Abstract

In the past two decades there has been a proliferation of medications collectively referred to as ‘atypical antipsychotics’ (AAP) which, as the name implies, are used to treat psychoses. Drugs in this group (e.g. clozapine, olanzapine, risperidone and quetiapine) are described as ‘atypical’ because they are less likely to cause extrapyramidal side effects compared to typical antipsychotics (e.g. chlorpromazine and haloperidol). But not all of these newer agents are equally efficacious, and in practice the majority fall short of the gold standard set by clozapine. For example, in the acute treatment of schizophrenia quetiapine has modest efficacy as an antipsychotic, and yet, this has not hindered its widespread uptake. This is partly because the side-effect profile of the more efficacious AAPs, such as clozapine and olanzapine, is particularly poor – especially when they are administered long term.
Slippery slope
Like many antipsychotics, quetiapine began life as a mimetic (Guzman, 2018). It was synthesised by scientists in 1985 with the aim of rivalling the efficacy of clozapine while producing far fewer side effects and, in particular, removing the risk of agranulocytosis. A decade later, in September 1997, the US Food and Drug Administration (FDA) approved the use of quetiapine for the treatment of schizophrenia and since then it has been prescribed worldwide. Pharmacologically, quetiapine binds to a range of receptors including adrenergic (α1) and histaminergic receptors, but of note, has a higher affinity for serotonergic receptors (5HT2A) compared to dopaminergic receptors (D1 and D2). Its low affinity for the latter, especially D2, and rapid reduction in receptor occupancy soon after binding, is thought to explain its low incidence of extrapyramidal side effects. Unfortunately, while quetiapine has slightly better tolerability than many of its contemporaries, it lacks the acute antipsychotic efficacy of other atypicals such as olanzapine (Chiappini and Schifano, 2018).
However, quetiapine’s modest efficacy did not prevent it from gaining an indication for schizophrenia (see Table 1), and indeed, some years later, it obtained a further indication for mania (Guzman, 2018). This is somewhat puzzling, although in practice some antipsychotics are quite effective in treating mania and are therefore used for this purpose (see Table 1). In other words, broadening the use of quetiapine from schizophrenia to mania is a step that is at least conceivable. However, this is not the case with its further expansion to the opposite pole encompassing first bipolar depression and then depression more generally.
FDA-approved indications and off-label uses for quetiapine.
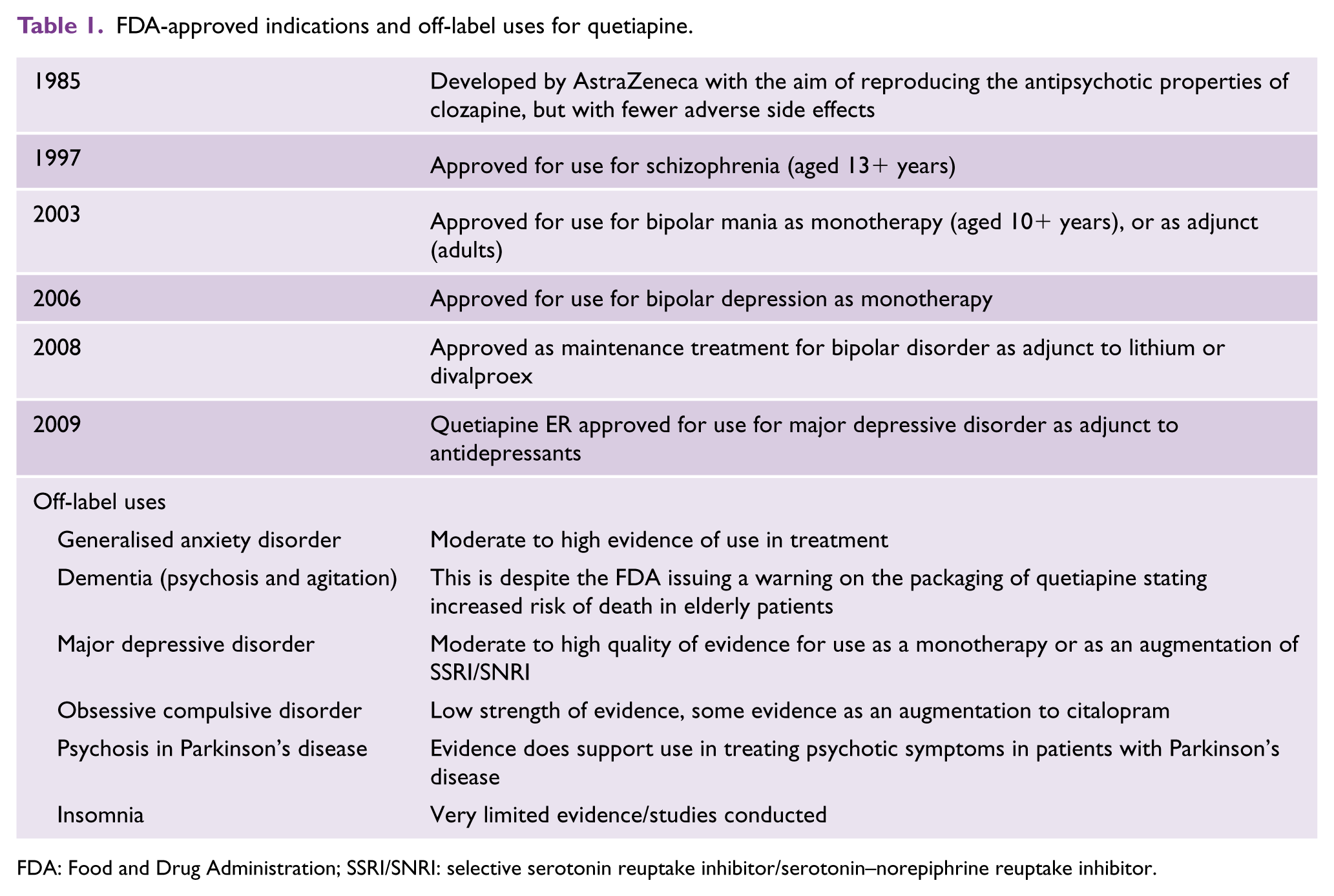
FDA: Food and Drug Administration; SSRI/SNRI: selective serotonin reuptake inhibitor/serotonin–norepiphrine reuptake inhibitor.
In 2006, quetiapine gained an indication for the treatment of bipolar depression. This is especially surprising because this particular indication allows for its use as monotherapy, suggesting that a single drug could treat schizophrenia, mania and depression. Soon after this, followed an indication; for the maintenance treatment of bipolar disorder – albeit adjunctively with lithium or valproate. But even these departures from its original indication are less astounding than its next feat – that of extending its reach to the treatment of major depressive disorder as an adjunct to antidepressants. This last quetiapine indication applied only to its extended release formulation. Nevertheless, in the space of 12 years, quetiapine achieved a phenomenal, all-encompassing range of successive indications that included schizophrenia, mania, bipolar depression, maintenance treatment of bipolar disorder and finally adjunctive treatment of major depressive disorder. But, remarkably, it did not end here because alongside its sanctioned expansion, the off-label use of quetiapine became equally ubiquitous. Once it had gained a foothold in the treatment of depression, its leakage into the mainstream treatment of major depressive disorder was perhaps to be expected. But its infiltration into the management of generalised anxiety and widespread uptake as an antidote to insomnia could not have been anticipated – at least not from its initial marketing as an ‘antipsychotic’. Although, the fact that quetiapine was developed from benzodiazepine-derived components was perhaps a clue.
Curing to quelling
The transdiagnostic migration of quetiapine – akin to the hermit crab’s quest for an ever bigger shell – evokes a perturbing question: how can one agent treat so many different psychiatric disorders?
To answer this question, we have to go back to the decades that preceded the prescription of quetiapine – namely the 1970s and 1980s. Research in this era was focused on psychological symptoms in the community, and was grappling with the problem of differentiating psychiatric ‘syndromes’ (such as depression) from more general expressions of distress. The latter, occasionally referred to as ‘general psychological distress’, was detected using screening instruments that tapped into feelings of nervousness, anxiety and restlessness – all of which appeared to be common. These symptoms were often accompanied by fatigue and sleep disturbance and mild feelings of sadness and low mood, and, collectively, they were referred to as general, psychological or even emotional distress.
The aetiology of this everyday distress (somewhat akin to neuroses) was unknown; however, it seemed reversible, often self-limiting and of a low intensity. Moreover, although psychiatric disorders such as anxiety and depression appeared to emerge from this milieu of distress, predicting which individuals would develop psychiatric disorders, and when, remained difficult. Research studies found that most screening questionnaires such as the GHQ (General Health Questionnaire) had very high sensitivity, much more so in fact than the clinical skills of primary care physicians (GPs) at the time, and that standardised measures used to rate symptoms of disorders – such as anxiety, depression, bipolar disorder and schizophrenia – all included items that captured psychological distress (Goldberg and Bridges, 1987). This was (and remains) a problem, because it means that in randomised controlled trials where an agent needs to demonstrate only modest benefit over placebo, it can achieve a seemingly significant difference – construed as a benefit – by simply ameliorating distress and anxiety without necessarily modifying the more specific symptoms of depression such as guilt and anhedonia, or the psychotic symptoms of schizophrenia such as hallucinations and delusions (Figure 1).
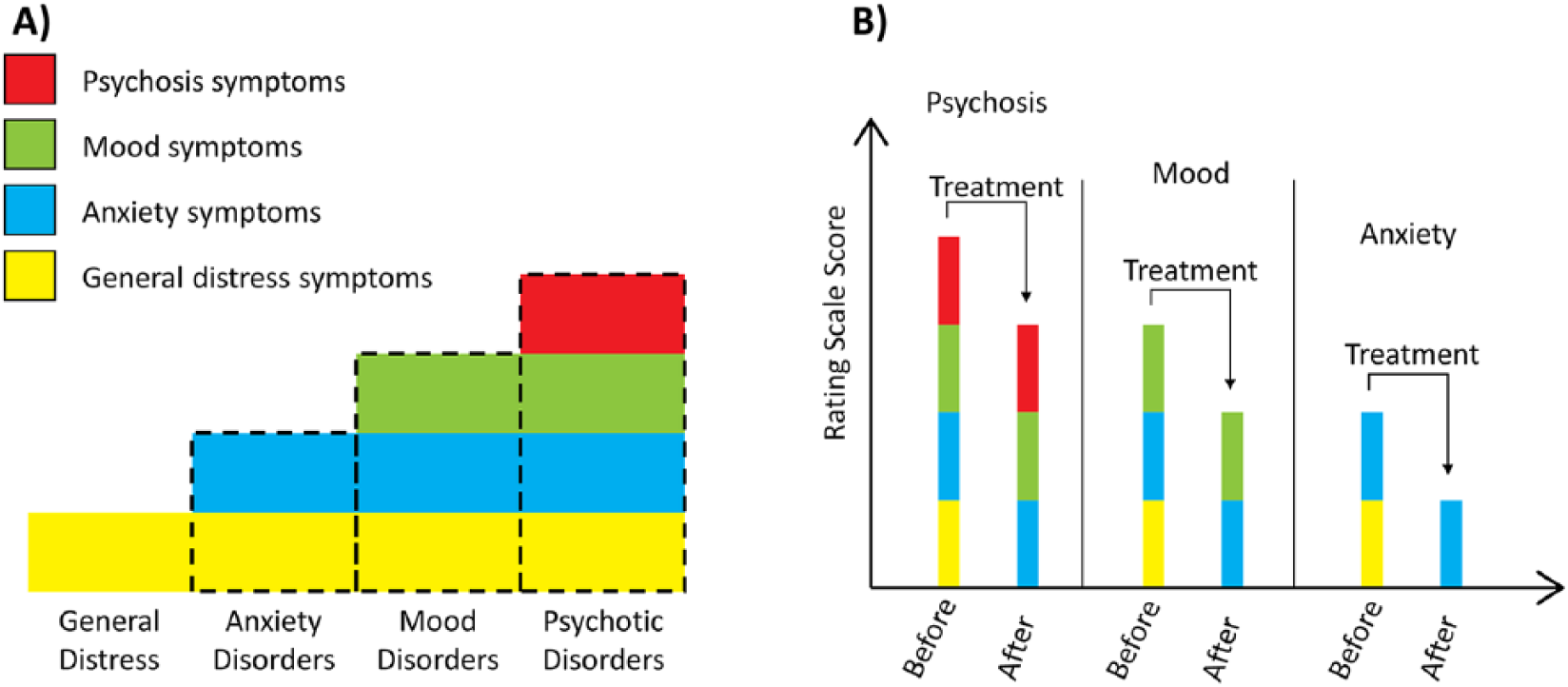
(A) A pyramid, the base of which is formed by symptoms of general distress. Above general distress sits anxiety symptoms, which in turn is situated below mood symptoms. Higher still, psychotic symptoms sit at the top of the pyramid, which at this point has narrowed considerably. General distress is thus pervasive and present across all disorders to a variable degree and underpins the syndromes of anxiety, mood and psychosis – which become successively more specific and subsume elements of syndromes beneath them. (B) Illustrates how the symptom ratings of various psychiatric disorders can appear to diminish when only measuring overall scores. The figure also shows how each disorder is a composite of various symptom components and how symptoms that are specific to a particular disorder, such as mood or psychosis, are not always significantly altered by the actions of a non-specific agent and hence remain relatively unchanged. In this manner an agent that can successfully diminish general distress and/or anxiety – can seemingly ‘treat’ multiple disorders.
This could perhaps explain why an agent such as quetiapine, which clearly has a strong sedative effect and is also an anxiolytic, performs well in trials designed to determine modest differences in the total scores of depression, mania or psychosis. By treating general distress and improving sleep, an ‘antipsychotic’ can appear to be an effective treatment for mood disorders or psychosis when in fact a significant proportion of any observed improvement is largely a consequence of changes in the ratings of non-specific symptoms. Indeed, original trials in bipolar depression acknowledge this by co-varying for one item (sleep) on a depression rating scale, but this technique cannot be expected to deal adequately with such non-specific effects (Calabrese et al., 2005).
Serial number
Another problem with the current framework for the management of chronic psychiatric disorders is the low expectation we have of pharmacotherapy. For example, STAR*D, a seminal study in the treatment of mood disorders, showed that at least two (30.6%), three (13.7%) and sometimes four (13.0%) medications are needed before an adequate response can be achieved (John Rush et al., 2006). This has reinforced clinical experience and set up an expectation that the first or second antidepressant treatment is unlikely to be effective and that this is an acceptable standard. But this expectation, apart from being poor practice, flies in the face of many definitions of treatment resistant depression, which require two treatment failures to designate an individual as having treatment-resistance. In practice, this has meant that trial and error is the dominant paradigm for antidepressant prescription, and that if this results in administering several treatments in succession this is regarded as acceptable, including combination, augmentation or even substitution with an atypical antipsychotic. In other words, serial treatment with any number of agents has become the norm.
An argument that is sometimes made in defence of an agent such as quetiapine and its broad range of indications is that the molecule has different actions at different doses. This may hold true to some extent. For example, the antidepressant venlafaxine has a largely serotonergic effect at low doses, but at higher doses also enhances noradrenergic neurotransmission. In addition, some agents, and again venlafaxine is a good example, have active metabolites (e.g. desvenlafaxine, which is now a medication in its own right). The properties of a metabolite can be very different from the parent molecule. One such active metabolite of quetiapine is N-desalkylquetiapine, a potential candidate for its putative antidepressant properties. Furthermore, though it is true that the doses of quetiapine used for sleep (25–50 mg per day) are markedly different from those administered for the treatment of schizophrenia or mania (200–800 mg per day), the variance in dosing may not be the only reason for differential clinical effects. An alternative explanation is that agents such as quetiapine essentially quell patients and do so across the board, irrespective of their syndrome or symptom profile. That is to say, they have a strong and immediate non-specific action, e.g. sedation, and this obfuscates more specific actions.
The fact that quetiapine has a very broad set of indications means that its use at some point in the treatment of a range of disorders is inevitable – especially because the selection of an appropriate therapeutic agent is a matter of trialling a series of drugs until some benefit emerges. This approach favours non-specific agents such as quetiapine, which can readily show some initial response but then struggle to maintain meaningful improvement. Hence, the paradigm of trialling non-specific medications that achieve little more than serial quelling needs to be re-examined.
Footnotes
Declaration of Conflicting Interests
G.S.M. has received grant or research support from the National Health and Medical Research Council, Australian Rotary Health, NSW Health, American Foundation for Suicide Prevention, Ramsay Research and Teaching Fund, Elsevier, AstraZeneca, Janssen-Cilag, Lundbeck, Otsuka and Servier, and has been a consultant for AstraZeneca, Janssen-Cilag, Lundbeck, Otsuka and Servier. E.B. declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article. R.J.P. has received grants from the NZ Health Research Council, Lotteries Health and Canterbury Medical Research Foundation and support for travel to meetings from Lundbeck and Servier.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
