Abstract
Background:
Clozapine is the most effective medication for treatment refractory schizophrenia, but is associated with cardiac adverse drug reactions. Myocarditis and cardiomyopathy are the most serious cardiac adverse drug reactions although reported rates of these conditions vary in the literature. We systematically reviewed and meta-analysed the event rates, the absolute death rates and case fatality rates of myocarditis and cardiomyopathy associated with clozapine.
Methods:
PubMed, EMBASE and PsycINFO were searched for studies that reported on the incidence of cardiomyopathy or myocarditis in people exposed to clozapine. Data were meta-analysed using a random effects model, with subgroup analysis on study size, time frame, region, quality, retrospective vs prospective, and diagnostic criteria of myocarditis or cardiomyopathy.
Results:
28 studies of 258,961 people exposed to clozapine were included. The event rate of myocarditis was 0.007 (95% confidence interval [CI] = [0.003, 0.016]), absolute death rate was 0.0004 (95% CI = [0.0002, 0.0009]) and case fatality rate was 0.127 (95% CI = [0.034, 0.377]). The cardiomyopathy event rate was 0.006 (95% CI = [0.002, 0.023]), absolute death rate was 0.0003 (95% CI = [0.0001, 0.0012]) and case fatality rate was 0.078 (95% CI = [0.018, 0.285]). Few included studies provided information on criteria for diagnosis of myocarditis and cardiomyopathy. Event rates of cardiomyopathy and myocarditis were higher in Australia.
Conclusion:
Clarity of diagnostic criteria for myocarditis remains a challenge. Observation bias may, in part, influence higher reported rates in Australia. Monitoring for myocarditis is warranted in the first 4 weeks, and treatment of comorbid metabolic syndrome and diabetes may reduce the risk of cardiomyopathy. The risks of myocarditis and cardiomyopathy are low and should not present a barrier to people with treatment refractory schizophrenia being offered a monitored trial of clozapine.
Introduction
Among people with treatment refractory schizophrenia, clozapine is the most effective agent for reducing positive symptoms (Siskind et al., 2016) and hospitalisations (Land et al., 2017). However, it may be underused (Bachmann et al., 2017) due to concerns regarding major adverse drug reactions (ADR) (Verdoux et al., 2018). Clozapine-associated myocarditis and cardiomyopathy were first recognised as a potential severe ADR following several related deaths that were reported in Australia (Kilian et al., 1999). Since then, numerous studies have reported the incidence of clozapine-associated cardiomyopathy and myocarditis with an estimated rate of between 0.03% and 8.5% for myocarditis (Reinders et al., 2004; Rohde et al., 2018) and 0.02% and 1% for cardiomyopathy (Kilian et al., 1999; Layland et al., 2009). However, there is substantial variation in observed rates between geographical regions particularly higher rates reported in Australia (Ronaldson et al., 2015).
Confirming a diagnosis of clozapine-associated myocarditis is challenging because the only definitive diagnosis involves cardiac biopsy (Aretz et al., 1987; Cooper, 2009). Given the invasiveness of cardiac biopsy, diagnostic criteria are often used, but the evidence for these is limited (Knoph et al., 2018). For instance, most published diagnostic criteria are consensus-based and reported by single institutions (Ronaldson et al., 2011; Youssef et al., 2016). Furthermore, these diagnostic criteria include non-specific symptoms such as dyspnoea, palpitations, tachycardia, fever, fatigue and chest pain (Citrome et al., 2016) that may be insufficiently specific for cardiac disease aetiologically related to clozapine.
Meta-analyses examining the epidemiology of clozapine-associated myocarditis and cardiomyopathy could therefore help to determine study factors which might explain the variability in reported event rates and establish a benchmark incidence for future research. We aimed to systematically review the current literature on myocarditis and cardiomyopathy rates among people on clozapine and to meta-analytically determine the event rates of these cardiac ADRs, as well as the absolute mortality and case fatality rate.
Methods
The methods are based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) (Moher et al., 2009). The PROSPERO registration number is CRD42018094658.
Searches
Systematic searches were conducted of publications indexed in PubMed, EMBASE and PsycINFO using the search terms (clozapine OR clozaril OR zaponex OR clopine) AND (cardiomyopathy OR myocarditis) (see Figure 1). The abstracts and titles of articles identified through electronic searches were reviewed by three authors (A.S., J.C. and Y.-T.C.) to identify studies that may have reported on the event rates of cardiomyopathy or myocarditis in people exposed to clozapine. Papers in all languages were considered for inclusion. No unpublished data or non-peer reviewed data were considered for inclusion.
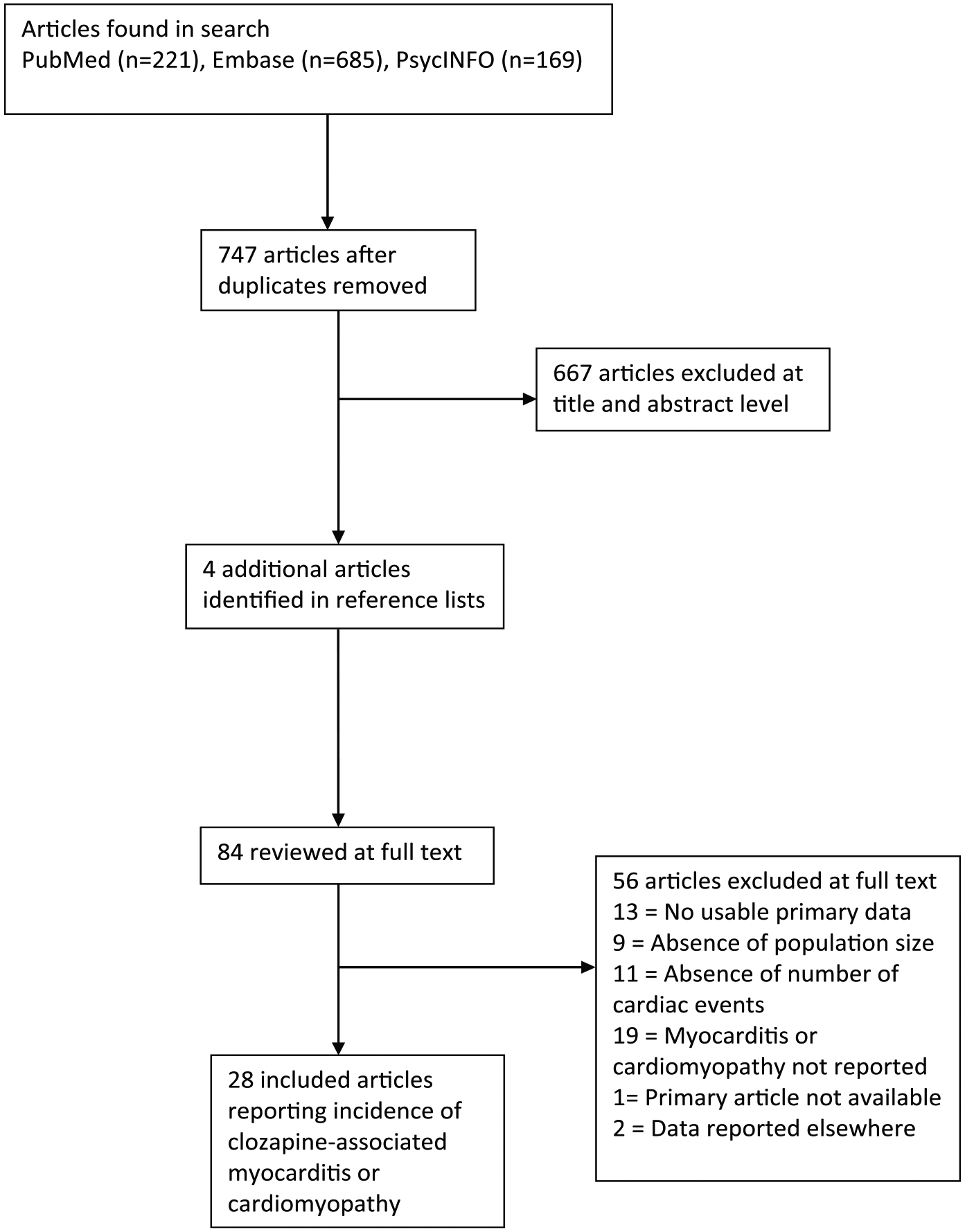
PRISMA diagram.
Inclusion criteria
Studies were included if they reported on cohorts followed up after exposure to clozapine that reported events of, and/or death from, myocarditis or cardiomyopathy at follow-up. We included randomised controlled trials (RCTs) or cohort studies of adults.
Exclusion criteria
Studies were excluded if they reported on cross-sectional data, as these studies may omit those who had ceased clozapine prior to cross-sectional data collection. Case reports and case series were excluded as were studies that included subjects aged less than 18 years.
Assessment of reporting strength
Considering that most studies suitable for inclusion would be observational studies, we used a modified Newcastle-Ottawa Quality Assessment Scale (Stang, 2010) for assessment of reporting strength. The maximum score was nine, and studies with a score of six or above were rated as high quality. We considered the following domains:
Representativeness of the group with the condition of interest – considered higher quality if they reported consecutively recruited subjects on clozapine who develop the cardiac condition of interest.
Representativeness of the group without the condition of interest – considered higher quality if they reported consecutively recruited subjects on clozapine who do not develop the cardiac condition of interest.
Ascertainment of exposure – considered higher quality if they reported subjects included as part of a prospective cohort study, and lower quality studies were defined as those that used registry data for ascertainment of exposure.
Demonstration that outcome of interest was not present at start of study – considered higher quality if baseline echocardiography and/or cardiac biomarker and/or other form of cardiac imaging was performed at study baseline to exclude cardiac disease prior to enrolment in the study.
Comparability – considered higher quality if they statistically controlled for one or more clinical factors for outcome reporting (studies that controlled for more than one factor were awarded two quality points).
Assessment of outcome – considered higher quality if a case was defined by pre-specified diagnostic criteria rather than registry data or hospital records.
Adequacy of follow-up – considered to have adequate follow-up for myocarditis with median follow-up of 2 months and for cardiomyopathy with median follow-up of 3 months consistent with previously published data (Curto et al., 2015).
Attrition – considered to have higher reporting strength if attrition was less than that reported in studies of clozapine discontinuation (Forrester et al., 2015) (i.e. <80% at 3 months, <75% at 6 months, <60% at 12 months and <50% at 2 years).
Definitions of clozapine exposure and case ascertainment
Clozapine exposure was defined according to the study protocol. Exposure could be defined as receipt of prescription in registry studies, prescription of clozapine according to hospital records in cohort studies or direct prescription of drug in RCTs. No minimum or maximum dose of clozapine was defined. We pre-specified no threshold for diagnosis of cardiomyopathy or myocarditis. Events were defined as per the study’s methodology regardless of the diagnostic criteria used. The diagnostic criteria used to define a case were collected for each study for narrative comparison.
Data extraction
Two authors independently extracted the data into an electronic spreadsheet, and disagreements were resolved by joint examination of the papers. The following data were extracted:
Sample size of subjects initiating clozapine.
Total number of myocarditis or cardiomyopathy events at follow-up to allow reporting of event rates of clozapine-associated cardiomyopathy or myocarditis.
Number of deaths attributed to clozapine-associated myocarditis or cardiomyopathy.
The following characteristics of each study were also recorded where possible:
Mean age of subjects included in the study.
The study type (cohort, case-controlled, RCT).
Whether data collection was prospective or retrospective.
The diagnostic criteria used to define a case of clozapine-associated cardiomyopathy or myocarditis.
Country in which the data were collected.
Length of follow-up for study subjects.
Was the majority of the cohort recruited before or after 2000 (this threshold being the date of publication of the seminal report on clozapine myocarditis, Kilian et al., 1999).
Mean time following exposure that clozapine-associated myocarditis or cardiomyopathy occurred in study subjects.
Data synthesis
The primary outcomes were the event rates (defined as number of people diagnosed with the specific condition among all included subjects), absolute death rates (defined as the number of people who died of the specific condition respectively among all included subjects) and case fatality rates (defined as the number of people who died of the specific condition among all people diagnosed with the specific condition) of clozapine-associated myocarditis and cardiomyopathy. Narrative reviews of outcomes, study quality and methodological characteristics of included studies were undertaken. Meta-analyses were conducted using Comprehensive Meta-Analysis (Version 3.3). Given the observational nature of primary studies and expected high rates of heterogeneity, a random effects model was used for all the analyses. Because of the large number of studies with zero events, a continuity correction was calculated for these studies based on the raw event rate across all studies multiplied by the study size of the zero-event studies (Myles et al., 2018; Sweeting et al., 2004).
Subgroup and sensitivity analysis
Subgroup analyses were undertaken on time period of data collection (dichotomised as before vs after the year 2000), the region of study (Australia vs rest of world), type of study (retrospective vs prospective) and diagnostic criteria of myocarditis of cardiomyopathy (specified vs not specified). Sensitivity analyses were undertaken and restricted to studies of higher quality and studies reporting at least one event.
Publication bias
Where meta-analyses included 10 or more studies, publication bias was tested using funnel plot asymmetry with Kendall’s Tau, where low p-values suggest publication bias (Barendregt and Doi, 2016). In studies where there was evidence of publication bias, Duval and Tweedie’s trim and fill method was used to examine the possible effect of hypothetically missing samples on the pooled estimate of effect.
Results
In total, 747 unique articles were identified after duplicates were removed. Of them, 667 articles were excluded on review of title and abstract. An additional four articles were found in reference lists. A total of 56 articles were excluded at full text review (see Figure 1 and Supplementary Table 1), with 28 articles included in the meta-analysis, with 258,961 subjects.
Myocarditis
Twenty-four studies consisting of 256,635 subjects (mean per sample = 10,693, SD = 38,419) reported on clozapine-associated myocarditis (Table 1) (Amodeo et al., 2016; Baptista et al., 2016; Butcher et al., 2015; Curto et al., 2015; Degner et al., 2000; Fernandez et al., 2004; Haas et al., 2007; Hägg et al., 2001; Hollingworth et al., 2018; Hyde et al., 2015; Ifteni et al., 2014; Joy et al., 2017; Khan et al., 2017; Kilian et al., 1999; Kishi et al., 2013; La Grenade et al., 2001; Murch et al., 2013; Nielsen et al., 2012; O’Connor et al., 2010; Poyraz et al., 2016; Reinders et al., 2004; Rohde et al., 2018; Tirupati, 2006; Youssef et al., 2016). Median follow-up was 24 months (interquartile range = 1–72 months). Overall, study quality was good, with 11 rated as high quality (score 6 or above) and 13 as low quality. Studies from Australia (n = 10) were the most common, with the remainder from Europe (n = 8), South America (n = 1), North America (n = 3), Turkey (n = 1) and Japan (n = 1). Eight studies collected data before the year 2000, with the remainder (n = 16) collected in the year 2000 or after. Four studies were prospective, while 20 were retrospective. Only seven studies provided any criteria for the diagnosis of myocarditis (Baptista et al., 2016; Butcher et al., 2015; Degner et al., 2000; Khan et al., 2017; La Grenade et al., 2001; Reinders et al., 2004; Youssef et al., 2016). Among the seven studies that reported diagnostic criteria for myocarditis, the following criteria were used: increased troponin I (n = 4), electrocardiogram (ECG) abnormalities (n = 5), changes in echocardiogram (n = 4), autopsy results (n = 2) and a temporal relationship to ceasing or commencing clozapine (n = 2). Eight studies reported the time between clozapine commencement and onset of myocarditis, with six of these studies reporting onset of myocarditis within 4 weeks (see Table 1).
A meta-analysis of 24 samples found that clozapine exposure was associated with a myocarditis event rate of 7 per 1000 people (event rate = 0.007, 95% confidence interval [CI] = [0.003, 0.016]) with high between-sample heterogeneity (p < 0.01, I2 = 98%) (Table 2). On sensitivity analysis, when restricted to the 18 samples that reported at least one event, the myocarditis event remained the same with slightly wider confidence intervals (event rate = 0.007, 95% CI = [0.003, 0.019]) and similar between-sample heterogeneity (p < 0.01, I2 = 99%). The event rate was higher than the summary effect when restricted to high-quality studies (event rate 0.016, 95% CI = [0.007, 0.035], p < 0.01, I2 = 74%). A meta-analysis of 18 samples found that clozapine exposure was associated with an absolute death rate from myocarditis of 4 per 10,000 people (event rate = 0.0004, 95% CI = [0.0002, 0.0009]) with moderate heterogeneity (p < 0.01, I2 = 71). A meta-analysis of 12 samples found that the case fatality rate from clozapine-associated myocarditis was 12.7% (event rate = 0.127, 95% CI = [0.034, 0.377]) with high heterogeneity (p < 0.01, I2 = 90%) (see Table 2 for details of meta-analysis and sensitivity analysis).
Included studies myocarditis.
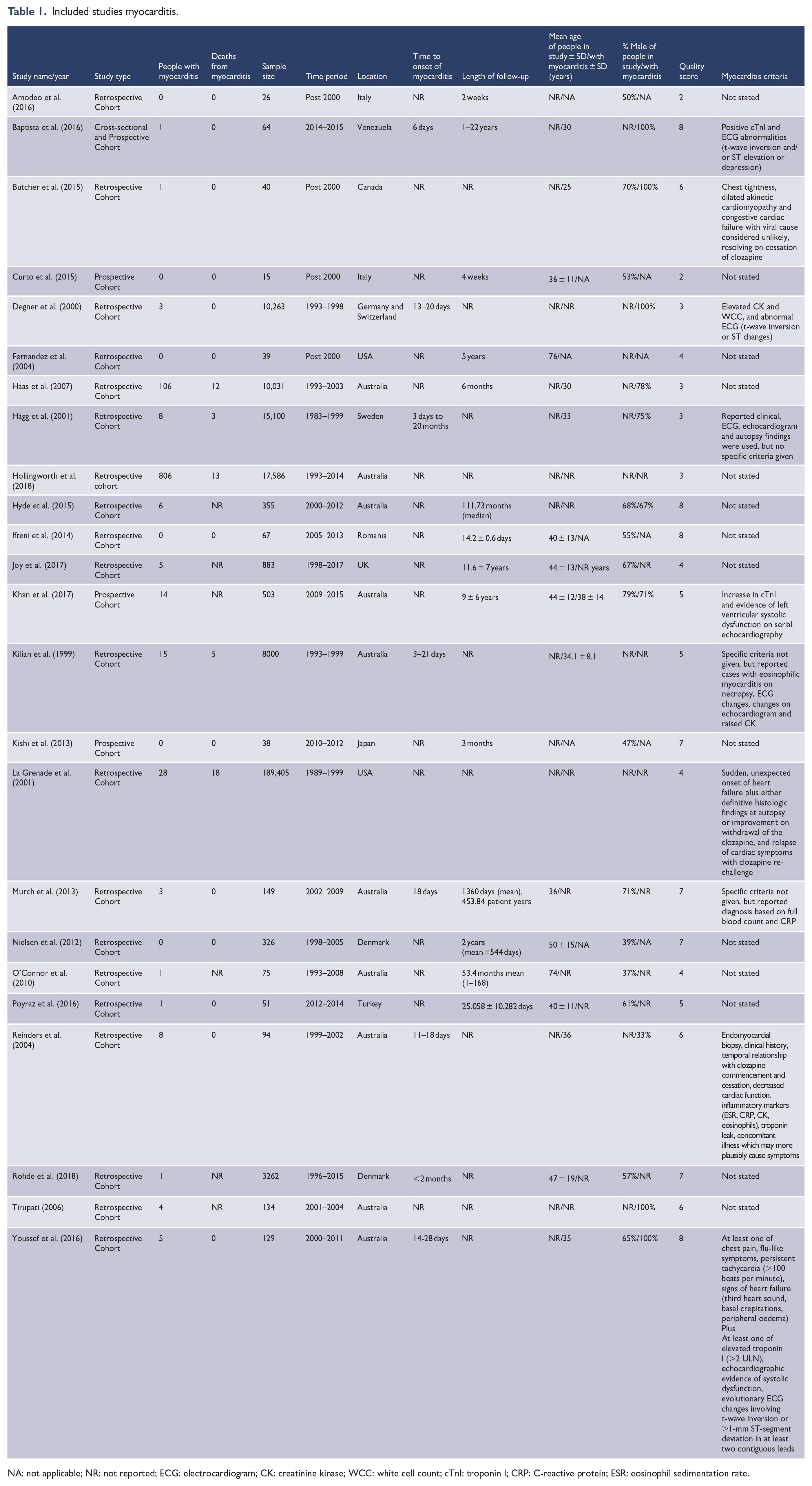
NA: not applicable; NR: not reported; ECG: electrocardiogram; CK: creatinine kinase; WCC: white cell count; cTnI: troponin I; CRP: C-reactive protein; ESR: eosinophil sedimentation rate.
Meta-analysis and sensitivity analysis of clozapine-associated myocarditis.
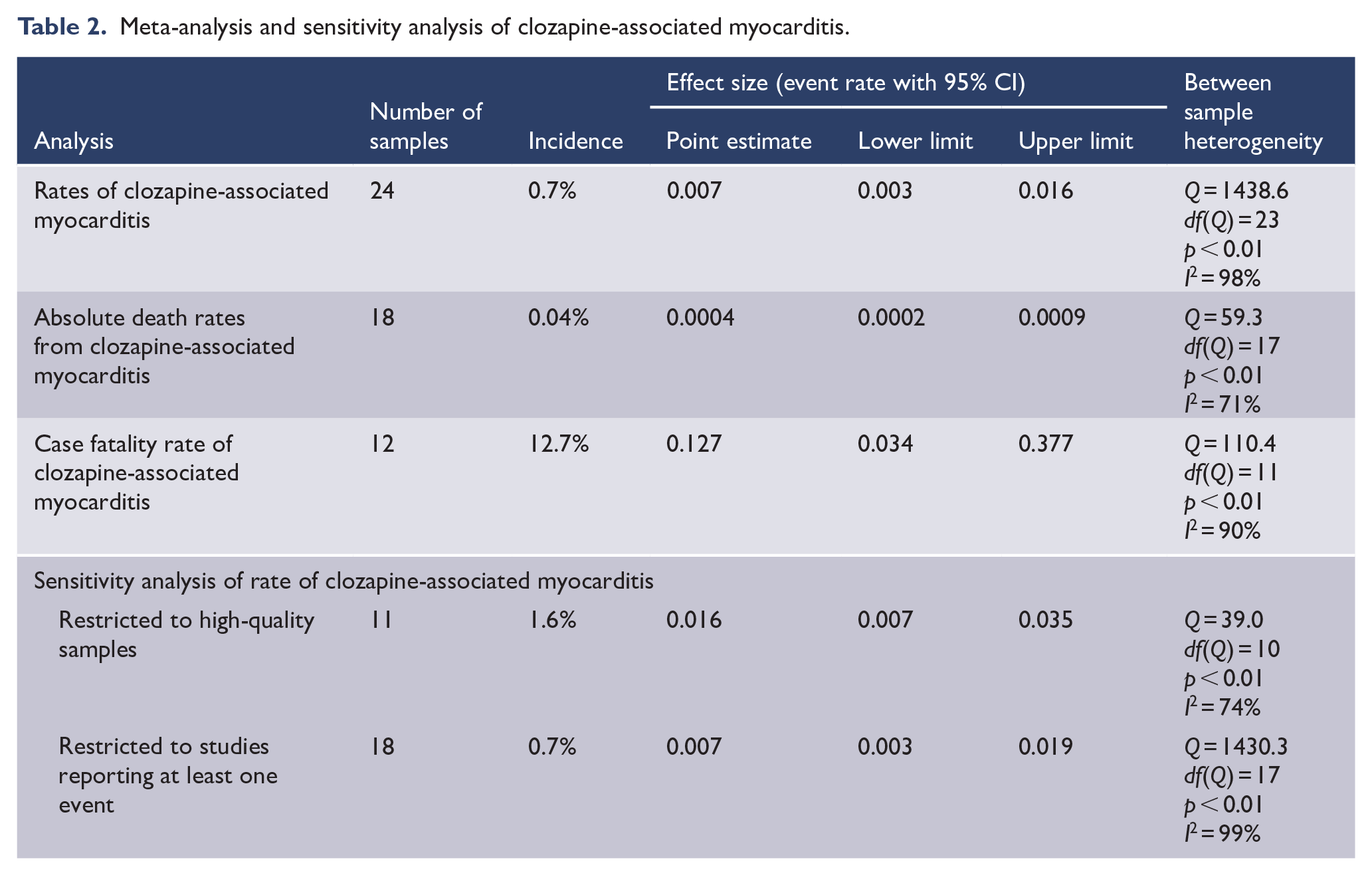
Subgroup analysis (Supplementary Table 2) demonstrated that the use of diagnostic criteria for myocarditis had no significant impact on effect size (Q = 0.3, p = 0.6) nor did data collection before or after the year 2000 (Q = 3.7, p = 0.06). The region in which data collection occurred explained a significant difference in between-study heterogeneity (Q = 6.9, p < 0.01) with Australian samples (nine samples, event rate = 0.020) reporting a significantly large effect size than samples collected elsewhere (15 samples, event rate = 0.003). Study methodology explained a significant difference in between-study heterogeneity (Q = 7.1, p < 0.01) with a higher event rate reported in prospective studies (four samples, event rate = 0.026) compared to retrospective studies (20 samples, event rate = 0.006).
There was no evidence of publication bias for analysis of absolute event rate (τ = 0.22, p = 0.13). Funnel plot is provided as Supplementary Figure 1. There was evidence of publication bias for the analysis of absolute death rate (τ = 0.42, p = 0.01), and trim and fill identified nine hypothetically missing studies to the right of mean reporting an adjusted event rate of 0.0007 (95% CI = [0.0002, 0.0020]). There was no evidence of publication bias for the analysis of case fatality rates (τ = −0.19, p = 0.41).
Cardiomyopathy
A total of 16 studies consisting of 220,493 subjects (mean per sample = 13,781; SD = 47,060) reported on clozapine-associated cardiomyopathy (Table 3) (Amodeo et al., 2016; Curto et al., 2015; Domenichetti et al., 2010; Hollingworth et al., 2018; Hyde et al., 2015; Joy et al., 2017; Kelly et al., 2009; Kidd et al., 2013; Kilian et al., 1999; La Grenade et al., 2001; Murch et al., 2013; Nielsen et al., 2012; O’Connor et al., 2010; Reinders et al., 2004; Rohde et al., 2018; Serrano et al., 2014; Youssef et al., 2016). Mean follow-up was 49 months (interquartile range = 19.5–73.5 months). Eight studies were from Australia, six from Europe, one from South America and two from North America. Three studies had the majority or all of their reporting period prior to the year 2000. Two studies were prospective with the remainder retrospective. All studies reporting time to cardiomyopathy reported that it was at least 2 months to the first event, with the majority over a year after commencement of clozapine. Four studies provided a diagnostic criteria of cardiomyopathy, with all citing echocardiogram findings, and three specifically defining an ejection fraction <50% as an essential criterion for diagnosis.
Included studies cardiomyopathy.
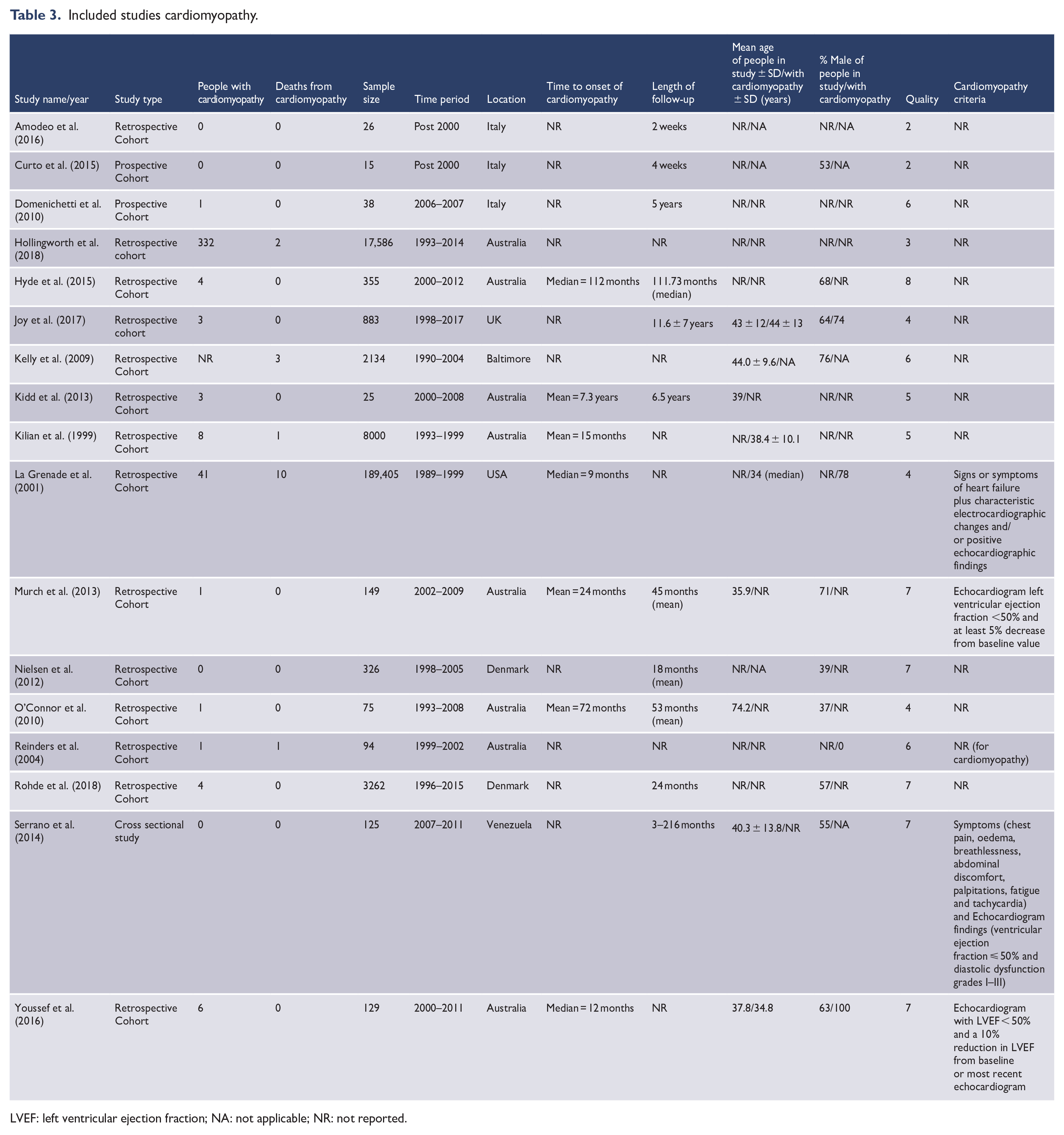
LVEF: left ventricular ejection fraction; NA: not applicable; NR: not reported.
Meta-analysis (Table 4) of 16 samples found that clozapine exposure was associated with a cardiomyopathy event rate of 6 per 1000 people (event rate = 0.006, 95% CI = [0.002, 0.023]) with high between sample heterogeneity (p < 0.01, I2 = 98%). On sensitivity analysis, the event rate was higher than the summary effect when restricted to the nine high-quality samples with slightly wider CIs (event rate = 0.011, 95% CI = [0.003, 0.036]) and similar between-sample heterogeneity (p < 0.01, I2 = 84%), and when restricted to studies reporting at least one event (event rate = 0.007, 95% CI = [0.002, 0.032]) with similar between-study heterogeneity (p < 0.01, I2 = 99%). Meta-analysis of 17 samples found that clozapine exposure was associated with an absolute death rate from cardiomyopathy of 3 per 10,000 people (event rate = 0.0003, 95% CI = [0.0001, 0.0012]) with moderate between-sample heterogeneity (p < 0.01, I2 = 64%). Meta-analysis of 12 samples found that the case fatality rate from clozapine-associated cardiomyopathy was 7.8% (event rate = 0.078, 95% CI = [0.018, 0.285]), with moderate between-sample heterogeneity (p < 0.01, I2 = 65%).
Meta-analysis and sensitivity analysis of clozapine-associated cardiomyopathy.
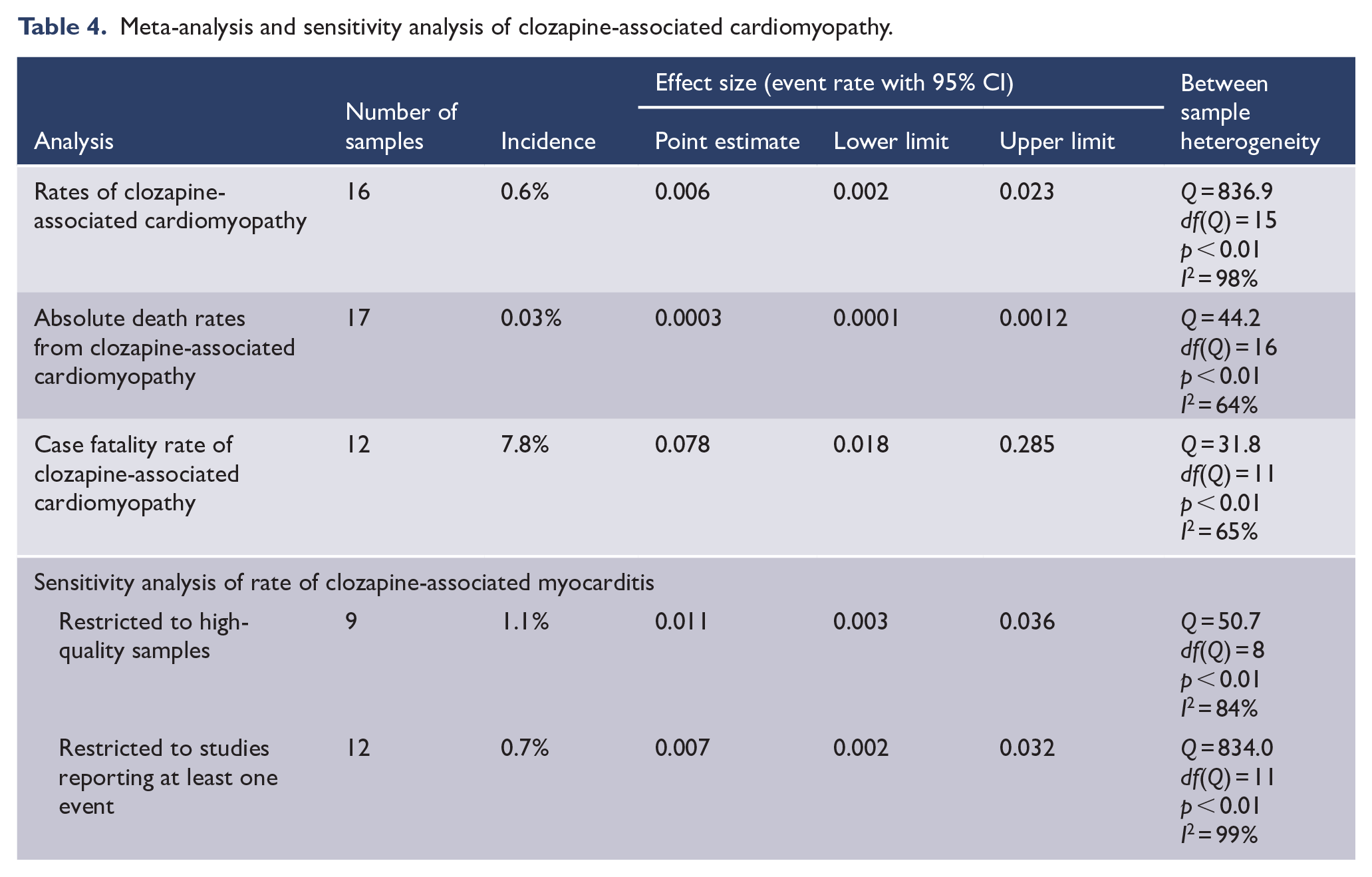
Subgroup analysis (see Supplementary Table 2) demonstrated that the use of diagnostic criteria of cardiomyopathy had no significant impact on effect size (Q = 0.2, p = 0.68), nor did retrospective vs prospective methodology (Q = 1.5, p = 0.22). Region in which data collection occurred explained a significant difference in between-study heterogeneity (Q = 4.7, p = 0.03) with Australian samples (eight samples, event rate = 0.014) reporting a significantly larger effect size than samples collected elsewhere (eight samples, event rate = 0.002). Data collection before or after year 2000 also explained a significant difference in between-study heterogeneity (Q = 4.7, p = 0.03) with data collection after the year 2000 reporting a higher effect size (14 samples, event rate = 0.011) than data collected before year 2000 (two samples, event rate = 0.0005).
There was no evidence of publication bias for analysis of absolute event rate (τ = 0.08, p = 0.65). Funnel plot is provided as Supplementary Figure 2. There was evidence of publication bias for the analysis of absolute death rate (τ = 0.60, p < 0.01), and trim and fill identified seven hypothetically missing studies to the right of mean reporting an adjusted event rate of 0.0004 (95% CI = [0.0001, 0.0020]). There was no evidence of publication bias for the analysis of case fatality rates (τ = 0.11, p = 0.63).
Discussion
This meta-analysis of more than 250,000 subjects indicates that both clozapine-associated myocarditis and cardiomyopathy are rare ADRs occurring in approximately 7 and 6 per 1000 people exposed to clozapine, respectively. The rates of death from these ADRs are even rarer with an absolute death rate from myocarditis of 4 in 10,000 people and from cardiomyopathy 3 in 10,000 people exposed to clozapine. It appears, however, that these events are associated with a moderate risk of mortality with a case fatality rate of 12.7% for myocarditis and 7.8% for cardiomyopathy. These estimates are robust given minimal evidence of publication bias across analyses and a similar effect size for cardiomyopathy on sensitivity analysis restricted to high-quality samples. One exception may be that the event rate of myocarditis may be underestimated given the higher effect size (16 in 1000 people) on sensitivity analysis restricted to high-quality samples. Regardless, the estimates reported here, which are the most comprehensive analysis of the literature to date, indicate event rates for both ADRs are lower than indicated in previous narrative reviews (Ronaldson et al., 2015).
There was a high degree of heterogeneity in our results. In regard to myocarditis, further exploration of heterogeneity using subgroup analysis indicated a significantly higher event rate in Australian data, studies that utilised prospective methodology and in studies with sample sizes less than 1000. The seminal paper describing cardiac ADRs used Australian data (Kilian et al., 1999), and subsequently, Australian authors have published the largest amount of literature describing clozapine-associated myocarditis. As a result, awareness of clozapine-associated myocarditis has increased in Australia to the point where cardiac screening is mandated in the routine care of clozapine patients in Australia. These factors indicate that observation bias may play a significant role in the reported rates of clozapine-associated myocarditis. Australian data sets may have been more likely to use diagnostic criteria than the rest of the world; however, report of diagnostic criteria had no significant impact on effect size in our analysis. Australian data sets had a similar likelihood of being prospective compared to the rest of the world. It is possible that the case fatality rate associated with myocarditis may not reflect current monitoring systems, as four of the five studies reporting any deaths from myocarditis collected all of their data prior to the year 2000.
Subgroup analysis of clozapine-associated cardiomyopathy similarly indicated that studies of Australian datasets and those utilising data collected after the year 2000 reported a significantly higher event rate. Overestimation of events is therefore likely due to a similar risk of observation bias. Some authors have attributed the increased rates of cardiac ADRs in Australia to unidentified genetic or environmental risk factors (Kilian et al., 1999). While plausible, these theories are nevertheless unlikely given there is no clear biological mechanism to aetiologically link clozapine and cardiac disease (Patel et al., 2019). It is most likely that an increase in rates of cardiomyopathy after the year 2000 reflects increased echocardiographic screening, particularly in Australia (Dawson et al., 2018) which is applied variably worldwide (Nielsen et al., 2016). In addition, clozapine use increased after being reintroduced in many jurisdictions in the early 1990s (Bachmann et al., 2017), and given cardiomyopathy can occur years after commencement of clozapine, higher rates after the year 2000 may reflect this delayed onset.
Perhaps, the largest limitation of the current literature reporting clozapine-associated cardiac ADRs is the inconsistent diagnostic criteria for both myocarditis and cardiomyopathy. Of the nine studies reporting diagnostic criteria for myocarditis, most relied on non-specific cardiac symptoms, ECG changes, echocardiographic changes or increased cardiac biomarkers, none of which are sufficiently sensitive for the diagnosis of myocarditis regardless of aetiology. Only three studies specified a temporal onset with clozapine and/or resolution with clozapine withdrawal, while only one specified histological confirmation which remains the gold standard for diagnosis. Knoph et al. (2018) reviewed the diagnostic criteria of myocarditis associated with clozapine and found a high degree of variation in suggested screening tests and diagnostic criteria (Knoph et al., 2018). They noted that myocarditis is not clinically well defined, with a heterogeneous pattern of symptoms and signs. The only definitive criteria involve cardiac biopsy (Aretz et al., 1987; Cooper, 2009). As cardiac biopsy is invasive, it makes creating rigorous alternative clinical diagnostic criteria challenging. Given clozapine is associated with fever and tachycardia, there is a possibility that these symptoms could be misattributed to myocarditis. In one published review of 20 clinical cases of people on clozapine diagnosed with clozapine-induced myocarditis, almost two-thirds were deemed to be highly unlikely to be clozapine-associated myocarditis on retrospective chart review, with concomitant viral illnesses or insufficient data to meet clinical criteria for myocarditis noted (Winckel et al., 2015).
The incidence of myocarditis in the general community is not well quantified (Cooper, 2009), while cardiomyopathy rates are estimated at 7.5–10 cases per 100,000 people per year (Friman et al., 1995). Given the mean follow-up for cardiomyopathy in our meta-analysis was 3.75 years, our reported rate of cardiomyopathy is still higher than the equivalent community rate.
Of the four studies reporting diagnostic criteria for cardiomyopathy all specified symptoms of congestive cardiac failure and echocardiographic features of either heart failure with reduced ejection fraction or heart failure with preserved ejection fraction, with most providing a left ventricular ejection fraction threshold of 50%. While echocardiography is the gold standard for diagnosis, it often remains difficult to aetiologically link clozapine and cardiomyopathy. Risk factors for ischaemic and hypertensive cardiomyopathy (including smoking, hypertension and type two diabetes) are extremely prevalent in people with schizophrenia (Firth et al., 2019), and it is probable that in some cases cardiomyopathy is misattributed to clozapine due to increased rates of screening (Bellissima et al., 2018).
While this meta-analysis is the largest and most comprehensive synthesis of literature reporting clozapine-associated cardiac ADRs, there remain several limitations to our analysis. First, there was a moderate to very high degree of heterogeneity across our meta-analyses. While use of a random effects model and subgroup analysis goes someway to addressing this limitation, the majority of heterogeneity remains unexplained in our analysis. This is likely to reflect the observational nature of the primary literature, issues with observation bias and wide variation in quality across the included studies. Second, few of the included studies used diagnostic criteria for either myocarditis or cardiomyopathy, and there was variation in the diagnostic criteria for myocarditis among studies that provided them. This issue may affect the statistical confidence of our summary effect size, but is also indicative of the broader difficulty in translating current research into screening and diagnostic pathways for patients at a clinical or service level. Third, there were a large number of studies that reported zero events that may have affected our statistical methods. There is no clear consensus on how to best approach the analysis of zero-value studies, and we used a continuity correction utilising a multiple of the raw event rate across all studies. This approach has the benefit of avoiding the omission of useable data (Sweeting et al., 2004), but may artefactually normalise between-study heterogeneity. Reassuringly, sensitivity analyses restricted to studies reporting at least one event returned very similar effect sizes and heterogeneity to the summary effect, indicating this approach was unlikely to have biased our results substantially. Fourth, a number of large studies reporting on cohorts in Australia collected data over a similar time period. As such, these studies may have reported data on the same individual patients and resulted in double counting. There is no way to determine if this is the case from the primary literature; however, it is reassuring that on sensitivity analysis of high-quality samples, which only included one of these potentially overlapping studies, effect size was not substantially different from the summary effect of myocarditis or cardiomyopathy which included all studies.
Taking into account these limitations, our results do have some important clinical and research implications. First, this analysis indicates clozapine-associated cardiac ADRs are very rare events, and while case fatality rates are high, the absolute risk of death is extremely low. While clinicians should be vigilant for these ADRs, the overall risk should be interpreted in the context of the demonstrated benefit of clozapine in psychiatric symptoms and all-cause mortality in treatment-resistant schizophrenia (Vermeulen et al., 2018). Second, there is a need for further research to define and validate a robust clinicopathological description of clozapine-associated myocarditis and cardiomyopathy. Current evidence is insufficient for specific recognition at a clinical level and current monitoring programmes run the risk of misclassifying suspected cases and unnecessarily withdrawing clozapine treatment. Harmonisation of diagnostic criteria would also have the benefit standardising the comparability of future research, would aid clinicians in providing accurate diagnoses and could avert unnecessary clozapine cessation. Third, because the clinical identification of cardiac ADRs may be insufficiently sensitive, it is important that suspected cases undergo work-up to exclude other causes. The preponderance of cardiac risk factors in schizophrenia makes it possible that ischaemic heart disease or hypertensive cardiomyopathy may mimic clozapine-associated cardiac ADRs, while the anticholinergic effects of clozapine itself may precipitate suspicious cardiac symptoms without underlying structural pathology. As such, it is logical that consultation with cardiology service for specialised investigations, such as cardiac magnetic resonance imaging (MRI) or endomyocardial biopsy, be considered when a diagnosis of myocarditis or cardiomyopathy is entertained to avoid incorrect diagnosis (Patel et al., 2019). Similarly, identifying a temporal onset is important, and thus in those people with cardiac risk factors, baseline assessment for cardiac disease and aggressive management of these risk factors should be undertaken prior to commencing clozapine. As a corollary, consultation with a cardiac service should be sought to inform diagnosis and safety of clozapine continuation or re-challenge in cases where the diagnosis is equivocal (Freudenreich, 2015; Patel et al., 2019). In cases with left ventricular dysfunction, cardiologist consultation regarding commencement of disease-modifying heart failure medications should occur (Patel et al., 2019).
The findings of this meta-analysis are that myocarditis and cardiomyopathy are rare events, and subsequent deaths from these conditions are very rare. Myocarditis, when it does occur, is usually within the first 2 months, while cardiomyopathy occurs months or years after commencing clozapine. Monitoring for myocarditis is warranted, and treatment of comorbid conditions associated with cardiomyopathy is part of good clinical care for people on clozapine. Given the effectiveness of clozapine for people with schizophrenia, and the absence of other evidence-based treatments, it is essential that people with treatment refractory schizophrenia be given a monitored trial of clozapine.
Supplemental Material
Supplementary_Figures – Supplemental material for Systematic review and meta-analysis of rates of clozapine-associated myocarditis and cardiomyopathy
Supplemental material, Supplementary_Figures for Systematic review and meta-analysis of rates of clozapine-associated myocarditis and cardiomyopathy by Dan Siskind, Ashneet Sidhu, John Cross, Yee-Tat Chua, Nicholas Myles, Dan Cohen and Steve Kisely in Australian & New Zealand Journal of Psychiatry
Supplemental Material
Supplementary_Tables – Supplemental material for Systematic review and meta-analysis of rates of clozapine-associated myocarditis and cardiomyopathy
Supplemental material, Supplementary_Tables for Systematic review and meta-analysis of rates of clozapine-associated myocarditis and cardiomyopathy by Dan Siskind, Ashneet Sidhu, John Cross, Yee-Tat Chua, Nicholas Myles, Dan Cohen and Steve Kisely in Australian & New Zealand Journal of Psychiatry
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: D.S. was funded, in part, by an NHMRC ECF APP 1111136.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
