Abstract

While meta-analyses are potentially helpful in determining the relative efficacy and tolerability of antidepressants for the treatment of major depressive disorder (MDD), their findings have been conflicting (Cipriani et al., 2018; Gartlehner et al., 2011), and apparent statistical advantages do not necessarily translate into clinical relevance. Although such equivalence of efficacy of antidepressants possibly holds true for Diagnostic and Statistical Manual of Mental Disorders (DSM-5) MDD, discernible differences across drug classes have been observed for melancholia – one example of a putatively more homogeneous subtype of depression. An important consideration is whether phenotypic heterogeneity of major depression prevents the emergence of true drug class difference in meta-analyses. Furthermore, in practice, given that systematic reviews and meta-analyses do not provide clear guidance as regards which antidepressants, for which patients, are more effective, how should clinicians select the optimal antidepressant?
First, clinicians commonly select an antidepressant based on recommendations for first-line antidepressant treatment based on guidelines (Malhi et al., 2015) despite a heterogenous clinical symptom presentation. Second, since clinical trial participants are not representative of ‘real world’ patients who invariably have comorbidities, the clinical question arises whether such efficacy study data can ever be translated to routine clinical practice. Third, how can the practical usefulness of antidepressants be evaluated in a clinically meaningful way when efficacy and tolerability data are considered separately when, in reality, clinicians generally adopt a ‘cost:benefit’ approach and consider both criteria jointly upon selection of an antidepressant. Importantly, tolerability is often the determining factor in the acceptability of, and adherence to, medication. Connecting these reasons has made it difficult to differentiate the effectiveness of antidepressants. However, another key problem has been that of evaluation. It is hardly surprising that antidepressants appear to have similar efficacy given that the measures of clinical outcome (e.g. Montgomery-Asberg depression rating scale (MADRS)) are designed to capture a broadly defined symptom-based phenotype namely, ‘MDD’.
One approach that could help achieve a more personalised and tailored approach in the treatment of MDD is to introduce additional symptom dimensions alongside mood that could be informed by efficacy and tolerability. Such an approach may reduce the need for the current process of ‘trial and error’.
In clinical practice, patients present with a variety of symptoms. These can be grouped into a number of dimensions such as cognition (e.g. deficit with executive function, concentration, memory, processing speed), mood (sad, low, irritable, anhedonia, suicidal ideation), anxiety (feeling anxious), activity (psychomotor-retardation, agitation), sleep (insomnia, hypersomnia) and other physical symptoms, demonstrating that patients may present with symptom dimensions in addition to mood disturbance during an acute episode of depression. For the clinical assessment of patients with likely complex symptom presentations, it would be necessary to assess such symptom dimensions to determine clinical treatment targets. In addition, the most distressing or disabling symptom dimensions from a patient perspective could be considered during treatment goal setting.
The dilemma is a poverty of adequate comparative studies of antidepressant efficacy that first define a broader symptom dimension as primary outcome (rather than symptom scales) and second conduct comparative analyses of efficacy of antidepressants A versus B versus C for their efficacy on such symptom dimensions. Only recently, a network meta-analysis approach has emerged that addresses some of these issues by comparing the efficacy of various antidepressants on the symptom dimension of cognition (Baune et al., 2018). As such, a network analysis approach may be a useful method of identifying the relevance of symptom dimensions to drug efficacy, using this study as an example (Baune et al., 2018). To further address such a comparative approach from a clinical perspective, a clinical consensus approach was chosen to examine the differential efficacy of antidepressants for symptom dimensions in MDD. Hence, a consensus was reached by a committee called the Treatment Algorithm Group (TAG) comprising Australia and New Zealand clinical and academic experts. TAG met in person in Sydney in November 2017 and reached consensus that differential efficacy and tolerability do exist among mainly first-line antidepressants and proceeded to capture clinical experience reflecting this differential efficacy – depicted in Table 1. While this approach is not directly comparative in nature, it does highlight that differences between first-line antidepressants across symptom dimensions do exist. The antidepressant efficacy across symptom dimensions as shown in Table 1 represents a consensus of subjective impressions regarding the relative properties of various antidepressants and is not based on clinical evidence.
Antidepressant efficacy across major depressive disorder symptom dimensions.

SSRIs: selective serotonin reuptake inhibitors; NaSSAs: noradrenergic and specific serotonergic antidepressants; NDRIs: norepinephrine-dopamine reuptake inhibitors; SNRIs: serotonin and norepinephrine reuptake inhibitors; NARIs: noreadrenaline reuptake inhibitors; Serotonin modulator: vortioxetine; MAOIs: monoamine oxidase inhibitor; TCAs: tricyclic antidepressants.
+/–: neutral activity; +: minor effect; ++ and +++: moderate effect; ++++ and +++++: strong effect.
It is important to note that differences in antidepressant effects may emerge from both direct effects of the drug on a specific symptom dimension and from indirect effects secondarily due to mood changes. Moreover, some differences in efficacy may result from differing side-effects that may be regarded either as beneficial, or in the absence of side-effects, as enablers of efficacy. The often reported benefits of well-tolerated drugs (e.g. agomelatine, escitalopram and sertraline) are contrasted with less well-tolerated drugs (e.g. Duloxetine) (e.g. Khoo et al., 2015); however, since side-effect assessment is commonly based on individual symptoms only, tolerability has not been investigated for additional effects on the above symptom dimensions. Evaluating efficacy and side-effects for their additional effects on symptom dimensions of MDD as well as the consideration of possible withdrawal symptoms based on pharmacokinetic properties of antidepressants would add value.
Taken together, a combination of efficacy and tolerability measures according to patient symptom dimensions in MDD probably best informs decision making when selecting an antidepressant (Figure 1). This may be superior in terms of clinical relevance as compared to the approach of meta-analyses-based recommendations that currently employ global clinical outcome measures. A combined efficacy plus tolerability concept that is based on symptom dimensions would provide clinicians with a broader choice of effective and well-tolerated antidepressant treatments. The ranking of antidepressants’ efficacy and tolerability, as shown in the recent Cipriani et al.’s (2018) network meta-analysis, would most likely change, if differential efficacy and tolerability based on symptom dimensions were considered. Head-to-head clinical trials and network meta-analyses are required to estimate the differential effects of antidepressants on symptom dimensions of depression.
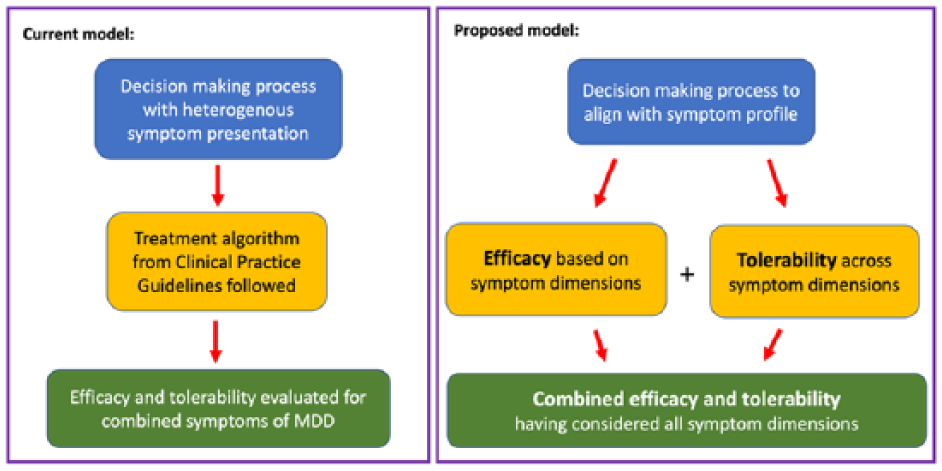
Model of symptom dimension-based efficacy and tolerability of antidepressants.
Footnotes
Acknowledgements
The Treatment Algorithm Group (TAG) was supported logistically by Servier who provided financial assistance with travel and accommodation for those TAG members travelling interstate or overseas to attend the meeting in Sydney (held on 18 November 2017). None of the committee were paid to participate in this project and Servier have not had any input into the content, format or outputs from this project.
Declaration of Conflicting Interests
B.T.B. and P.B. have received consultation fees, sponsorship and speaker fees from Servier; is a member of the advisory board for Lundbeck, Eli Lilly, AstraZeneca and Janssen; has received speaker fees from Lundbeck, AstraZeneca and Janssen and has received funding for a clinical trial from Brain Resource Company and Ferring Pharmaceuticals. M.H. has received grants and personal fees from Servier; personal fees from Janssen-Cilag, Lundbeck, Eli Lilly, Hahn, Sequiris, Bionomics and Mundipharma. G.P. has spoken at meetings or been on advisory boards for the following companies: Servier, Lundbeck and Otsuka. R.J.P. uses software for research at no cost from Scientific Brain Training Pro. A.S. has been a speaker for Servier, Lundbeck and Otsuka; has equity in CNSDose LLC and ABC Life Pty Ltd. G.S.M. has received grant or research support from AstraZeneca, Eli Lilly, Organon, Pfizer, Servier and Wyeth; has been a speaker for AstraZeneca, Eli Lilly, Janssen-Cilag, Lundbeck, Pfizer, Ranbaxy, Servier and Wyeth and has been a consultant for AstraZeneca, Eli Lilly, Janssen-Cilag, Lundbeck and Servier.
Funding
The study was funded by the National Health and Medical Research Council (Grant/Award Number: 1037196).
