Abstract
Objective:
Withdrawal from psychoactive medication such as quetiapine is a well-documented phenomenon. Despite the extensive use of quetiapine, there have been few studies into the presence of discontinuation symptoms. We therefore performed a systematic review of published literature for evidence of quetiapine withdrawal or symptoms associated with discontinuation.
Methods:
We searched PubMed, Embase, CINAHL, Medline, Web of Science, PsycINFO for articles containing the terms ‘Quetiapine’ AND ‘withdraw$’ OR ‘discontinue$’. We included all study types that reported on somatic withdrawal symptoms and had no language restrictions. We excluded studies where there was withdrawal from multiple medications or any other psychoactive substance, or where the only symptoms were psychological such as rebound psychosis or craving.
Results:
We included 13 papers, all of which were individual case reports. The quality of the individual case reports was sub-optimal, as assessed by the CARE Case Report Guidelines. There was an association between rapid cessation of quetiapine and onset of somatic symptoms such as nausea, vomiting, agitation, restlessness, diaphoresis, irritability, anxiety, dysphoria, sleep disturbance, insomnia, tachycardia, hypertension and dizziness. Three studies also reported the onset of a withdrawal dyskinesia characterised by abnormal choreiform movements as well as confusion and speech disturbance in some cases. However, these findings were limited by the number and quality of case reports identified.
Conclusion:
Discontinuation symptoms are an uncommon side effect of quetiapine cessation, which may have clinical implications. Clinicians should therefore be alert to the possibility of quetiapine withdrawal in individuals who present with somatic symptoms or choreiform movements. However, large prospective studies are required to clarify this association.
Introduction
Withdrawal symptoms associated with antipsychotic discontinuation have previously been discussed in the literature (Cerovecki et al., 2013; Gardos et al., 1978; Salomon and Hamilton, 2014). Less well-documented are withdrawal symptoms associated with the specific discontinuation of quetiapine.
The first case report of somatic withdrawal symptoms attributed solely to quetiapine cessation appeared in 2004 (Jiménez-Caballero et al., 2004). Following this, quetiapine withdrawal has been sporadically mentioned in the literature.
Quetiapine was synthesised by scientists in 1985 as a clozapine mimetic, in the hope it would have fewer adverse effects (Guzman, 2018). In the United States, the immediate-release formulation was initially approved in 1997 for the treatment of schizophrenia (Guzman, 2018). The extended-release formulation followed and indications have widened to include bipolar and major depression, either as sole or adjuvant therapy (Guzman, 2018). The immediate- and extended-release formulations have similar indications in the United Kingdom (Datapharm Ltd, 2017).
In Australia, the immediate-release formulation is approved by the Therapeutic Goods Administration (TGA) for use in adults for treatment of adults with bipolar affective disorder (BPAD) for maintenance treatment of bipolar I disorder, as monotherapy or in combination, treatment of depressive episodes and for treatment of acute mania as monotherapy or in combination with mood stabilisers. It is also indicated for the treatment of schizophrenia in individuals aged 13 or above (AstraZeneca, 2018a) and forgeneralised anxiety disorder (AstraZeneca, 2018b).
The use of atypical antipsychotics in Australian adults is increasing (Hollingworth et al., 2010). In the case of quetiapine, there was an 82% increase in prescription between 2008 and 2011 with the result that 38% of all patients on a Pharamceutical Benefits Scheme (PBS)-subsidised antipsychotic in 2013 were prescribed quetiapine (Drug Utilisation Sub-Committee [DUSC], 2013). Of particular concern was the use of low-dose preparations; two-thirds of original prescriptions for quetiapine 25 mg were written by General Practitioners, suggesting that they were prescribed for conditions other than schizophrenia (DUSC, 2013). This is mirrored in a recent study from the United Kingdom where an increase in prescribing rates has been noted (Marston et al., 2014). Off-label prescribing, including prescription of 25 mg quetiapine outside of approved indications for titration in schizophrenia and BPAD, may explain the increases in prescription in both countries (DUSC, 2013). For instance, in the United Kingdom, quetiapine is widely used for the treatment of insomnia (Anderson and Vande Griend, 2014) and expansion into the management of generalised anxiety disorder has also been noted (Malhi et al., 2019).
There is also concern regarding the abuse potential of quetiapine (Bogart and Ott, 2011; Cubala and Springer, 2014). Quetiapine may be used in combination with other psychoactive substances, such as opiates or marijuana, to intensify sedative and anxiolytic effects (Pirog-Balcerzak et al., 2015). Examples include inhalation of quetiapine with marijuana, referred to as a ‘maq ball’ (Haridas et al., 2010), and the intravenous injection of quetiapine with cocaine, referred to as a ‘q ball’ (Waters and Joshi, 2007). It has also attracted various street names including ‘Quell’, ‘Suzie Q’ or ‘Baby Heroin’ (Sansone and Sansone, 2010). Its listing on street Rx, a website where users post the price they paid for individual tablets, is further evidence of its street value. In October 2019 users paid between US$36 and US$100 for a single tablet of quetiapine 100 mg IR in Sydney and rural NSW, respectively. In forensic settings, prisoners may orally and nasally inhale powdered tablets to obtain sedation and anxiolysis (Pierre et al., 2004). Feigned symptoms of psychosis in order to obtain quetiapine have also been documented in the literature; in one study, 30% of prisoners were suspected of this behaviour (Caniato et al., 2009; Pierre et al., 2004). The risk of abuse, diversion and cost led to the removal of quetiapine from a correctional formulary in the United States (Tamburello et al., 2012).
Despite a widening of the PBS indications for quetiapine, a sharp increase in its use and increasing concerns regarding the potential for abuse, there has been limited research into the possibility of discontinuation symptoms. Symptoms associated with the discontinuation of psychotropic drugs can be separated into three different types: withdrawal symptoms, rebound symptoms and super-sensitivity syndromes (Cerovecki et al., 2013). For the purposes of this article, we will focus on somatic withdrawal symptoms, which are characterised by the appearance of somatic symptoms such as nausea, vomiting and insomnia (AstraZeneca, 2018a). This decision was made due to the inherent difficulties in distinguishing psychological withdrawal from drug-seeking behaviour, rebound psychosis or super-sensitivity syndrome. Somatic symptoms typically begin in the first few days after drug continuation, peak during the first week and subside by the end of the second week (Gardos et al., 1978). Most patients only have one or two symptoms, which are generally only mild enough to elicit on direct questioning (Gardos et al., 1978). Hence, it is possible that withdrawal symptoms are more prevalent than previously appreciated.
The main receptor-mediated effects of quetiapine occur via dopaminergic, serotonergic and histaminergic pathways. The half-life of quetiapine is 7 hours, and the half-life of its active metabolite nor-quetiapine is 12 hours (AstraZeneca, 2018a, 2018b). The pharmacokinetics of quetiapine and nor-quetiapine are linear across the approved dose range, with peak plasma concentrations of quetiapine being achieved 6 hours after administration of the extended-release version (AstraZeneca, 2018b). Withdrawal symptoms such as insomnia, nausea and vomiting implicate the histaminergic and serotonergic pathways. However, there are almost no data available on withdrawal phenomena associated with the differing serotonin receptors (Cerovecki et al., 2013).
Methods
Search strategy
The protocol for this systematic review was registered with PROSPERO (registration number CRD42019119318) and we followed guidelines from the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (Moher et al., 2009). We sought advice from a research librarian prior to the commencement of this search. Articles were located by searching the terms ‘Quetiapine’ AND ‘withdraw$’ OR ‘discontinue$’ in following databases from inception to July 2019: Medline, Embase, CINAHL, PubMed, Web of Science and PsycINFO. There were no language restrictions. Search terms included quetiapine and withdrawal. Specific search terms are provided in Table 1. Ethical approval was not required for this systematic review, as all included primary data had previously been published.
Search strategy.

Two of the authors (K.M. and J.C-S.) independently screened records and abstracts. Where there was a lack of consensus, a third reviewer was consulted (S.K.). The reference lists of selected retrieved papers were screened to identify additional studies that met inclusion criteria.
Inclusion criteria
We included studies of humans where individuals developed somatic withdrawal symptoms when quetiapine was ceased. We included patients where quetiapine was used for any condition on the premise that withdrawal could occur regardless of psychiatric diagnosis. This included BPAD, rapid cycling BPAD, schizophrenia, schizoaffective disorder, Parkinson’s disease psychosis, post-traumatic stress disorder, depression with psychotic features, alcohol dependence in remission, benzodiazepine dependence in remission and anti-social personality disorder. We included all study designs such as randomised controlled trials, case series, cohort studies, case–control and cross-sectional studies.
Exclusion criteria
We excluded the following papers:
Studies on non-human subjects.
Studies where patients were withdrawing simultaneously from other psychoactive medication or substances or had ongoing comorbid substance use disorders including quetiapine abuse, diversion or misuse.
Studies where the only symptom of quetiapine withdrawal was rebound psychosis, as this was thought to be due to the natural pathophysiology of psychotic illness, or where the only symptom of quetiapine withdrawal was craving for quetiapine.
Studies where only an abstract could be found or that were narrative reviews of antipsychotic discontinuation. However, the reference lists of these papers were hand searched for additional articles.
Study quality
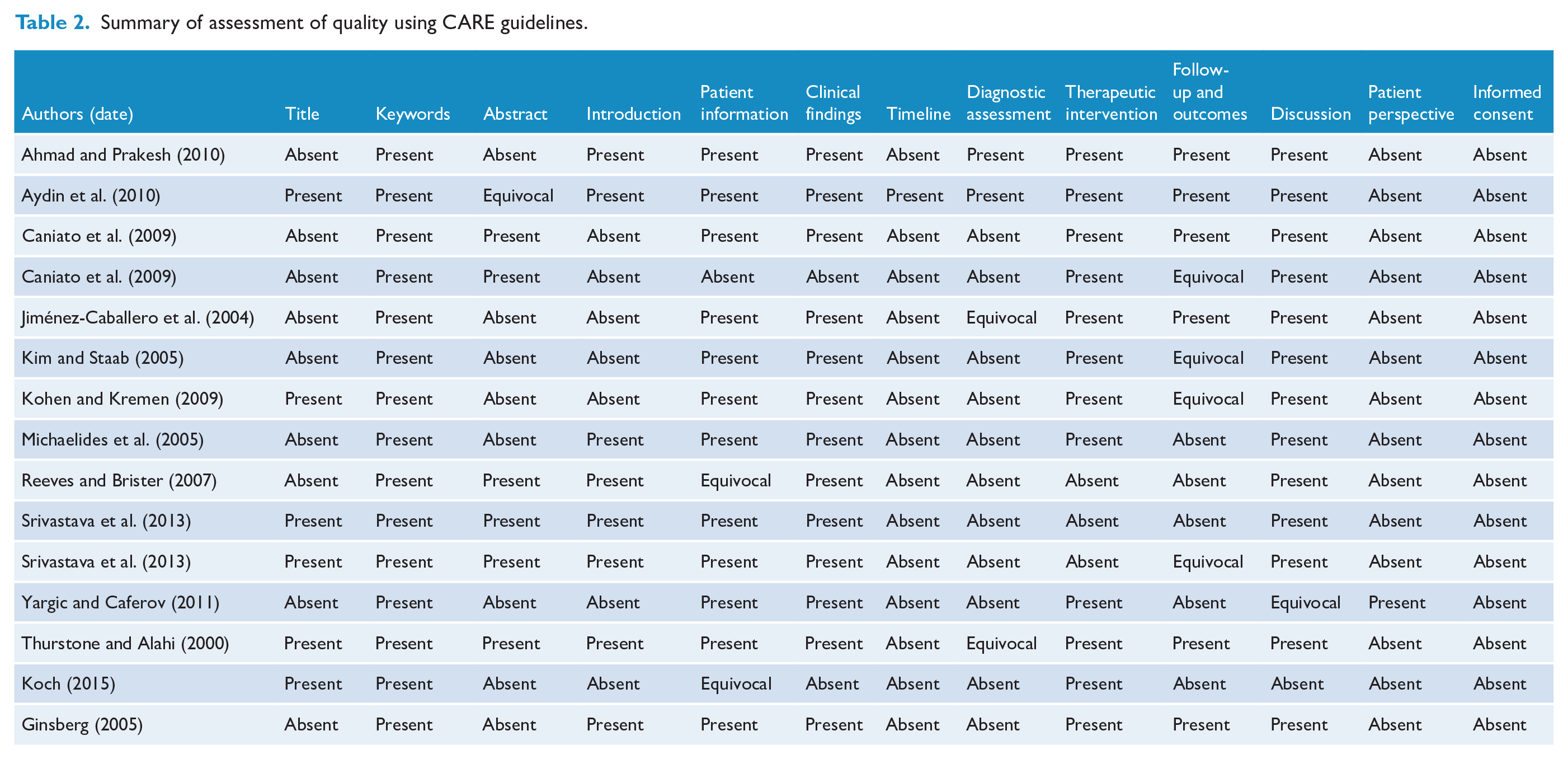
All studies identified for inclusion were case reports. We assessed the quality of these studies using the CARE (CAse REport) guidelines (Gagnier et al., 2013). The CARE guidelines were developed in 2013 by expert consensus with the aim of providing a systematic reporting guideline to guide clinical practice or to inform clinical study design. They consist of a 13-item checklist with up to 4 subcategories within each of the 13 items. We scored the studies as ‘present’ for an item in the checklist if the majority of the items in the subcategories were present in the case report. Likewise, if the minority of items were present, the study was scored as ‘absent’. If there was an equal number of subcategories present and absent, this item was scored as ‘equivocal’. Detailed scoring can be seen in Table 2.
Summary of assessment of quality using CARE guidelines.
Statistical analysis
We were unable to combine data from studies given the lack of randomised controlled trials, case series, cohort studies, case–control and cross-sectional studies.
Results
We identified 8437 studies in the databases; 4433 in Embase, 2779 in Medline, 338 in PubMed, 405 in Web of Science, 369 in PsycINFO and 115 in CINAHL. An additional 11 papers were identified via screening of references (Figure 1). After removing duplicates there were 6889 papers that were screened at title and abstract level. A total of 76 full-text papers were then assessed for inclusion, of which we included 13 case reports on 15 patients (Table 2) (Ahmad and Prakash, 2010; Aydin et al., 2010; Caniato et al., 2009; Ginsberg, 2005; Jiménez-Caballero et al., 2004; Kim and Staab, 2005; Koch, 2015; Kohen and Kremen, 2009; Michaelides et al., 2005; Reeves and Brister, 2007; Srivastava et al., 2013; Thurstone and Alahi, 2000; Yargic and Caferov, 2011). Further details of included studies can be seen in Table 3. We did not find formal observational studies. This has resulted in an inability to combine data for meaningful statistical analysis.
Summary of included case reports.
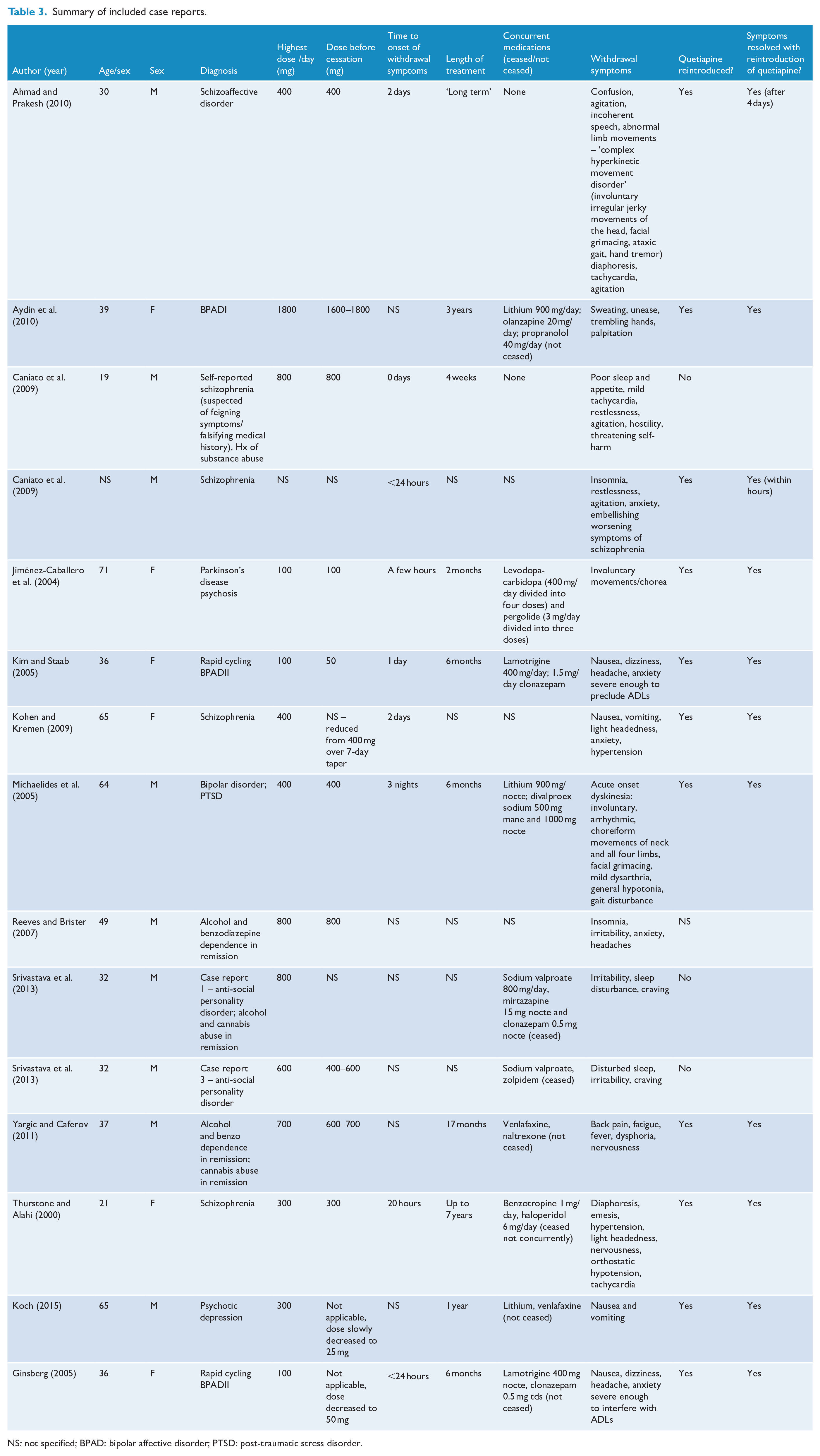
NS: not specified; BPAD: bipolar affective disorder; PTSD: post-traumatic stress disorder.
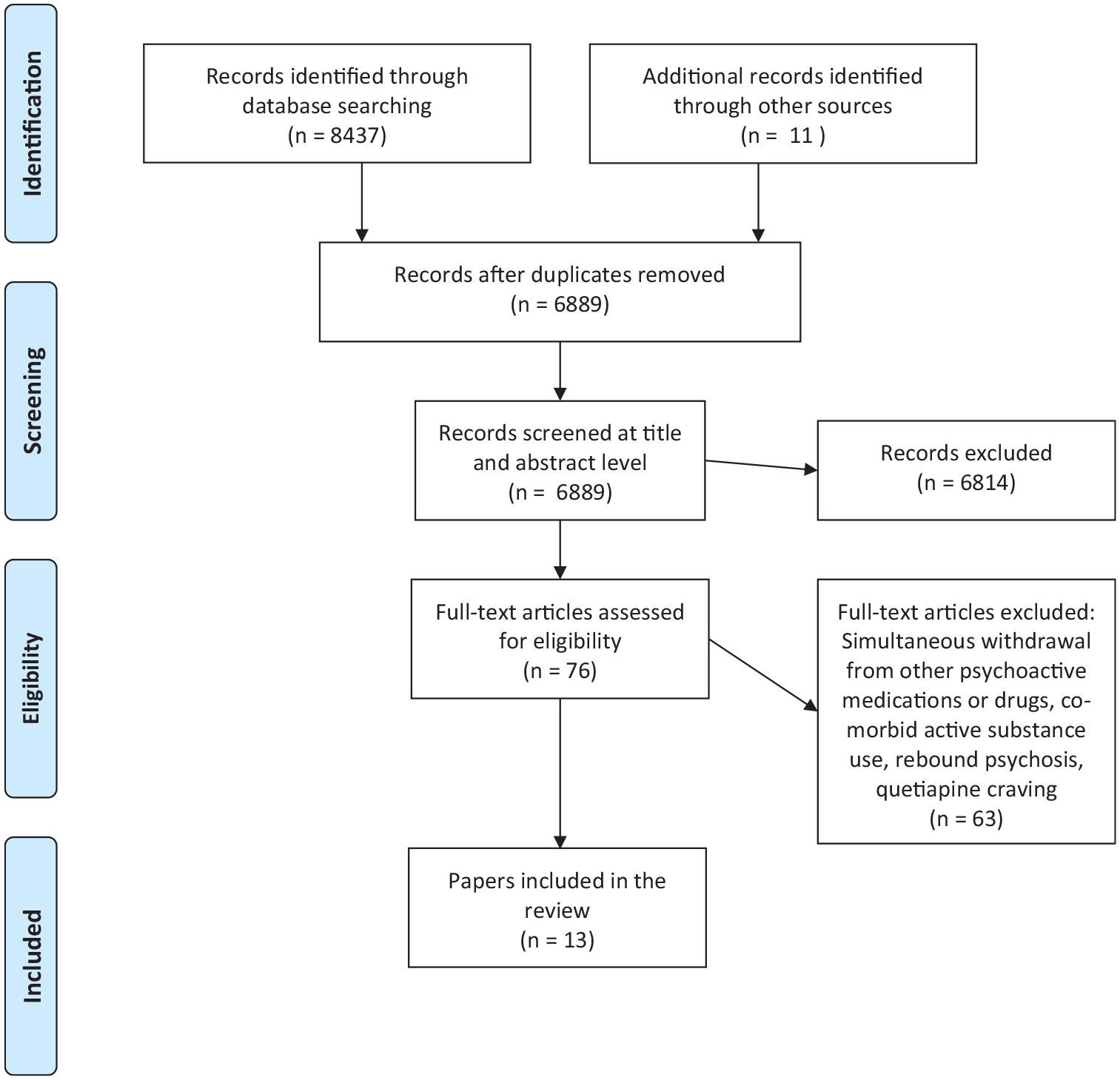
PRISMA flow chart of included and excluded studies.
Quetiapine was used for a range of conditions, which were occasionally comorbid, including BPAD (n = 2), rapid cycling BPAD (n = 2), schizophrenia (n = 4), schizoaffective disorder (n = 1), Parkinson’s disease psychosis (n = 1), post-traumatic stress disorder (n = 1), depression with psychotic features (n = 1), alcohol and benzodiazepine dependence in remission (n = 2) and anti-social personality disorder (n = 2). The mean age at presentation was 42 years old (standard deviation = 17, range = 19–71), although the age was not specified in one patient. The majority were male (60%). The mean quetiapine dose was 543 mg/day (standard deviation = 444, range = 100–1800 mg/day) with the median dose being 400 mg/day. No papers specified whether the preparation of quetiapine was either in the immediate or modified release form. The average time to onset of discontinuation symptoms was 33 hours (standard deviation = 22, range = ‘a few hours’–3 nights), which is consistent with known half-life of quetiapine and its metabolites of between 7 and 12 hours (AstraZeneca, 2018a, 2018b). The timing was not specified in the other 40% (n = 6) of cases. The mean dose prior to cessation of quetiapine was 595 mg, with ranges from 25 to 1800 mg, although this was not specified in three of the cases. There appeared to be no correlation between dose prior to cessation and severity of reported withdrawal symptoms.
Symptoms
A third of patients (n = 5) reported nausea and vomiting, while 46% (n = 7) of cases displayed symptoms related to the cardiovascular system including palpitations, tachycardia, dizziness, light headedness, hypertension and/or orthostatic hypertension (Figure 2).
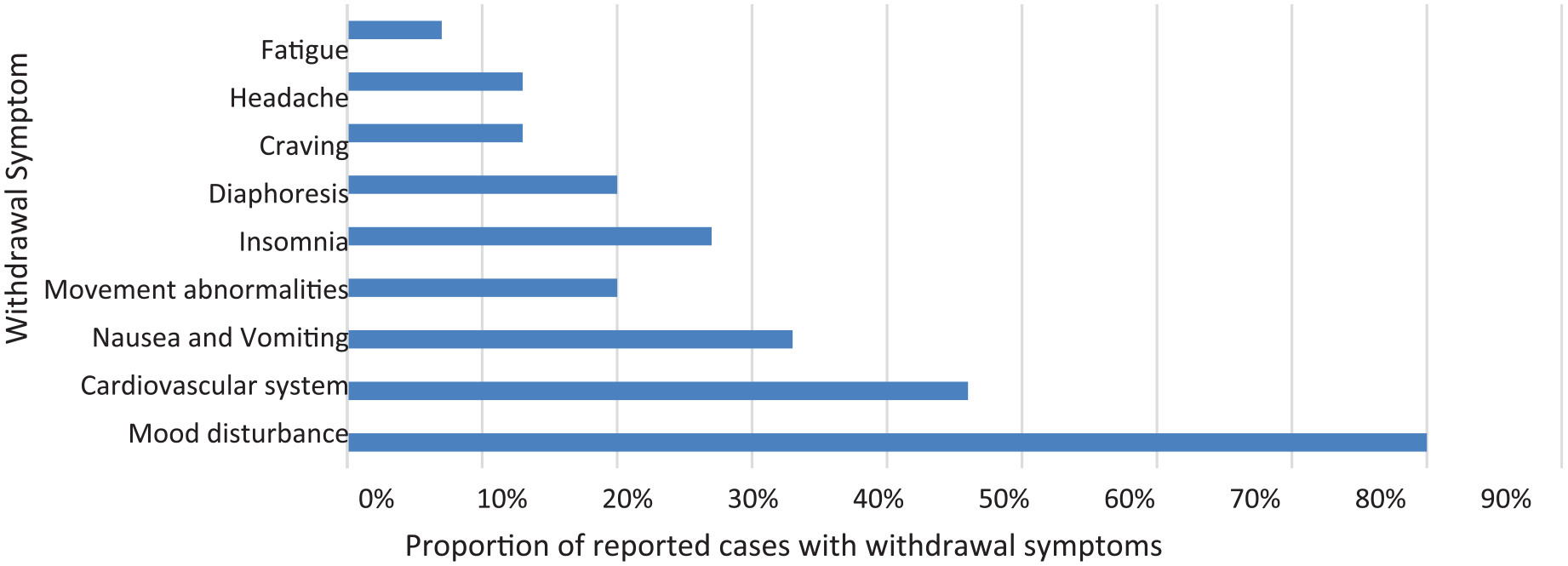
Summary of withdrawal symptoms after cessation of quetiapine.
Three patients also developed acute onset movement abnormalities including one who experienced ‘involuntary movements’, and two acute onset dyskinesias. These were characterised by involuntary irregular jerky movements of the head, facial grimacing, ataxic gait and involuntary, arrhythmic, choreiform movements of neck and limbs, facial grimacing, mild dysarthria, general hypotonia and gait disturbance. In one case, there was hand tremor.
A total of 27% of patients (n = 4) developed insomnia or sleep disturbance, while 20% (n = 3) developed diaphoresis. There were also occasional reports of craving, headache or fatigue.
There was one case of agitation, hostility and threats of harm to self with associated sleep and appetite disturbance, as well as mild tachycardia in a 19-year-old patient who was believed by the original authors to be deliberately feigning symptoms of psychosis to obtain quetiapine.
Although this was not the focus of this study, the most common withdrawal symptom was actually mood disturbance, with 80% (n = 12) of patients reporting dysphoria, irritability, a feeling of unease, agitation, nervousness, anxiety and/or restlessness. These ranged from a mild subjective experience, to sufficiently severe to preclude activities of daily living.
Management of withdrawal
In three-quarters of cases (n = 11), quetiapine was reinstated with an associated resolution of withdrawal symptoms. One case did not specify if quetiapine was reinstated: a 49-year-old male, on parole after a recent incarceration (Reeves and Brister, 2007). He had obtained quetiapine on the street and developed withdrawal symptoms when unable to obtain this any longer. In the remaining three cases, quetiapine was not recommenced. One was a 19-year-old male in a forensic setting who was suspected of feigning psychotic symptoms in order to gain access to quetiapine. When he was reviewed 7 days later, he displayed no mood disturbance, restlessness or agitation, although he continued to complain of insomnia. An alternative psychopharmacological agent was commenced (Caniato et al., 2009). The second was a 32-year-old male, also in a forensic setting, with a history of alcohol and cannabis abuse in remission. On enquiry, he revealed that since there was no availability of other substances in the prison, he used quetiapine to alleviate agitation. He reported that it ameliorated his cravings for alcohol and cannabis (Srivastava et al., 2013). Yet another was a 32-year-old male who was also in a forensic setting. He was admitted to a psychiatric facility for detailed psychiatric evaluation which revealed no mood features, psychotic symptoms or insomnia. He was found to be lying and showing manipulative behaviour in keeping with a personality disorder. Sodium valproate was commenced in order to assist with irritability and impulsivity. He continued to obtain to obtain quetiapine from other inmates and continued to request this from medical practitioners within the forensic system (Srivastava et al., 2013).
Study quality
Overall, the quality of the case reports was sub-optimal, as assessed using the CARE guidelines. The average number of items completed from the 13-item checklist in the CARE guidelines was 6.4 (standard deviation = 1.8, range = 3–10, median = 6) (Table 3).
Discussion
There have been sporadic case reports of withdrawal following quetiapine cessation; however, this is the first systematic review of the topic. Although we found a limited number of reports, there was evidence of withdrawal symptoms that could not be explained by rebound or super-sensitivity syndromes. Withdrawal symptoms are of particular concern given increasing rates of prescribing within Australia (PBAC, 2018) and internationally (Marston et al., 2014) due to a widening of indications (Malhi et al., 2019; Marston et al., 2014), as well as off-label prescribing (DUSC, 2013).
There may be a number of explanations for the relatively small number of papers we found despite high rates of quetiapine prescription worldwide. One is that withdrawal symptoms, may be mild and occur infrequently. However, it is also possible that these case reports are an under-estimate of the true extent of the problem given information from other sources such as the Monthly Index of Medical Specialities (MIMS) electronic database, the relevant Consumer Medicines Information Leaflet and adverse drug reactions reported to Australia’s Therapeutic Goods Agency (TGA) (AstraZeneca, 2018a; MIMS, 2018). Although these sources do not generally specify whether symptoms occurred in the context of withdrawal, there is a possibility that some of the symptoms described as side effects may be part of withdrawal. For instance, tachycardia, palpitations and vomiting are listed as common adverse reactions of quetiapine treatment in MIMS, occurring in ⩾1% to ⩽10% of adults (AstraZeneca, 2018a; MIMS, 2018). In addition, there were 1270 reports to the TGA of adverse reactions associated with quetiapine over the 20 years of use in Australia. These included 11 reports of quetiapine withdrawal but also 237 of symptoms similar to those found in the case reports in this study that could possibly be linked to withdrawal (TGA, 2019). Somatic symptoms of withdrawal may therefore go unrecognised and be attributed to the underlying psychiatric condition, a process that is sometimes termed ‘diagnostic overshadowing’ (Giddings, 2013).
Clinical implications
Clinicians should be aware of the risk of withdrawal when initiating treatment and advise patients of the possibility when reducing doses. In particular, consideration should be given to the presence of discontinuation symptoms if nausea, vomiting, mood or cardiovascular symptoms develop in the context of abrupt cessation. Prescribers should also be alert to the possibility of quetiapine abuse or diversion, especially when working in closed settings such as inpatient wards, jails or prisons. A higher level of caution should be exercised when prescribing to those with a previous history of substance abuse; however, quetiapine abuse and dependence has been noted in individuals without a history of substance abuse (Cubala and Springer, 2014). It may be prudent to reconsider the prescription of quetiapine as a first-line antipsychotic for patients with a previous history of substance abuse. Although we found no evidence in our results of a difference in the occurrence of withdrawal symptoms between the immediate- and extended-release forms, given this information was not provided, it might still be expected that the latter might be a better choice when prescribing.
Our findings also raise concerns regarding current guidelines for the treatment of schizophrenia and non-affective first episode psychosis. For instance, the RANZCP clinical practice guideline lists quetiapine as a first-line option for the treatment of non-affective first episode psychosis (Galletly et al., 2016). However, reports of the abuse potential of quetiapine, as well as withdrawal from quetiapine, raise concern that quetiapine may not be an appropriate first-line psychotropic agent in individuals with a history of substance abuse or sensitivity to medication cessation, and perhaps should be relegated to a second-line option for such individuals.
Clinicians should also be aware of the possibility of withdrawal symptoms following abrupt changes in use and not attribute them to the underlying psychiatric condition (Giddings, 2013). If quetiapine withdrawal is suspected, reinstatement of previous quetiapine dose is likely to produce resolution of withdrawal symptoms. Gradual reduction of dose and tapering to cease is recommended to avoid the possibility of and lessen the severity of somatic withdrawal symptoms. Pharmacotherapy for symptoms of withdrawal, such as anti-emetics, short-term hypnotics or sedative antihistamines, may help to alleviate some distress associated with withdrawal. Theoretically, changing to an extended-release formulation of quetiapine and gradually reducing the dose should minimise withdrawal symptoms, although we found no documented difference in the half-life between formulations (AstraZeneca, 2018b).
Strengths and limitations
By systematically reviewing the literature, we report the first systematic review on the topic. We searched all relevant databases, had no language restrictions, and hand searched for relevant papers.
However, there were also several limitations which limit generalisability of these findings to clinical practice. First, we identified a limited number of case reports rather than formal observational studies. As a result, these findings may under-estimate the magnitude of the issue and we were unable to combine data from studies for statistical analysis. Second, the quality of the case reports was poor, as assessed using the CARE guidelines.
Third, none of the papers identified whether an immediate- or extended-release formulation of quetiapine was used. In addition, the time till onset of somatic symptoms was poorly documented, and only one of the case reports used a timeline to illustrate important information from the patients’ history (Aydin et al., 2010). Most reports did not specify diagnostic methods, challenges, reasoning or prognostic characteristics as suggested in the CARE guidelines. Furthermore, most reports gave poorly detailed accounts of patient follow-up, perspectives or consent.
In addition, several papers reported symptoms such as insomnia, irritability and craving that could well be described by concomitant personality disorder and previous poly-substance dependence. Finally, the limited number of papers identified meant that a clear association between abrupt quetiapine cessation and somatic withdrawal symptoms could not be reliably established.
Conclusion
There is preliminary evidence of the presence of quetiapine withdrawal symptoms. Clinicians should therefore be aware of the possibility following abrupt changes in use and be alert to the risk of ‘diagnostic overshadowing’. However, further research is also required to expand the evidence base. Such studies would ideally be large, randomised, multi-centred and controlled, with use of consistent tools to assess for the presence and severity of withdrawal symptoms. Studies should also differentiate between the immediate- and extended-release forms. In addition, any future case reports should adhere to the CARE guidelines for reporting of case reports in order to standardise reporting and better inform clinical practice or study design.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
