Abstract

The use of lithium has recently declined, despite it being the only medication that has specificity for the treatment of bipolar disorder and worldwide affordability (Tondo et al., 2019). Part of the reason for this is the diagnostic distortion of manic-depressive illness to create a family of ‘bipolar’ disorders, alongside depressive disorders. It is important to bear in mind that, in Diagnostic and Statistical Manual of Mental Disorders (5th ed.; DSM-5) for example, ‘bipolar disorder’ is a statistical (and largely theoretical) construct that came into being for classificatory purposes and that it has been sustained partly because of prescriptive, legal and arguably economic reasons. Furthermore, in both DSM-5 and International Classification of Diseases (ICD) 10/11, the construction of bipolar disorder disproportionately favours polarity, at the expense of recurrence. Thus, in the current taxonomy, bipolar disorder is principally a cross-sectional entity upon which categorical status has been foisted.
It is interesting to note that unlike bipolar disorder, manic-depressive illness (its forerunner) emphasised recurrence more so than polarity and, in doing so, captured those patients with highly recurrent depression. A significant number of these patients would develop mania later in the course of their illness – a natural developmental transition, but one that is now in a system which prioritises polarity, characterised erroneously as a capricious transmutation of diagnosis from major depression to bipolar disorder. In sum, the classification of mood disorders in current classificatory systems lacks specificity with respect to predicting long-term course. But perhaps even more significant is the loss of diagnostic patterns that held treatment specificity.
In addition to diagnostic equivocation obscuring the target of lithium therapy, the other major reason why lithium has fallen out of clinical favour is that unlike its competitors it has lacked widespread marketing. In contrast, all new agents that have gained an indication for the treatment of bipolar disorder in the past two decades have had robust sponsorship and have been extensively promoted. At the same time, discussions on the long-term management of bipolar disorder have emphasised the inconvenience of monitoring, poor tolerability and long-term serious side effects of lithium, many of which have been found to be exaggerated. Together these factors, a lack of promotion and excessive concerns regarding the side effects of lithium, have discouraged its prescription. To rectify this unjust state of affairs and restore lithium to its rightful position as the foremost agent in the pharmacotherapy of bipolar disorders, and arguably the only true mood stabiliser, we have briefly outlined five important reasons why lithium must be given consideration in the management of bipolar disorder.
One
Lithium is the only agent with proven long-term mood-stabilising properties and is effective in treating bipolar disorder, both acutely and long term, with somewhat better efficacy in addressing mania than depression. However, maintenance of the euthymic phase of bipolar disorder is perhaps the most important period of the illness, as it affords the greater opportunity for patients to undertake psychological and lifestyle preventive measures. It is also when patients regain functioning by re-establishing relationships and social connections. Only lithium has unequivocally demonstrated an ability to maintain euthymia and have a long-term preventive effect over decades. And we can all agree that the prophylactic property of an agent prescribed for a chronic illness is its most fundamental action. Unfortunately, in practice, the focus of management is often biased towards acute treatments and, instead of prioritising efficacy, centres excessively on tolerability. Of course, side effects are important and need to be carefully considered, but they should not be given priority over determining whether a medication works (see below).
Two
While lithium has some side effects, it is generally well tolerated with few long-term adverse effects. Rates of renal dysfunction, the greatest concern, are low; as noted in a recent systematic review with rates of renal failure of 0.5%, and while thyroid dysfunction has an odds ratio of 5.78, it is correctable (Shine et al., 2015). The second-generation antipsychotics (SGAs), on the other hand, can be over sedating and lead to weight gain as part of a more extensive and serious metabolic syndrome, which drive premature mortality, and, contrary to common perceptions, can still cause extrapyramidal side effects (including akathisia). Therefore, just as with efficacy, when considering tolerability, lithium is to be favoured over many widely used SGAs.
Three
Lithium has true specificity. It is rarely used outside of mood disorders and it has been shown to be particularly effective in treating bipolar disorder. Comparing lithium to the SGAs for the treatment of bipolar disorder is akin to the use of a narrow- versus broad-spectrum antibiotic. The latter is a blunderbuss approach that kills indiscriminately ‘good’ and bad bacteria, whereas with appropriate dosing, lithium allows targeted therapy with less side effects. For instance, lithium is not used in schizophrenia or other psychotic disorders and does not have sedative or anxiolytic effects. Indeed, some have argued that lithium responsivity may be one of the best biomarkers available, in parsing out ‘Cades Disease’, which may differ biologically from other forms of mood disorder.
Four
The fact that the plasma levels of lithium can be easily and reliably measured allows it to be titrated carefully and ensure adherence. This means both its efficacy, and any potential tolerability issues, can be gauged accurately (see Box 1). This should not be regarded as an inconvenience or limitation, either for the patient or the doctor, and instead it should be considered an important advantage, one that SGAs do not afford. Most other treatments for bipolar disorder are prescribed somewhat ‘blind’ with no objective means of determining whether the patient has taken the medication, and if they have, whether the dose is sufficient. Being able to measure the serum level in relation to its clinical effects is an extraordinary benefit and one that allows pharmacotherapeutic precision (see Figure 1). Furthermore, an added advantage of regular monitoring is it ensures regular clinical contact that encourages clinical review (along with symptom monitoring) and assessment of progress and outcomes. Given the importance of the therapeutic alliance in the management of long-term chronic illnesses, this is a critical benefit.
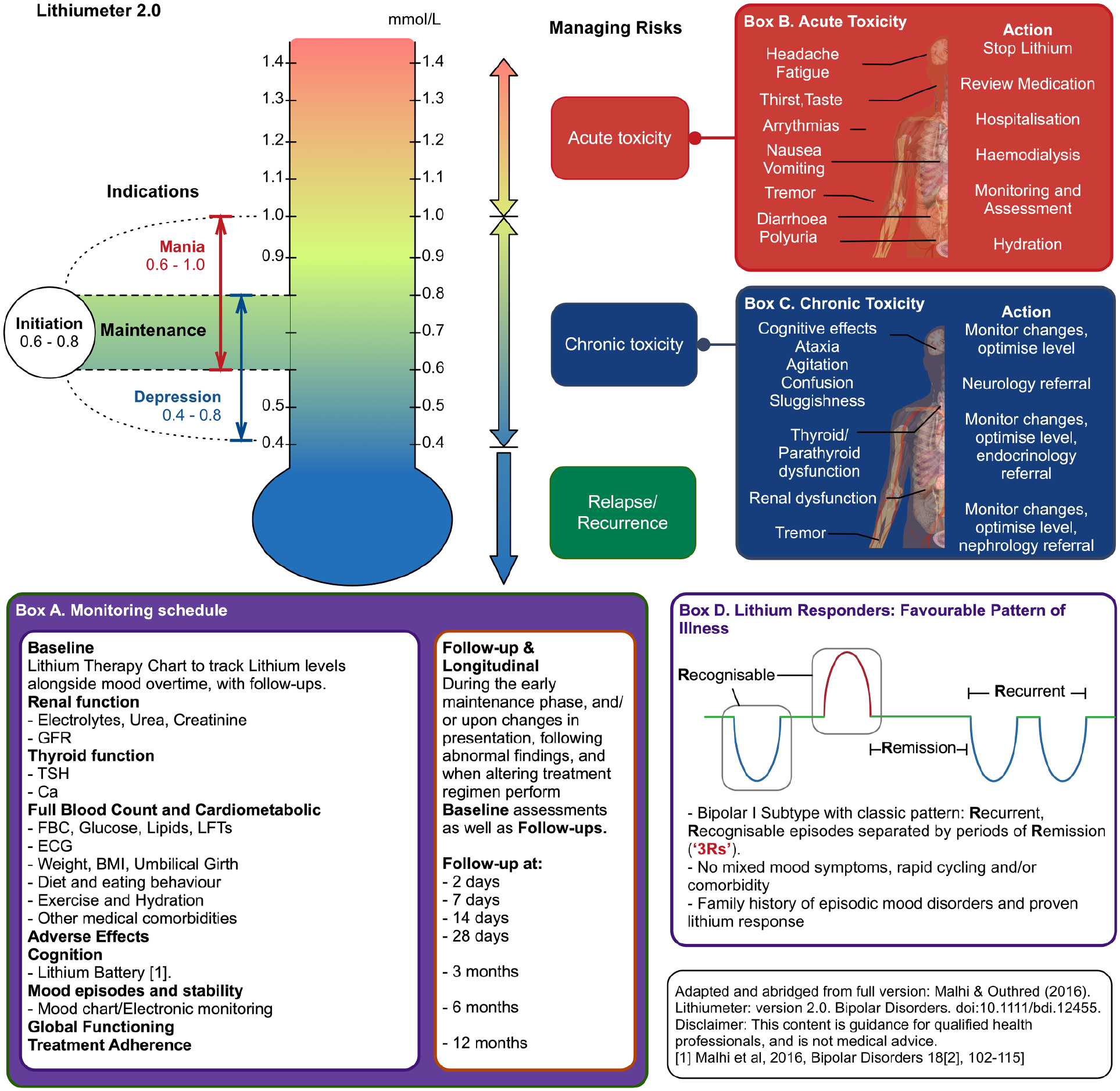
Abridged version of the Lithiumeter. This schematic illustrates the ideal plasma levels of lithium for each indication within bipolar disorder. The schematic also outlines a proposed monitoring schedule (Box A), the symptoms and management strategies for both acute lithium toxicity (Box B) and chronic lithium toxicity (Box C). Finally, the schematic outlines the pattern of illness that is most likely to respond to lithium therapy (Box D). For further explanation, see Malhi et al. (2016).
Practical guidance for lithium therapy.

Source: Adapted from Malhi et al. (2016) and Tondo et al. (2019).
ACE: angiotensin-converting enzyme.
Where possible should be conducted every 3–6 months.
Especially in hot climates and when inadequately hydrated.
Five
Lithium has some unique protective properties. First is its protection against suicide in mood disorders. This is thought to occur at all levels (therapeutic and subtherapeutic) and clearly reducing the likelihood of suicide is a significant benefit, but especially so in the management of bipolar disorder, where the risk of suicide is greater than any other psychiatric conditions. In practice, there is often concern that lithium is toxic if ingested in large amounts as in an overdose, but this has to be balanced against its significant long-term benefits. The degree of suicidal intent an individual is experiencing has to be determined carefully and is an important consideration when prescribing lithium. However, suicidal thoughts, in and of themselves, should not be a contraindication to the prescription of lithium. Second, lithium has a neuroprotective effect arising from its ability to protect against white matter and grey matter loss, encourage neuronal regeneration and diminish potential progressive cognitive decline. This is once again pertinent to patients with bipolar disorders as they are at increased risk of dementia, and lithium appears to be clearly neuroprotective in this regard.
In contrast, and of note, some SGAs might cause neuronal cell loss, although this remains uncertain. Third, lithium has several general medical health benefits, for example, in terms of all-cause mortality, and specifically cardiovascular mortality, which is of critical benefit given the increased mortality of patients with bipolar disorder. Furthermore, many antidepressants and antipsychotics cause osteoporosis, and lithium is the only agent that enhances bone integrity and reduces fracture risk (Liu et al., 2019). Finally, recent data suggest that lithium may uniquely reduce the risk of cancer (Huang et al., 2016).
Thus, in practice, lithium should be prescribed to all patients with bipolar disorder, unless there is a specific reason why an alternative is preferable, or better suited. Typically, lithium is best suited to ‘classic’ bipolar disorder, where there are clear episodes of mania and depression separated by periods of remission. However, it is also useful in the treatment of bipolar disorder more broadly and should be given far greater consideration whenever managing bipolar disorder.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: G.S.M. has received grant or research support from National Health and Medical Research Council, Australian Rotary Health, NSW Health, American Foundation for Suicide Prevention, Ramsay Research and Teaching Fund, Elsevier, AstraZeneca, Janssen-Cilag, Lundbeck, Otsuka and Servier; and has been a consultant for AstraZeneca, Janssen-Cilag, Lundbeck, Otsuka and Servier. R.J.P. has received support for travel to educational meetings from Servier and Lundbeck and uses software for research at no cost from Scientific Brain Training Pro. P.B. has received research support from the NHMRC, has received speaker fees and educational grants from Servier and educational support from Lundbeck. M.H. has received grant or research support in the last 5 years from the National Health and Medical Research Council, Medical Research Future Fund, Ramsay Health Research Foundation, Boehringer-Ingleheim, Douglas, Janssen-Cilag, Lundbeck, Lyndra, Otsuka, Praxis and Servier; and has been a consultant for Janssen-Cilag, Lundbeck, Otsuka and Servier. M.B. has received Grant/Research Support from the NIH, Cooperative Research Centre, Simons Autism Foundation, Cancer Council of Victoria, Stanley Medical Research Foundation, Medical Benefits Fund, National Health and Medical Research Council, Medical Research Futures Fund, Beyond Blue, Rotary Health, A2 milk company, Meat and Livestock Board, Woolworths, Avant and the Harry Windsor Foundation; has been a speaker for Astra Zeneca, Lundbeck, Merck and Pfizer; and served as a consultant to Allergan, Astra Zeneca, Bioadvantex, Bionomics, Collaborative Medicinal Development, Lundbeck Merck, Pfizer and Servier – all unrelated to this work. RM has received support for travel to education meetings from Servier and Lundbeck, speaker fees from Servier, and Committee fees from Janssen. E.B., P.H., D.B., R.A.B., B.L. and G.M. declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: M.B. is supported by a NHMRC Senior Principal Research Fellowship (1059660 and 1156072).
