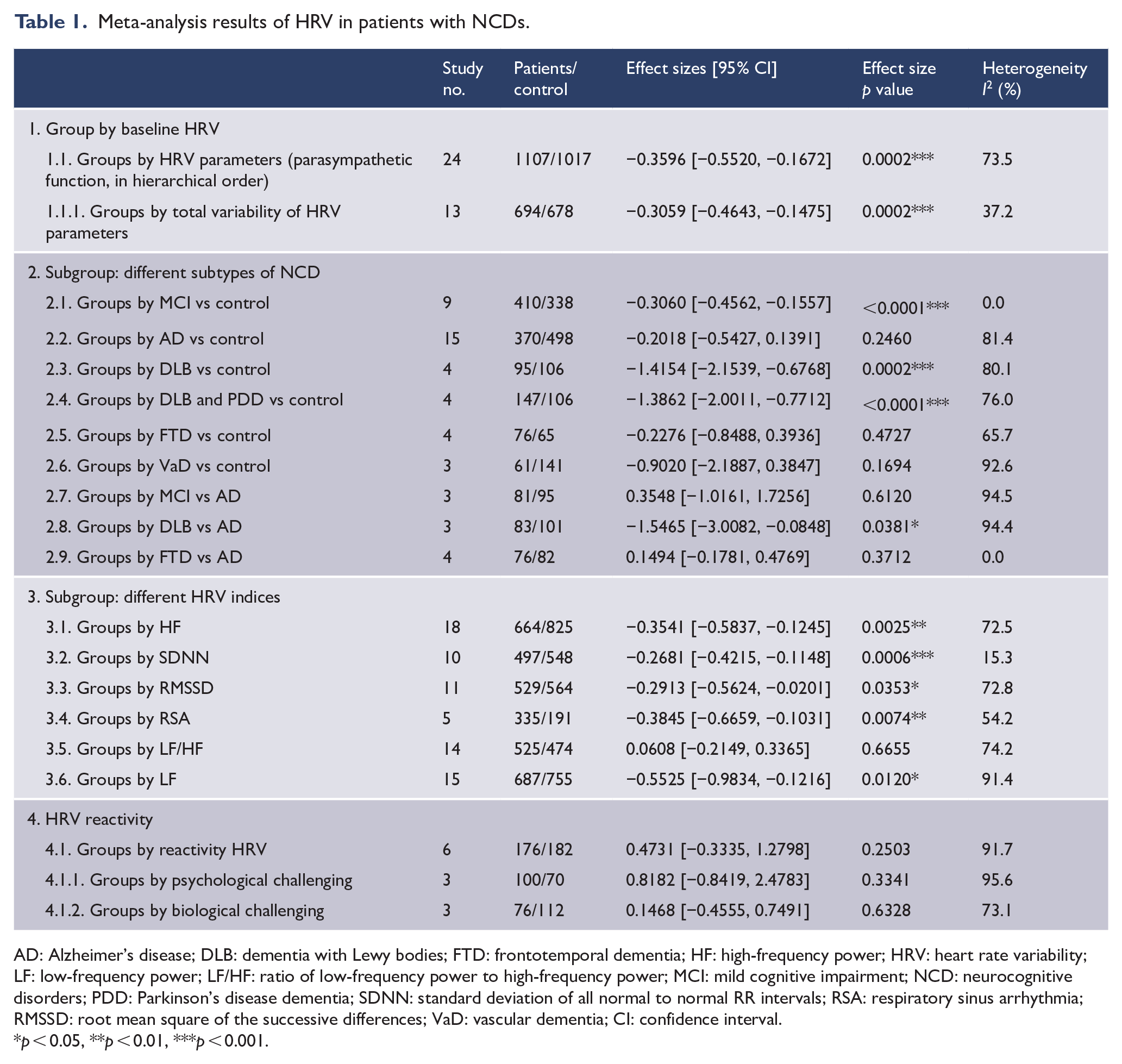
AD: Alzheimer’s disease; DLB: dementia with Lewy bodies; FTD: frontotemporal dementia; HF: high-frequency power; HRV: heart rate variability; LF: low-frequency power; LF/HF: ratio of low-frequency power to high-frequency power; MCI: mild cognitive impairment; NCD: neurocognitive disorders; PDD: Parkinson’s disease dementia; SDNN: standard deviation of all normal to normal RR intervals; RSA: respiratory sinus arrhythmia; RMSSD: root mean square of the successive differences; VaD: vascular dementia; CI: confidence interval.
p < 0.05, **p < 0.01, ***p < 0.001.
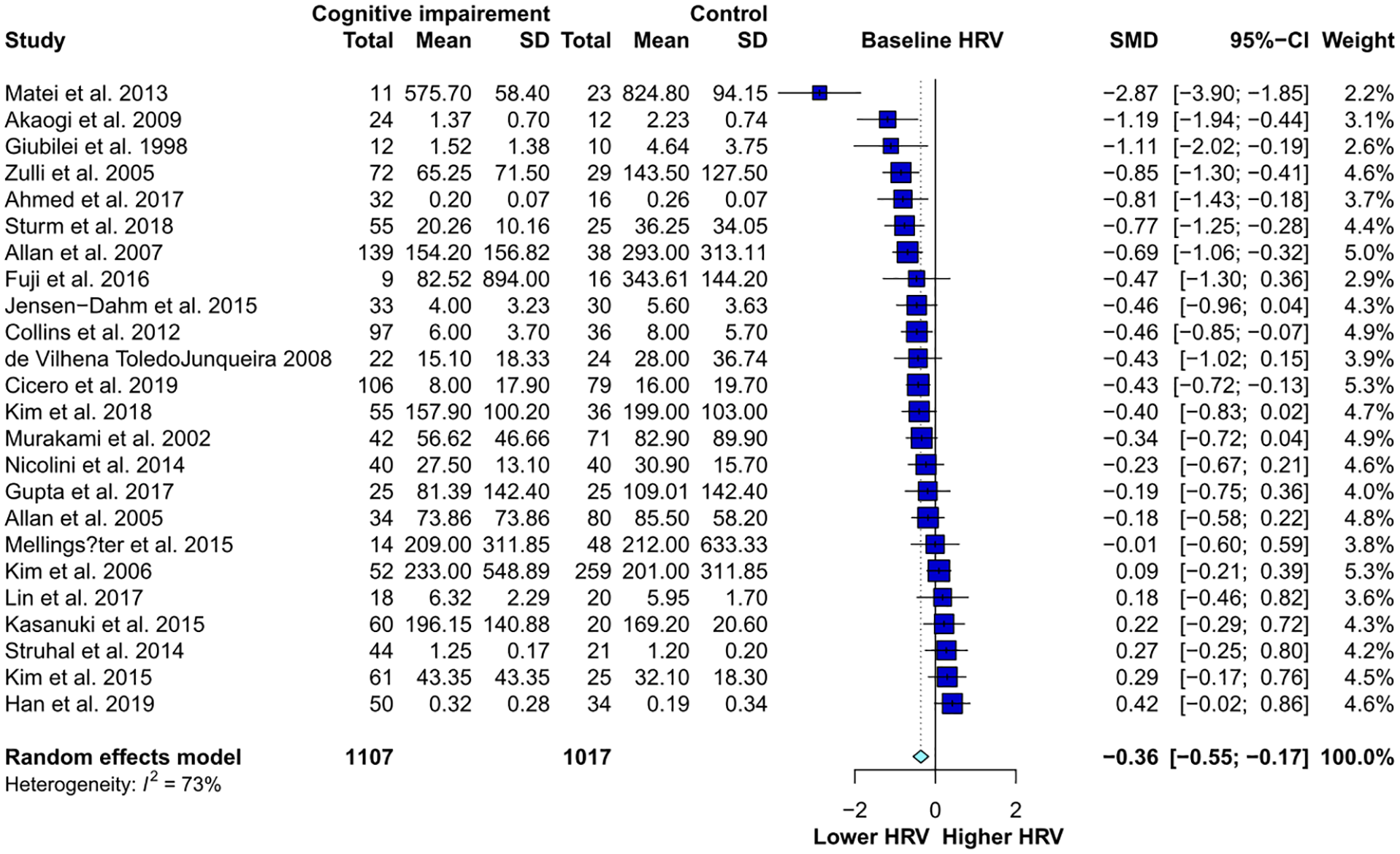
Comparison of HRV parasympathetic indices (in hierarchical order) in NCD patients and controls. HRV is significantly lower in NCD patients than in the controls.
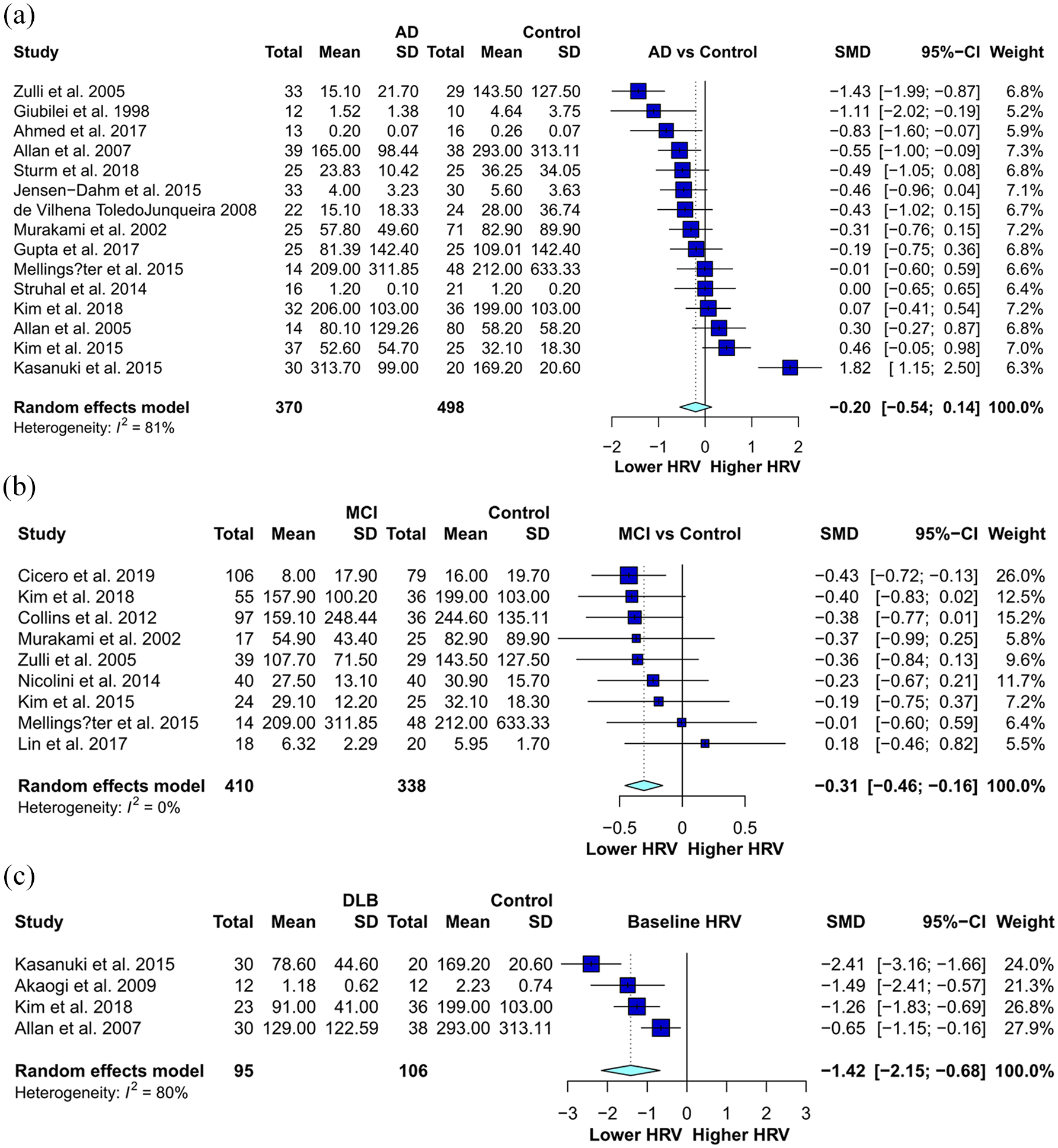
Comparison of HRV parasympathetic indices in hierarchical order: (a) in AD patients and controls there is no significant inter-group difference; (b) in MCI patients and controls the inter-group difference is significant; and (c) in DLB patients and controls there is a significant inter-group difference.
Subgroup analysis of resting HRV in different subtypes of dementia/NCD
Diagnostic subgroup analysis was repeated for comparison between different subtypes of dementia/NCD patients and healthy controls (AD vs control, VaD vs control, DLB with/without PDD vs control, FTD vs control, MCI vs control; Figure 3) and between different subtypes of NCD (DLB vs AD, FTD vs AD, MCI vs AD). Relative to healthy controls, HRV was significantly lower in patients with MCI (nine studies: Hedges’ g = −0.3060, 95% CI = [−0.4562, −0.1557], p < 0.0001) and in patients with DLB (four studies: Hedges’ g = −1.4154, 95% CI = [−2.1539, −0.6768], p = 0.0002). Combining DLB and PDD into one group, the pooled HRV remained significantly lower relative to healthy controls (four studies: Hedges’ g = −1.3862, 95% CI = [−2.0011, −0.7712], p < 0.0001), whereas the pooled HRV in AD (nine studies: Hedges’ g = −0.2018, 95% CI = [−0.5427, 0.1391], p = 0.2460), VaD (three studies: Hedges’ g = −0.9020, 95% CI = [−2.1887, 0.3847], p = 0.1694) and FTD (four studies: Hedges’ g = −0.2276, 95% CI = [−0.8488, 0.3936], p = 0.4727) was non-significant.
Compared to AD, resting HRV was significantly lower in patients with DLB (three studies: Hedges’ g = −1.5465, 95% CI = [−3.0082, −0.0848], p = 0.0381), whereas the pooled HRV did not reach statistical significance in patients with MCI (three studies: Hedges’ g = 0.3548, 95% CI = [−1.0161, 1.7256], p = 0.6120) or in patients with FTD (four studies: Hedges’ g = 0.1494, 95% CI = [−0.1781, 0.4769], p = 0.3712).
Meta-analyses of HRV reactivity in individuals with dementia/NCDs
Overall, HRV reactivity revealed no significant difference between subjects with dementia/NCDs and controls (Hedges’ g = 0.4731, 95% CI = [−0.3335, 1.2798], p = 0.2503; Table 1). On separating HRV reactivity into biological and psychological challenges, the result remained non-significant (Table 1).
Sensitivity analysis
The stability of the meta-analysis and subgroup analyses was tested through a leave-one-out sensitivity analysis; therefore, the following meta-analysis results should be treated with caution: subgroup analyses of RMSSD and RSA; subgroup analyses of comparisons between FTD and control, AD and control, DLB and AD, and FTD and control; and HRV reactivity during the biological challenge (Supplementary Table 3).
Meta-regression
The following moderating variables were examined: age, MMSE score, proportion of male participants, body mass index, risk of study bias and duration of illness. The proportion of male participants exhibited significant associations on Hedges’ g (gender: p = 0.0128, R2 = 31.74%), whereas the other covariates showed no significant associations (Table 2), indicating that gender may influence the main results.
Meta-regression of pre-defined variables of interest.
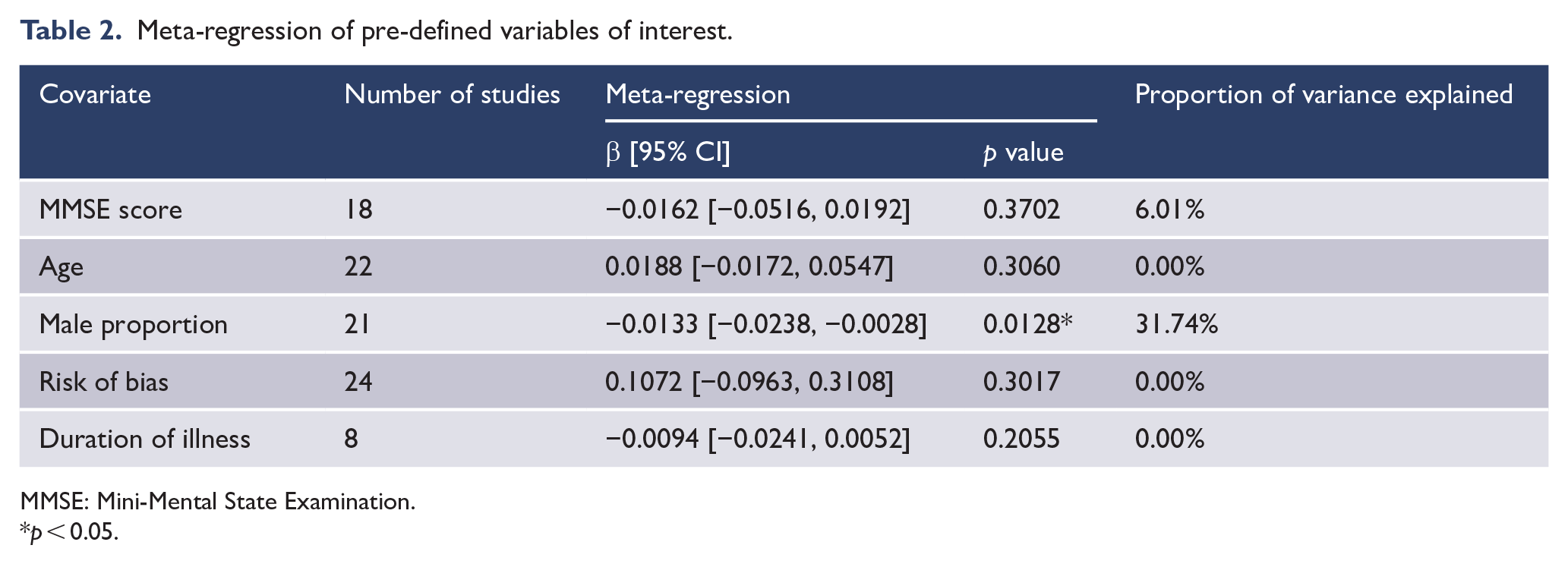
MMSE: Mini-Mental State Examination.
p < 0.05.
Discussion
For the several pre-selected aims, we have the following findings. First, compared with the controls, the dementia/NCD population revealed significantly lower HRV values. Second, separating different types of dementia and comparing with the controls, only the DLB and MCI populations showed significantly lower HRV; AD, VaD and FTD patients did not show significantly different HRV compared with healthy controls. Third, when comparing HRV between patients with different types of dementia, only the dyad DLB–AD revealed a significant difference (HRV in DLB subjects was lower). Fourth, on separating the different HRV indices, HF, RMSSD, RSA and SDNN were all significantly lower in the dementia group than in the healthy group. Fifth, HRV reactivity was not significantly different between the dementia and healthy populations. Finally, gender proportion is significantly associated with the effect size of the inter-group difference in meta-regression.
Combining the data from all eligible studies, the baseline HRV in dementia/NCD patients was significantly lower than that in the controls. There are several possible pathways to connect dementia and low HRV. Dementia causes reduced psychological flexibility and maladaptation, which may present with low parasympathetic activity (Bar, 2015). Impaired autonomic function is often found in cardiovascular diseases and may be a risk factor for developing dementia/NCDs (Justin et al., 2013). Vagal activity is considered to be related to the regulation of emotion and attention (Porges, 1995), both of which are involved with several prefrontal or associated brain regions, such as the anterior cingulate, orbitofrontal cortex, insula and amygdala (Etkin et al., 2006; Thayer et al., 2012). Thus, degeneration of any of the above brain regions may manifest as both cognitive decline and reduced HRV (Bar, 2015; Thayer et al., 2012). A previous review points out that high sympathetic and low parasympathetic activity is associated with cognitive impairment (Bar, 2015). Our present study provides further quantitative evidence for the parasympathetic connection with dementia. For detecting the contribution of DLB/PDD to low HRV in the main analysis, we removed studies on DLB and PDD and performed the analysis again. The result is shown in Supplementary Figure 17, where a significant inter-group difference can be seen. Therefore, the lower HRV in patients with dementia than in controls seems robust and does not originate just from the DLB/PDD effects.
After separating patients with different types of dementia/NCDs, not all of them showed significantly lower HRV than the healthy population. The strongest HRV-lowering effect is related to DLB/PDD and MCI. Among the neurodegenerative disorders, multiple system atrophy, DLB and Parkinson’s disease are those frequently reported to have autonomic failure (Koga et al., 2015); therefore, the significantly lower HRV in DLB/PDD patients than in controls was not a surprising finding. In those diseases with α-synucleinopathies, the dysautonomia in multiple system atrophy is the most obvious and can present with orthostatic hypotension and bladder and sexual dysfunction (Kirchhof et al., 2003). The autonomic dysfunction in Parkinson’s disease and DLB is usually not as severe as that in multiple system atrophy; therefore, some autonomic examinations (such as the thermoregulatory sweat test) are used in the clinical field to distinguish Parkinson’s disease from multiple system atrophy (Lipp et al., 2009). On the contrary, AD patients did not show significantly lower HRV than the controls in our analysis. This implies that the dysautonomia in DLB/PDD is more obvious than in AD. DLB/PDD and AD have some shared mechanisms, such as the change of cholinergic transmission, but the affected cholinergic receptors and brain regions are not totally the same (Bertrand and Wallace, 2020; Francis and Perry, 2007).
The nucleus ambiguus, dorsal motor vagal nucleus, solitary nucleus and ventrolateral medulla are the main brainstem regions of autonomic control (Agassandian et al., 2002). These structures are affected by alpha-synuclein immunoreactive inclusion pathologies in Parkinson’s disease and DLB (Kay et al., 2015) but seem not to be common regions affected by the pathologies of AD, which may explain why the low HRV in PDD/DLB patients is more obvious than in AD patients. As for the sympathetic modulation, Toledo et al. (2019a) found that in patients with heart failure, reactive oxygen species are associated with norepinephrine spillover and sympathoexcitation, which may cause cognitive decline. The mechanism may be associated with pre-sympathetic catecholaminergic neurons in the rostral ventrolateral medulla; noradrenergic locus coeruleus projections to the hippocampus and Wnt/β-catenin signalling may also have a role to play (Andrade et al., 2019; Toledo et al., 2019a, 2019b). However, because the sympathetic findings are not as robust as the parasympathetic findings in our analysis, further research is needed to clarify the sympathetic features and mechanisms of the cognitive impairment.
Another interesting finding in our study is the significantly lower HRV in MCI patients than in controls. This means that the tendency for low HRV in MCI individuals is even more obvious than that in AD patients; however, this HRV difference was not significant when directly comparing the values between MCI and AD groups, indicating that cognitive function should not be the most influential factor for HRV in neurodegenerative diseases. A possible explanation is that MCI includes amnestic and non-amnestic types; the former tends to develop into AD and the latter into other types of dementia, including DLB (Ferman et al., 2013). Therefore, if the MCI patients with non-amnestic type in our included studies were common, they should not be viewed as the prodromal state of AD individuals. DLB/PDD patients may present reduced HRV in the early stage of cognitive decline, thus conforming to non-amnestic MCI. This finding indicates a probable clinical use: HRV may be adopted to differentiate subtypes of future dementia during the MCI period. Further investigation of this hypothesis is warranted.
Considering the different HRV indices, HF, RMSSD, RSA and SDNN all revealed inter-group differences between dementia patients and controls. Only LF/HF showed a non-significant difference. The result is quite intuitive because the physiological meanings of HF, RMSSD and RSA are similar: they are all considered to be specific parasympathetic indices (Alvares et al., 2016; Shaffer and Ginsberg, 2017). Although SDNN is a total variability index and includes partial sympathetic activity, it is usually viewed as a parasympathetic index (Cheng et al., 2020; Shaffer and Ginsberg, 2017). On the contrary, the clinical meaning of LF/HF is controversial, sometimes considered as a sympathovagal balance index and sometimes as reflecting sympathetic activity; this is quite different to the clinical meaning of most other HRV indices (Kuo et al., 1999; Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology, 1996). Furthermore, no significant inter-group difference was found for HRV reactivity; this may be due to the heterogeneous approaches and the low number of studies (three physiological and three psychological reactivity designs) (Lin et al., 2017; Nicolini et al., 2014). Currently, there is not enough evidence to support that HRV measurement under specific conditions corresponds to cognitive impairment.
Our results indicate that LF/HF did not have enough clinical value for separating individuals with and without cognitive impairment. LF/HF is often viewed as an index of sympathovagal balance or mainly sympathetic modulation. To investigate the sympathetic activity in dementia patients more clearly, we performed another two meta-analyses: LF, an index reflecting both sympathetic and parasympathetic modulation, was significantly lower in dementia patients than in controls (Supplementary Figure 18); and skin conductance, the specific non-HRV index of sympathetic activity, does not show a significant inter-group difference (Supplementary Figure 19). Combining these results, the low vagal activity in dementia patients should be more robust than the sympathetic feature.
The meta-regression results indicated that gender was significantly associated with the effect size of the inter-group difference. The high proportion of male subjects was related to a non-significant inter-group HRV difference. This finding can be interpreted along with the findings on DLB and MCI. The male predominance in the several DLB studies indicates that the DLB results were not likely to be confounded by the gender issue. If we only considered the AD and MCI studies (which had relatively large sample sizes), gender was not significantly associated with effect size. This implies that the significant inter-group difference of MCI was not likely to be confounded by gender proportion; however, the association between gender proportion and HRV inter-group difference in other dementia subtypes cannot be excluded. The associations between gender and dementia and between gender and HRV have been investigated. A high proportion of female patients with AD have been reported in the literature; women are considered more susceptible to developing AD than men (Mouton et al., 2018; Rocca et al., 2014). PDD was found to be more common in men, whereas DLB tended to be gender balanced (Mouton et al., 2018). With regard to HRV, in the middle-aged population, it was usually sympathetic predominant for men and vagal predominant for women, but this gender difference diminishes with older age (Kuo et al., 1999). Hormonal status, such as oestrogen level and menopausal symptoms, has been found to be associated with HRV (Lee et al., 2011). Because of the age range, most women included in this analysis are menopausal or post-menopausal. Whether the sex hormone plays a role (such as a moderator) in the association between cognitive decline and autonomic activity awaits further clarification. As a whole, most included studies in the present analysis revealed comparable features of gender proportion between dementia and control individuals; therefore, the meta-regression findings regarding gender cannot be explained with the above gender patterns of dementia or with HRV directly. However, this finding indicates that the clinical meaning of HRV for assisting in dementia evaluation may be different for males and females and is worthy of further investigation.
Several limitations of this study should be discussed. The heterogeneity of the study population and HRV data is the most important limitation of this kind of meta-analysis. The definition of dementia and its method of diagnosis in the included studies are not always identical. For example, a comprehensive survey of biological and psychological features (including neuroimaging studies) was adopted in some studies, whereas scores of cognitive tests were the only basis in other studies (Kasanuki et al., 2015; Kim et al., 2006; Negami et al., 2013; Zulli et al., 2005). Furthermore, the measuring duration and adopted indices are heterogeneous for HRV. We tried to separate some conditions (such as the different dementia subtypes and HRV indices) and manage them with subgroup analyses. However, not all heterogeneity issues can be analysed with this approach. Another limitation is that publication bias was indicated in some subgroup analyses, with some comparisons not passing the sensitivity analyses. This implies two possibilities: that the results of the non-significant inter-group difference were underreported and also affected by outliers. Furthermore, not all factors with potential influence on HRV were comprehensively reported or excluded across studies. For example, psychiatric disorders (e.g. depression, anxiety, somatic symptoms; Alvares et al., 2016; Cheng et al., 2020; Koch et al., 2019), physical illness (e.g. diabetes, cardiovascular diseases; Colhoun et al., 2001; Liao et al., 1995) and medications (e.g. anti-arrhythmic agents, multi-acting receptor-targeted antipsychotics, tricyclic antidepressants; Alvares et al., 2016; Huang et al., 2013, 2016) may affect the values of HRV. However, the influence of these factors cannot be analysed via subgroup analyses or meta-regressions. Other potentially influential factors, such as function, cannot be analysed with a meta-regression due to the limited data and distinct measurements. Although a negative association between duration of illness and the result was found, only eight articles contained data on the duration of illness. Finally, the respiratory rate affects HRV; therefore, if HRV is measured with ventilation control, this may influence the results (Cheng et al., 2020). We checked the statement about respiration in the included studies and only four of them briefly mentioned respiration; therefore, our results may not be representative enough for HRV under ventilation control (or paced breathing).
Conclusion
This study is a new attempt to combine HRV data in dementia/NCD individuals. Our results strongly suggest that the dementia/NCD population, especially those with DLB, tends to have lower parasympathetic activity than healthy individuals. This finding may be used to assist in the diagnosis of dementia and DLB. The difference can be observed using several resting HRV indices, such as HF, RMSSD, RSA and SDNN. Furthermore, the low HRV value in MCI subjects indicates that HRV measurement may be helpful for early detection of dementia subtypes, but this needs further exploration.
Supplemental Material
sj-pdf-1-anp-10.1177_0004867420976853 – Supplemental material for Heart rate variability in patients with dementia or neurocognitive disorders: A systematic review and meta-analysis
Supplemental material, sj-pdf-1-anp-10.1177_0004867420976853 for Heart rate variability in patients with dementia or neurocognitive disorders: A systematic review and meta-analysis by Ying-Chih Cheng, Yu-Chen Huang and Wei-Lieh Huang in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
The authors thank Yi-Ling Lin and Huei-Mei Ma for their administrative work during manuscript preparation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
