Abstract
Objective:
The aim of this study was to estimate whether neurocognitive deficits are predictors of the long-term clinical course of patients with bipolar disorder.
Methods:
A total of 76 outpatients with bipolar disorder performed a neurocognitive assessment at baseline and were followed for a period of at least 48 months. The clinical course during the follow-up period was documented by two measures: (1) number of affective episodes and (2) time spent ill.
Results:
Patients had lower performance than controls in the domains of verbal memory and executive functions, and they were followed-up for a mean period of 70.73 months. Global cognitive deficits (performance 1.5 standard deviations below the mean in two or more cognitive domain) were independent predictors of both hypo/manic episode density and time spent with hypo/manic symptoms during follow-up. On the contrary, no neurocognitive measure showed a relationship with depressive morbidity during follow-up.
Conclusion:
Our findings suggest that cognitive deficits could be useful for predicting hypo/manic morbidity in the long-term clinical course. Theoretical and clinical implications of these findings are discussed.
Introduction
Bipolar disorder (BD) is a chronic and recurrent illness that affects 1% to 2% of the general population (Fagiolini et al., 2013). More than half of the patients have a recurrence within 2 years since their first episode and more than 90% experience at least one new episode throughout their lifetime (Solomon et al., 1995). Even in maintenance treatment programs, the rates of relapse (return of the original episode) and recurrence (occurrence of a new episode) are highly variable. The risk of relapses and recurrences has been related to several clinical features such as persistence of residual or subclinical symptoms (De Dios et al., 2012; Judd et al., 2003; Perlis et al., 2006), number of previous episodes (De Dios et al., 2012; Perlis et al., 2006; Tohen et al., 1990), mixed episodes in the past (Peselow et al., 2016; Sportiche et al., 2017), or psychiatric comorbidity (Perlis et al., 2006; Simon et al., 2004; Tohen et al., 2003). The relevance of identifying predictors of long-term clinical course lies in the possibility of establishing more personalized care needed to prevent or reduce recurrences.
Nowadays, it is well known that patients with BD exhibit cognitive deficits in domains of verbal memory, attention and executive functions even during euthymic periods (Mann-Wrobel et al., 2011; Torres et al., 2007). Beyond this general pattern, more recent studies have consistently replicated that cognitive deficits are heterogeneous, with some patients showing preserved performance, others having selective deficits and others presenting global deficits (Burdick et al., 2014; Cullen et al., 2016; Martino et al., 2014). In a previous study, the presence of clinically significant cognitive deficits was also an independent predictor of recurrences in patients with euthymic BD (Martino et al., 2013). Although the risk of recurrence is routinely used as a surrogate of long-term clinical course, it might be insufficient to assess its complexity. Thus, the use of measures such as episode density or time spent ill over an extended follow-up period could provide valuable additional information. However, to the best of our knowledge, there are no previous studies evaluating the predictive capacity of neurocognitive functioning on these additional measures of over the long-term clinical course. Therefore, the aim of this study was to assess whether neurocognitive functioning allows predicting the long-term clinical course in BD.
Materials and methods
A total of 76 subjects were consecutively selected from the outpatient population of the INCyT-Favaloro University according to the following inclusion criteria: age between 18 and 65 years, diagnosis of BD type I or type II using Structured Clinical Interview for DSM-IV, euthymic (Hamilton Depression Rating Scale ⩽ 8 and Young Mania Rating Scale ⩽ 6) for at least 8 weeks and a period of follow-up of more than 48 uninterrupted months. In addition, 50 healthy controls without personal history of psychiatric disorders and without history of psychotic or affective disorders in first-degree family members were included. Exclusion criteria for all participants were history of substance abuse, neurological disease or any other unstable clinical condition that could affect cognitive performance.
All subjects performed at baseline a neuropsychological battery selected to assess the following cognitive domains: (1) Attention: Forward Digit Span (Wechsler, 1955) and Trail Making Test part A (Reitan, 1958), (2) Verbal memory: List learning of the Memory Battery of Signoret (Signoret and Whiteley, 1979), (3) Language: Boston Naming Test (Kaplan et al., 1983) and (4) Executive functions: Wisconsin Card Sorting Test (Heaton, 1981), Trail Making Test part B (Reitan, 1958) and Phonological Fluency (Benton et al., 1983). In addition, estimated premorbid IQ was calculated with the WAIS-R vocabulary subtest at baseline (Wechsler, 1955). Vocabulary has been identified as the single best measure of premorbid IQ (Lezak, 1995).
Demographic and clinical information at baseline was obtained from clinical charts and direct patient interview. During the follow-up period, clinical course of each patient was assessed by two measures: (1) time spent ill (both clinical and subclinical symptoms) documented at each visit (with intervals usually around 1–2 months) with a modified life charting technique rated by the treating psychiatrist on a weekly basis. This life chart technique was used in previous studies by our group (Martino et al., 2017) and was developed without the knowledge or purpose of the present work. (2) Density of affective episodes (number of DSM-5 defined depressive and hypo/manic episodes per year of follow-up). All patients included in our program are evaluated neurocognitively and followed through this mood-chart method as routine practices. Therefore, the sample of this study is larger although partially overlaid with that of previous research (e.g. Martino et al., 2018a). Exposure to antidepressants, mood stabilizers, antipsychotics and benzodiazepines at baseline was assessed by Clinical Scale of Intensity, Frequency and Duration of Psychopharmacological Treatment (Peralta and Cuesta, 2002). This scale provides a quantitative measure of current exposition to different groups of psychotropic medications in a 0–5 points range (0 = no medication, 1 = sporadic low dose, 2 = continue low dose; 3 = middle dose, 4 = high dose and 5 = very high dose).
The Hospital Ethics Committee approved the study and all subjects gave written informed consent. The investigation was carried out in accordance with the latest version of the Declaration of Helsinki.
Data analysis
Raw score of neurocognitive performance was transformed to Z scores from normative data of each test. Based on previous studies (Martino et al., 2014), subjects were categorized as having clinically significant cognitive deficits (if they showed a performance in any test of one cognitive domain below 1.5 standard deviations [SD] from the mean) and global cognitive deficits (if the 1.5 SD performance below the mean affected tests of two or more cognitive domains). Finally, an overall value was obtained for each cognitive domain averaging the performance in each included test.
Since most continuous variables such as number of episodes during follow-up or time spent ill were skewed, non-parametric tests were used. Comparison between groups in clinical-demographical and neurocognitive variables was made using Mann–Whitney or chi-square tests as appropriate. Bivariate relationships between continuous clinical-demographical and neurocognitive variables were assessed using Spearman correlation. Clinical-demographical and neurocognitive variables with significant association—at level p < 0.05—in the bivariate analysis were considered potential predictors of clinical course in multiple linear regression models. The presence of multicollinearity in the multiple regression models was assessed by means of the variance inflation factor (values below 10 were considered adequate).
Results
Clinical and demographical features of patients and healthy controls are shown in Table 1. At baseline, 100% of patients were medicated with mood stabilizers, 56.6% with antipsychotics, 42.1% with benzodiazepines and 40.8% with antidepressants. Patients had lower performance than controls in the domains of verbal memory and executive functions (Table 1). There were more patients than controls with clinically significant cognitive deficits (66% vs 24%; χ2 = 21.07, p < 0.001) and with global cognitive deficits (33% vs 16%; χ2 = 4.45, p = 0.035).
Clinical and demographical characteristics of bipolar patients and healthy controls at baseline.
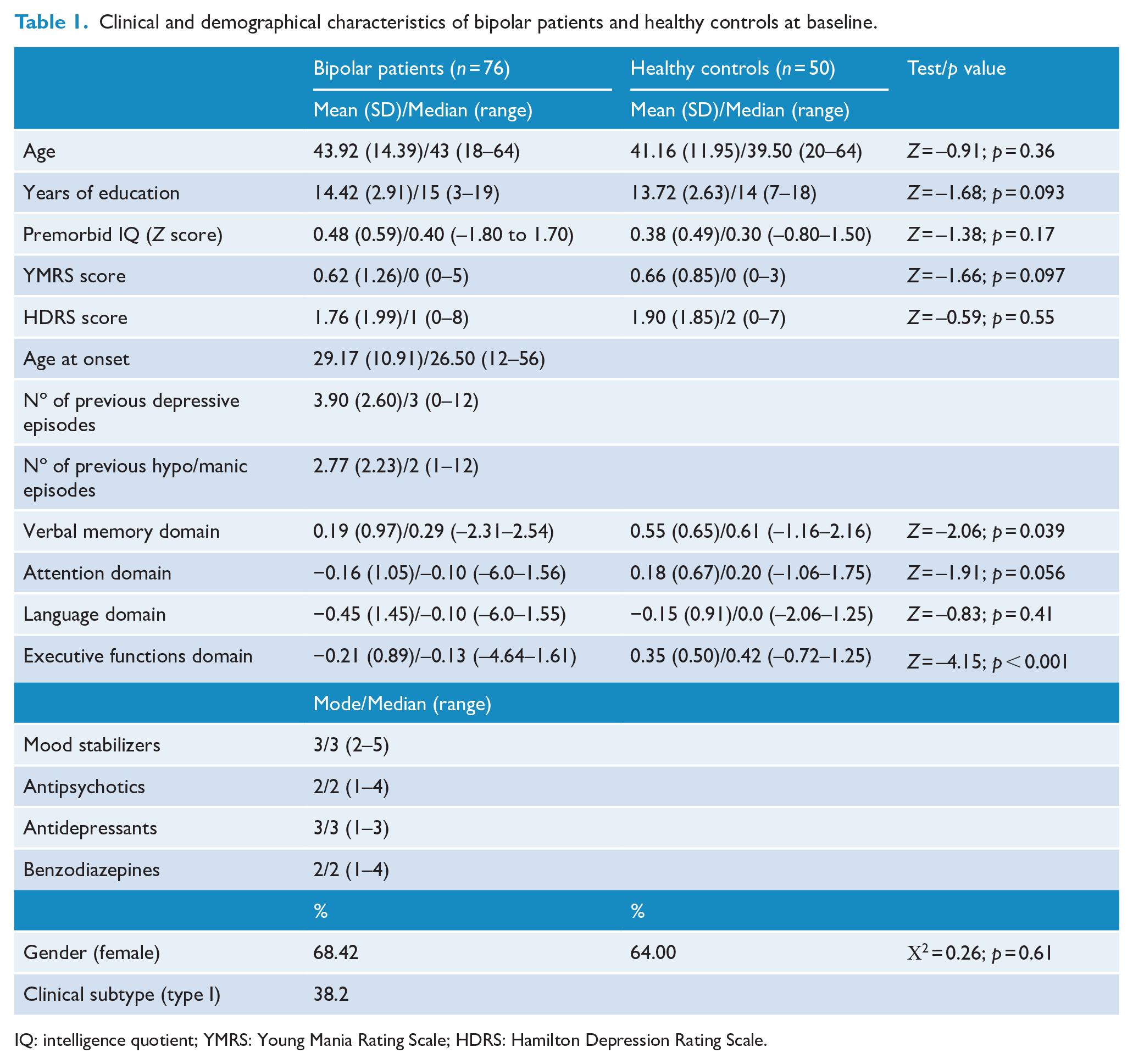
IQ: intelligence quotient; YMRS: Young Mania Rating Scale; HDRS: Hamilton Depression Rating Scale.
The period of follow-up was 70.73 (SD = 20.92, median = 68, range = 48–137) months during which patients experienced a mean annual rate (episode density) of 0.35 (SD = 0.31, median = 0.28, range = 0–1.06) depressive episodes and 0.22 (SD = 0.27, median = 0, range = 0–1.00) hypo/manic episodes. On average, patients spent 78.30% of the follow-up euthymic, 15.95% (range = 0–46.20) with depressive symptoms and 5.39% (range = 0–28.51) with hypo/manic symptoms.
The density of hypo/manic episodes during follow-up was associated with BD type I (Z = −3.09, p = 0.002), history of hypo/manic episodes (R = 0.38, p = 0.002), exposure to antipsychotics at baseline (Z = −2.46, p = 0.014), performance in verbal memory (R = −0.25, p = 0.031) and executive function domains (R = −0.27, p = 0.029) at baseline and global cognitive deficits (Z = −2.52, p = 0.012). These variables were entered in different multiple linear regression analyses, and the most parsimonious model was one that retained the BD type I (β = −0.32, t = −2.95, p = 0.004) and global cognitive deficits (β = 0.31, t = 2.87, p = 0.005), allowing to explain about 19% (adj. R2 = 0.187) of the variance of density of hypo/manic episodes during the follow-up (F = 9.28, p < 0.001).
The density of depressive episodes during follow-up was related only to BD type II (Z = −3.62, p < 0.001) and history of depressive episodes (R = 0.28, p = 0.021). In a linear regression model, only BD type II (β = 0.43, t = 4.08, p < 0.001) was an independent predictor of the density of depressive episodes during the follow-up period, explaining 18% of the variance (adj. R2 = 0.177, F = 16.65, p < 0.001).
Time spent with hypo/manic symptoms during follow-up was related to BD type I (Z = −3.08, p = 0.002), exposure to antipsychotics at baseline (Z = −3.21, p = 0.001), premorbid IQ (R = −0.24, p = 0.046), history of depressive episodes (R = −0.25, p = 0.046), presence of clinically significant cognitive deficits (Z = −2.20, p = 0.027) and global cognitive deficits (Z = −2.41, p = 0.016). The best linear regression model was the one that included BD type I (β = −0.385, t = −3.56, p = 0.001) and global cognitive deficits (β = 0.24, t = 2.17, p = 0.033), accounting for around 20% of variance of the time spent with hypo/manic symptoms (adj. R2 = 0.197, F = 9.48, p < 0.001).
Time spent with depressive symptoms during follow-up period was related to BD type II (Z = −2.83, p = 0.005) and history of previous depressive episodes (R = 0.47, p < 0.001). In a linear regression model, the number of previous depressive episodes (β = 0.38, t = 3.24, p = 0.002) was the only independent predictor of time spent with depressive symptoms explaining 13% of the variance (adj. R2 = 0.129, F = 10.50, p = 0.002).
Discussion
To the best of our knowledge, this is the first study to evaluate the role of cognitive deficits as predictors of long-term clinical course in BD. No relationship was found between neurocognitive performance with the density of depressive episodes or the time spent with depressive symptoms over an average of almost 6 years of follow-up. These outcomes were predicted by the subtype of BD (type II rather than type I) and the history of previous depressive episodes, which is consistent with previous studies (De Dios et al., 2012; Perlis et al., 2006; Vieta et al., 1997).
On the contrary, cognitive impairments at baseline were independent predictors of both density of hypo/manic episodes and time spent with hypo/manic symptoms along the follow-up period. Although deficits in domains of verbal memory and executive functions were associated with the density of hypo/mania, the presence of global cognitive deficits was the best predictor of both hypo/manic outcomes (density and time spent ill). These findings would suggest that the extent of deficits, rather than the compromise of any particular cognitive domain, could be a risk marker for the development of hypo/manic symptoms in the long-term clinical course.
The relationship between cognitive deficits and manic episodes dates back to the first cross-sectional studies in the BD (for a review, see Robinson and Ferrier, 2006) and was reproduced in subsequent investigations (López-Jaramillo et al., 2010; Torres et al., 2010). It was interpreted as evidence that cognitive deficits increase as a function of successive episodes (López-Jaramillo et al., 2010; Torres et al., 2010) and was presented as one of the cornerstones of the concept of neuroprogression and staging proposed for the BD (Berk et al., 2007; Kapczinski et al., 2009). However, meta-analysis of longitudinal studies in recent-onset, middle-age and later-life BD patients did not find evidence of decline in neurocognitive functioning (for review, see Bora and Özerdem, 2017; Samamé et al., 2014; Szmulewicz et al., 2019). Likewise, neurocognitive studies conducted on older adults with BD indirectly also did not support the view of a cognitive decline (for a review, see Samamé et al., 2013). Therefore, although limited by the relatively short follow-up periods of longitudinal studies and risk of selection bias in older adults, the evidence currently available suggests stable neurocognitive functioning throughout the course of the BD. In this context, the relationship found between neurocognition and manic morbidity in this study and previous studies could suggest the existence of subgroups or profiles of BD patients rather than the progression of deficits over time.
In fact, several studies using different methodologies have consistently reported the existence of subgroups of patients with preserved cognitive functioning, others with selective deficits and others with global deficits (Burdick et al., 2014; Cullen et al., 2016; Martino et al., 2014; Roux et al., 2017; Volkert et al., 2015). This pattern tends to be reproduced in different age groups (Martino et al., 2018b) and to remain stable over time (Martino et al., 2017). However, the assessment of the relationship of these cognitive subgroups with number of episodes is incipient and the results inconclusive. While some cross-sectional studies show a tendency toward fewer previous affective episodes in the preserved cognitive functioning group (Burdick et al., 2014; Martino et al., 2018a), other studies do not show differences between subgroups (Lima et al., 2019; Roux et al., 2017; Volkert et al., 2015). Based on our results, we propose that episode density more than the number of episodes could be a more accurate measure of the clinical course to assess this relationship. Of course, longitudinal studies also have the advantage of a lower risk of recall bias, which could be higher among patients with more cognitive impairments. The results of this study agree with the findings of the only previous longitudinal study that evaluated the relationship between cognitive subgroups and clinical course. In that study, Frías et al (2017) using latent class analysis identified three cognitive subgroups in youth BD, and patients in the ‘low’ cognitive functioning subgroup spent more time with manic and depressive symptoms during a follow-up period of 2.5 years. Thus, these first longitudinal studies seem to show the existence of profiles of patients with more and less cognitive deficits and manic morbidity. If these profiles respond only to a continuum of severity of the BD or if they show underlying pathophysiological differences, it should be elucidated in future studies (Martino et al., 2018a).
Several considerations must be taken into account to interpret the results of this study. First, the sample size is relatively small limiting especially subgroup analysis. Second, we included only patients with a follow-up period of more than 48 uninterrupted months, which could imply a potential selection bias. However, we compared the sample of patients included in this study with a random sample of patients of our database not included and there were no differences between in any clinical or neurocognitive variables at baseline (all p > 0.05). Moreover, our sample had a relatively benign course based on the shortest time spent ill compared with previous studies (Judd et al., 2002, 2003). This might be a consequence of having included patients with strict criteria of euthymia at baseline. Finally, all patients were taking psychotropic medications. Hence, although exposure to medications was included in our regression models when necessary, drug-related effects cannot be completely excluded from the interpretation of the findings.
Notwithstanding these limitations, this study contributes to understand the relationship between neurocognition and long-term clinical course in BD. Our results suggest that global cognitive deficits could be an independent predictor of hypo/manic morbidity. Future research is necessary to clarify the nature of this association and to determine how improvements in neurocognitive functioning could be of benefits for the clinical course of patients with BD.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
