Abstract

To the Editor
Fragile X syndrome (FXS), a common form of inherited Intellectual Disability, is caused by a mutation or expansion of the fragile X mental retardation 1 (FMR1) gene, which produces Fragile X mental retardation protein (FMRP) (Mila et al., 2017). FMRP plays an important role in synaptic development and plasticity (Mila et al., 2017). Pre-mutation carriers (55–200 trinucleotides CGG repeats) may develop associated disorders such as Fragile X-associated tremor ataxia syndrome (FXTAS), a neurodegenerative disease characterized by neurological symptoms including kinetic tremor and cerebellar ataxia (Mila et al., 2017). The prevalence of FXS in the general population is approximately 1 in 4000–7000 compared to pre-mutation carriers with a prevalence of approximately 1 in 130–250 females and 1 in 250–810 males (Lozano et al., 2014). Of those pre-mutation carriers approximately 40% of men and 16% of women are affected by FXTAS (Lozano et al., 2014). Cognitive assessments of FXTAS demonstrate impairment in attention, intelligence, working memory, executive function, declarative learning and memory, information processing speed and temporal sequencing, with language and verbal comprehension spared (Grigsby et al., 2008). Progression to dementia occurs in an estimated 50% of patients (Grigsby et al., 2008).
We report a 60-year-old married computer analyst, diagnosed with FXTAS at the age of 58 with no familiar history of FXTAS. Symptoms included kinetic tremor, foot drop, vestibular dysfunction, ataxia, peripheral neuropathy, swallowing deficit, intractable hiccups and generalized anxiety with panic attacks. He was referred to a memory clinic with concerns about evolving dementia. Cognitive concerns included inattention, poor concentration, anomia, mistakes at work and poor frustration tolerance. Genetic analysis revealed a trinucleotide CGG repeat of 85. Magnetic resonance imaging revealed bilateral symmetrical hyper intensities in the middle cerebellar peduncles with no associated restricted diffusion, associated mild middle cerebellar peduncle atrophy and mild cerebellar atrophy (Figure 1), consistent with FXTAS (Grigsby et al., 2008). Cognitive assessment identified mildly reduced attention, inefficiencies in sustained concentration and attentional capacity. There was variable but inferior performance in information processing speed and mild anomia. Learning of new verbal information and visual learning was mildly inefficient. Working memory and language abilities were intact with no evidence of primary impairment of memory or visuospatial dysfunction. Our patient’s cognitive profile was generally consistent with that reported in the literature with inconsistencies possibly explained by the low trinucleotide repeat.
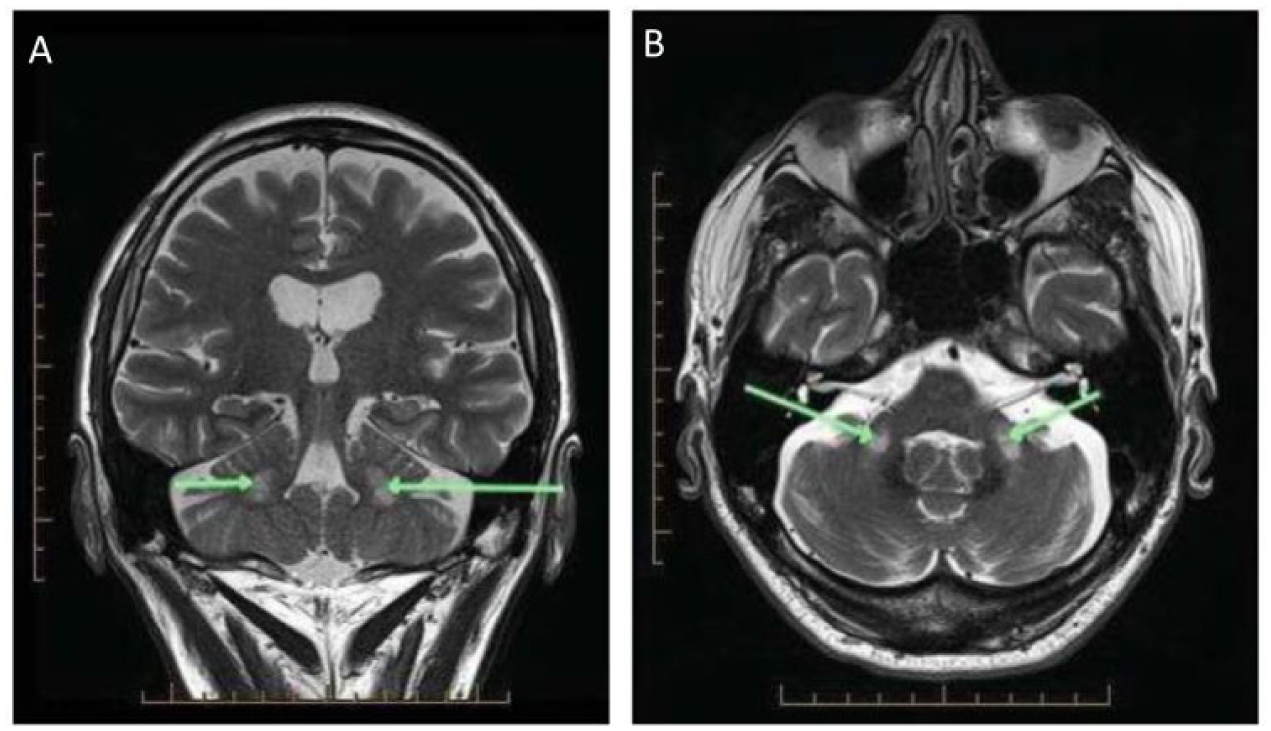
T2-weighted MRI of our 60-year-old patient showing bilateral high signal in the middle cerebellar peduncles (green arrows; (A) coronal, (B) axial).
Our case highlights the need to consider FXTAS in patients presenting to memory clinics with concurrent neurological symptoms. This case report adds to the limited knowledge of the nature of cognitive deficits in FXTAS with implications for further research to enhance our understanding, recognition and treatment of this syndrome.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
