Abstract
Objective:
Catatonia can be life-threatening unless timely identified and treated. Lorazepam’s ubiquitous response has led to its universal acceptance as being the first-line management of catatonia and alludes to catatonia’s neurobiological underpinnings. Lorazepam challenge test (LCT) is widely used to either confirm a catatonia diagnosis or determine lorazepam sensitivity. It has a proposed schedule for administering lorazepam. However, efficacy of recommended LCT doses lack systematic evidence, resulting in variable LCT doses used in clinical and research settings contributing to findings that are challenging to generalize or assist with developing standardized lorazepam treatment protocols for catatonia. Given the same, this study aimed to objectively compare the response between two groups receiving different LCT doses and factors influencing the same.
Methods:
The 6-month study in a psychiatric emergency setting at a tertiary neuropsychiatric center in India evaluated 57 catatonia patients, before and after administration of single 2 mg (n = 37; LCT-2) or 4 mg (n = 20; LCT-4) lorazepam dose, applying Bush Francis Catatonia Rating Scale (BFCRS), Mini International Neuropsychiatric Interview (MINI 5.0) and obtaining sociodemographic, clinical data.
Results:
No between-group differences (LCT-2 vs LCT-4) for sociodemographic, clinical profiles or BFCRS severity score changes to lorazepam on Mann–Whitney U test were noted. Applying Wilcoxon signed rank test comparing individual sign severity demonstrated response variability, with significant response noted to both doses (stupor, mutism, staring, posturing, withdrawal, ambitendency, automatic obedience) and others selectively to 2 mg (echolalia, rigidity, negativism, mitgehen). Notably, sign resolution (present/absent) only to 2 mg was significant for stupor, mutism, staring, posturing, echolalia, rigidity, negativism and mitgehen.
Conclusion:
This study suggests 2 mg lorazepam may be an optimal LCT dose, given significant response to most catatonic signs thereby ensuring accurate detection and preventing misinterpretation of response. It offers future studies direction for standardizing lorazepam dosing schedules for catatonia management and exploring neurobiological underpinnings for individual catatonic signs that may be potentially different, given these findings.
Keywords
Introduction
Currently, catatonia is best conceptualized as an independent syndrome, observed in psychiatric, medical and neurological conditions (Fink et al., 2010). Greater insights from research into catatonia over the 21st century have resulted in a paradigm shift nosologically, being accorded an independent status in the Diagnostic and Statistical Manual of Mental Disorders, 5th Edition (DSM-5) (American Psychiatric Association, 2013). Specifically, in psychiatric disorders associated with catatonic presentations, various studies across the world have estimated its prevalence to be ranging from 1.64% to 10% (Grover et al., 2015; Ishida et al., 2016; Narayanaswamy et al., 2012; Solmi et al., 2018; Subramaniyam et al., 2019; van der Heijden et al., 2005).
Catatonia carries a potential risk of patient morbidity and mortality unless treated in a timely manner (van der Heijden et al., 2005). Irrespective of underlying diagnosis, benzodiazepines, particularly lorazepam, remain the mainstay of treatment in catatonia given that evidence suggests a 70–80% remission rate of catatonia with lorazepam (Bush et al., 1996b; Fink and Taylor, 2003; Rosebush and Mazurek, 2010). Hence, lorazepam is generally accepted to be the first-choice drug having the highest frequency of use, ideally administered initially intravenously (iv) for better absorption and improved rate of response as well as ensuring adherence (Hawkins et al., 1995; Sienaert et al., 2014). Studies have shown that most patients attain remission (complete resolution of catatonia signs and symptoms) within 3–7 days of initiating lorazepam with doses from 8 to 24 mg lorazepam per day commonly used and well tolerated in catatonia patients (Dhossche et al., 2016; Hung and Huang, 2006; Tibrewal et al., 2010). Although other benzodiazepines like diazepam, oxazepam, zolpidem and clonazepam have also been reported to be effective in catatonia, the evidence for their use remains poor (Lin et al., 2017; McEvoy and Lohr, 1984; Schmider et al., 1999). A recent systematic review of catatonia treatment noted that reasons for selective response to lorazepam in comparison with other benzodiazepines remain to be understood, given studies with other benzodiazepines were too few and their varying study designs limited more generalized interpretations of their efficacy in treating catatonia (Sienaert et al., 2014). The same review highlighted that overall the evidence base to develop or propose a standard protocol for treatment of catatonia was not possible due to heterogeneous study designs, assessment tools used and populations in which these studies were conducted. It, however, stated that the existing studies’ findings were adequate to encourage future systematic research that could overcome the limitations noted in the review.
Recent research exploring the pathophysiology of catatonia suggests that various psychomotor abnormalities noted are possibly attributable to dysregulation of complex pathways that modulate motor initiation, cessation as well as context-based motor function such as evaluating the need, nature and extent of spontaneous or external stimulus-based motor responses. These motor responses are additionally influenced by associated aspects such as emotion and cognition (Walther et al., 2019). Three major motor circuits have been implicated in psychosis as per the Research Domain Criteria (RDoC) (Mittal et al., 2017). The first is a cortico-subcortico-cortical loop, proposed to modulate movement initiation and inhibition. The second, a cortico-thalamo-cerebello-subcortical loop is proposed to modulate movement dynamics and timing, while the third, cortico-cortical, mainly a horizontal loop, is proposed to regulate the speed and organization of motor activity. Therefore, the above indicates a high possibility that catatonic symptoms could be attributable to dysfunction of these circuits, although which circuits specifically, remain a matter of ongoing debate (Hirjak et al., 2015; Walther et al., 2019). The same may possibly be attributable to variations in response noted in catatonia to varying lorazepam doses as potentially individual catatonic signs may have distinct neurobiological underpinnings.
Lorazepam challenge test (LCT) is often used to validate the diagnosis of catatonia in patients suspected with catatonia, wherein, one (or two) doses of 1–2 mg lorazepam is administered iv and patient is assessed after 5 minutes for the response (Bush et al., 1996b; Fink and Taylor, 2003). A marked reduction (e.g. at least 50%) of catatonic signs and symptoms using a standardized rating scale is considered a positive LCT (Sienaert et al., 2014). While several rating scales are available to diagnose catatonia and measure response, the Bush Francis Catatonia Rating Scale (BFCRS) is most commonly used in clinical settings (Bush et al., 1996a; Lee et al., 2000; Sienaert et al., 2011).
Although the LCT is widely accepted and clinically used for both confirming a suspected case of catatonia and predicting response to lorazepam, we were unable to identify any prior studies that had systematically investigated as to what extent in LCT, did the proposed 2 or 4 mg lorazepam doses reduce overall catatonic symptoms or if there was a dose-dependent differential response of individual catatonic symptoms to the above LCT doses. Furthermore, to our knowledge, no prior studies have examined the level of reduction in individual catatonic signs severity associated with variable doses as part of LCT or the predictors of response to LCT. The aim of this study was to estimate the extent of response of both, severity and frequency of catatonic signs with variable first-dose lorazepam and to also identify the factors predicting response to these variable doses.
Methods
Setting
This study was conducted over a 6-month period (December 2018–May 2019) at the National Institute of Mental Health and Neurosciences (NIMHANS), Bengaluru, India. It is a 930 bedded tertiary care neuropsychiatric center offering inpatient, outpatient and emergency services. Patients who are acutely ill and require immediate psychiatric intervention are received in Emergency Psychiatry and Acute Care (EPAC) services. Those evaluated in outpatient department (OPD) requiring immediate care are referred to EPAC services with a management plan. Patients ranging anywhere from 8000 to 8500 patients per year present to EPAC services, either directly or referred from OPD. On an average, in a month, 10–15 patients with catatonia are received in EPAC services.
Procedure
This was a cross-sectional observational study using a continuous sampling strategy. Recruited patients were those who had a priori diagnosis of catatonia following an evaluation by the OPD team and referred to EPAC services for acute management that included lorazepam administration. Once the patient arrived to EPAC services, a psychiatrist independently confirmed the diagnosis of catatonia and implemented the management plan advised by the OPD team. As shown in Figure 1, patients who met these criteria and were medically stable were included for the purposes of this study. To minimize bias and ensure the observational nature of study, those patients who arrived to EPAC services directly and diagnosed with catatonia by EPAC team (study investigators) were not included for this study as were those referred from OPD for EPAC management without a catatonia diagnosis but determined to have the same by the EPAC team. This was because for the latter two patient groups, initial dose for lorazepam treatment was determined by the EPAC team and hence were not included in the study. Patients with organicity were also excluded. A total of 57 patients who met the inclusion criteria were recruited during the study period.
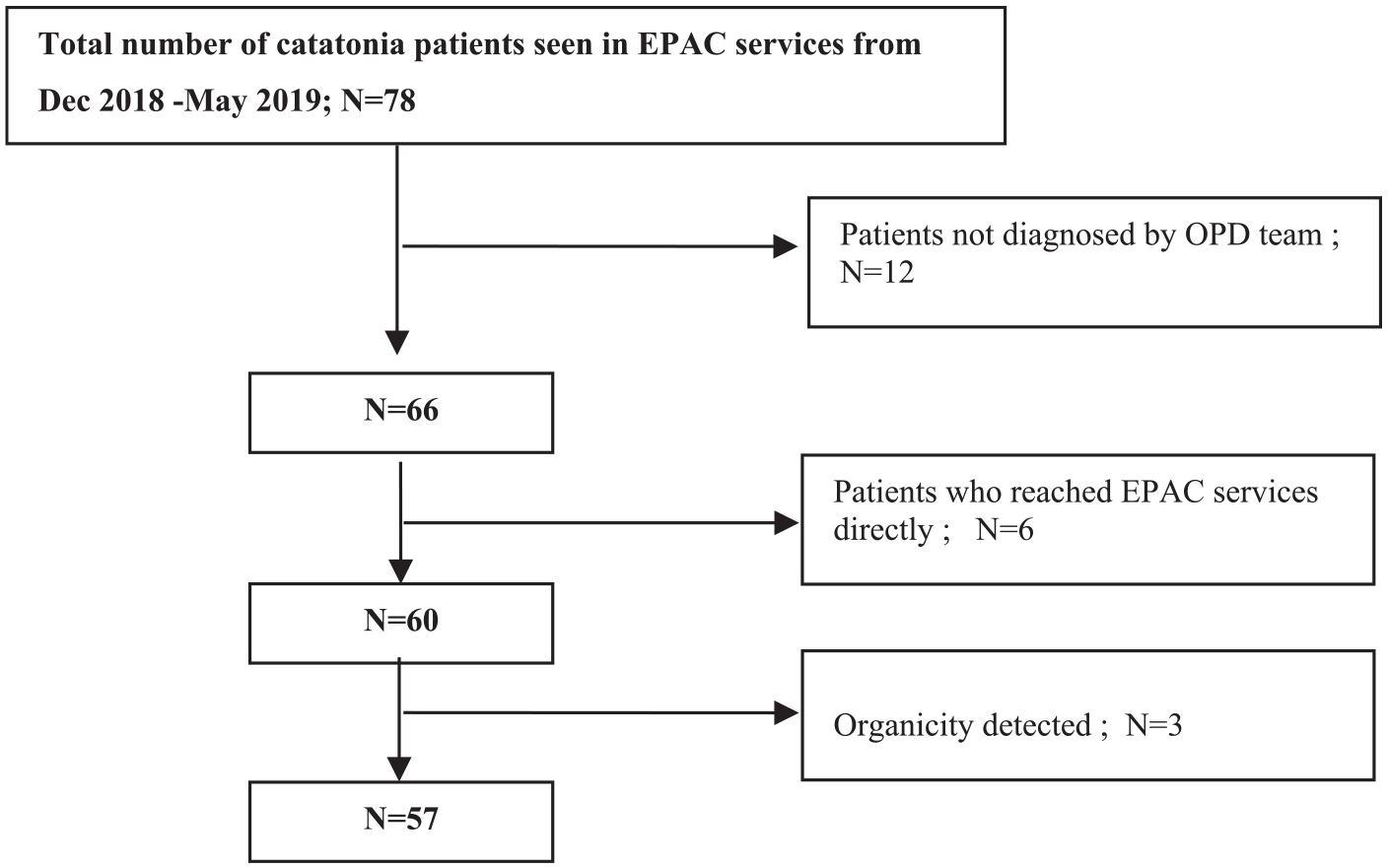
Study sample.
Written informed consent was obtained from patients or from caregivers of patients who were not capable of providing informed consent at the time of study due to acute catatonic symptoms and subsequently obtained from the patient when they were able to provide the same. Socio demographic data and clinical details were documented in a semi-structured proforma. BFCRS was applied in the EPAC just before administering intravenous lorazepam injection. All subjects received a single intravenous lorazepam in doses as prescribed by the OPD team and were reassessed after 5 minutes with BFCRS for determining extent of response to first-dose lorazepam (i.e. LCT). The investigator who applied the pre–post BFCRS was blinded to lorazepam doses administered. All the patients received same quality of drug from the hospital supply. Once the patient improved and clinically determined to be capable to provide a valid interview, Mini International Neuropsychiatric Interview (MINI 5.0) (Lecrubier et al., 1997; Sheehan et al., 1998) was administered to assess comorbid psychiatric disorders as per International Classification of Diseases, 10th Revision or ICD-10 (WHO,1992). Institute ethics committee approval was granted for the study.
Statistics
Test of normality was conducted using Shapiro–Wilk test. Sociodemographic profile and clinical variables of the 2 mg LCT group and 4 mg LCT group were compared using chi-square test for categorical data and Mann–Whitney test for continuous variables. Wilcoxon signed rank test was conducted to compare BFCRS severity and frequency scores of pre-LCT and post-LCT in both the groups. The data were analyzed using a licensed version of Statistical Package for Social Sciences (SPSS) 25.
Results
A total of 78 patients diagnosed with catatonia were seen in EPAC services during the study period. Of these, 12 patients not diagnosed with catatonia by the OPD team, 6 patients who arrived at EPAC services directly and 3 having organicity, were excluded. Among the 57 patients recruited, 37 patients and 20 patients received single lorazepam iv doses of 2 and 4 mg, respectively (dose determination was by the treating clinician) as LCT. Shapiro–Wilk test of normality showed that the sociodemographic, clinical variables and BFCRS severity and frequency were not normally distributed. Patient median age in years was 33.0 (range: 18–60) and 24.5 (18–35) in 2 mg (LCT-2) and 4 mg (LCT-4) groups, respectively. While this indicated that the LCT-4 group was significantly younger in age, when LCT-2 and LCT-4 were categorized into three age groups as shown in Table 1, one-way analysis of variance (ANOVA) with Tukey post hoc analysis conducted to check for the distribution between two groups, demonstrated no significant difference in age between the two. With the Mann–Whitney U test, no significant between group differences were observed with respect to gender, duration of total psychiatric illness, duration of current catatonic episode, psychiatric diagnosis and drug naive status as depicted in Table 1. Past history of catatonia between the two groups also was not significant, although a trend toward significance (0.08) was noted for the 4 mg group (Table 1).
Socio demographic, clinical profile and BFCRS score between LCT groups.
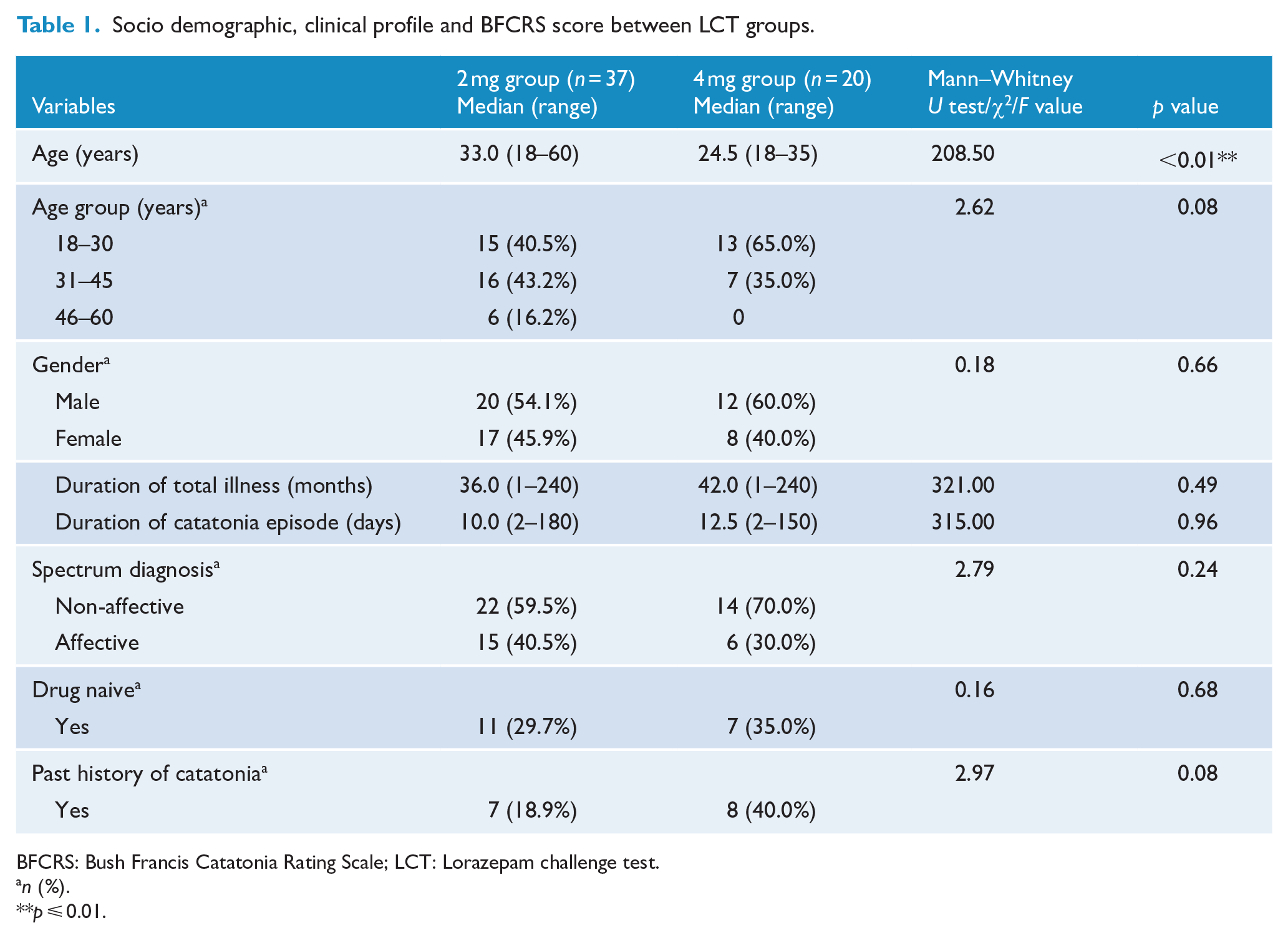
BFCRS: Bush Francis Catatonia Rating Scale; LCT: Lorazepam challenge test.
n (%).
p ⩽ 0.01.
As shown in Table 1, median duration of current catatonia episode was 10 and 12.5 days while median duration of illness was 36 and 42 months in LCT-2 and LCT-4, respectively, with no significant statistical difference between groups noted. Median BFCRS score before administering lorazepam in the LCT-2 group was 12.0 (7–28) decreasing to 6.0 (0–22) after 5 minutes of receiving lorazepam, whereas in the LCT-4 group, the median scores decreased from 15.0 (9–27) to 7.5 (0–18) (Table 2 and Figure 2). No significant difference was noted between the two groups with regard to pre-LCT and post-LCT BFCRS severity scores and frequency of signs. Figure 3 shows the extent of drop in BFCRS severity scores before and after administering lorazepam 2 mg dose for individual patients, while Figure 4 shows the scores of each patient who received lorazepam 4 mg dose. As demonstrated (Figures 3 and 4) for each dose, there was a wide variation in extent of response noted across individuals that was not dependent on the severity of catatonia or dose used.
Response to first-dose lorazepam.
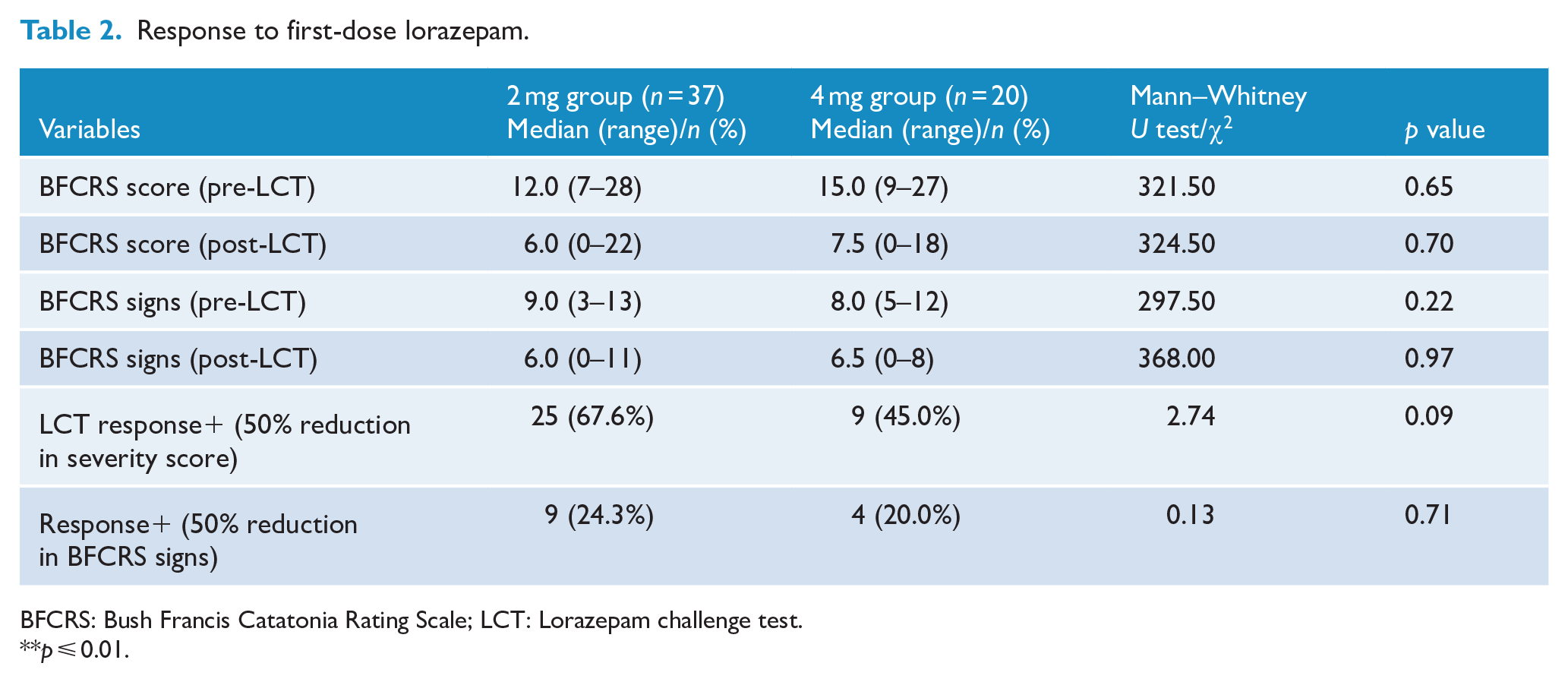
BFCRS: Bush Francis Catatonia Rating Scale; LCT: Lorazepam challenge test.
p ⩽ 0.01.
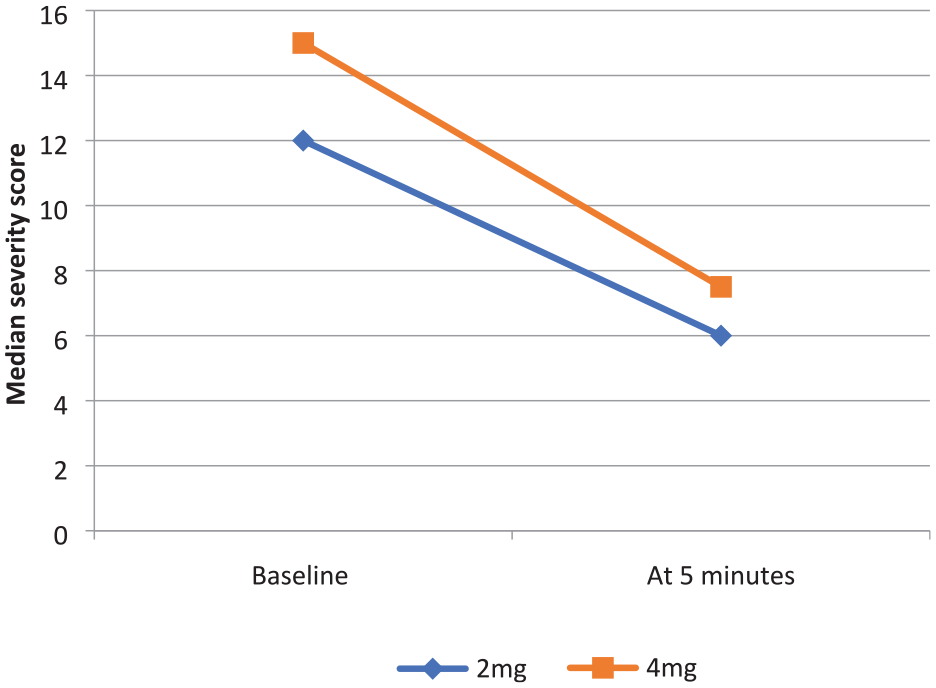
BFCRS median severity scores.
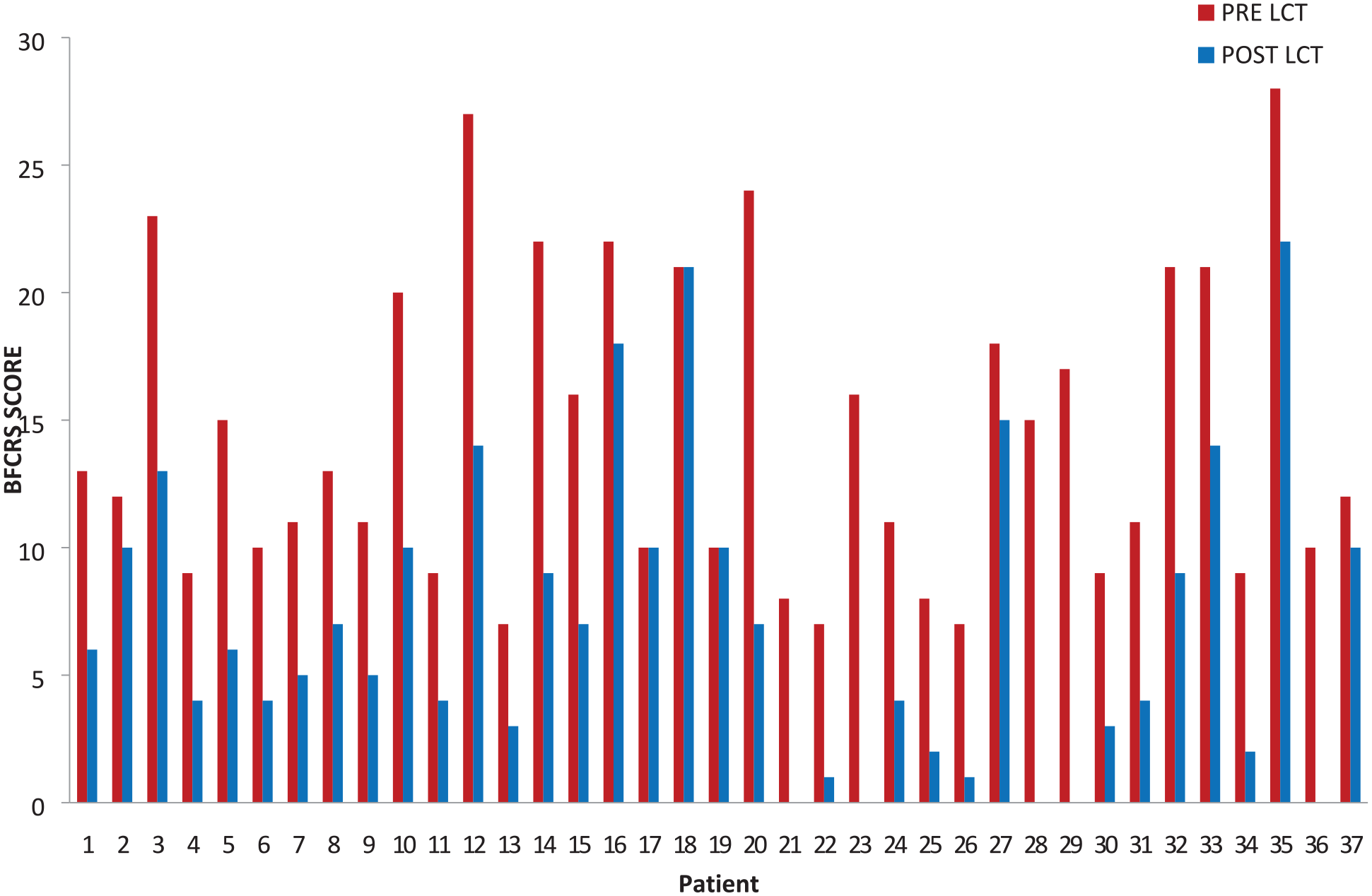
Pre-LCT and post-LCT response with 2 mg dose.
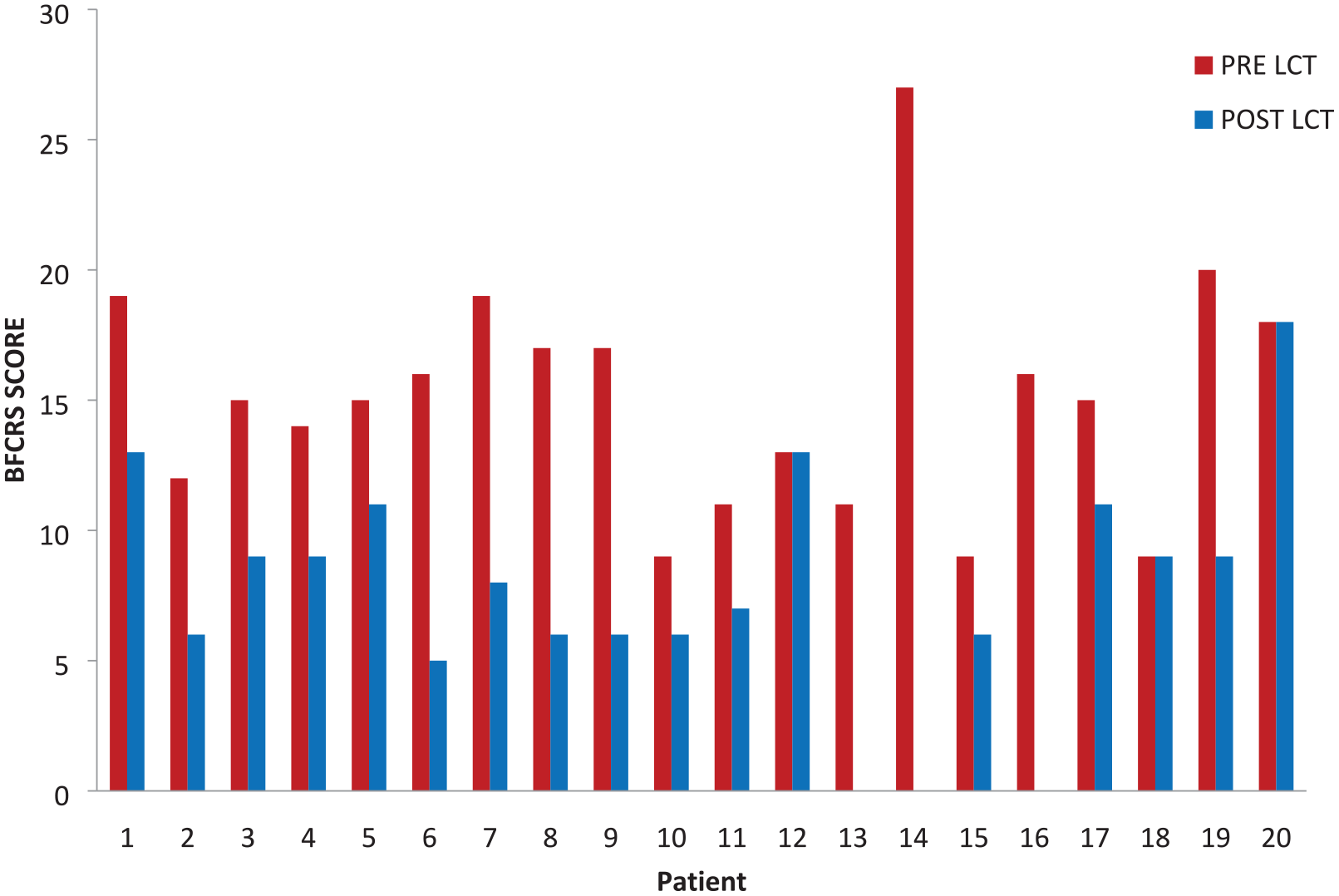
Pre-LCT and post-LCT response with 4 mg dose.
As defined in LCT, wherein, 50% reduction in severity of scores (BFCRS) was considered as a cut-off for response to lorazepam, the response rate was 67.6% to LCT-2, whereas 45% showed response to LCT-4 (Table 2).
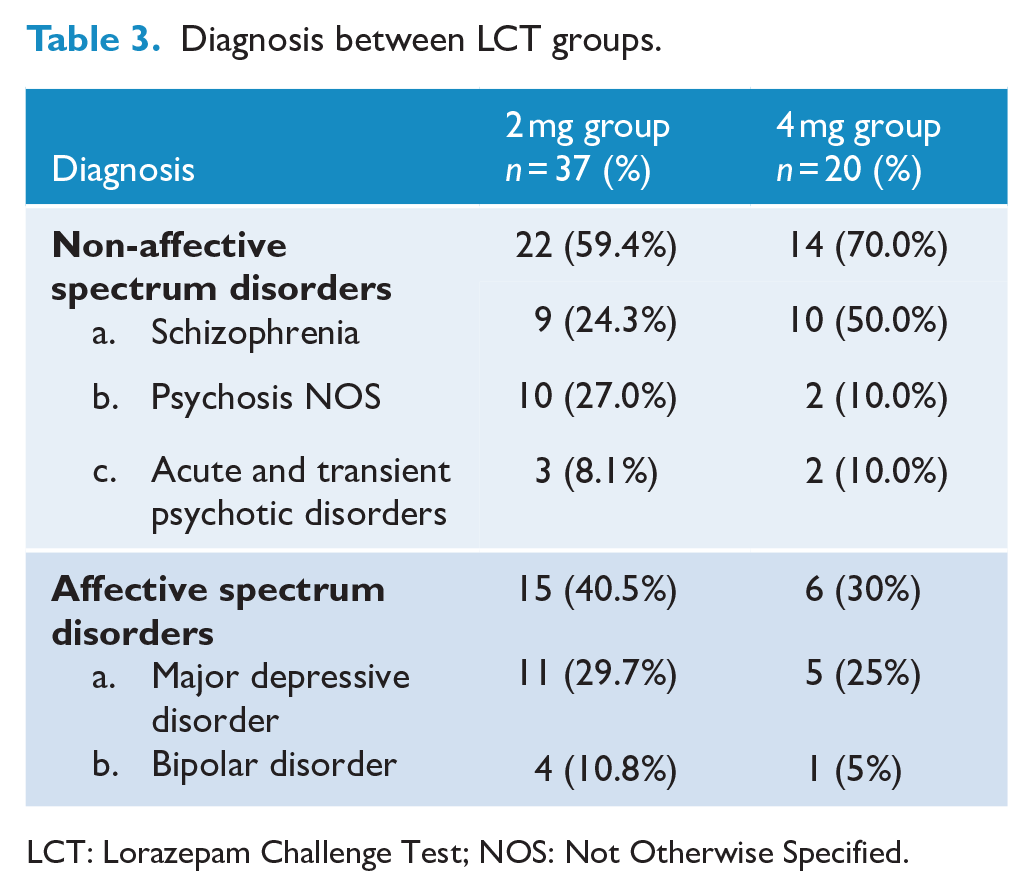
As depicted in Table 3, among the subjects recruited, dominant diagnosis was non-affective disorders (like psychosis-not otherwise specified, schizophrenia and acute transient psychosis) compared to affective disorders (major depressive disorder, bipolar disorder) in both groups. Table 4 lists individual catatonic signs observed, with high severity scores for mutism, staring, withdrawal, posturing and stupor noted in both LCT-2 and LCT-4 groups, whereas BFCRS items with least severity scores observed were combativeness, verbigeration, perseveration, grasp reflex, mannerism, grimacing and stereotypy. Wilcoxon signed rank test was applied for both the groups to analyze the significant difference between pre-LCT and post-LCT severity scores for individual catatonic signs. Stupor, mutism, staring, posturing, withdrawal, ambitendency and automatic obedience responded equally well to the first-dose intravenous lorazepam in LCT-2 and LCT-4 groups. However, echolalia, rigidity, negativism and mitgehen responded significantly only to LCT-2, while gegenhalten showed a selective response to LCT-4.
Diagnosis between LCT groups.
LCT: Lorazepam Challenge Test; NOS: Not Otherwise Specified.
Severity scores of individual catatonic signs: pre-LCT and post-LCT.
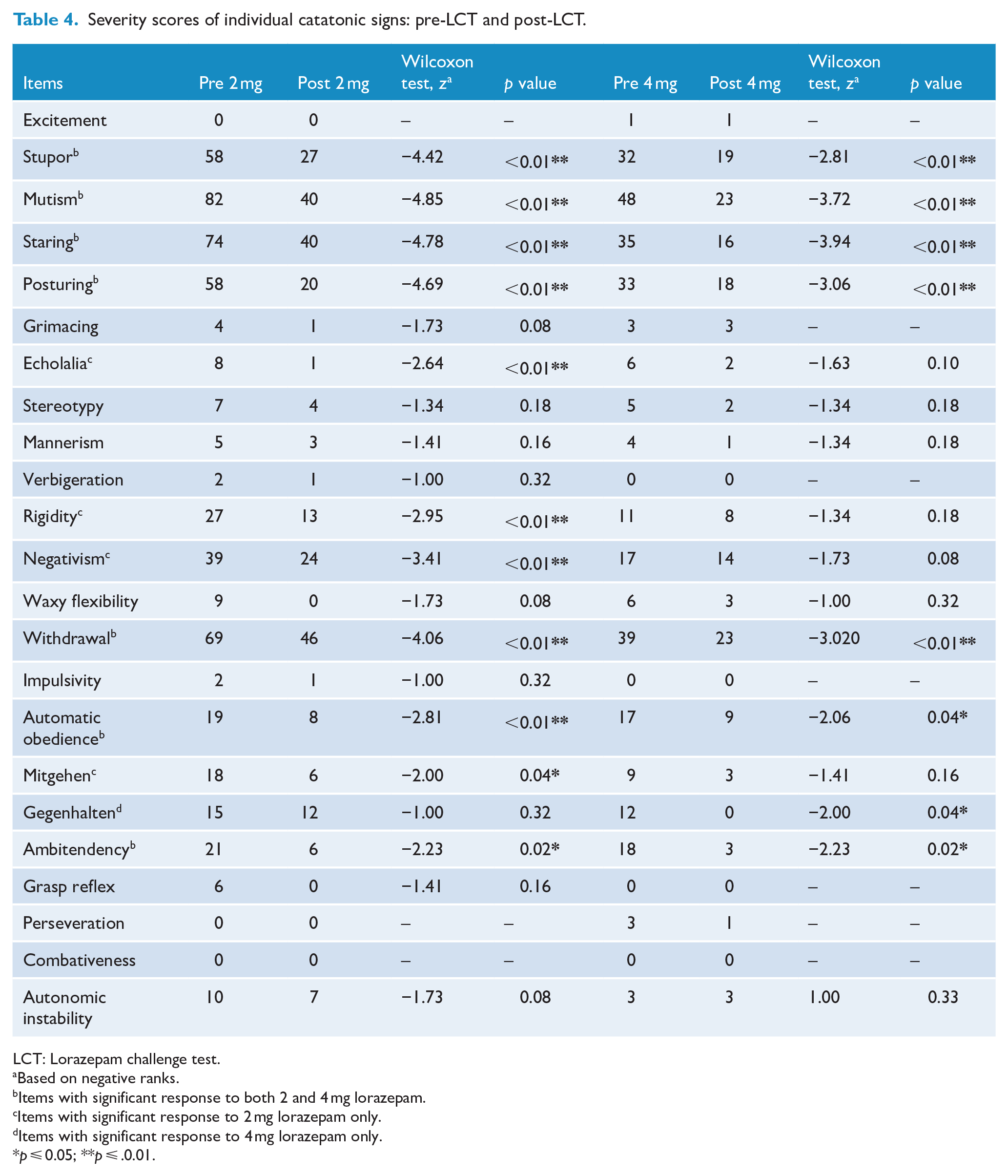
LCT: Lorazepam challenge test.
Based on negative ranks.
Items with significant response to both 2 and 4 mg lorazepam.
Items with significant response to 2 mg lorazepam only.
Items with significant response to 4 mg lorazepam only.
p ⩽ 0.05; **p ⩽ .0.01.
Given that remission is defined as the complete absence of catatonic signs, in addition to their severity we also evaluated the frequency of items based on either the presence or absence of catatonic signs (resolution) following LCT as shown in Table 5. Staring, mutism, withdrawal, stupor followed by posturing was the most frequent signs noted at baseline. Wilcoxon signed rank test demonstrated that stupor, mutism, staring, posturing, echolalia, rigidity, negativism and mitgehen showed a statistically significant response in terms of complete resolution with LCT-2 in comparison with LCT-4, while gegenhalten showed selective resolution with LCT-4. Withdrawal, automatic obedience and ambitendency resolved equally with both LCT-2 and LCT-4.
Frequency of individual catatonic signs: pre-LCT and post-LCT.
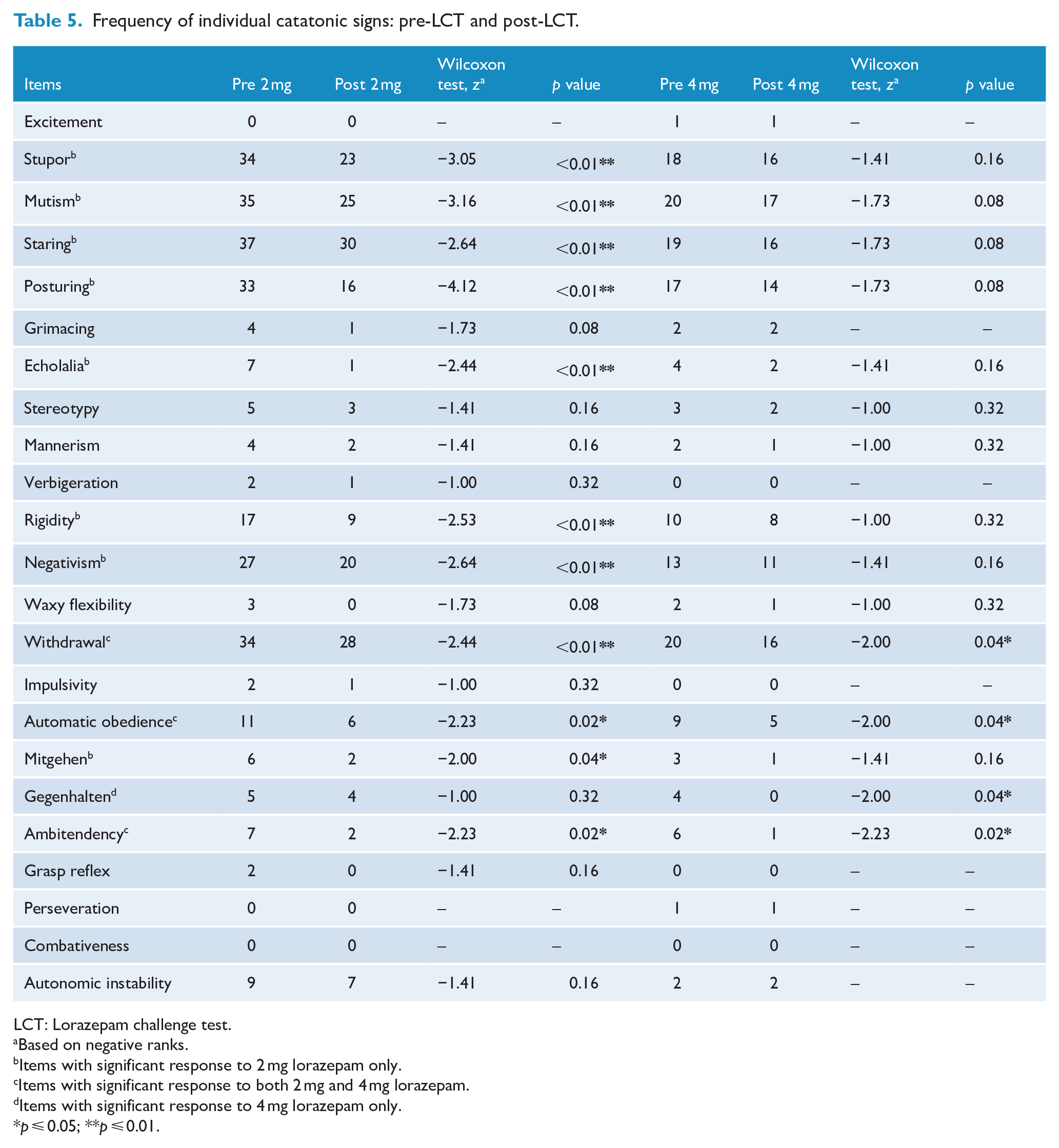
LCT: Lorazepam challenge test.
Based on negative ranks.
Items with significant response to 2 mg lorazepam only.
Items with significant response to both 2 mg and 4 mg lorazepam.
Items with significant response to 4 mg lorazepam only.
p ⩽ 0.05; **p ⩽ 0.01.
On binomial logistic regression analysis, none of the clinical or sociodemographic variables predicted the response.
Discussion
In our study, both groups were comparable across all sociodemographic and clinical correlates (including severity and frequency of catatonic signs). Age and prior history of catatonia demonstrated a trend toward significance in the 4 mg group, possibly accounting for the reason for having been clinically prescribed a higher first dose of lorazepam. Interestingly, the above findings should have enhanced the likelihood of a better response to lorazepam 4 mg, given that earlier studies have suggested that prior history of catatonia and younger age are factors associated with a better response for lorazepam in catatonia (Sienaert et al., 2014; van Waarde et al., 2010). We, however, did not find the same in our study. The median duration of catatonic episodes in both groups (10 and 12.5 days, respectively) was similar to the mean (4.6 days; SD: 6.7) duration of catatonia noted in the original study by Bush et al. (1996b) from where the LCT was proposed. With respect to clinical diagnosis, non-affective disorders (63%) were overrepresented compared to affective disorders (37%) in both groups in our study, similar to that noted in a recent study from a different center in India (Grover et al., 2019). While consistent with the recent literature published from our center and other Indian studies as well (Grover et al., 2011; Ramdurg et al., 2013; Subramaniyam et al., 2019), the preponderance of psychosis in our sample is at variance with the view that catatonia is more frequently associated with affective disorder and that catatonia in schizophrenia is rare nowadays (Abrams and Taylor, 1976; Bush et al., 1996a; Rosebush et al., 1990). Importantly, the range of diagnostic distribution in our sample also attests to the acknowledgment in DSM-5 that several psychiatric conditions are associated with catatonia (American Psychiatric Association, 2013; Heckers et al., 2010). It was also noted that the diagnostic distribution did not influence treatment response between 2 and 4 mg groups.
Mutism, staring, withdrawal, posturing and stupor were overrepresented compared to other BFCRS items in our study. Combativeness was not noted in our sample, while perseveration, grasp reflex, impulsivity, verbigeration, stereotypy, mannerism and grimacing had low severity scores. These findings are similar to the available literature indicating that the group in our study did not differ significantly with respect to commonly noted catatonic signs in other studies (Chalasani et al., 2005; Grover et al., 2011; Lee et al., 2000; Subramaniyam et al., 2019; Wilson et al., 2015). In a recent publication, we provided a three-factor solution for catatonia, namely, ‘retarded’, ‘excited’ and ‘aberrant volitional’ (Aandi Subramaniyam et al., 2020) wherein catatonia was an objectively derived representative group from a large sample of acute inpatients that allowed for accurate determination of the catatonia dimensions. In contrast, a similar three-factor solution could not be derived using the current sample, as it comprised patients who were a priori diagnosed with catatonia clinically (based on ICD-10 criteria for catatonic schizophrenia) and consequently overrepresented with retarded catatonia, similar to that noted in most other studies.
In our study, all patients diagnosed with catatonia received intravenous lorazepam as the first dose for treating catatonia. Benzodiazepines have been reported to be effective in 70% of the cases. Lorazepam has the highest frequency of use, with a wide range of response rates extending up to 80%, as well as complete response (Hung and Huang, 2006; Pommepuy and Januel, 2002). In these studies, response was defined as complete resolution of symptoms and average time period for resolution of symptoms was between 3 and 7 days (Lee et al., 2000; Tibrewal et al., 2010). We have only examined the response at 5 minutes to the variable first dose of lorazepam administered, in accordance with Fink and Taylor who also studied responses at the end of 5 minutes (Bush et al., 1996b; Fink and Taylor, 2003). In our study, when 50% reduction in severity scores was considered as a cut-off for response, 60% of the entire sample responded to lorazepam or a demonstrated a positive LCT. Importantly, no significance was noted between the 2 and 4 mg groups indicating that a lower first-dose lorazepam (LCT) is equally effective irrespective of catatonia severity scores. In addition, a significant number of individual catatonic signs resolved completely (absent) with lorazepam 2 mg although the same extent of response was not noted for most signs using lorazepam 4 mg barring a few. A study by Rosebush et al. (1990) demonstrated that 13 of 15 episodes of catatonia showed significant response to lorazepam within 2 hours and 80% responded completely and dramatically. In Ungvari et al. series, 89% of patients had significant improvement following a 48-hour trial of benzodiazepines with average mean dose of 11.8 mg (Ungvari et al., 1994). Findings from our study are consistent with findings from literature wherein dramatic and significant reductions are noted with lorazepam and that lower and more frequent doses rather than specific daily doses demonstrate better response (Lee et al., 2000; Yassa et al., 1990). This is of particular relevance, given that a large number of catatonic episodes resolve dramatically within a few hours to a few days and the need for high daily lorazepam doses may be potentially unnecessary in a large proportion of catatonic patients (Rosebush et al., 1990; Tibrewal et al., 2010). Our study suggests using a low first-dose of lorazepam for catatonia, either as LCT or predicting treatment response may allow for accurate detection (positive LCT) as well as more efficient, effective and optimal dose titration, ensuring more accurate estimation of daily lorazepam doses required for treatment of same. Here it is also important to highlight that other studies have noted that despite dramatic improvements noted within a few hours of lorazepam administration, catatonic symptoms re-emerge unless treatment with lorazepam is continued (Ungvari et al., 1994; Yassa et al., 1990). These contradictory findings from literature have possibly led to the current lack of a treatment protocol available for lorazepam use (Pelzer et al., 2018). Adopting our proposed strategy of low first-dose lorazepam in studies across different clinical settings may be one approach that could possibly offer more consistent findings for developing standardized treatment protocols for catatonia treatment in the future.
Furthermore, presence of stupor, mutism, staring, posturing, withdrawal, ambitendency and automatic obedience predicted good response to intravenous lorazepam in our study irrespective of 2 or 4 mg initial dose. While these catatonic signs in patients indicated a better response rate to lorazepam, our findings suggest no particular advantage for a higher first dose of lorazepam in patients with the above catatonic signs. This is similar to the previous study which reported mutism, immobility and withdrawal were high in responders, whereas posturing, grimacing and waxy flexibility were more common in non-responders (Ungvari et al., 1994). However, this is in contrast to a retrospective study which reported presence of waxy flexibility and grasp reflex as good predictors of benzodiazepine response, although the authors of that study highlighted the need for a prospective study to evaluate for predictors of response (Narayanaswamy et al., 2012). Drawing interpretations of response to lorazepam for waxy flexibility and grasp reflex from our study are limited given the few numbers in both groups noted for waxy flexibility and absence of grasp reflex noted in the 4 mg group. Although it is worthwhile mentioning that waxy flexibility and grasp reflex, where noted in our study, did respond to lorazepam.
In addition, in our study, selective signs like echolalia, rigidity, negativism and mitgehen showed better response with 2 mg intravenous lorazepam compared to 4 mg dose, whereas gegenhalten responded better to 4 mg. These findings are of particular interest in that they suggest that lower first doses of lorazepam selectively improve certain catatonic signs that may not necessarily improve with high first-dose lorazepam. Thus, from a clinical perspective, using higher first dose of lorazepam may lead to incorrect or premature interpretation of poor response to lorazepam in patients demonstrating the following catatonic signs, namely, echolalia, rigidity, negativism and mitgehen. The diagnostic validity of some of these signs for catatonia is a matter of ongoing debate, termed by Rogers as a ‘conflict of paradigms’, due to overlaps of catatonic signs having similarities to signs observed in neurological signs and psychiatric disorders conceptually (Rogers, 1985).
The selective preferential response of echolalia, rigidity, negativism and mitgehen to 2 mg lorazepam and gegenhalten response to 4 mg may also indicate different pathophysiological underpinnings for these catatonic signs. Given that evolving nosological frameworks and development of varying clinical rating scales for catatonia over time have prevented a clear delineation of the concept of catatonia, the selective response noted for the above signs to variable dose lorazepam may offer an opportunity to guide research that could address these challenges. This approach has been demonstrated in offering insights into the neurobiological underpinnings of catatonia in one study (Northoff et al., 2000). Imaging studies have demonstrated that motor dysfunction and emotional disturbances noted in catatonic patients result from low GABAergic receptor density in the sensorimotor cortex and reduced perfusion in the prefrontal cortex and parietal regions as well as changes to functioning of these regions with lorazepam (Northoff et al., 1999). The application of insights from the understanding of various neuronal networks involved in motor acts (Hirjak et al., 2015; Mittal et al., 2017) to catatonia is essential, given shifts and overlaps continue for conceptual frameworks of motor abnormalities in disorders including catatonia (Van Harten et al., 2017). The selective response of individual signs to varying lorazepam doses could offer a direction in the future for enhancing our understanding of the neurobiological basis for various catatonic signs.
In summary, our study has demonstrated that an adequate and significant reduction in severity as well as frequency of most catatonic signs was noted with 2 mg lorazepam for determining response to lorazepam or a positive LCT, indicating the limited role for 4 mg lorazepam for the same. Importantly, a 2 mg first-dose lorazepam predicted a better response for most signs and would potentially allow for more optimal daily dose determinations for resolving catatonia effectively and quickly. In this study, on binomial regression analysis, none of the sociodemographic and clinical variables predicted the response to intravenous lorazepam further reinforcing that lorazepam is an effective treatment for catatonia irrespective of other factors. The selective response of certain catatonic signs to 2 mg lorazepam as well as 4 mg lorazepam also offers clues that could potentially be explored that could shed light on the pathophysiology of these signs.
To our knowledge, this study is the first to evaluate the response of individual catatonic signs to different initial doses of intravenous lorazepam or LCT in patients diagnosed with catatonia. Randomized control trials and prospective studies with larger sample sizes are required to understand the response of different catatonic signs to different doses of lorazepam. Strengths of our study include all patients being evaluated in detail clinically using a standardized instrument to analyze the severity of catatonia as well as profile of catatonic signs consistent with earlier studies that allow for drawing broader interpretations of the findings from the same. In addition, to minimize bias, the investigator was blinded when applying BFCRS pre-post lorazepam and all patients in the study sample received same brand of intravenous lorazepam. Limitations include the absence of sequential monitoring of patients prospectively both after the first dose and subsequent dose administrations. This would have offered a clearer perspective on individual doses warranted, frequency of daily dosing and average duration of treatment required for complete resolution of catatonia. In addition, prospective observations would have permitted a greater understanding of the correlation of various catatonic signs to lorazepam dose responses longitudinally. While the MINI was applied to determine the clinical diagnosis, the diagnostic stability of the same may be uncertain given that the study was done in an emergency setting. The observational nature of the study possibly contributed to retarded catatonia presentations being over represented in the sample, given that only those clinically diagnosed a priori with catatonia were recruited. Under diagnosis of catatonia is common in clinical setting wherein catatonia presentations other than that retarded variant are often overlooked, resulting in the over representation of this subtype in most studies. The above could have been overcome by objectively evaluating all patients presenting to EPAC services for catatonia, thereby allowing for a more accurate representation of a catatonia sample in the emergency setting. However, the large numbers presenting to EPAC services daily made it challenging to evaluate all for catatonia.
Conclusion
This study demonstrates that in almost two-thirds of acute catatonia patients greater than 50% reduction in the severity of signs occurred within 5 minutes following single-dose intravenous lorazepam 2 mg. Significantly, this study highlights that irrespective of sociodemographic and clinical variables, using 4 mg lorazepam for LCT in catatonia offers no specific advantages given the comparable reductions in severity of catatonia noted with 4 mg and 2 mg doses. Importantly, using 4 mg as LCT, may possibly lead to incorrect interpretations of lorazepam ‘response’ as certain signs selectively responded to low-dose lorazepam 2 mg. Prospective intervention studies with larger sample sizes would provide a more comprehensive understanding of the effects of dose variations and offer insights into the predictors to treatment for catatonia. These findings may also help guide future studies exploring the possible pathophysiological basis for individual signs observed in catatonia.
Footnotes
Author Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by H.H.S., B.A.S., V.S.K.R. and K.P.M. The first draft of the manuscript was written H.H.S. and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
All authors hereby declare that they
1. Made substantial contributions to the conception or design of the work; the acquisition, analysis or interpretation of data;
2. Drafted the work or revised it critically for important intellectual content;
3. Approved the version to be published; and
4. Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Code Availability (Software Application or Custom Code)
SPSS 25-715f46a035b8b041a073 (January 2019–January 2020).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
