Abstract

Introduction
We report a case of lurasidone use in a mother who was exclusively breastfeeding, with no signs of infant toxicity for 39 days post-partum, and a relative infant dose (RID) of 0.29%. Lurasidone is a novel second-generation antipsychotic, with a favourable metabolic profile (Javed et al., 2019). Given the ever-clearer link between some second-generation antipsychotics and gestational diabetes (Galbally et al., 2020), there is a search for alternatives in the peripartum period. Antipsychotic lactation safety data is urgently required in order to safely prevent and treat post-partum illness episodes in patients with mood and psychotic disorders and to allow maintenance of breastfeeding for infants and their mothers.
Lurasidone may sequester in breast milk as it is a small molecule, lipophilic and a weak base, with a fairly long elimination half-life of 18–40 hours. On the contrary, it is highly protein bound at 99%. It has fairly low oral bioavailability compared to other antipsychotics, plasma levels peak at 1–3 hours and steady state is reached within 7 days. Lurasidone undergoes hepatic metabolism via cytochrome P450 3A4, which does not significantly vary in effect between individuals (Greenberg and Citrome, 2017). Rat data indicate breast milk-to-maternal serum ratio of 7:1 at 1 hour, 13:1 at 2 hours and 4:1 at 24 hours. There are no data for rat pup serum levels (Sunovion Pharmaceuticals Inc, 2009).
Methods
The mother gave written consent for publication. The ACT Health Human Research Ethics Committee determined that ethics review was not required.
Peak maternal and infant blood samples and maternal breast milk were collected 5 days post-partum. Steady-state maternal blood and breast milk samples were collected after 41 days. Timing of collection was necessarily influenced by clinical considerations: repeated blood and milk tests overnight would have allowed for a curve to be plotted, but were clinically unwise. Day 5 sampling offered the best compromise between establishment of milk production and impending hospital discharge.
Lurasidone concentrations were determined using a validated ultra-performance liquid chromatography (UPLC) tandem mass spectrometry method. Samples were deproteinated with acetonitrile containing Lurasidone-d8 (internal standard) and 2 µL of supernatant injected onto a UPLC reverse phase column. The Lurasidone and Lurasidone-d8 were detected and quantitated using analyte-specific mass transitions. The range of the assay was 0–50 µg/L. Intra-assay and inter-assay coefficient of variation was <5%.
The mother was clinically examined throughout the study period by a psychiatrist. The infant was examined at birth and in the post-natal ward by a neonatology fellow and then followed up in child health nurse clinics.
Maternal characteristics
The mother was 34 years with a 3-year-old child, in her second pregnancy. She suffered from schizoaffective disorder, depressive type, predating her first pregnancy. She had discontinued medications during the first pregnancy, and, unmedicated, developed psychotic depression 3 months post-partum. She achieved remission with lurasidone 80 mg nocte and desvenlafaxine 50 mg mane, after failing to tolerate other medicines. After a detailed prenatal exploration of treatment options, including a discussion of options with a stronger safety record, she elected to proceed with a second pregnancy unmedicated, and to recommence psychotropics post-partum. Gestational diabetes was controlled by diet. After birth, she recommenced desvenlafaxine 50 mg mane and lurasidone 40 mg nocte. Exclusive breastfeeding was successful. She reported mild sedation from the lurasidone, but it did not impair her ability to wake and feed her baby. She remained well through the study period.
Results
Infant clinical outcomes
The infant was a healthy male born at 37 + 6 weeks gestation through spontaneous vaginal delivery weighing 3330 g (between 50th and 90th centile), and 3220 g at 5 days post-partum. His length was 50 cm (between 50th and 90th centile) and his head circumference was 36 cm (90th centile). Apgar was 8 at 1 minute and 9 at 5 minutes of age. He had an isolated temperature of 38 degrees on day 1 which resolved spontaneously. His C-reactive protein level and full blood count were in the normal range for his age. Neonatal examination after birth and on day 3 was unremarkable. Neurological examination at day 5 did not reveal any abnormalities. Follow-up in child health nurse clinics in the following days and weeks revealed normal weight gain and appropriate initial development. At 39 days, he was exclusively breast fed, feeding on demand every 2–3 hours, sleeping 4–5 hours overnight, bowels open regularly, gaining 377 g/week, between the 50th and 85th percentile for weight and length and at the 85th percentile for head circumference. He was noted by a child health nurse to be alert and responsive, with positive interactions between mother and son.
Laboratory measures
Milk and plasma lurasidone levels are given in Table 1.
Milk and plasma lurasidone levels at 5 and 41 days post-partum.
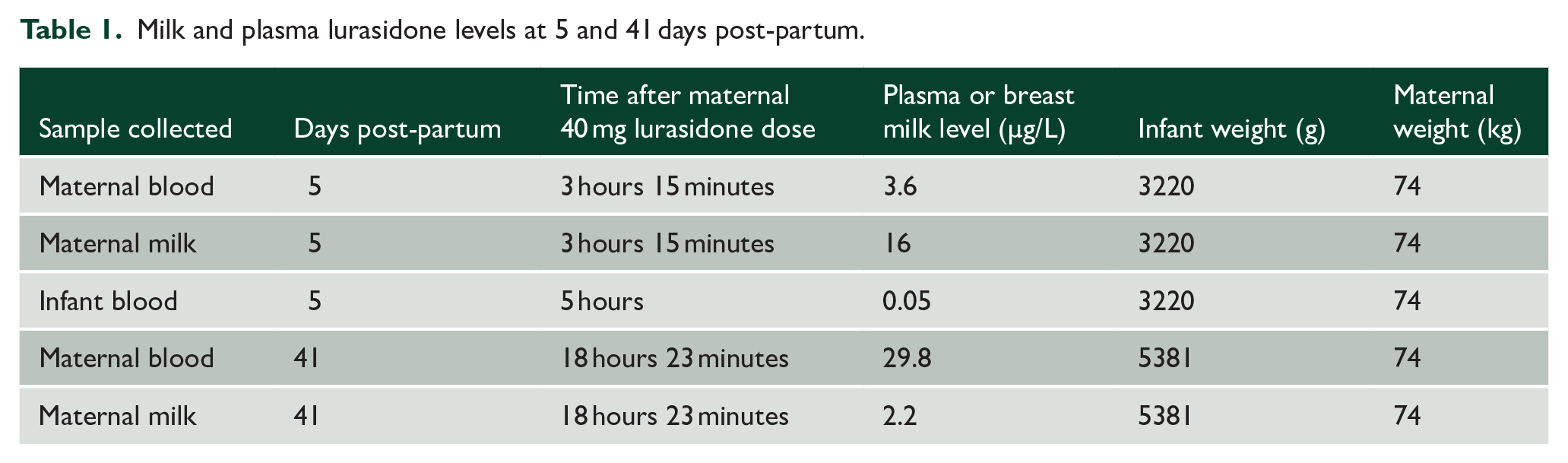
Previously published adult lurasidone serum levels reach a maximum concentration 2 hours post ingestion, at 48 µg/L for a 40 mg dose (Naccarato et al., 2016). Based on our maternal milk levels of 16 µg/L, and assuming 100 mL/kg/day infant milk ingestion, the infant would ingest 0.0016 mg/kg/day. This would give an RID of 0.29%. The relatively high maternal plasma lurasidone level at 41 days does not seem in keeping with known lurasidone adult pharmacokinetics and raises the possibility that the patient did not take the medication as prescribed. This reduces our confidence in using these data for calculations.
Discussion and conclusion
Serum lurasidone levels in a breastfed infant were negligible 5 hours after maternal ingestion, despite some drug sequestering in breast milk. This may be accounted for by low RID and low oral bioavailability. Although there were no immediately observed impacts on infant growth and development, a limitation of this report is that a standardised developmental assessment such as a Bayley Scale or the Ages & Stages Questionnaire, Third Edition, was not conducted. There were insufficient data points to plot a curve for maternal milk levels or infant blood levels of lurasidone, and so we were not able to calculate area under the curve concentrations, accurate mean milk concentrations, or derive peak milk or infant lurasidone levels. All these results merit repetition.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
