Abstract
Objective:
To provide practical clinical guidance for the treatment of adults with panic disorder, social anxiety disorder and generalised anxiety disorder in Australia and New Zealand.
Method:
Relevant systematic reviews and meta-analyses of clinical trials were identified by searching PsycINFO, Medline, Embase and Cochrane databases. Additional relevant studies were identified from reference lists of identified articles, grey literature and literature known to the working group. Evidence-based and consensus-based recommendations were formulated by synthesising the evidence from efficacy studies, considering effectiveness in routine practice, accessibility and availability of treatment options in Australia and New Zealand, fidelity, acceptability to patients, safety and costs. The draft guidelines were reviewed by expert and clinical advisors, key stakeholders, professional bodies, and specialist groups with interest and expertise in anxiety disorders.
Results:
The guidelines recommend a pragmatic approach beginning with psychoeducation and advice on lifestyle factors, followed by initial treatment selected in collaboration with the patient from evidence-based options, taking into account symptom severity, patient preference, accessibility and cost. Recommended initial treatment options for all three anxiety disorders are cognitive–behavioural therapy (face-to-face or delivered by computer, tablet or smartphone application), pharmacotherapy (a selective serotonin reuptake inhibitor or serotonin and noradrenaline reuptake inhibitor together with advice about graded exposure to anxiety triggers), or the combination of cognitive–behavioural therapy and pharmacotherapy.
Conclusion:
The Royal Australian and New Zealand College of Psychiatrists clinical practice guidelines for the treatment of panic disorder, social anxiety disorder and generalised anxiety disorder provide up-to-date guidance and advice on the management of these disorders for use by health professionals in Australia and New Zealand.
Keywords
Executive summary
Anxiety is normal
Anxiety can be good for us. Moderate levels of anxiety make us alert and improve performance, and even high levels of anxiety will be appropriate when they are consistent with the demands of the situation. For example, moderate levels of anxiety before a sporting event, an exam or a job interview will increase alertness and performance, while high arousal in situations of real danger will enable people to focus on the threat and act quickly to escape or ward off the danger.
The problem is that high anxiety can reduce a person’s capacity to think, plan and do complex tasks that also need attention in difficult situations. It is normal for a person’s current level of anxiety to affect their ability to perform. However, people with anxiety disorders experience very pronounced states of anxiety, often against a background of constant fear and worry. These severe states of anxiety can be disabling.
Anxiety disorders involve unhelpful thinking patterns
Having an anxiety disorder is not just a matter of being too anxious. People with anxiety disorders have fears and worries about ‘what might happen if …’, and those fears and worries persist on and off for months and years, causing distress and disability. It is the months or years of distress and disability that drive people to treatment.
The continuing fears and worries, which most patients recognise as somewhat irrational but nevertheless dread, are the basis for making a diagnosis of an anxiety disorder (Figure 1) and prescribing treatment.
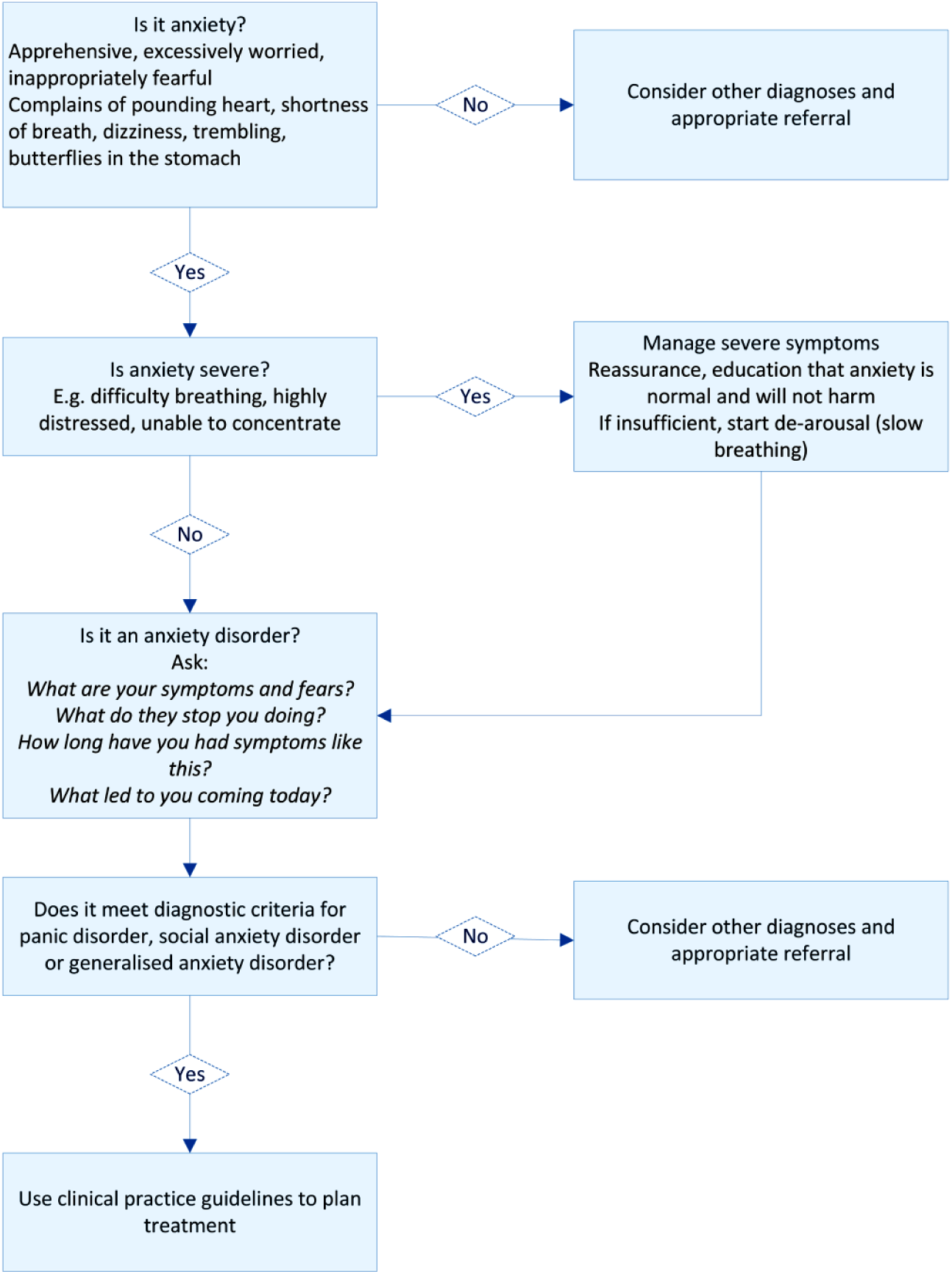
Initial assessment for a patient presenting with anxiety symptoms.
Each of the anxiety disorders covered in these guidelines is characterised by specific thoughts and behaviours:
Panic disorder – sudden attacks of fear or anxiety (usually brief, but which may be so severe that the person thinks they might collapse or die), concern about the attacks recurring and avoidance of situations in which they might recur.
Social anxiety disorder (SAD) – fear and avoidance of situations where the person thinks they might be the centre of attention, concern about doing or saying something embarrassing, and that others might notice the anxiety and be critical.
Generalised anxiety disorder (GAD) – months of excessive worry over everyday things, avoiding or seeking reassurance about situations where the outcome is uncertain, and being overly concerned about things that could go wrong.
Anxiety disorders can be treated
Effective treatments are available (Figure 2) but they take time to work. In practice, most people have had their disorder for years before seeking help and do not expect to get better instantly. Their most acute need is appropriate reassurance that their disorder has been recognised and that help will be forthcoming.
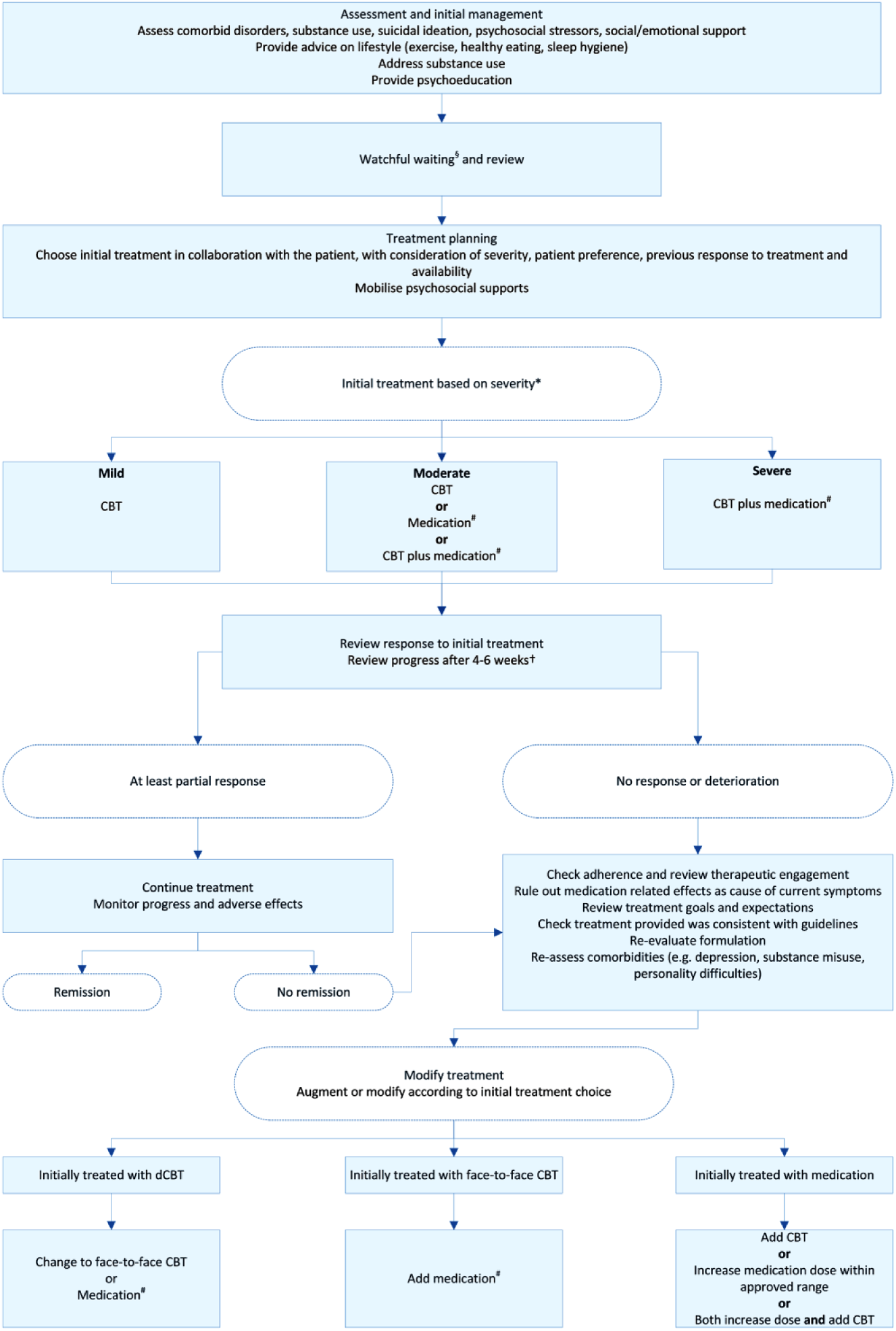
Overview of the management of anxiety disorders.
Initial treatment should be selected in collaboration with the patient, based on the severity of the disorder, previous response to treatment, availability and the person’s preference.
It will usually take 4–6 weeks to see improvement, whether cognitive–behavioural therapy (CBT) or an antidepressant is used, and most people can tolerate this. Only rarely is there any need to prescribe medication for the acute relief of symptoms.
What can a clinician do to help someone who is acutely anxious?
Just sitting in the waiting room often helps to reduce anxiety because people know that a doctor is near.
If the person is too anxious to be able to concentrate, get another staff member to use a second hand on a watch to supervise them as they do the slow breathing technique (Centre for Clinical Interventions, 2016): the person breathes in for 4 seconds, holds their breath for 2 seconds and then breathes out slowly over 6 seconds, for example: Shall we try? Breathe in for 4 seconds: one, two, three, four. Hold your breath for 2 seconds: one, two. Breathe out slowly for 6 seconds: six, five, four, three, two, one. Repeat for a minute then, if necessary, repeat the whole procedure.
When you see them and they are calmer, sit them down and say:
Tell me about being anxious.
What has made you so anxious?
What do you fear will happen?
What does it stop you doing?
Remember:
Anxiety often fluctuates and you may be seeing the person at their worst, which is reassuring for them but anxiety provoking for you.
Resist the urge to write a script immediately – many people are instantly reassured just by knowing, from you, that they have a recognisable and treatable condition.
How severe and disabling is the anxiety?
While anxiety always produces distress, anxiety disorders can be disabling. Data from the Australian National Survey of Mental Health and Wellbeing (Slade et al., 2009b) demonstrate significant disruption to everyday life among people who met criteria for current panic disorder, SAD or GAD as their principal complaint; the number of days unable to work or do normal tasks due to anxiety during the previous month was reported as less than 1 day by approximately half, 1–7 days by one-third, and more than 7 days by one-sixth. These three groups can be considered to have a mild, moderate or severe anxiety disorder, respectively.
Introduction
These clinical practice guidelines for the treatment of panic disorder, SAD and GAD were developed by the Royal Australian and New Zealand College of Psychiatrists (RANZCP). They update and replace the previous RANZCP guidelines for panic disorder and agoraphobia (Royal Australian and New Zealand College of Psychiatrists Clinical Practice Guidelines Team for Panic Disorder and Agoraphobia, 2003).
Purpose and scope
These clinical practice guidelines amalgamate evidence-based knowledge with clinical knowledge to advise health professionals on the treatment of adults with panic disorder, SAD or GAD. The main target population is adults aged 18–65 years, but evidence for other age groups was included where available.
The guidelines cover the management of mild, moderate and severe disorder including treatment-refractory disorder, for which there is limited evidence to guide practice.
Although anxiety disorders are the most prevalent mental health conditions in the community, many people with anxiety symptoms do not seek treatment. We hope these guidelines will help address this issue by clearly outlining the conditions and evidence for their treatment.
These guidelines are intended for use by psychiatrists, physicians, general practitioners (GPs) and psychologists in primary care, community mental health centres or specialist practices in Australia and New Zealand. Most patients with these anxiety disorders first present for treatment to primary care, where effective treatment can be provided or arranged. Those with severe or chronic anxiety disorders usually need specialist psychiatric and psychological treatment.
These guidelines do not provide recommendations on the care of people who experience anxiety in the setting of:
Other internalising disorders such as obsessive–compulsive disorder (OCD), trauma-related disorders such as post-traumatic stress disorder (PTSD), or mood disorders, although many elements of these guidelines will be relevant to the treatment of anxiety in these other contexts (Andrews et al., 2009; Goldberg et al., 2009).
Separation anxiety.
Other disorders (e.g. psychosis, cognitive impairment, substance use disorders or personality disorders), for which the approach to treatment will differ.
Working group
The RANZCP Clinical Practice Guidelines Team for Panic Disorder, Social Anxiety Disorder and Generalised Anxiety Disorder (working group) was appointed in 2014, composed of health care academics and clinicians from Australia and New Zealand (Appendix 2). The working group represented a diverse range of expertise, opinion and adherence to particular therapeutic approaches.
Development process
Clinical recommendations were based on systematic collection and appraisal of evidence (see section ‘Methods’).
The working group met face-to-face in December 2014 and in December 2017 and corresponded extensively by email. For each diagnosis, a subgroup of working group members synthesised and summarised the evidence and then modified this summary for consistency with a framework developed by the full working group. Recommendations were developed by the whole group through considerable frank and robust discussion to reach agreement.
The working group implemented several approaches to minimise the potential for bias towards any particular therapeutic approach. The composition of the working group ensured that a range of opinions were represented. All working group members participated equally in discussions. Peer-reviewed clinical papers and the highest quality evidence available were prioritised during decision-making, and discussion continued until consensus was reached.
The draft manuscript was circulated repeatedly, feedback was sought and areas of controversy were identified and resolved in an iterative process. At the final meeting, controversial areas were reconsidered with careful evaluation of the evidence, and revisions were drafted and finalised by teleconference and email.
Consultations and external review
A draft version of these guidelines was reviewed by national and international expert advisers (see Acknowledgments). The working group revised the manuscript in response to their suggestions.
A revised version of the guidelines was released for public consultation during 30 June–30 July 2017. To encourage wide participation, the RANZCP invited review by its committees and members, and by key stakeholders, including professional bodies and special interest groups. Respondents were asked to review the guidelines and answer four key questions related to each section of the guidelines via Survey Monkey:
Are there any significant gaps (of topic, literature, other)?
Are there errors in the content?
Is the structure logical and easy to use?
Do you have any other comments?
During the consultation period, a draft was publicly available on the RANZCP website. A total of 31 submissions were received during the consultation period from both stakeholder organisations and individuals with either a professional background or a lived experience of anxiety disorders (see ‘Acknowledgements’ for a list of organisations that provided a submission). The working group met by teleconference to consider all responses. For each response, the working group agreed on whether to revise the manuscript and recorded their decision. Several amendments were made during this revision process.
The amended draft was reviewed by the following RANZCP committees:
Committee for Evidence-Based Practice;
Practice, Policy and Partnerships Committee;
Corporate Governance and Risk Committee.
A draft was approved for publication by the RANZCP Board in August 2018.
Methods
Evidence collection and synthesis
Search strategy
Phase 1: The systematic literature review focused on meta-analyses and systematic reviews from 2000 to 2014, to include any relevant trials since the previous RANZCP anxiety guidelines (Royal Australian and New Zealand College of Psychiatrists Clinical Practice Guidelines Team for Panic Disorder and Agoraphobia, 2003). We searched PsycINFO, Medline, Embase and Cochrane databases using the search terms ‘panic disorder’ OR ‘agoraphobia’ OR ‘social anxiety disorder’ OR ‘social phobia’ OR ‘generalized anxiety disorder’ AND ‘treatment guidelines’ OR ‘systematic review’ OR ‘meta-analysis’.
Reference lists of identified articles and grey literature were also searched for relevant studies.
Phase 2: After initial consultation and revision of the draft, the database search was repeated in May 2017, restricted to systematic reviews and meta-analyses.
Phase 3: After expert review of the draft, the database search was repeated in December 2017, again restricted to systematic reviews and meta-analyses.
Study selection
A total of 736 papers were obtained from phase 1 database searches. An additional 25 papers were identified by members of the working group. After removing duplicates, the title and abstract of 531 citations were examined against pre-specified inclusion and exclusion criteria by two independent raters. Disagreements were resolved by a third independent rater and discussion. This resulted in exclusion of 388 articles, leaving a final 143 quantitative studies to be included in the qualitative synthesis. The following inclusion criteria were applied:
Papers on panic disorder (with and without agoraphobia), SAD or GAD;
Papers with level I evidence for intervention studies (i.e. meta-analyses or systematic reviews of randomised controlled trials [RCTs]);
Papers published in English.
Studies were excluded if the focus was not on the relevant disorders or if insufficient details were provided to allow synthesis.
For each included study, the level of evidence was assessed according to the Australian National Health and Medical Research Council (NHMRC) classification for intervention studies (Table 1; National Health and Medical Research Council, 2009).
Levels of evidence for intervention studies.
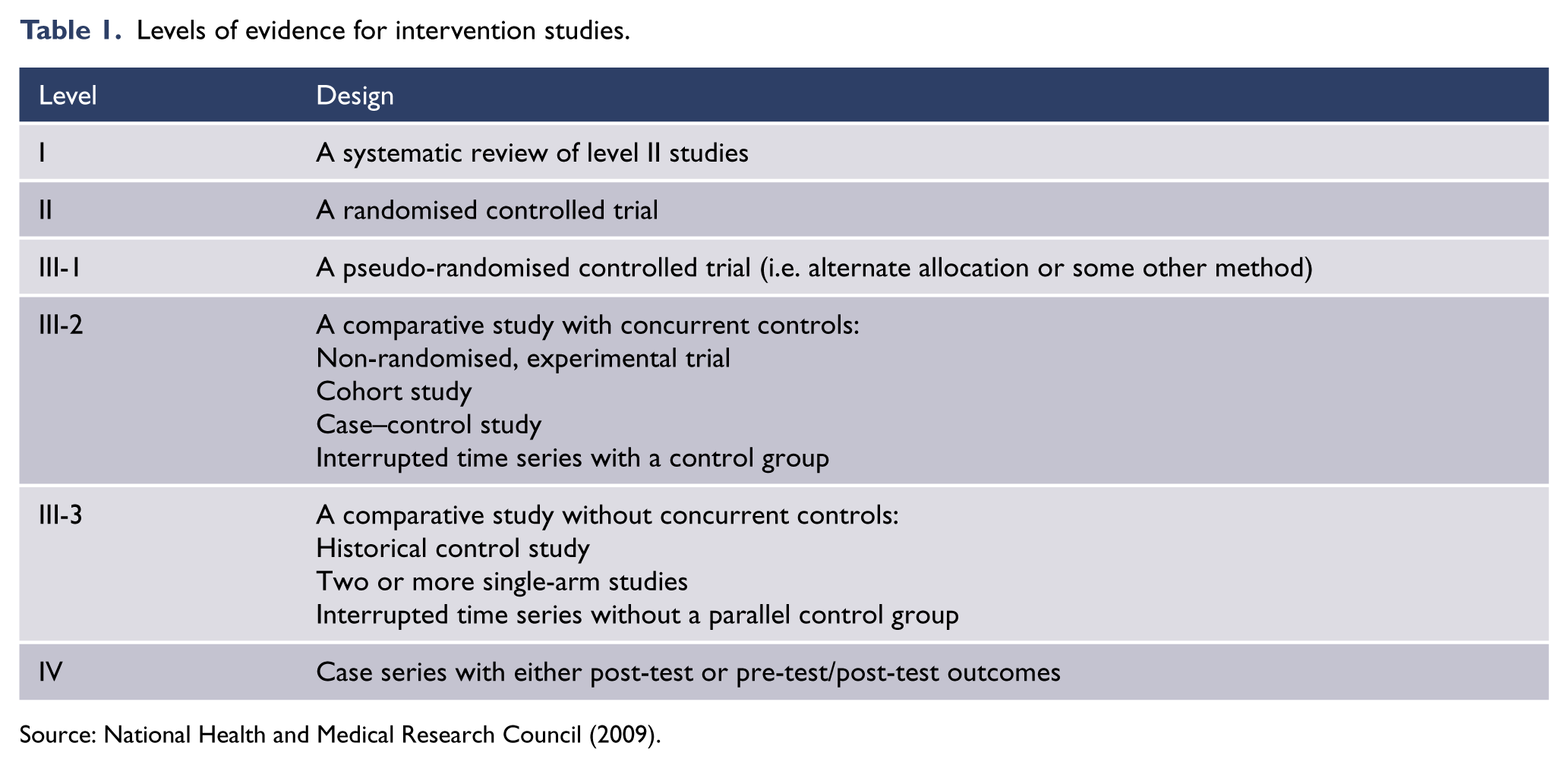
Working group members also identified relevant replicated RCTs (level II evidence) that were not included in systematic reviews.
Data analysis
We used two statistics to summarise the overall treatment effect: number needed to treat (NNT) and effect size.
NNT is an estimate of the number of people who would need to be treated for one of them to achieve designated treatment success. For example, a pooled NNT of 3 means that a clinician needs to treat three people to achieve the outcome of one patient no longer meeting criteria for diagnosis. Thus, a NNT of 2 is better than a NNT of 6.
The effect size illustrates the extent of improvement in the average patient. It is the difference between outcome measures of the treatment group (t) and control group (c) divided by the pooled standard deviation (SD) units [(dt – dc)/(SDt + SDc/2)], thereby allowing comparisons that are independent of the properties of different outcome measures. An effect size of 1.0 indicates that the average treated person would be better than 86% of untreated patients. By convention, a Cohen’s d of 0.2 is considered to represent a small effect size and not likely to be important, while 0.8 is considered to represent a large effect size and is always important (Cohen, 1988).
Limitations of the evidence
General limitations in the body of evidence for psychiatric interventions include potential bias arising from the funding of clinical trials, from the fact that clinical trials evaluating a particular psychological therapy are often carried out by the same clinicians who devised the intervention or its mode of delivery (such that similar results might not be expected when the intervention is delivered by other clinicians), from publication bias, and from clinical trial design that results in samples unrepresentative of clinical populations (Malhi et al., 2015).
The use of different outcome measures makes direct comparisons between studies difficult, although authors have addressed this in various ways, including by calculating standardised mean differences (SMDs). Some measures, such as the Clinical Global Impression scale (CGI), appear to give rise to generally higher effect sizes than other measures, raising the possibility of inflation in those studies that rely on, or report only this measure.
A significant limitation of the pharmacological research is the degree of improvement that occurs with pill placebo that tends to underestimate the effect of medication.
In trials evaluating psychological therapies, bias or distortion may also arise from the design of control conditions. Studies of CBT generally use wait-list controls, which of themselves have a pre–post effect size of about 0.2, hence tending to overestimate the effect of the CBT comparator.
Changes in clinical trial recruitment strategies and secular trends in the general population over recent decades may result in samples that do not accurately reflect the clinical population. In addition, no trials have been conducted in Indigenous populations or other locally relevant culturally and linguistically diverse populations.
There is very limited evidence for the comparative efficacy of treatment options to manage treatment-refractory anxiety.
Developing the recommendations
Clinical practice recommendations were formulated after appraising the evidence.
Evidence-based recommendations (EBRs) were formulated when there was sufficient evidence on a topic. They were based mainly on evidence from systematic reviews, but at the minimum, replicated treatments from two RCTs and from two research groups were also considered as sufficient evidence.
Where there was insufficient evidence to make an EBR (few studies or poor-quality studies), the committee considered whether to make a consensus-based recommendation (CBR). CBRs were based on the working group’s collective clinical and research knowledge and experience.
In formulating the recommendations, the working group considered a range of factors in addition to evidence from efficacy studies. These included evidence of effectiveness from results in routine practice, accessibility and availability of treatment options in Australia and New Zealand, the degree to which interventions implemented in practice are likely to adhere to the treatment models evaluated in clinical trials (fidelity), acceptability of treatment options to patients, safety considerations, and the costs of treatments to patients and to health systems.
Off-label prescribing
In this guideline, some therapies identified as effective for the treatment of anxiety disorders on the basis of available evidence may not be approved for such use in Australia and/or New Zealand. The use of such therapeutic agents outside their approved product information indication(s) is sometimes referred to as ‘off-label’ use and may result in out-of-pocket expenses for patients. Please refer to the RANZCP Professional Practice Guideline 4: ‘Off-label’ prescribing in psychiatry for more information.
General issues in the recognition and management of anxiety disorders
Framework
Dimensional and cluster models of psychiatric conditions
In preparing these guidelines, we considered two important concepts. First, that all disorders exist on a dimension from subthreshold to severe cases. While guidelines apply to above-threshold cases, they are applicable to people with subthreshold cases who are at risk of developing a threshold disorder (Helzer et al., 2009). Second, that there are clusters of mental disorders that share causes and remedies, so that guidelines for one disorder will resemble guidelines for related disorders (Andrews et al., 2009).
Both these issues are pertinent to the anxiety disorders. Subthreshold cases do warrant treatment and the internalising disorders (major depression, panic and phobias, GAD, OCD and PTSD) do respond to similar therapies (Goldberg et al., 2009) and to transdiagnostic therapies (Newby et al., 2015).
Desired health outcomes
The desired health outcome is simple; that people recover, stay well and get on with a productive life. Attaining such a goal requires increased access to care, increased prescription of effective treatments and decreased use of ineffective treatments. This is particularly important, as anxiety is under-recognised in general practice (Jameson and Blank, 2010) and perhaps only 30–60% of people who see their GP for an anxiety disorder receive a treatment regarded as adequate (Slade et al., 2009b; Stein et al., 2004b).
The systematic implementation of anxiety disorder guidelines has been shown to result in earlier treatment gains and shorter treatment times (van Dijk et al., 2015).
Treatment factors
These guidelines cover the use of psychological treatments and pharmacotherapy. Of the psychological treatments, we focus on CBT because the evidence base for benefit is much more extensive than with other structured psychotherapies, because the pool of experienced practitioners is larger and because CBT is available in Australia and New Zealand in an automated form over the internet. Of the medication options, we focus on the selective serotonin reuptake inhibitor (SSRI) and serotonin and noradrenaline reuptake inhibitor (SNRI) classes of antidepressant medicines because the evidence base of benefit is larger, the side effect profile is better and the experience of practitioners is wider than with other classes of medication.
Literature on the treatment of major depressive disorder (but not yet on the treatment of these three anxiety disorders) supports the idea that, while patient preference is important to adherence, some patients will only respond to CBT and others only to antidepressants. Therefore, persisting with either when there is no response may not be beneficial, and switching to the alternative may be very productive (Cuijpers et al., 2014b; Dunlop et al., 2017; Karyotaki et al., 2016; Wiles et al., 2013).
Recommendations take into account a range of factors likely to affect treatment outcomes in addition to efficacy demonstrated in RCTs (Table 2).
Factors considered in making recommendations about treatment with unguided self-help, face-to-face CBT, guided digital CBT (dCBT) and medications.
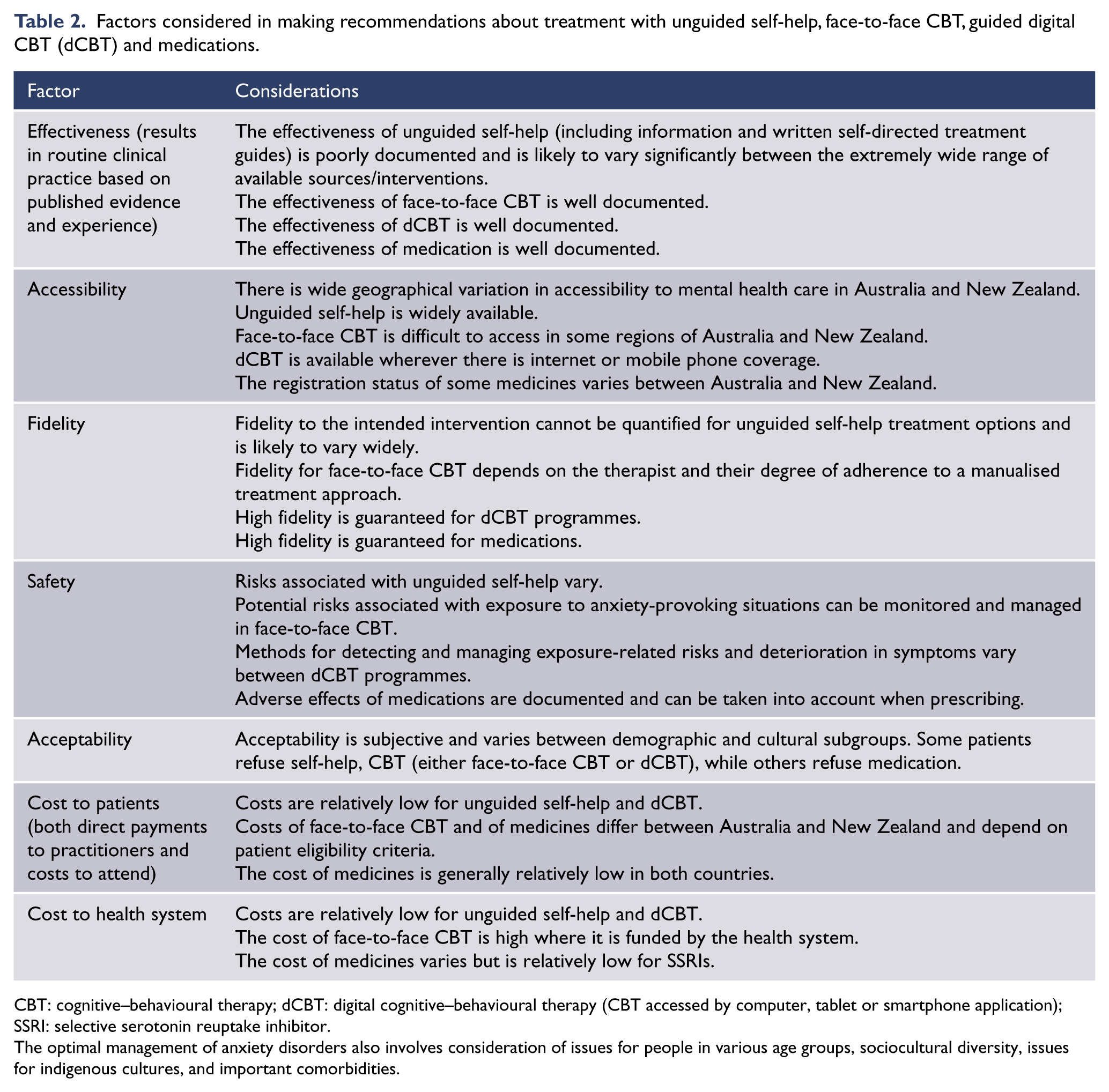
CBT: cognitive–behavioural therapy; dCBT: digital cognitive–behavioural therapy (CBT accessed by computer, tablet or smartphone application); SSRI: selective serotonin reuptake inhibitor.
The optimal management of anxiety disorders also involves consideration of issues for people in various age groups, sociocultural diversity, issues for indigenous cultures, and important comorbidities.
Epidemiology
Anxiety disorders are common, chronic mental disorders (Beard et al., 2010; Kessler et al., 2005a, 2005b; Wittchen, 2002b). They form the most common class of mental disorders, with one in seven adults suffering from an anxiety disorder in any year: 14.4% in Australia and 14.8% in New Zealand (Slade et al., 2009a; Wells, 2006a). SAD is the most prevalent anxiety disorder, followed by GAD and panic disorder/agoraphobia (Table 3). Anxiety disorders are more common in women than men and in people who are separated, divorced or widowed, less educated or unemployed. Demographic status may be both a consequence and a cause of anxiety disorders (Slade et al., 2009b).
Twelve-month prevalence of anxiety disorders in Australia and New Zealand.

Sources: Australian 2007 National Survey of Mental Health and Wellbeing (Slade et al., 2009a), Te Rau Hinengaro: the New Zealand Mental Health Survey 2006 (Wells et al., 2006).
PTSD: post-traumatic stress disorder; OCD: obsessive–compulsive disorder.
Anxiety disorders typically start early in life, especially for SAD (Table 4), and prevalence declines with age (Lampe, 2015; Slade et al., 2009a). Developing an anxiety disorder after the age of 40 years is uncommon, and so when a person over 40 presents with an anxiety disorder for the first time, alternative causes of anxiety such as mood or substance use disorders, physical illness or its treatment should be considered (Lampe, 2015; McEvoy et al., 2011).
Typical age at onset of anxiety disorders.

Sources: Australian 2007 National Survey of Mental Health and Wellbeing (Slade et al., 2009a), Te Rau Hinengaro: the New Zealand Mental Health Survey 2006 (Wells et al., 2006).
Anxiety disorders are associated with high levels of distress, disability and service use (Slade et al., 2009b), yet only a minority of people with anxiety disorders get adequate treatment (Harris et al., 2015). Fewer than half seek treatment by visiting a health professional. Those who do, commonly attend primary care and only a third receive minimally adequate treatment. The rest receive counselling or inadequate advice (Boyce et al., 2015; Harris et al., 2015).
Diagnostic issues
Is it an anxiety disorder?
Anxiety is a normal and healthy reaction to stress and is associated with the activation of the fight-or-flight response – the physical, mental and behavioural changes that allow one to deal with threat or danger. Moderate levels of anxiety may improve performance, and quite severe levels of anxiety can be experienced as normal when they are consistent with the demands of the situation.
Anxiety disorders, like all mental disorders, lie on dimensions that extend from transient symptoms, to symptoms that are severe, disabling and persist for years. The threshold on this dimension at which a disorder is defined is specified in the diagnostic criteria listed by International Statistical Classification of Diseases and Related Health Problems, 10th revision (ICD-10; World Health Organization, 1993, 2016) and Diagnostic and Statistical Manual of Mental Disorders, fifth edition (DSM-5; American Psychiatric Association, 2013).
Anxiety disorders are typified by variants of excessive worry and the urge to avoid situations that are the focus of this worry. The disability thresholds at which the diagnoses are made vary between the three anxiety disorders, with the disability at the threshold for diagnosing GAD being high and that for SAD being lower. People who meet criteria for an anxiety disorder usually have a pre-existing anxious temperament, measured as neuroticism, and are sensitive to additional stress, becoming anxious and upset very quickly.
Becoming anxious does not, itself, constitute a disorder. It is the extent of the fear and avoidance that defines the disorder; perceiving a threat of some type (physical, social, financial, other) leads to high anxiety, which in turn triggers diagnostically specific fears of negative consequences of the anxiety itself, and triggers related avoidance behaviours, which can be disabling.
Individuals seek to escape from or avoid situations that trigger these anxieties. This strategy reduces anxiety in the short term, but promotes avoidance as a preferred strategy for managing threat and anxiety in the longer term, which results in the considerable disability and distress associated with an anxiety disorder. These self-reinforcing cycles of anxiety and avoidance are a key target of treatment.
Treatment should aim to reduce the emotional sensitivity to stress, the anticipatory anxiety about outcomes and the avoidance behaviours related to specific situations (Andrews et al., 2003).
Discriminating between disorders
One of the most important ways to discriminate between different anxiety disorders is to examine the content of the associated cognitions. People with panic disorder worry that their panic will result in physical or mental harm, people with SAD worry that they will be judged negatively and those with GAD worry that disaster will occur across a variety of contexts. There is a range of self-report measures that can assist with symptom assessment and monitoring (Appendix 1).
Structured clinical interviews
There are four well-established diagnostic interviews that generate a reliable and valid diagnosis:
Structured Clinical Interview for Axis 1 DSM-IV Disorders (First et al., 1997);
Anxiety Disorders Interview Schedule (Brown and Barlow, 2014);
Composite International Diagnostic Interview (Kessler and Üstün, 2004);
Mini-International Neuropsychiatric Interview (Sheehan et al., 1998).
These interviews often take well over an hour to complete. They are mostly used in research settings and almost never used in clinical practice, even though their use has been shown to reduce treatment duration and improve treatment outcome (Andrews et al., 2010a).
Assessment
The aims of assessment are as follows:
To establish a good therapeutic relationship;
To consider differential diagnoses and establish a primary diagnosis;
To identify comorbid disorders and risks that may affect treatment and outcome;
To assist treatment planning;
To assess psychosocial and lifestyle factors that could predispose and perpetuate the anxiety disorder;
To assess the capacity of the individual to benefit from self-help material independent of the clinician.
The clinician should develop a detailed biopsychosocial formulation based on comprehensive assessment, for example:
The nature, severity and duration of symptoms;
The underlying cognitions;
Behavioural and cognitive responses to anxiety (e.g. avoidance, worry, reassurance-seeking, safety behaviours);
Precipitants for anxiety;
The degree of distress and functional impairment;
The presence of any comorbid mood disorders or anxiety disorders, substance use disorders, personality disorders or medical conditions;
The presence of suicidal ideation;
Experience with previous treatment for the disorder, including therapeutic response and adverse effects;
Personal and family history of mental disorders;
Social life and circumstances (e.g. quality of interpersonal relationships, social media presence, living conditions, employment, immigration status);
Factors that could be maintaining the disorder or preventing the individual from recovering.
Comorbidity
There are high rates of comorbidity between an anxiety disorder and other anxiety and depressive disorders (Andrews et al., 2002; Brown et al., 2001; Comer et al., 2011). The presence of comorbid anxiety and depressive disorders should be routinely assessed, as comorbidity indicates increased severity, functional impairment and economic costs (Andrews et al., 2002; Moffitt et al., 2007; Tyrer et al., 2004; Zhu et al., 2009).
Comorbidity has been associated with a slower rate of recovery and reduced response during active treatment. However, the finding of comorbidity should not lead to undue pessimism about the value of treatment and prospects for eventual recovery, especially as both antidepressant medication and transdiagnostic CBT are effective for the anxiety and depressive disorders (Newby et al., 2014, 2015).
Suicide risk
A meta-analysis that assessed rates of suicidal thoughts and behaviours among patients participating in prospective studies of anxiety disorder (Bentley et al., 2016) found that people with panic disorder, SAD and GAD were 50% more likely to experience suicidal ideation and suicide attempts. There were insufficient data to estimate the risk of suicide associated with these three disorders. The risk of suicide is greatest when there is comorbid major depression.
All patients with anxiety disorders should be assessed for suicidal thinking and the risk of self-harm.
Treatments for anxiety disorders
Psychological interventions
CBT with an experienced therapist has been studied more than other psychological therapies and is supported by numerous meta-analyses (Craske and Stein, 2016). Related psychological therapies, such as problem-solving, relaxation, interpersonal therapy, cognitive bias modification, mindfulness or psychodynamic approaches, appear to be of benefit but the evidence base is smaller.
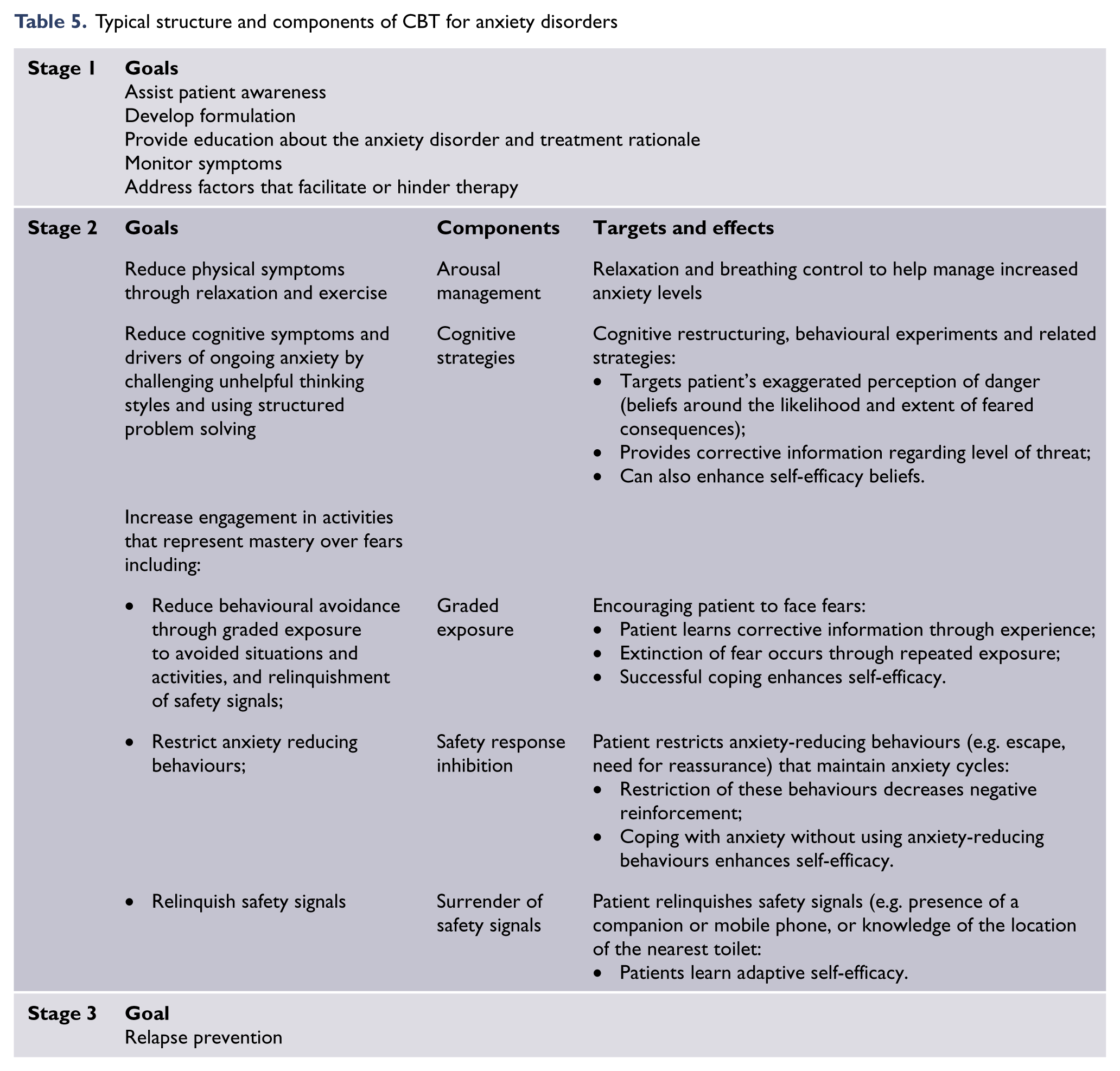
CBT is typically staged and involves education about the condition, arousal management, graded exposure, safety response inhibition, surrender of safety signals and cognitive strategies (Table 5). It should be noted, however, that CBT is a broad term encompassing a variety of component strategies. Specific CBT programmes can vary and not all are equally efficacious for a given disorder (Mayo-Wilson et al., 2014).
Typical structure and components of CBT for anxiety disorders

Efficacy of CBT
Effect size superiority is, as expected, greatest when CBT is compared with wait-list or no treatment, and less when compared to a psychological or pill placebo, or treatment as usual. For about half of the participants in clinical trials of CBT, symptoms improve to the point that they no longer meet criteria for the disorder. Disability decreases and quality of life improves.
Mode of delivery of CBT
CBT can be delivered face-to-face (individual or group), through digital CBT (dCBT) accessed by computer, tablet or smartphone application, or through self-guided CBT books for patients (self-help books).
Face-to-face delivery of CBT (particularly individual therapy) has been the most extensively studied with efficacy supported by meta-analyses (Craske et al., 2005).
dCBT is a rapidly growing field and there is now an evidence base for dCBT. Compared with CBT with a therapist, dCBT appears to be equally beneficial, with equivalent reductions in symptoms and disability and equivalent improvement in quality of life (Andrews et al., 2010a, 2018; Olthuis et al., 2016) and has the advantages of reduced cost, broader availability and consistent fidelity to the manual since delivery does not depend on the individual therapist. While CBT is part of a skill set of a well-trained clinical psychologist, dCBT for many is relatively new (see Overview of digital CBT).
Although one RCT has reported that the use of a CBT self-help book was equivalent to dCBT in the treatment of depression (Smith et al., 2017), the comparative efficacy of CBT self-help books with dCBT or face-to-face CBT in the treatment of these anxiety disorders has not been assessed in appropriately designed studies.
RCT: randomised controlled trial; CBT: cognitive–behavioural therapy; dCBT: digital cognitive–behavioural therapy; SAD: social anxiety disorder; GAD: generalised anxiety disorder.
Not available in New Zealand (as at date of publication).
Likely to be directly available in New Zealand in 2019.
Adverse effects of CBT
While the potential risks of CBT have not been systematically evaluated in published literature, a number of problems and barriers may be encountered. Effective face-to-face CBT requires considerable therapist training and expertise. Poorly conducted or poorly paced CBT may be ineffective or emotionally distressing, both of which may lead to treatment discontinuation and negative attitudes to further trials of CBT. Cost and access are frequently problematic for patients.
CBT requires emotional effort and persistence on the part of patients. Anxiety associated with exposure tasks can be distressing. CBT may increase symptomatic distress in the short term, and there is a dropout rate similar to that of antidepressant pharmacotherapy. However, the body of evidence suggests that CBT (delivered face-to-face by an experienced clinician or as guided digital CBT) produces no serious adverse effects.
Pharmacotherapy
The advantages of pharmacotherapy are that the recommended medications are easy for a primary care physician to prescribe, they are widely available and of relatively low cost, and the quality of medicines is assured.
Antidepressants, especially the SSRIs and, to a lesser extent the SNRIs, are the first-line medications for panic disorder, SAD and GAD on the basis of efficacy evidence from pill placebo-controlled RCTs (Craske and Stein, 2016; Ravindran and Stein, 2010), overall safety and low misuse potential (see sections ‘Panic disorder and agoraphobia’, ‘Social anxiety disorder’ and ‘Generalised anxiety disorder’).
Pharmacotherapy for anxiety disorders should always be accompanied by instructions for graded exposure to feared situations.
SSRI and SNRI antidepressants
For about half of the participants in clinical trials of SSRIs or SNRIs, symptoms improve to the point that they no longer meet criteria for the disorder. Disability decreases and quality of life improves.
Overall, evidence does not indicate that any one of these medications is to be preferred and selection should be made on the basis of previous success with the individual patient, patient preference and clinician familiarity with the medication.
SSRI and SNRI antidepressants can have adverse effects including initial exacerbation of anxiety (particularly where there is a history of panic attacks), nausea, headache, sleep disruption and sexual dysfunction. Although the tolerability profiles of SSRIs and SNRIs in patients with anxiety disorders are not fully established, systematic reviews of studies in depressed patients suggest that the SNRIs, duloxetine and venlafaxine, may be less well tolerated than the SSRIs (Cipriani et al., 2012; Schueler et al., 2011).
People with anxiety (particularly those with panic disorder, but also those with other anxiety disorders) are very aware of, and concerned about bodily symptoms. They should receive careful education about likely adverse effects and warned that side effects usually occur early in treatment, before benefits are seen (see section ‘Dosing of SSRIs/SNRIs’).
In addition, in younger people, there has been an association of SSRIs with suicidal thoughts, but not completed suicide (Bridge et al., 2007; Hammad et al., 2006). Clinicians should use particular caution in prescribing any antidepressant in childhood or adolescence.
Other antidepressant classes
Tricyclic antidepressants (TCAs) have demonstrated efficacy in the treatment of panic disorder and GAD, but their use raises concerns about side effects, tolerability and danger in overdose. TCAs should generally be reserved for patients who have not responded to, or been unable to tolerate, SSRIs and SNRIs.
The irreversible monoamine oxidase inhibitors (MAOIs) have proven efficacy in SAD and panic disorder. However, their use in the treatment of these disorders has been limited, due to significant potential adverse effects, the need for dietary restrictions, toxicity in overdose and important pharmacokinetic interactions. Moclobemide, a reversible inhibitor of monoamine oxidase A (RIMA), has been demonstrated to be effective in the treatment of SAD, and there is also limited evidence that it is effective in the treatment of panic disorder.
Dietary restrictions are not required with low doses in patients with normal dietary habits (e.g. those who do not consume an excess of tyramine-rich foods), but may be required at higher doses.
Very few studies have evaluated mirtazapine in the treatment of anxiety disorders. There is some evidence to support the use of agomelatine in GAD, but not the other anxiety disorders.
Drug–drug interactions with antidepressants
As with all medications, interactions with other drugs can occur because of pharmacokinetic or pharmacodynamics effects. Prescribers are strongly advised to check for the risk of these interactions when combining medications (Andrews et al., 2014).
Some antidepressant agents are potent inhibitors of cytochrome P450 (CYP) isoenzymes, which can have implications when prescribing for other conditions. For example, fluoxetine and paroxetine are potent inhibitors of the activity of CYP2D6. As a result, toxic levels or increased adverse effects may result if other drugs that are substantially metabolised via this pathway (e.g. TCAs, some antiarrhythmic agents and beta blockers) are used concomitantly.
Careful management is also needed when changing or adding another serotoninergic medication or any synergistic medications, due to the risks of serotonin syndrome. Adequate washout time is required, taking into account elimination half-life of the particular SSRI or SNRI. Fluoxetine and its metabolites have a very long half-life which means that a washout period of at least a week is required before starting another antidepressant (and 5 weeks if switching to an MAOI). Combinations of antidepressants are generally to be avoided and combining an MAOI with another antidepressant is contraindicated.
Dosing of SSRIs/SNRIs
To reduce the likelihood and severity of side effects, it is advisable to start treatment with antidepressants at a low dose (approximately half of the starting dose given to depressed patients) and to titrate slowly (increase as tolerated to therapeutic effect within the approved dose range).
Patients need to know that the medication must be taken daily as prescribed (and not just when they feel anxious) and that it can take up to 6 weeks to feel a benefit from the medication, or even up to 12 weeks for full benefit.
There is inconsistent evidence for a dose–response relationship. However, some patients who have not responded to lower doses may respond to higher doses within the therapeutic dose range.
Little evidence is available to guide duration of treatment. If the desired response is achieved, medication should be continued for 12 months and discontinuation should be gradual over weeks to months to minimise discontinuation symptoms.
Stopping and switching antidepressants
Prescribing information for each medication should be consulted when switching between medications.
When ceasing some SSRIs and SNRIs (especially paroxetine and venlafaxine), many individuals experience a discontinuation syndrome, with flu-like symptoms, shock-like sensations, dizziness, insomnia, vivid dreams, irritability and crying spells, as well as excessive sweating, and myalgia. Fluoxetine and its metabolites have a very long half-life, which means that discontinuation symptoms are unlikely, but this also means that a washout period of at least a week is required before starting another antidepressant (and 5 weeks if switching to an MAOI).
Abrupt discontinuation of TCAs may cause similar symptoms. Risk factors for a discontinuation syndrome include agents with a shorter half-life, experience of significant side effects on starting the medication, taking medication for longer than 8 weeks and concomitant use of other centrally acting medications such as centrally acting antihypertensive agents, antihistamines and antipsychotic agents (Taylor et al., 2015).
To minimise the potential for discontinuation symptoms, it is recommended that clinicians downward-titrate medication slowly over weeks to months while monitoring effects.
It is important to note that MAOIs inhibit the monoamine oxidase enzyme for 2 weeks even after discontinuation of medication, which means that a washout period of 2 weeks is needed before starting another antidepressant or other medication.
Benzodiazepines
Benzodiazepines have well-established anxiolytic effects, but there is concern about their use because of adverse effects (e.g. cognitive impairment, falls and sedation), tolerance and dependence (Gale and Millichamp, 2011). There is also a potential for abuse (Williams et al., 2017). While it is difficult to predict which patients will develop long-term problems, benzodiazepines should be avoided in those with a previous or current history of substance abuse.
Because of these concerns, benzodiazepines should not be used as first-line agents, but reserved for patients whose symptoms have not responded to other treatments. Benzodiazepines may have a favourable adverse effect profile in the management of treatment-refractory anxiety disorders, compared with atypical antipsychotic agents.
Comparing CBT with antidepressants
Medications are usually compared against pill placebo, with the progress of both treatment groups assessed by interview using assessor rating scales. In CBT trials, active treatment is usually compared with wait-list (untreated control group) and assessed by self-report scales.
In research studies, pre–post changes due to pill placebo are surprisingly large (effect size approximately 1.3; Bandelow et al., 2015), in part due to natural remission and in part due to the expectation that one might be receiving the active medication. In a review of 96 studies of antidepressants in depression (Rief et al., 2009), the pill placebo effect sizes assessed by rating scales were three times greater than those from self-report scales.
This imbalance in favour of the rating scales has been increasing over the years, not due to greater symptom reduction, but due to reduced variance in the assessor ratings. This effect is important, as the effect size is the ratio of the change in symptoms divided by the SD of the measure used. The benefits of being on an untreated wait-list control group are small (effect size approximately 0.2) due to natural remission, perhaps being tempered by the knowledge that one has to stay symptomatic if one is to receive treatment after the waiting period.
Therefore, it has been difficult to compare the benefits of medication versus pill placebo assessed by rating scales, with the benefits of CBT versus wait-list controls assessed by self-report. Studies in anxiety disorders that compare medication and CBT with the same control group, whether pill placebo, or wait-list, are rare and somewhat atypical. Accordingly, comparing medication with psychotherapies using control groups specific to the intervention could well put medication at a disadvantage.
These findings of placebo studies simply remind us that good clinical care involves encouragement and instilling hope, as well as the prescription of a specific remedy. In the management of depression, it is clear that the nonspecific elements are more important than the specific elements, as reflected in the response to pill placebo. The management of anxiety disorders is likely to be similar.
The US Agency for Healthcare Research and Quality (Agency for Healthcare Research and Quality, 2015) has issued a clinician advisory that CBT and medication are equally effective for mild, moderate and severe depressive disorder, and this finding probably extends to the three anxiety disorders covered in this guideline. The best treatment to prescribe for an individual patient should be decided in consultation with the patient, taking into consideration prior responses to treatment.
Combination of CBT and pharmacotherapy
Despite its common use in clinical practice, there is currently limited evidence to support the routine combination of CBT and pharmacotherapy for anxiety disorders. To date, few clinical trials have evaluated the combination, and their findings have been conflicting. The state of the evidence for specific disorders is outlined below.
Panic disorder
Four studies, including two meta-analyses (Bandelow et al., 2007; Cuijpers et al., 2014a) and two RCTs (Roy-Byrne et al., 2005; Van Apeldoorn et al., 2013) found that the combination of SSRIs and CBT was better than SSRIs alone in the treatment of panic disorder. Another RCT (van Apeldoorn et al., 2008) and a narrative review (Würz and Sungur, 2009) concluded that CBT was equally effective as the combination of medication and CBT, suggesting that CBT contributed the greater benefit in combination therapy.
In contrast, two meta-analyses (Bandelow et al., 2007; Hofmann et al., 2009) concluded that the combination of medication and CBT was better than CBT, and one RCT (van Apeldoorn et al., 2008) found medication equally effective as the combination, suggesting that medication contributed the greater benefit.
SAD
A meta-analysis of RCTs comparing psychopharmacological and psychological treatment for anxiety disorders (Bandelow et al., 2007) found that the combination of medication and CBT was better than either single treatment in the treatment of SAD. However, only three studies have compared combined CBT and pharmacotherapy against monotherapies for SAD and only one of these has used SSRI, so conclusions must be considered tentative.
GAD
A meta-analysis investigating the benefits of adding pharmacotherapy to CBT in the treatment of anxiety disorders (Hofmann et al., 2009) found that the combination of medication and CBT was better than CBT in the treatment of GAD. In contrast, a narrative review (Würz and Sungur, 2009) found the combination of medication and CBT to be equal to CBT in the longer term.
Digital CBT combined with medication
There are no substantive studies of dCBT compared to medication or when used in conjunction with medication. In these guidelines we accept that there is evidence that both medication and CBT (and dCBT) are effective when used separately, and given the possibility that they target different individuals, we recommend the use of both medication and CBT together when a person has not responded or when the condition is severely disabling.
General principles of treatment
Collaborative pragmatic approach
These guidelines recommend a pragmatic approach to selecting therapy in collaboration with the patient – beginning with psychoeducation and advice on lifestyle factors, followed by specific treatment. Selection of treatment should be based on evidence of efficacy, patient preference, accessibility, cost, tolerability and safety, with consideration of symptom severity. Recommended initial treatment options include CBT (face-to-face or dCBT), medication with an SSRI (or an SNRI if SSRIs are ineffective or not tolerated) accompanied by instructions for graded exposure to anxiety triggers, or a combination of CBT plus medication.
When possible, the patient’s family or significant others should be involved in management planning decisions and in supporting the person through their treatment.
Education
All patients should be given education about anxiety, especially the adaptive aspects; an increase in alertness and anxiety facilitates problem-solving, whereas severe anxiety impairs ability to problem-solve and can be debilitating (Yerkes-Dodson curve). This information is often extremely beneficial because the person often feels that what they have been experiencing is frightening and unique to them. Education about management involves an outline of fear-reinforcement cycles as an explanation of why anxiety has persisted and the need to eventually confront what is feared. It also includes promotion of healthy behaviours (e.g. healthy eating, good sleep, regular exercise and reduced use of caffeine, tobacco and alcohol).
Reliable, plain-language information for patients is available at www.yourhealthinmind.org.
Self-monitoring
Active self-monitoring of symptoms encourages patients to become aware of the triggers to anxiety and their typical responses, including thoughts and feelings, and actions they take to try to cope (e.g. escape, avoidance, reassurance-seeking, use of medications, substances or over-the-counter preparations). This information will later be of assistance in treatment planning, as well as being important in helping patients become more aware of fear-reinforcement cycles.
Discussing treatment options with the patient
The treatment options, and the likely cost, duration and content of treatment and expected outcome should be discussed. The treatment options are as follows:
Face-to-face CBT provided by an experienced clinician;
Guided dCBT accessed by computer or mobile phone – guidance can consist of regular contact and encouragement to complete the course and does not have to be conducted by a clinician. Unguided dCBT is associated with lower adherence or compliance;
Medication with an antidepressant, accompanied by instructions for graded exposure to anxiety triggers.
The choice should be made in collaboration with the patient with one proviso: people with severe anxiety disorders or with severe comorbid major depression should be advised to consider a combination of antidepressant medication plus CBT.
Follow-up and monitoring effects of treatment
The onset of beneficial effects typically occurs 4–6 weeks after starting treatment with either CBT or medication. Treatment should be reviewed after 4–6 sessions of weekly CBT or after 4–6 weeks of pharmacotherapy with advice on graded exposure to feared situations.
Patients should initially be seen weekly to monitor adherence, adverse effects, and to identify any worsening of symptoms until there is a response and symptoms have stabilised. Rating scales (Appendix 1) can be used to monitor change and are often helpful for both patient and clinician.
Practical guidance for clinicians
Talking to patients about anxiety and its treatment
Although doctors readily understand the detail in these guidelines, it can be difficult to formulate how they might talk to a patient. As an example, this is what a doctor might say to a patient newly diagnosed with GAD:
We’ve agreed that you have generalised anxiety disorder – a disorder of excessive worry about everyday things, and that you have been like this for a long time and that your worry is wearing you down.
So, let’s talk about treatment. Good treatment should enable you to worry over things that need to be worried about and not worry over things that don’t. Furthermore, good treatment should mean that with a few months of treatment your life is no longer dominated by worry and avoidance. You can just be yourself.
It is important to provide information about treatment options that will allow individuals to make informed choices. For example, a doctor might say something like:
There are two treatments that work in the longer term: cognitive–behavioural therapy (usually called ‘CBT’), which can be delivered either face-to-face by a clinician or in a digital format (over the internet, on your computer or smartphone), and medication with an antidepressant medicine.
I know that may sound strange, but antidepressants have been shown to be very good for anxiety, even in people who are not depressed.
With CBT, 8 out of 10 people will improve, and 5 out of 10 will recover completely and will stay well. The benefits from medication are comparable, but there may be a higher incidence of adverse effects and higher relapse rates after the medication is stopped. However, CBT can be quite hard work.
Either treatment will take about 4–6 weeks to start to work for you. Which treatment would you prefer?
Managing the doctor’s own anxiety
As with all illnesses, patients with anxiety disorders come when their symptoms are severe. This high level of anxiety may be interpreted by the doctor as an urgent need to do something.
There are two rules to follow: once you have made a diagnosis, tell the patient you have a treatment for it. Second, do not let your own anxiety lead you to prescribe sedatives or over-investigate for all possible differential diagnoses.
Our anxiety as doctors comes from our own uncertainty about being able to relieve a patient’s distress, a mistaken sense that this needs to be done urgently and excessive concern not to miss a physical cause of anxiety.
Practical issues with CBT
A practical approach to assessing fidelity of delivered therapy to the intended treatment model (if provided by another therapist) and the patient’s adherence to treatment might include the following questions:
Can you show me how you do your slow breathing?
Can you show me your mood diary? (Can you upload your mood app output to share it with me?)
What have you been working on in your CBT? (Can you show me your homework?)
It can also be helpful to contact the therapist to check fidelity and adherence.
Practical issues in pharmacotherapy
Careful discussion with the patient about the risks and benefits of antidepressants is important. People with anxiety are often hypervigilant to physical symptoms and adverse effects. Most adverse effects are likely to be minor and time-limited.
The main issue is not the adverse effects themselves, but the potential to misinterpret these as signs of serious illness. This can be managed through careful discussion of what to expect, including that the adverse effects will commonly occur before the benefits are seen and starting treatment at a low dose (approximately half the starting dose used in patients with depression). Infrequently, medication may lead to activation/agitation in the initial stage of treatment. Rarely, it can be accompanied by suicidal ideation. It is therefore advisable to arrange an early review, for example, within a week.
The patient can be advised to increase to the minimal therapeutic dose within the approved dose range as tolerated (e.g. usually within 1–2 weeks). There is usually no need to prescribe a sedating medication concurrently.
Response times are longer than for depression. It can take up to 6 weeks for benefits to start to be seen. There is no advantage to increasing the minimal therapeutic dose prior to this time.
In the treatment of panic disorder and agoraphobia, SAD or GAD, available evidence does not consistently support the use of higher doses of antidepressant medicines than those recommended as standard for the treatment of depression. However, some patients who have not responded to lower doses may require higher doses within the therapeutic range.
There is a paucity of evidence regarding optimal duration of treatment. Expert consensus suggests continuing for approximately 6–12 months after optimal response has been achieved. When medication is discontinued, expert advice is to taper the dose over weeks to months.
Lack of response.
Patients who do not respond to CBT and two adequate trials of antidepressant medication (12 weeks of treatment at a suitable dose) should be referred to a psychiatrist for a second opinion on the validity of the diagnosis and treatment plan, adherence with instructions and the utility of other treatment options.
Panic disorder and agoraphobia
Diagnosis
People with panic disorder:
Experience recurrent, unexpected panic attacks;
Are persistently concerned about having another panic attack or the consequences of a panic attack (e.g. that they are having a heart attack or losing control);
May change their behaviour in ways which are designed to avoid having further panic attacks (e.g. avoiding situations from which escape may be difficult or where help might not be available in case of a panic attack).
A panic attack is an abrupt surge of intense fear or discomfort that reaches a peak within minutes (Table 6). The person often feels that they have to do something urgently (e.g. escape to a safer place).
Essential features of panic disorder and agoraphobia.

For diagnostic criteria for panic disorder and for agoraphobia, refer to DSM-5 pages 217 and 218 (American Psychiatric Association, 2013) and ICD-10 diagnostic criteria for research pages 91 and 94 (World Health Organization, 1993).
DSM-5: Diagnostic and Statistical Manual of Mental Disorders, 5th edition; ICD-10: International Statistical Classification of Diseases and Related Health Problems, 10th revision.
The requirement that panic attacks need to be recurrent for a diagnosis of panic disorder is to ensure a high threshold for diagnosis, because non-recurrent panic attacks are relatively common in the population (Culpepper, 2003). The surge of anxiety that occurs in the context of exposure to a dangerous situation is not conceptualised as a panic attack, despite it having similar physical symptoms, as the ‘catastrophising’ cognitive symptoms do not occur.
The term unexpected to describe panic attacks means that they seem to occur ‘out of the blue’ and are not associated with known situational precipitants. This is in comparison with expected panic attacks in response to obvious precipitants: people with other anxiety disorders (e.g. specific phobia, SAD or PTSD) can have panic attacks, but this response is expected when a person is exposed to their feared situation.
People with panic disorder can have both unexpected and expected panic attacks, with unexpected panic attacks usually occurring during the initial stages of the condition. The frequency of panic attacks can vary widely from several times per day to one every 6 months, but the anticipatory anxiety does not abate.
The type and combination of symptoms can vary. In panic disorder the person’s principal concern is physical (e.g. that they are having a heart attack or seizure, or that they will choke), mental (that they are going to ‘lose control’) and, to a lesser extent, social (embarrassment that they will be judged negatively because of the visible panic symptoms). They often feel that no one can understand them and that their panic experiences set them apart from others. People with panic disorder have a persistent fear of having another panic attack (and/or of certain symptoms during an attack).
The changes in behaviour that may occur in panic disorder represent attempts by the person to minimise or avoid further panic attacks. For example, by avoiding physical exercise, restricting activities or avoiding situations where escape may be difficult, or in which it may be difficult to access help in the event of a panic attack. These situations include public transport, enclosed spaces, open spaces (e.g. bridges, parks), being in crowds, being alone outside of the home or even being home alone.
In DSM-5 (American Psychiatric Association, 2013), a separate diagnosis of agoraphobia, in addition to panic disorder, is given if the patient also meets criteria for agoraphobia. This approach to diagnostic classification is similar to that of ICD-10 (World Health Organization, 1993, 2016; agoraphobia with panic attacks) and represents a change from Diagnostic and Statistical Manual of Mental Disorders, fourth edition (DSM-IV) (American Psychiatric Association, 2000), in which the diagnostic options were ‘panic disorder without agoraphobia’, ‘panic disorder with agoraphobia’ and ‘agoraphobia without a history of panic disorder’.
Natural history
Prevalence
The prevalence of panic attacks is high, with around one in nine people reporting a panic attack in any year (American Psychiatric Association, 2013).
In the most recent (2007) Australian National Survey of Mental Health and Wellbeing, the 12-month prevalence of panic disorder was 2.3% for males, 2.9% for females (2.6% overall); and for agoraphobia, 2.1% for males, 3.5% for females (2.8% overall; Slade et al., 2009a). In Te Rau Hinengaro: The New Zealand Mental Health Survey (Wells et al., 2006), the prevalence of panic disorder was 1.7% (male 1.3%, female 2.0%).
In community samples, one-third to one-half of those diagnosed with panic disorder also have agoraphobia (Bienvenu et al., 2006; Eaton et al., 1994; Goodwin et al., 2005), but higher rates are found in clinical samples (Weissman et al., 1997). As with the other anxiety disorders, panic disorder and agoraphobia are each more common in women than men, in ratios approximating 2–2.5:1 (Eaton et al., 1994; Wells, 2006). When panic disorder is associated with agoraphobia, the female-to-male ratio is higher (2.5–4:1; Eaton et al., 1994; Yonkers et al., 1998). Panic disorder and agoraphobia are more common in people who are separated, divorced or widowed, less educated or unemployed (Hunt et al., 2002).
Many people vividly remember their first panic attack and, even though the attack itself was unexpected, often report a prodrome of symptoms or significant life events prior to the onset of panic disorder (De Loof et al., 1989). The location or circumstances of the first panic attack often determines the person’s subsequent response in terms of avoidance of that or similar locations.
Course and prognosis
The median age of onset of panic disorder is 30 years (McEvoy et al., 2011). Panic disorder can have onset prior to puberty, but this is relatively uncommon. Panic symptoms in childhood and adolescence are frequently a predictor of later onset psychiatric disorders (Goodwin et al., 2004). Onset after the age of 45 is unusual and would suggest the presence of an organic disorder or other mental disorder, such as depression.
Panic disorder often follows a chronic course with waxing and waning. Some people have episodic outbursts with periods of remission in between. Some experience a fluctuating course with exacerbations often precipitated by life-event stress, excess caffeine, sleep disruption, physical illness such as acute infections, or hormonal changes, while others have more chronic course. Only a few people have complete, sustained remission without relapse (Andersch and Hetta, 2003; Roy-Byrne et al., 2006; Swoboda et al., 2003).
Distress, disability and impairment
Panic disorder causes a significant burden of disease. It is associated with significant personal distress, disability and impairment, both directly and through the burden of comorbidity, and often puts a significant strain on the patient’s family or support network. Compared with healthy controls, patients with panic disorder have greater impairment on measures of quality of life (Barrera and Norton, 2009; Comer et al., 2011) and an increased likelihood of suicide attempts (Nepon et al., 2010).
Panic disorder is associated with substantial economic costs due to decreased work productivity and absenteeism (mean 36 days/year; de Graaf et al., 2012), increased health care utilisation and high medical costs (Batelaan et al., 2007; Gros et al., 2011).
Service use
Relative to people with other mental health disorders, people with panic disorder seek help more frequently, although only about one-third of those affected seek help within a year of onset of the disorder (Boyd, 1986; Wang et al., 2005). When seeking help, people often go to medical specialists or emergency departments, probably because of the predominance of physical symptoms and the catastrophic concerns about these (Deacon et al., 2008; Fleet et al., 1998; Gerdes et al., 1995; Katerndahl and Realini, 1995). They may be referred for unnecessary investigations to rule out possible medical causes for the symptoms. Yet, evidence suggests that panic disorder is undertreated in both primary (Stein et al., 2004) and secondary care (Bruce et al., 2003; Burton et al., 2011; Goisman et al., 1999).
Assessment
The aims of assessment are as follows:
To establish a good therapeutic relationship;
To establish a primary diagnosis of panic disorder (including distinguishing between normal and pathological anxiety) and to determine whether agoraphobia is also present;
To rule out differential diagnoses;
To identify comorbid disorders that may affect treatment and outcome;
To identify predisposing, precipitating and perpetuating biopsychosocial and lifestyle factors.
Comprehensive assessment includes obtaining information about all of the following:
The nature, severity and duration of symptoms, avoidance behaviours and use of safety behaviours;
The degree of distress and functional impairment;
The presence of comorbid anxiety or mood disorders, substance use (including tobacco, illicit substances, prescribed and over-the-counter medications and other substances such as caffeine and ‘energy’ drinks) and medical conditions;
Personal and family history of mental health disorders, and personal history of chronic health problems, domestic violence or sexual abuse;
Experience of, and response to, past treatments;
The quality of interpersonal relationships, and social support network, living conditions, social isolation, employment status including work environment, and immigration status;
Safety, including suicide risk;
Medical evaluation including system review and appropriate physical examination and blood tests including, at a minimum, thyroid function tests, urea and electrolytes (U&E), full blood count (FBC), and blood glucose level (BG), electrocardiography (ECG) if cardiac symptoms or relevant family history (e.g. arrhythmias);
The patient’s goals and expectations of treatment.
Differential diagnosis
Distinguishing panic disorder from physical disorders
It is important to exclude substance-induced and medical conditions that can present with panic attacks or panic-like symptoms (Table 7). However, not all patients need to undergo extensive diagnostic testing for all of these conditions; investigations need to be tailored to the clinical presentation.
Common conditions associated with panic attacks or panic-like symptoms.

Distinguishing panic disorder from other mental disorders
A crucial first step is to determine whether the panic attacks are better understood as part of another disorder.
Identify precipitants for the panic attacks, where possible. In panic disorder, panic attacks are, at least initially, reported as unexpected or uncued.
A further helpful component of the differential diagnosis is establishing whether there is persistent concern or behavioural change because of fear of further panic attacks. This may differentiate panic disorder from panic attacks associated with depression and bipolar disorder.
Panic disorder needs to be differentiated from illness anxiety disorder, in which people are overly preoccupied with somatic symptoms and focused on their body. However, in panic disorder, people are most concerned about these symptoms during the panic attacks, report symptoms of autonomic hyperactivity in particular, and the focus of their catastrophic fears is immediate (e.g. heart attack). In illness anxiety disorder, these fears are more distant (e.g. cancer).
Instruments for assessment and differential diagnosis are listed in Appendix 1.
Comorbidity
Panic disorder is often associated with comorbidity with other mental health disorders, particularly other anxiety disorders, mood disorders, psychotic disorders and substance use disorders (Roy-Byrne et al., 2006). Major depressive disorder is common, occurring in approximately 35–40% of people with panic disorder (Kessler et al., 2006), is associated with greater impairment in functioning and can result in poorer outcome (Scheibe and Albus, 1994). Alcohol use and dependence have been associated with panic disorder and may also complicate treatment (Kessler et al., 1997; Otto et al., 1992).
There is also significant comorbidity with medical disorders, particularly thyroid disease, cardiac disease, respiratory conditions, migraine, irritable bowel syndrome, arthritis, various allergies, chronic pain and cancer (Korczak et al., 2007; Roy-Byrne et al., 2006; Yamada et al., 2011). Using a timeline approach to the onset of disorders may be helpful for establishing the chronological onset of the disorders and identifying the primary disorder (National Institute for Health and Care Excellence, 2011a).
It is important to determine the presence of psychiatric comorbidity because this has been shown to increase the severity of the illness, functional impairment, economic costs, and is associated with worse outcomes. It is also important in terms of treatment planning in order to incorporate treatment of the comorbid condition, in addition to the panic disorder. However, medical comorbidity is not associated with poorer outcomes across anxiety disorders (Olatunji et al., 2010). The effectiveness of anxiety interventions is not significantly affected by the presence of multiple medical comorbidities, with the exception of migraine sufferers, who display less improvement at long-term follow-up (Campbell-Sills et al., 2013).
Monitoring progress
It is important to monitor and review progress. This involves monitoring the distress caused by the panic attacks, their frequency, avoidance of feared situations and associated functional impairment.
Self-report questionnaires
Two scales have been developed that assess the components of panic disorder (panic attacks, anticipatory anxiety and avoidance, health related concerns and disability): the Panic Disorder Severity Scale (Houck et al., 2002) and the Panic and Agoraphobia Scale (Bandelow, 1995). Both are designed to be used by clinicians, but there are also self-report versions (Table 8).
Self-report measures for panic disorder.
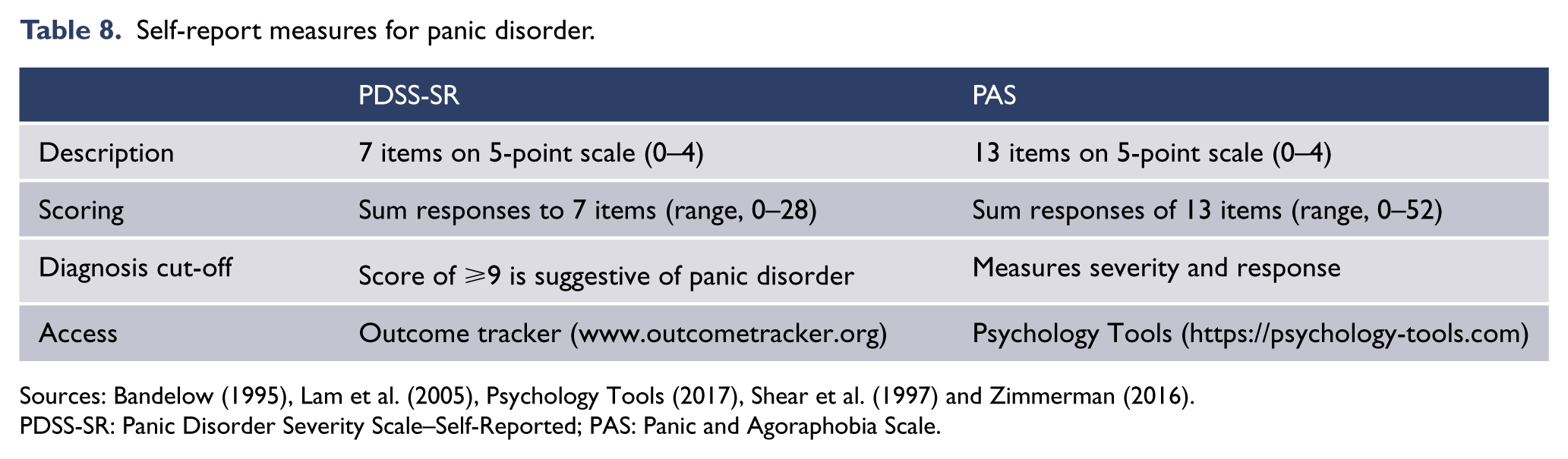
Sources: Bandelow (1995), Lam et al. (2005), Psychology Tools (2017), Shear et al. (1997) and Zimmerman (2016).
PDSS-SR: Panic Disorder Severity Scale–Self-Reported; PAS: Panic and Agoraphobia Scale.
Assessment instruments
Additional instruments have been developed for assessing aspects of anticipatory anxiety (Appendix 1). These include the Agoraphobic Cognitions Questionnaire, which measures the frequency of thoughts about catastrophic consequences of anxiety and panic (Chambless et al., 1984), the Anxiety Sensitivity Index (Reiss et al., 1986), which measures symptoms of anxious arousal; and the Mobility Inventory which measures avoidance of typical agoraphobic situations (Chambless et al., 1985).
Treatment
Overview
A collaborative, pragmatic approach is recommended, beginning with psychoeducation and advice on lifestyle factors (see section ‘General issues in the recognition and management of anxiety disorders’), followed by specific treatment. In addition to efficacy, selection of initial treatment should take into account severity, patient preference, accessibility, cost, tolerability and safety.
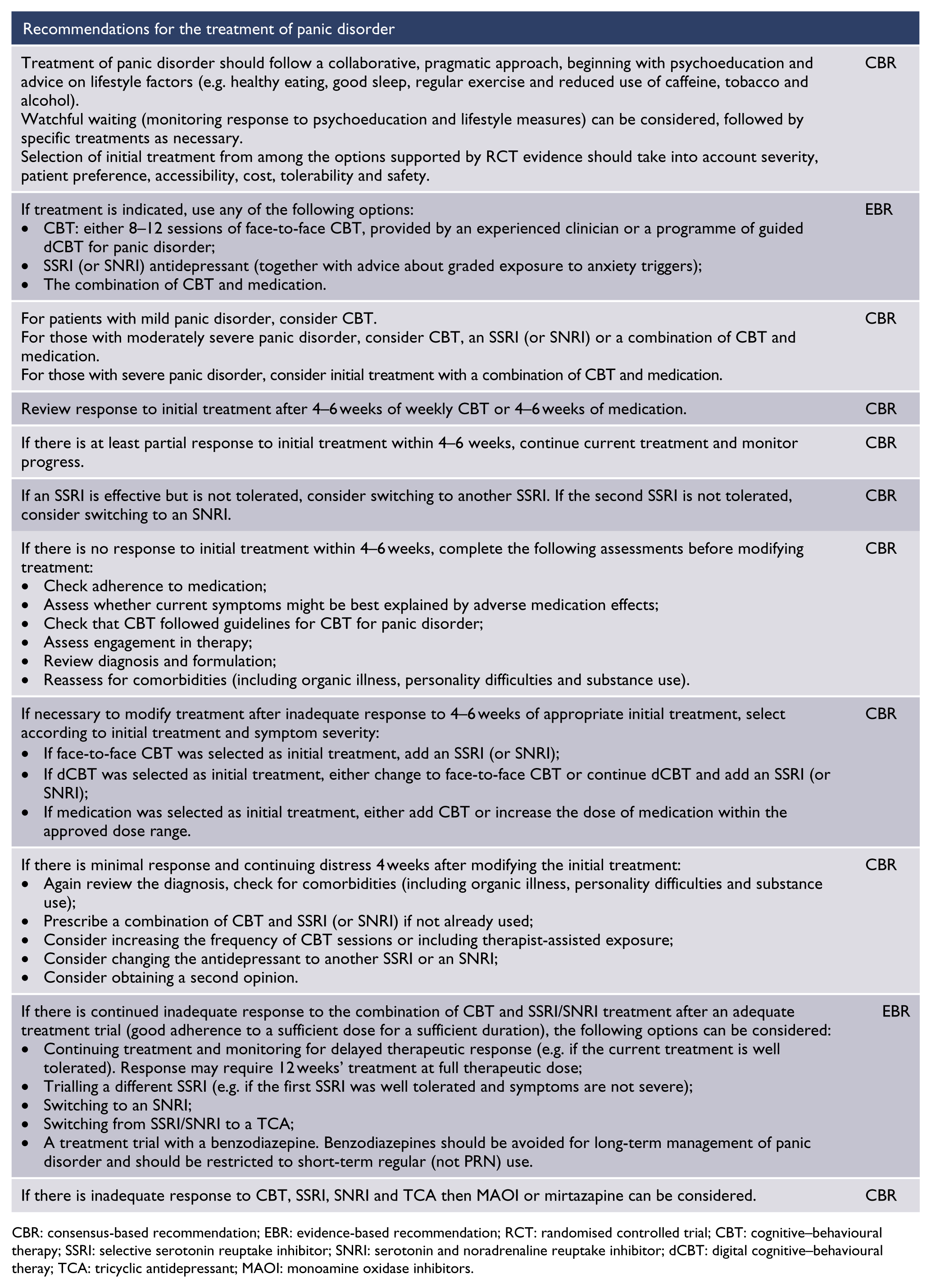
CBR: consensus-based recommendation; EBR: evidence-based recommendation; RCT: randomised controlled trial; CBT: cognitive–behavioural therapy; SSRI: selective serotonin reuptake inhibitor; SNRI: serotonin and noradrenaline reuptake inhibitor; dCBT: digital cognitive–behavioural theray; TCA: tricyclic antidepressant; MAOI: monoamine oxidase inhibitors.
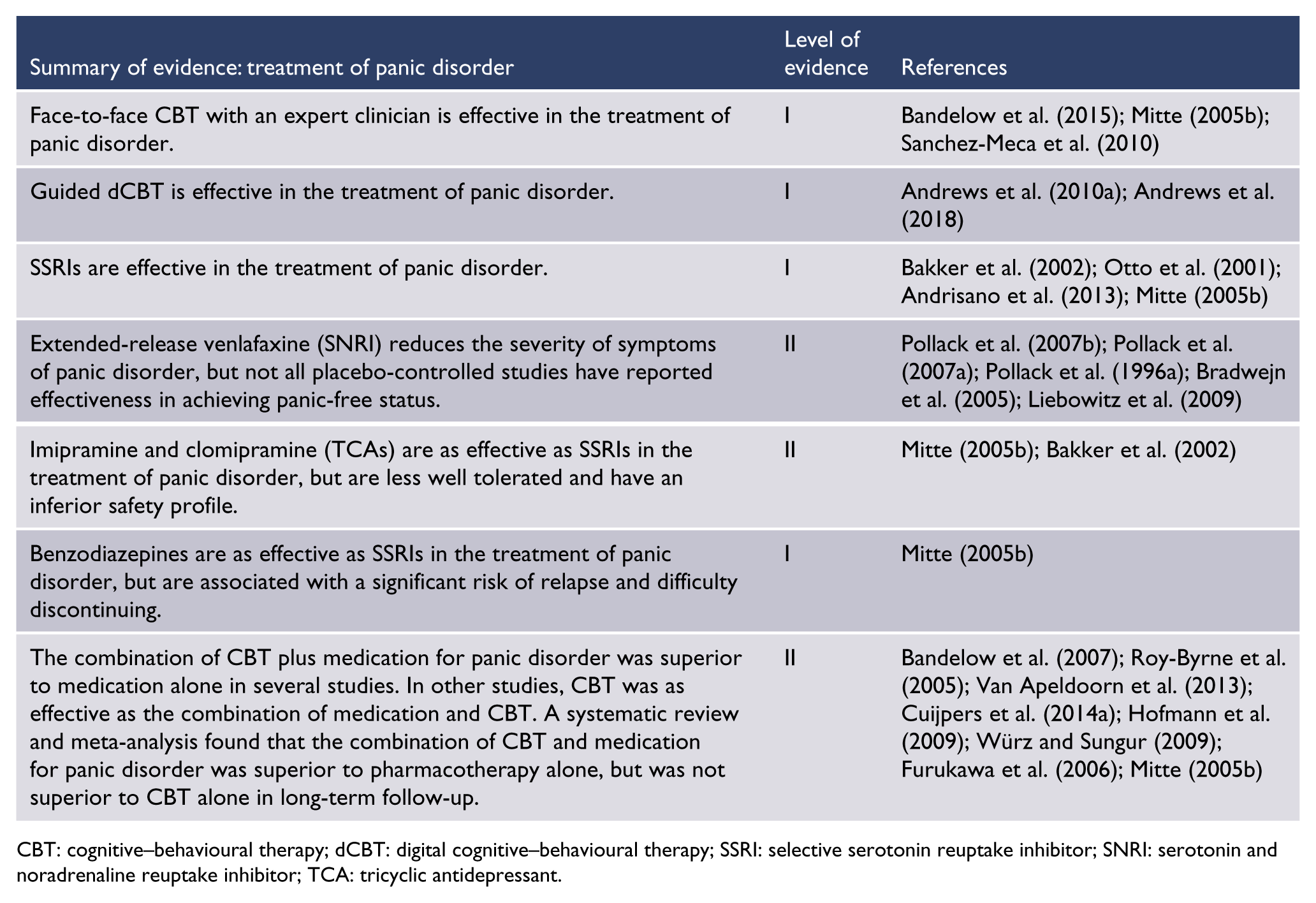
CBT: cognitive–behavioural therapy; dCBT: digital cognitive–behavioural therapy; SSRI: selective serotonin reuptake inhibitor; SNRI: serotonin and noradrenaline reuptake inhibitor; TCA: tricyclic antidepressant.
A large body of level I evidence demonstrates the efficacy of CBT, antidepressant pharmacotherapy with SSRIs, SNRIs or TCAs and benzodiazepines for the treatment of panic disorder. There is limited or lower quality evidence for other psychological therapies, other antidepressant classes and other medication classes. Effect sizes are generally moderate.
Initial treatment options are CBT (face-to-face CBT with an expert clinician, usually a clinical psychologist; or guided dCBT), medication with an SSRI (or an SNRI if SSRIs are ineffective or are not tolerated) in combination with graded exposure to anxiety triggers, or a combination of CBT plus medication. Initial treatment should be selected in collaboration with the patient, based on the severity of the disorder, previous response to treatment, availability and the person’s preference.
CBT for panic disorder
CBT has been shown to be efficacious in the treatment of panic disorder. It has been extensively studied in studies comparing CBT with a control, pharmacotherapy, and the combination of CBT and pharmacotherapy.
Structure and components
Typical CBT programmes address the physical, cognitive and behavioural symptoms of panic disorder and aim to prevent relapse in three stages (Table 9).
Structure and content of CBT for panic disorder.
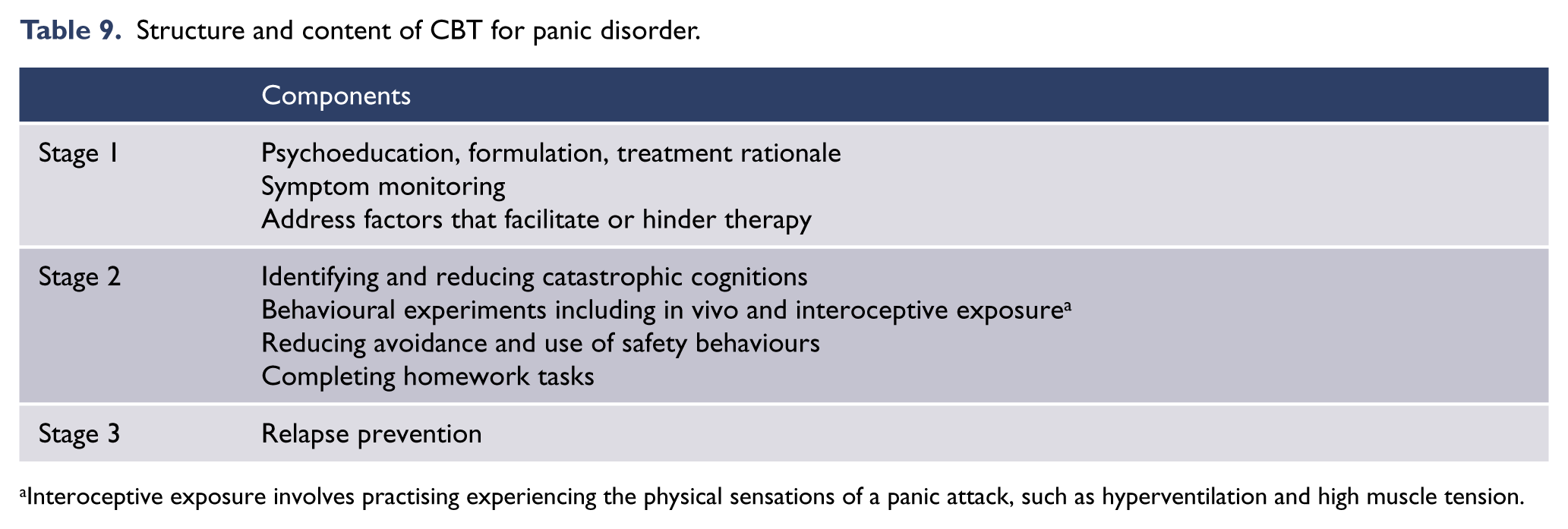
Interoceptive exposure involves practising experiencing the physical sensations of a panic attack, such as hyperventilation and high muscle tension.
The first stage includes psychoeducation (explaining about anxiety and the symptoms of panic disorder), formulation (or case conceptualisation), treatment rationale, symptom monitoring and addressing factors that facilitate or hinder therapy. Motivational interviewing and education of the person’s family or members of their social support network should also be considered, and written information or links to reliable online information should be provided.
The second stage includes identifying and reducing cognitive symptoms through challenging unhelpful thinking, particularly about catastrophic cognitions, using behavioural experiments and in vivo exposure to test hypotheses, with the aim of reducing safety behaviours and avoidance, and interoceptive exposure to feared physical sensations.
The final stage is relapse prevention that includes identifying potential precipitants for setbacks, identifying the patient’s early warning signs and developing a plan to manage setbacks and prevent relapse.
The optimal duration of CBT for panic disorder is 7–14 hours, usually delivered in weekly sessions (Clark et al., 1999; Marchand et al., 2009; National Institute for Health and Care Excellence, 2011a; Roberge et al., 2008a). Intensive treatment has also been shown to be effective (Bohni et al., 2009; Deacon and Abramowitz, 2006).
Several studies support the efficacy of exposure treatment as a component of CBT, in reducing panic and agoraphobic symptoms (Ito et al., 2001; Marks et al., 1993; Ost et al., 1993; Swinson et al., 1995). Interoceptive exposure has been shown to be superior to relaxation (Siev and Chambless, 2007). A meta-analysis of three studies of cognitive therapy (in which exposure to situations or sensations which were known to induce panic or behavioural experiments were actively discouraged) also emphasised the importance of exposure, since cognitive therapy without exposure was not very effective at reducing symptoms of panic (SMD = 0.34; 95% confidence interval [CI] = [–0.25, 0.93]) or agoraphobia (SMD = 0.13; 95% CI = [–0.97, 0.71]; Sanchez-Meca et al., 2010). Breathing retraining has not been found to contribute any incremental benefit over other CBT components and some evidence suggests a poorer outcome (Craske et al., 1997; Schmidt and Woolaway-Bickel, 2000). Factors that improved outcome were inclusion of homework and a follow-up programme (Sanchez-Meca et al., 2010).
Efficacy
Meta-analyses (Bandelow et al., 2007; Cuijpers et al., 2013; Mitte, 2005b) report that the efficacy of CBT is equivalent to that of pharmacotherapy. On average, response rates are 50–70%.
A meta-analysis of 19 studies that assessed the efficacy of CBT for panic disorder compared with control (wait-list, psychological control or pill placebo) reported large effect sizes on measures of both panic disorder and agoraphobia (Sanchez-Meca et al., 2010). Other meta-analyses of clinical trials have similarly concluded that CBT significantly reduces panic disorder symptoms, is markedly more effective than placebo or wait-list control, and benefits are maintained at 6 and 12-month follow-up (Butler et al., 2006; Clum et al., 1993; Gould et al., 1995; Hofmann et al., 2010; Mitte, 2005b; Sanchez-Meca et al., 2010; Westen and Morrison, 2001).
The average dropout rate for CBT from meta-analyses was 12.7%. On average, response rates are 50–70%. The response rate is similar to that of medication, although some report it as better than medication (Roshanaei Moghaddam et al., 2011).
The benefits of CBT have been shown to be long lasting (Barlow et al., 2000; Brown et al., 1995; Clark et al., 1999; Fava et al., 1995; Marchand et al., 2009; Ruwaard et al., 2010). A study in which participants were followed up for 2–14 years (median, 8 years) reported that only 23% had a relapse at any time over this period (Fava et al., 2001b).
Predictors of poorer outcome to CBT include severity of panic disorder, presence of agoraphobia, earlier age of onset of panic disorder and comorbid social anxiety (Dow et al., 2007; Haby et al., 2006).
Mode of delivery
CBT can be delivered face-to-face (individual or group), accessed by computer, tablet or smartphone application (dCBT), or through self-help books. CBT delivered face to face (particularly individual) is the most traditional form of delivery and the most widely available, hence has been the most extensively studied.
Studies of CBT delivered in group formats report that group CBT was comparable to individual therapy (Lidren et al., 1994; Néron et al., 1995; Telch et al., 1993).
dCBT has been an area of rapid development and study over recent years. There have been 12 RCTs of automated guided dCBT, which show a mean effect size of 1.31 (95% CI = [0.85, 1.76]) at a mean follow-up of 7.8 months post intervention, and an average adherence of 74%. Three of the studies included a comparison with face-to-face CBT and showed no difference in efficacy (Andrews et al., 2018). Other meta-analyses have reported similar outcomes (Olthuis et al., 2016). All studies used a wait-list or information only control. One study examined long-term outcomes over 3 years and reported no evidence of relapse in the first year (Ruwaard et al., 2010). Patients in dCBT studies reported that they were very satisfied with treatment, stressing convenience, privacy and ability to work at their own pace.
Other psychological therapies and interventions
There is currently much interest in other potentially useful therapies such as mindfulness, and Acceptance and Commitment Therapy (ACT). However, there is insufficient evidence from meta-analyses specific to panic disorder to make conclusions at this time.
Exercise as a form of treatment has been reported to be less effective than medication and no more effective than relaxation (Broocks et al., 1998; Wedekind et al., 2010). Exercise is, nevertheless, recommended by National Institute for Health and Care Excellence (NICE) as part of general health care for people with panic disorder (National Institute for Health and Care Excellence, 2011a). Often, patients with panic disorder have stopped exercising because they catastrophically misinterpret the bodily sensations produced such as increased heart rate, shortness of breath and sweating. This can be managed by careful explanation and encouraging gradual introduction of exercise.
Panic-focused psychodynamic psychotherapy delivered twice a week in a 12-week manualised treatment programme has been shown to be effective in one RCT (Milrod et al., 2007).
Two studies of Eye Movement Desensitisation and Reprocessing (EMDR) have reported equivocal or unsustained benefits and do not support the use of EMDR for panic disorder (Feske and Goldstein, 1997; Goldstein et al., 2000).
Pharmacological treatment
Pharmacotherapy has been extensively studied and shown to be efficacious in the treatment of panic disorder.
Before starting any medication, psychoeducation should be provided about anxiety and panic symptoms, and advice given about lifestyle factors that may be contributing or perpetuating symptoms.
The timeframe for change should also be discussed as response is relatively slow, as indicated by findings that antidepressants separate from placebo most convincingly at 12 weeks and that response rates continue to increase over follow-up periods extending to 6 months. Potential adverse effects of medication should also be discussed carefully. This is particularly important for patients with panic disorder, who are very focused on somatic symptoms.
Throughout treatment, the patient should be encouraged to gradually increase exposure to feared situations.
Overview of efficacy
Medication with SSRIs, SNRIs, TCAs and benzodiazepines has been shown to be efficacious in the treatment of panic disorder. These medications have been extensively studied in RCTs comparing medicines with pill placebo control, CBT, and the combination of CBT and pharmacotherapy.
On average, rates of response to medication are 50–70%. Meta-analyses report equivalent efficacy for SSRIs, TCAs and benzodiazepines. These studies also report no differences in attrition and dropout rates for the different medications (SSRIs 23.1%, TCAs 23.5% and benzodiazepines 17.7%). MAOIs are effective in managing the symptoms of panic disorder (Batelaan et al., 2012), but their use is limited by their safety and tolerability profile.
First-line agents: SSRIs and SNRIs
Meta-analyses (Andrisano et al., 2013; Bakker et al., 2002; Otto et al., 2001), systematic reviews (Batelaan et al., 2012) and RCTs support the use of SSRIs as first-line pharmacotherapy for panic disorder. Moderate effect sizes (0.41, NNT = 8; 95% CI = [6, 11]) have been reported for all SSRIs (Mitte, 2005b).
Several RCTs have demonstrated that the SNRI, extended-release venlafaxine, reduced the severity of panic disorder symptoms (Bradwejn et al., 2005; Liebowitz et al., 2009; Pollack et al., 1996a, 2007a, 2007b). SNRIs are recommended as alternative first-line agents on this basis. However, two of these studies observed no difference in the rates of panic-free patients in the treatment and placebo groups (Bradwejn et al., 2005; Liebowitz et al., 2009; Pollack et al., 2007a, 2007b).
Randomised fixed-dose studies have reported superior efficacy for some SSRIs and SNRIs (paroxetine, fluoxetine; Ballenger et al., 1998; Michelson et al., 1998) than others (citalopram, venlafaxine; Pollack et al., 2007a; Wade et al., 1997).
Long-term, open-label follow-up studies over 6 months to 3 years have demonstrated maintenance of improvements with continued use of citalopram (Dannon et al., 2007; Lepola et al., 2003), fluoxetine (Tiller et al., 1999), sertraline (Lepola et al., 2003), fluvoxamine and paroxetine (Dannon et al., 2007; Lecrubier et al., 1997; Nardi et al., 2012). Studies have also shown that there is an increase in overall treatment response with increased duration of treatment from 12 to 52 weeks (Ballenger et al., 1998; Batelaan et al., 2012).
Current evidence does not support the recommendation of one SSRI over another, or venlafaxine over an SSRI in terms of efficacy, onset of action, side effect profile or dropout rates.
Second-line agents: TCAs
Imipramine and clomipramine have demonstrated efficacy in panic disorder, with an effect size of 0.41 (NNT = 6; 95% CI = [5, 8]; Mitte, 2005b), which is similar to those seen with SSRIs. Other more noradrenergic TCAs have also been shown to be efficacious in acute treatment in some studies (Batelaan et al., 2012), although others have reported that the noradrenergic TCAs are less effective than SSRIs (Den Boer and Westenberg, 1988). However, SSRIs show superior tolerability (dropout rates for SSRIs 18% compared with 30% for TCAs; Bakker et al., 2002) and safety profile. Therefore, TCAs should be considered only when SSRIs are not effective or appropriate.
Long-term, open-label follow-up studies over 6 months to 3 years have demonstrated maintenance of improvements with both clomipramine (Lecrubier et al., 1997; Lepola et al., 1998) and imipramine (Curtis et al., 1993; Mavissakalian and Perel, 1992). They have also reported increased treatment response with increased duration of treatment (Lecrubier and Judge, 1997).
Starting and treatment doses for SSRIs, SNRIs and TCAs
Some people with panic disorder are very sensitive to medication side effects and anxiety exacerbation at treatment initiation (Mavissakalian and Perel, 1985; Zitrin et al., 1983). It is therefore recommended that starting doses of SSRIs, SNRIs and TCAs are given at half the usual daily dose for depression (Louie et al., 1993). In general, good-quality psychoeducation about the likely adverse effects of medication, reassurance that these symptoms are not dangerous and sufficient clinician support often renders adjunctive benzodiazepine treatment unnecessary at initiation of treatment.
Findings from fixed-dose, placebo-controlled studies suggest that higher daily doses may be superior to lower doses for some antidepressants such as paroxetine and fluoxetine (Ballenger et al., 1998; Michelson et al., 1998) but not for others such as citalopram and venlafaxine (Pollack et al., 2007b; Wade et al., 1997). However, the evidence to support dose escalation after an initial lack of response to lower doses is only limited (Michelson et al., 2001) or negative (Simon et al., 2009). Few studies have addressed the optimal dose of TCAs. Available studies suggest final daily doses of 150–300 mg for imipramine (Mavissakalian and Perel, 1995) and 25–150 mg for clomipramine (Cassano et al., 1988; Modigh et al., 1992). We recommend starting antidepressants at a low dose and increasing the dose as tolerated until a therapeutic response is achieved, but allowing for the slower onset of action. This may require doses at the higher end of the indicated range in some people (National Institute for Health and Care Excellence, 2011a).
Time to response with SSRIs, SNRIs and TCAs
Although there is lack of clear evidence for what constitutes an adequate trial of medication, consensus expert recommendation is to wait for at least 6 weeks with at least 2 weeks at the full dose (Lecrubier et al., 1997; National Institute for Health and Care Excellence, 2011a; Stein et al., 2009a), however the recently updated British Association of Psychopharmacology guidelines recommend 12 weeks (Baldwin et al., 2014).
Duration of treatment with SSRIs, SNRIs and TCAs
The optimal duration of treatment has not been well studied. Most guidelines refer to expert consensus recommendations and suggest continuation for at least 6 months (National Institute for Health and Care Excellence, 2011a) to a year (Bandelow et al., 2008; Landelijke Stuurgroep Multidisciplinaire Richtlijnontwikkeling (LSMRG), 2009). It is important that avoidance behaviour has been overcome before medications are withdrawn, to avoid an increased risk of relapse (Mavissakalian and Perel, 2002).
The decision to remain on long-term antidepressants needs to be carefully discussed with the patient, weighing up the risks and benefits. It may be that some people do need to remain on antidepressants for some years.
Stopping treatment with SSRIs, SNRIs and TCAs
When medication is discontinued, consensus advice is to taper medication down over weeks to months to reduce the risk of discontinuation symptoms (American Psychiatric Association, 2013; Baldwin et al., 2014; LSMRG, 2009). Better outcomes are achieved when medication is discontinued only after attaining remission of symptoms, rather than just improvement.
Relapse rates after discontinuation vary, with about a third of people relapsing within 6 months of discontinuation of antidepressant medication (Mavissakalian and Perel, 2002) and two-thirds over the course of 3 years (Toni et al., 2000). A greater number may relapse after discontinuation of benzodiazepines (Noyes et al., 1991).
Benzodiazepines
Benzodiazepines (alprazolam, clonazepam, diazepam, lorazepam) have been shown to be efficacious for the treatment of panic disorder (Batelaan et al., 2012; Mitte, 2005b) with an effect size of 0.40 (NNT = 5; 95% CI = [4, 7]) (Mitte, 2005b) – approximately equal to that of SSRIs and TCAs. Benzodiazepines have a rapid onset of action. Alprazolam is the most extensively studied of the benzodiazepines and was the benzodiazepine used in the Cross National Collaborative Panic Study (CNCPS Second Phase Investigators, 1992). It is effective in the treatment of panic disorder but is no longer recommended due to safety concerns, mainly a high risk of dependency, and difficulty discontinuing.
Despite their efficacy, benzodiazepines are not recommended as first-line treatment options, largely because of the risk of side effects (particularly sedation and cognitive impairment), tolerance and dependence (especially with alprazolam). Their use in combination with CBT also has potentially detrimental effects. Because of these concerns, recommendations are for benzodiazepines to be used short term and to be dosed regularly rather than ‘as required’ (Katzman et al., 2014; Stein et al., 2009).
They may also be infrequently required short term to reduce any exacerbation of anxiety associated with the initiation of SSRI treatment, which usually lasts from a few days to 2 weeks. Three controlled studies have shown that short-term (4–6 weeks) addition of benzodiazepines (alprazolam or clonazepam) to antidepressants produces a more rapid response, although patients on alprazolam had difficulty discontinuing (Goddard et al., 2001; Pollack et al., 2001; Woods et al., 1992).
Occasionally, benzodiazepines may be useful in an emergency setting for short-term management of severe agitation or anxiety and for the management of an acute panic attack.
They should not be used as a treatment for panic disorder in people with a history of substance use disorder.
Other medications
Traditional MAOIs have been shown to be effective in the treatment of panic disorder (Batelaan et al., 2012), but are not recommended as first-line antidepressants because of poor tolerability, the requirement for dietary restrictions, interactions and risks in overdose. When prescribing MAOIs, careful monitoring for adverse effects is needed, and a washout period is necessary if switching from an SSRI/SNRI.
Medications for which there is less empirical support include the antidepressants mirtazapine (Ribeiro et al., 2001), duloxetine (Simon et al., 2009), milnacipran (Blaya et al., 2007), moclobemide (Kruger and Dahl, 1999) and bupropion (Simon et al., 2003), and the anticonvulsants divalproex (Baetz and Bowen, 1998; Keck et al., 1993; Primeau et al., 1990; Woodman and Noyes, 1994), levetiracetam (Baetz and Bowen, 1998; Keck et al., 1993; Primeau et al., 1990; Woodman and Noyes, 1994) and gabapentin (Pande et al., 2000). There is limited evidence supporting the use of these medications either as monotherapies or adjunctive therapies. Their role may be limited to people who have failed to respond to standard treatments.
A Cochrane review of second-generation (atypical) antipsychotic agents (Depping et al., 2010) reported no clear benefits for their use in panic disorder.
Current evidence does not support the use of beta blockers or buspirone for panic disorder.
Combination of CBT and antidepressant medication
Meta-analyses and a systematic review found that combined treatment was no better than psychotherapy alone in long-term follow-up, although it was superior to pharmacotherapy alone (Furukawa et al., 2006; Mitte, 2005b). Combination treatment does not, therefore, appear to be significantly superior to standard monotherapies for most people. It may be useful where there is insufficient response to either CBT or pharmacotherapy alone, where it is difficult to conduct CBT because of the high level of anxiety or panic attacks, and where there is comorbidity. It may provide benefits in relapse prevention following combination therapy especially following medication withdrawal.
A controlled combination study (Barlow et al., 2000), which compared imipramine, CBT, placebo, the combination of CBT and placebo, and the combination of CBT and imipramine, reported equivalent outcomes at 3 months for CBT, imipramine, and the combination of CBT and imipramine, compared with placebo. At 12-month follow-up, after treatment withdrawal, CBT was found to have a greater response rate than CBT plus imipramine (Barlow et al., 2000).
In SSRI studies, the combination of paroxetine plus CBT and the combination of fluvoxamine plus exposure treatment were both superior to CBT plus placebo, and to exposure alone (de Beurs et al., 1995; Oehrberg et al., 1995). A study in primary care showed that CBT plus pharmacotherapy was more effective than pharmacotherapy alone (Craske et al., 2005; Roy-Byrne et al., 2005).
A systematic review of all controlled studies (23 in total, of which 21 involved behavioural therapy or CBT) compared combined treatment with psychotherapy alone or antidepressants alone (Furukawa et al., 2007). It reported advantages for combination treatment in the short term versus antidepressants (relative risk [RR] = 1.24; 95% CI = [1.02, 1.52]) and versus psychotherapy (RR = 1.17; 95% CI = [1.05, 1.31]). These benefits were sustained for as long as medication continued. However, after stopping medication, the advantage remained for combination treatment versus antidepressants alone (RR = 1.61; 95% CI = [1.23, 2.11]) but not for combination treatment versus psychotherapy (RR = 0.96; 95% CI = [0.79, 1.16]).
Several studies have reported that the combination of CBT and an SSRI was superior to medication alone (Bandelow et al., 2007; Cuijpers et al., 2014a; Roy-Byrne et al., 2005; Van Apeldoorn et al., 2013a) or CBT alone (Bandelow et al., 2007; Hofmann et al., 2009; Würz and Sungur, 2009) in the treatment of panic disorder. A review reported that the combination of CBT plus medication for panic disorder was superior to CBT alone in the short term but equivalent in the long term (Würz and Sungur, 2009).
A study that compared CBT alone, SSRI alone and combination of the two over 1 year and for 1 year after treatment discontinuation (van Apeldoorn et al., 2010) reported that patients treated with the SSRI (whether alone or with CBT) had faster treatment gains but no significant differences at 1-year follow-up. Those on SSRIs (again whether alone or in combination with CBT) showed maintenance of gains to the same extent as those treated with CBT at 1 year after discontinuation (van Apeldoorn et al., 2010). The same authors reported that for patients with moderate or severe agoraphobia, the combination of CBT plus SSRI was associated with more rapid improvement than either treatment alone (Van Apeldoorn et al., 2013).
The addition of CBT to medication has been shown to be beneficial in patients with panic disorder that are resistant to medication treatment alone (Heldt et al., 2003; Pollack et al., 1994). Adding in medication to patients unsuccessfully treated with CBT alone has also been found to be beneficial (Hoffart and Martinsen, 1993; Kampman et al., 2002).
Other treatment guidelines for panic disorder
NICE guidelines recommend the use of CBT over first-line pharmacotherapy and that pharmacological interventions should only be routinely offered to people who have not benefitted from psychological interventions (National Institute for Health and Care Excellence, 2011b, 2012).
Other guidelines, including Canadian clinical practice guidelines (Katzman et al., 2014), those by the American Psychiatric Association (Stein et al., 2009) and the British Association of Psychopharmacology (Baldwin et al., 2014), suggest that the choice of treatment (between CBT and pharmacotherapy) should be based on the person’s preferences, previous response to treatment, comorbidity and availability of treatment options.
Social anxiety disorder
Diagnosis
People with SAD are fearful of being negatively judged by others. They become anxious in personally relevant situations that may involve possible or actual scrutiny by other people, interaction with other people or performance before other people (Table 10).
Essential features of social anxiety disorder.

SAD: social anxiety disorder; DSM-5: Diagnostic and Statistical Manual of Mental Disorders, 5th edition; ICD-10: International Statistical Classification of Diseases and Related Health Problems, 10th revision.
For diagnostic criteria for SAD, refer to DSM-5 page 202 (American Psychiatric Association, 2013) and ICD-10 diagnostic criteria for research page 92 (World Health Organization, 1993).
The person’s underlying fear is of acting in a way that is seen as socially awkward or inappropriate, to the extent (in their mind) that other people might think badly of them or be offended.
The manner in which individuals with SAD fear that social inappropriateness followed by negative evaluation may happen is highly variable and depends on the specific underlying cognitions. Typical fears include showing anxiety (especially visible signs of anxiety such as blushing, sweating, shaking or appearing awkward) when under scrutiny or in the performance of some task (such as public speaking, but also eating, drinking, signing a document or using the telephone). Typically, people with SAD also fear making conversation with others, because they worry about saying something foolish or having nothing to say.
Natural history
Prevalence
Within Australia and New Zealand, it has been estimated that 8.4% of the population would meet criteria for SAD at some time in their life and that 4.2–6.8% of the population would meet criteria for SAD at some time over a 12-month period (McEvoy et al., 2011; Wells, 2006). SAD might be severe in 30–40% of these people, moderately severe in 35–50% and mild in about 20–30% (Kessler et al., 2005a; Slade et al., 2009a; Wells, 2006a).
Reported estimates of the lifetime prevalence of SAD in primary care settings have ranged from 12% to 30% (Demertzis and Craske, 2006).
Australian and New Zealand data indicate that women are affected at about 1.2–1.5 times the rate of men (Slade et al., 2009b). SAD is over-represented in separated, divorced and never-married people, and there is also an association with unemployment (Lampe et al., 2003).
In the 2007 Australian National Survey of Mental Health and Wellbeing, respondents who met criteria for SAD in the past 12 months reported an average of 4.7 days per month where they were unable to function in life roles or had to cut back on what they did; this compares to a mean of 6.4 days per month for major depression (Slade et al., 2009a).
Course and prognosis
Typically, SAD has its onset in adolescence. In Australia, the estimated median age at onset is 13 years, which represents a relatively early onset for an anxiety disorder and is much earlier than the median age of onset of mood disorders (McEvoy et al., 2011).
SAD is mostly a chronic disorder. Periods of remission are most common in milder, non-generalised SAD, in non-clinical samples, and in those with fewer social fears (Cox et al., 2011; Ruscio et al., 2008; Vriends et al., 2014). The US National Comorbidity Survey Replication estimated that only 20–40% of those with social phobia recover within 20 years of onset and only 40–60% recover within 40 years (Ruscio et al., 2008). The Harvard Brown Anxiety Disorders Research Programme found a 0.63 probability that participants had remained in the index episode of SAD 12 years after intake (Bruce et al., 2005).
Distress, disability and impairment
SAD causes a significant burden of disease that has been poorly recognised. It is associated with significant personal distress, disability and impairment, both directly and through the burden of comorbidity. The burden of disease is comparable to that of chronic physical disorders: a community study estimated a burden of disease for SAD that was intermediate between that of asthma and diabetes (Andrews et al., 1998).
The level of distress, disability and impairment increases with the number of social fears (Acarturk et al., 2008; Ruscio et al., 2008). Even subthreshold illness has been associated with impairment in social and occupational functioning (Fehm et al., 2008). Individuals with greater symptom severity at pre-treatment tend to show lower end-state functioning than those who begin treatment less impaired (Eskildsen et al., 2010). Thus, severity is relevant in informing treatment expectations.
Service use
There is frequently a long delay (9–28 years) between developing symptoms of SAD and seeking professional help (Green et al., 2012; Thompson et al., 2008), with a median of 16 years reported in the US National Comorbidity Survey Replication (Wang et al., 2005) and 28 years in Te Rau Hinengaro: The New Zealand Mental Health Survey (Oakley Browne et al., 2006; Wells, 2006). It is likely that delays are longer in rural and regional areas (Green et al., 2012).
Australian epidemiological data identified that about 62% of those with SAD had sought help at some time in the previous 12 months. The data suggested that many did not persevere with treatment: only about 21% were currently receiving treatment at the time of the survey. Only 30–40% received a treatment likely to be effective (Issakidis and Andrews, 2002; Issakidis et al., 2004). GPs are the most commonly consulted health practitioner.
Assessment
The aims of assessment are as follows:
To establish a good therapeutic relationship;
To establish a primary diagnosis of SAD;
To rule out differential diagnoses;
To identify comorbid disorders that may affect treatment and outcome;
To provide a foundation for treatment planning.
Comprehensive assessment includes obtaining information about all of the following:
The nature, severity and duration of symptoms, avoidance behaviours and use of safety behaviours;
The degree of distress and functional impairment;
The presence of substance use disorders and medical conditions;
The presence of comorbid depressive or anxiety disorders;
Personal and family history of mental disorders;
Experience of, and response to, past treatments;
The quality of interpersonal relationships, living conditions and employment;
The patient’s goals and expectations.
Differential diagnosis
Distinguishing SAD from normal social anxiety and shyness
Normal social anxiety and shyness can occur across a range of situations involving performance, scrutiny and interaction and is differentiated from SAD by the lack of persistence, generalisation, impairment and significant distress.
Shyness is common and appears to be somewhat heterogeneous in nature, showing some overlap with SAD, in that social fears and impairment in social performance may be present to an extent intermediate between SAD and non-shy, but shyness does not appear to be associated with comorbid psychiatric conditions or substance use disorders (Burstein et al., 2011; Heiser et al., 2009). In an epidemiological survey of youth in the United States, 46.7% of respondents self-identified as shy, whereas a lifetime diagnosis of SAD was assigned in 8.6% (12.4% of those who endorsed shyness; Burstein et al., 2011). The key to differentiating shyness from SAD is a careful assessment for social fears and to determine whether the individual meets the distress and impairment criteria for SAD. Instruments for assessment and differential diagnosis are listed in Appendix 1.
Distinguishing SAD from other mental disorders
SAD is most commonly confused with agoraphobia because of the overlap in feared situations, especially crowded situations in which escape would be difficult such as shops, public transport, cinemas and restaurants. The disorders can be differentiated by determining the principal underlying fear in each case: negative evaluation in SAD, and fear of coming to mental or physical harm in agoraphobia. A fear of embarrassment may be a factor in agoraphobia but is not the chief concern.
Major depression may present with social anxiety and loss of confidence. In such cases, the longitudinal history of distressing and impairing social evaluative concerns dating from adolescence will be absent.
Individuals with body dysmorphic disorder may present with social anxiety due to concerns that others will notice a body part perceived to be repugnant. It can be challenging to elicit these concerns due to high levels of shame that are commonly associated with such beliefs.
Occasionally, individuals with narcissistic personality traits may present with self-reported social anxiety. This is usually related to fears that in limited social interactions others may not appreciate the superiority of the person’s talents or abilities. The desire of the person with SAD to simply be ‘average’ and in fact to hide from scrutiny provides a useful contrast to assist in differentiating these disorders.
Avoidant personality disorder may be differentiated by the more globally negative self-view, and a debilitating fear of rejection that leads to extensive avoidance. It is both a differential diagnosis and also a commonly comorbid condition. Many researchers have questioned whether these disorders are variants of the same condition (Bogels et al., 2010), although there appear to be relevant clinical differences (Eikenaes et al., 2013; Rettew, 2000). Comorbid avoidant personality disorder is associated with slower response and with regard to outcome there are mixed findings. Some studies report less likelihood of remission from SAD (Fava et al., 2001a) and failure to reach normative levels of functioning (Alden, 1989; Eikenaes et al., 2006), but some have reported no effect on treatment outcome (Hope et al., 1995; Kamaradova et al., 2014; van Velzen et al., 1997).
GAD can include worries about social evaluation. Consider GAD in preference to SAD when worries cover a broader range than just social situations, centre on minor daily matters, are constant and intrusive, and a source of distress in and of themselves.
Eating disorders include many social concerns, and SAD will often include poor body image. Consider eating disorders when self-concept is integrally centred on body image, and when excessive activity focuses around weight and dietary control.
Comorbidity
People with SAD show high levels of comorbidity with other disorders (Ruscio et al., 2008). Rates of comorbidity with other anxiety disorders are especially high (50–60%), and there is very high overlap with depressive disorders (30–50%) and substance use disorders (20–40%; Andrews et al., 2002).
A European epidemiological study reported that major depressive disorders were found in 19.5% of participants with SAD (Ohayon and Schatzberg, 2010). Co-occurrence of another anxiety disorder was increased when a major depressive disorder was present (65.2%).
The likelihood of having comorbid anxiety and mood disorders, and suicidal ideation, increases with the number of social fears (El-Gabalawy et al., 2009; Ruscio et al., 2008). Comorbidity with alcohol use disorders does not appear to be related to the severity of SAD (El-Gabalawy et al., 2009; Fehm et al., 2008; Ruscio et al., 2008).
A comorbid diagnosis of avoidant personality disorder can be made in about one-third of individuals (Lampe and Sunderland, 2015) and shows considerable overlap in symptomatology and level of impairment. However, studies suggest that avoidant personality disorder may be associated with greater impairments in interpersonal functioning, self-identity and self-esteem, and greater attachment pathology (Lampe, 2016). Comorbid depression, avoidant personality disorder or GAD may be associated with worse outcome (Ansell et al., 2011; Blanco et al., 2011; Bruce et al., 2005; Cox et al., 2011; Ledley et al., 2005; Mululo et al., 2012).
SAD most commonly precedes comorbid disorders (Fehm et al., 2008), as would be expected from its relatively early age of onset. One study reported the odds of developing a major depressive episode 2 years after the first appearance of SAD at 5.74:1 (Ohayon and Schatzberg, 2010).
DSM-5 allows a diagnosis of SAD in the presence of medical conditions, if the anxiety is out of proportion to what would normally be expected. High rates of SAD are found among people who stutter (Craig and Tran, 2006; Iverach and Rapee, 2014). Medical conditions that include highly visible symptoms might be expected to increase risk for SAD, but the best evidence to date is for Parkinson’s disease, where high rates of comorbid SAD have been reported and are associated with depression and GAD (Kummer et al., 2008). Neural mechanisms common to both SAD and the underlying disorder cannot be excluded.
The presence of comorbidities is relevant to treatment selection and may affect prognosis. Patients with comorbid depression may not engage well with CBT (Ledley et al., 2005).
Screening questions
General screening questions for SAD include the following:
Do you worry about looking anxious or being embarrassed in social situations?
Do you worry a lot about what people think of you?
A number of freely available measures assess social evaluative concerns and social avoidance (Appendix 1). The Social Phobia Inventory (SPIN) was designed to provide a brief assessment of the fears and avoidance characteristic of SAD (Connor et al., 2000). It includes 17 items, with good psychometric properties. For rapid screening, the mini-SPIN comprises three items that can identify and distinguish SAD from non-clinical populations with 0.88 sensitivity and 0.98 specificity (Connor et al., 2001; Seeley-Wait et al., 2009).
Self-report questionnaires
Detailed assessment can be made using the Social Phobia Scale (SPS) and Social Interaction Anxiety Scale (SIAS) that together comprise 40 items covering fears of public scrutiny and social interactions (Mattick and Clarke, 1998; Peters, 2000). A short (12-item) form of these companion scales has recently also been developed (Peters et al., 2012). The Liebowitz Social Anxiety Scale (LSAS; Baker et al., 2002) includes both a reflection of social fear and a measure of avoidant behaviour (reflecting impairment). It is scored to provide a total score, but can also provide separate social interaction and performance subscales. Both clinician-administered and self-reported versions have been validated (Baker et al., 2002; Fresco et al., 2001). The Social Phobia and Anxiety Inventory (SPAI) provides a more comprehensive measure comprising over 45 items (Turner et al., 1989). Other instruments for assessment and monitoring are listed in Appendix 1.
Treatment
Overview
CBT and antidepressant pharmacotherapy with SSRIs, SNRIs or MAOIs have been demonstrated to be effective by a large body of level I evidence. There is also evidence that dCBT (accessed by computer, tablet or smartphone application) is effective. There is limited or lower quality evidence for other psychological therapies, other antidepressant classes and other medication classes.

SAD: social anxiety disorder; CBR: consensus-based recommendation; EBR: evidence-based recommendation; RCT: randomised controlled trial; CBT: cognitive–behavioural therapy; dCBT: digital cognitive–behavioural therapy; SSRI: selective serotonin reuptake inhibitor; SNRI: serotonin and noradrenaline reuptake inhibitor; MAOI: monoamine oxidase inhibitor.
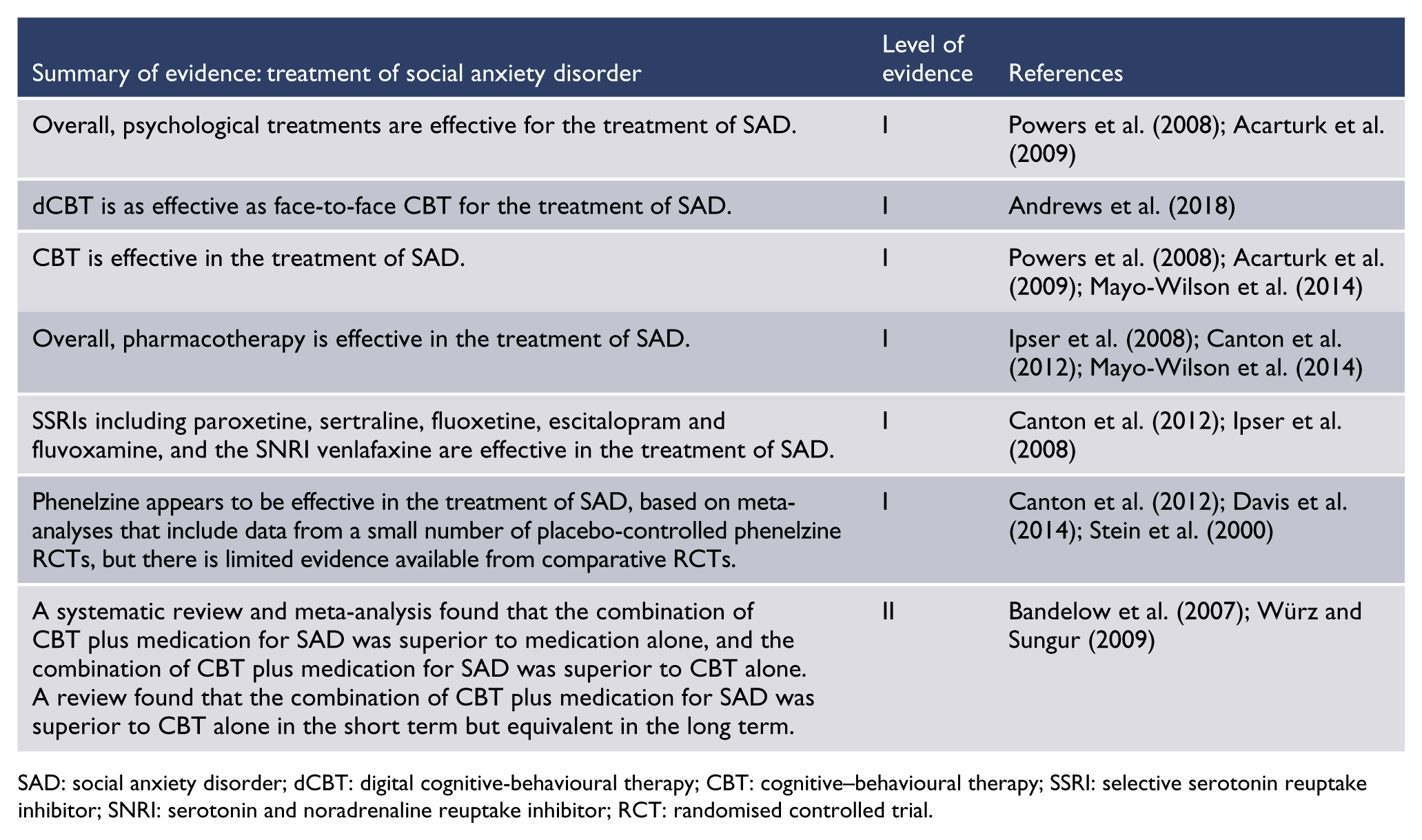
SAD: social anxiety disorder; dCBT: digital cognitive-behavioural therapy; CBT: cognitive–behavioural therapy; SSRI: selective serotonin reuptake inhibitor; SNRI: serotonin and noradrenaline reuptake inhibitor; RCT: randomised controlled trial.
A collaborative, pragmatic approach is recommended, beginning with psychoeducation and advice on lifestyle factors (see section ‘General issues in the recognition and management of anxiety disorders’), followed by specific treatment. Initial treatment options are CBT (face-to-face CBT with an expert clinician, usually a clinical psychologist; or guided dCBT), medication with an SSRI (or an SNRI if SSRIs are ineffective or are not tolerated) in combination with graded exposure to anxiety triggers, or a combination of CBT plus medication. Selection of initial treatment should take into account severity, patient preference, accessibility, cost, tolerability and safety.
An SSRI may be used instead of CBT or added to CBT for patients who have a comorbid depression interfering with their ability to engage with or implement CBT, or who have not had a satisfactory response to CBT. Alternatively, or in the case of inadequate response, venlafaxine or duloxetine can be used.
Benzodiazepines have a limited role for patients who have failed to respond to, or tolerate other therapies. Buspirone, antipsychotic agents and beta blockers should not be used.
Response to pharmacological treatment for SAD is relatively slow, as indicated by findings that antidepressants separate from placebo most convincingly at 12 weeks and that response rates continue to increase over follow-up periods extending to 6 months. Given the absence of a clear dose–response relationship, pharmacotherapy should be given at the minimum therapeutic dose for an initial period of 4–6 weeks before assessing response.
Overall efficacy of psychological treatments
Two meta-analyses have examined overall effect sizes for treatment of SAD with any psychological intervention (Acarturk et al., 2009; Powers et al., 2008). Overall effects are large when psychological treatments are compared with wait-list (effect size = 0.86; NNT = 2.19). As expected, effect sizes are markedly smaller when active treatments are compared with credible psychological placebo (effect size = 0.36–0.38; NNT = 4.42–5.00), although these effects are still significant and moderate.
The significant effects of treatment last in the longer term (typically 3–12 months), with reported effect sizes of 0.90 versus wait-list (NNT = 2.10) and 0.47 versus placebo (NNT = 3.85; Powers et al., 2008).
CBT for SAD
Structure and components
Typical CBT programmes (Table 11) address the physical, cognitive and behavioural symptoms of SAD and aim to prevent relapse in three stages. Traditional CBT programmes include psychoeducation, cognitive restructuring and in vivo exposure to a wide variety of social situations.
Components of CBT for social anxiety disorder.

CBT: cognitive–behavioural therapy.
The first stage includes psychoeducation (explaining about anxiety and the symptoms of SAD, formulation [or case conceptualisation]), treatment rationale, symptom monitoring and addressing factors that facilitate or hinder therapy. Motivational interviewing and education of the person’s family or members of their social support network should also be considered, and written information or links to reliable online information should be provided.
The second stage includes identifying and reducing cognitive symptoms through challenging unhelpful thinking, especially focused on reducing catastrophising around the consequences of negative evaluation, and recognition that negative evaluation is unlikely, and using behavioural experiments and in vivo exposure to test hypotheses, with the aim of reducing safety behaviours and avoidance.
The final stage is relapse prevention that includes identifying potential precipitants for setbacks, identifying the patient’s early warning signs and developing a plan to manage setbacks and prevent relapse.
In some programmes, training in social skills is also incorporated. In addition to these components, the newer CBT programmes based on theoretical models of SAD also include performance feedback (e.g. via video) to provide a more realistic appraisal of personal performance and abilities, in vivo exposure and cognitive restructuring to the consequences of being evaluated negatively, training in attention refocusing during social events, dropping of safety behaviours, and identification and modification of core negative schemas.
Efficacy
By far the majority of research on psychological therapies has evaluated the efficacy of CBT. Overall, CBT is efficacious in the reduction of SAD symptoms, both immediately following treatment (effect size = 0.61–1.19, NNT = 1.67–2.99; Acarturk et al., 2009; Fedoroff and Taylor, 2001; Gil et al., 2001; Mayo-Wilson et al., 2014; Powers et al., 2008) and longer term (effect size = 0.95, NNT = 2.01; Fedoroff and Taylor, 2001; Gil et al., 2001).
Many so-called ‘dismantling’ studies have compared different components of CBT, especially cognitive therapy, exposure and combined exposure plus cognitive therapy. However, the definitions and parameters of what is included within these highly overlapping aspects of CBT vary widely. Unsurprisingly, meta-analyses have failed to show consistent differences between these components.
One meta-analysis showed that applied relaxation (effect size = 0.51, NNT = 3.55) and social skills training (effect size = 0.64, NNT = 2.86) were associated with smaller effects than exposure plus cognitive restructuring (effect size = 0.84, NNT = 2.23; Fedoroff and Taylor, 2001). Another analysis showed that treatments that include some form of in vivo exposure are associated with slightly larger effects than treatments without exposure (exposure effect size = 0.79, NNT = 2.36; non-exposure effect size = 0.61, NNT = 2.99), although the difference was not statistically significant (Acarturk et al., 2009).
A more recent development in CBT delivery is the inclusion of additional treatment components based on theoretical models of the disorder (Clark and Wells, 1995; Rapee and Heimberg, 1997). In the largest and most comprehensive meta-analysis, individual CBT that was based on these theoretical models was associated with the largest effect sizes (effect size, intervention vs wait-list 1.56, NNT = 2.0). A meta-analysis of treatments for anxiety disorders (Bandelow et al., 2015) found that individual therapy was superior to group CBT for SAD. Group CBT based on theory also showed a somewhat larger effect (effect size = 1.10, NNT = 1.77) than group therapy based on more traditional CBT (effect size = 0.80–0.85, NNT = 2.21–2.34), although no statistical comparison could be conducted. Both individual and group CBT based on theory were significantly superior to psychological placebo (Mayo-Wilson et al., 2014).
Mode of delivery
Self-help CBT, either delivered over the internet or through printed materials, has also shown significant benefits for SAD (Cuijpers et al., 2009, Mayo-Wilson et al., 2014). However, effects have not been shown to be superior to psychological placebo (Mayo-Wilson et al., 2014). Effect sizes are generally slightly larger for therapist-assisted self-help (effect size = 0.86, NNT = 2.19) than for pure self-help (effect size = 0.75, NNT = 2.48).
A recent meta-analysis of 11 studies of guided dCBT versus wait-list or psychological placebo controls showed an effect size superiority of 0.92 (95% CI = [0.76, 1.0]; NNT = 2.07, mean adherence = 57%) and was compared to face-to-face CBT in 2 of the 11 studies and efficacy found to be comparable (Andrews et al., 2018).The benefits were evident at a mean follow-up of 9.8 months post treatment (Andrews et al., 2018).
Other psychological therapies
Considerably fewer controlled trials have evaluated psychological therapies other than CBT. Significant effects have been shown for psychodynamic therapy versus wait-list (effect size = 0.62, NNT = 2.96), although its effects are similar to those shown by psychological placebo (effect size = 0.63, NNT = 2.91; Mayo-Wilson et al., 2014).
Other psychological therapies, including mindfulness, supportive therapy and interpersonal therapy, have failed to show effects that are significantly better than wait-list (combined effect size = 0.36, NNT = 5.00; separate effect size = 0.26–0.43; Mayo-Wilson et al., 2014). In one meta-analysis, CBT programmes showed somewhat larger effect sizes (0.71, NNT = 2.60) than non-CBT programmes (0.55, NNT = 3.31), although this difference was not statistically significant (Acarturk et al., 2009). However, a more recent analysis showed that individual CBT was significantly more efficacious than psychodynamic therapy (effect size = 0.56, NNT = 3.25; Mayo-Wilson et al., 2014).
Pharmacological treatment
Pharmacotherapy has been extensively studied and shown to be efficacious in the treatment of SAD.
Before starting any medication, psychoeducation should be provided about anxiety and SAD symptoms, and advice given about lifestyle factors that may be contributing to or perpetuating symptoms. The timeframe for change should also be discussed as response is relatively slow, as indicated by findings that antidepressants separate from placebo most convincingly at 12 weeks and that response rates continue to increase over follow-up periods extending to 6 months. Potential adverse effects of medication should also be discussed carefully.
Throughout treatment, the patient should be encouraged to gradually increase exposure to feared situations.
Overall efficacy and safety
Numerous RCTs demonstrate the efficacy of pharmacological agents in the short-term (typically 12–14 weeks) treatment of SAD. Some studies have also demonstrated protection against relapse in the longer term: an analysis of four datasets (Stein et al., 2000) calculated the RR of relapse compared with placebo at 0.33 (95% CI = [0.22, 0.49]; NNT = 3.7–5.7).
Analysis of three trials that reported dropout rates (Stein et al., 2000) showed little difference between dropout rates among patients who received active treatment and those who received placebo (RR = 0.61; 95% CI = [0.27, 1.36]).
Effective medications not only reduced symptoms of social anxiety but also associated disability and comorbid depressive symptoms (Stein et al., 2000).
A systematic review of pharmacotherapy for SAD (Ipser et al., 2008) estimated the NNT for medication overall as 4.2 and the number needed to harm as 14.4. Differences in tolerability were noted between classes and individual agents (Ipser et al., 2008).
Comparative efficacy and safety
Studies were generally unable to draw conclusions about comparative efficacy between medications. A meta-analysis of double-blind placebo-controlled trials of SSRIs in the treatment of SAD (Hedges et al., 2007) found no differences between the medications studied, but there were relatively few studies permitting comparisons. A systematic review and meta-analysis (Hansen et al., 2008) found no differences in two studies of venlafaxine versus paroxetine or in one study comparing paroxetine against citalopram.
A systematic review of pharmacotherapy for SAD reported that the number needed to harm showed substantial variation between individual medicines and pharmacological classes: 87.2 for moclobemide, 83.1 for phenelzine, 14.9 for SSRIs, 10.1 for venlafaxine and 9.3 for brofaromine (Ipser et al., 2008).
A comparatively large number of trials have evaluated SSRIs (total n = 3677) and venlafaxine (total n = 1318) in the treatment of SAD. Two meta-analyses (Blanco et al., 2003; Canton et al., 2012) found stronger evidence of efficacy for the SSRIs than for phenelzine. Another systematic review and meta-analysis (Mayo-Wilson et al., 2014) concluded that there is no evidence of differential effectiveness between medication classes in the treatment of SAD.
The choice of pharmacological agent should be guided by the strength of the evidence, together with the tolerability and safety profile. Given the relative safety of SSRIs and SNRIs, tolerability for the individual patient is likely to be the most important factor guiding selection of a specific agent. Choice of agent if trials of SSRI and SNRI antidepressants have failed to show sufficient benefit should be guided by the weight of evidence, tolerability and safety. Throughout treatment, the patient should be encouraged to gradually increase exposure to feared situations that involve actions while being observed.
First-line agents: SSRIs and SNRIs
SSRIs show equivalent, but not superior, efficacy to phenelzine and venlafaxine in the treatment of SAD, but have a superior safety and/or tolerability profile (Blanco et al., 2003; Mayo-Wilson et al., 2014). SSRIs are also effective in treating comorbid depression (Stein et al., 2000). These findings support the recommendation of SSRIs as first-line pharmacological agents in the treatment of SAD.
Some authors recommended SNRIs as equal first-line agents; venlafaxine has been well studied in RCTs (Canton et al., 2012; Ipser et al., 2008) and a pilot study of duloxetine reported a positive finding (Simon et al., 2010).
A meta-analysis of second-generation antidepressants in the treatment of SAD (de Menezes et al., 2011), in which three quarters of the included studies evaluated SSRIs, reported that active treatment overall was associated with a 62% increase in treatment response (defined as final CGI <2), compared with placebo (RR = 1.62; 95% CI = [1.44, 1.81]). Post-treatment SMDs on various outcome measures were calculated for paroxetine (–0.31 to –0.63; NNT = 2.9–5.7), fluvoxamine (–0.28 to –0.67; NNT = 2.7–6.4), sertraline (–0.29 to –0.85; NNT = 2.2–6.1), fluoxetine (0.03 to –0.57; NNT ⩾3.1), and venlafaxine (–0.40 to –0.63; NNT = 2.9–4.5). The combined SMD for the second-generation antidepressants was –0.43 (95% CI = [–0.49, –0.37]; NNT = 4.2). Reliable conclusions could not be drawn about relative efficacy between the agents.
A systematic review and meta-analysis (Canton et al., 2012) reported efficacy for paroxetine (odds ratio [OR] = 3.43; 95% CI = [2.51, 4.69]; NNT = 2.97–11.6), sertraline (OR = 2.48; 95% CI = [1.82, 3.37]; NNT = 8.2–10.6), escitalopram (OR = 2.0; 95% CI = [1.47, 2.86]; NNT = 12.2–14.1), fluoxetine (OR = 1.98; 95% CI = [1.0, 3.67]; NNT = 10.3–18.2) and fluvoxamine (OR = 2.73; 95% CI = [1.67, 4.48]; NNT = 4.0–9.1) in the treatment of SAD.
Current evidence does not support differences in efficacy between individual SSRIs for the treatment of SAD.
MAOIs
Meta-analyses that included trials of phenelzine generally conclude that it may be the most effective agent in treating SAD, with the highest effect sizes or SMDs against placebo (Canton et al., 2012; Davis et al., 2014; Stein et al., 2000). However, data are available from only a few clinical trials of phenelzine (Canton et al., 2012), and the findings are heavily influenced by one study.
Despite evidence of efficacy, phenelzine is not recommended as first-line treatment due to its considerable burden of adverse effects, difficulty in use due to strict dietary limitations and potential for toxicity through food and drug interactions and in overdose. When prescribing MAOIs, careful monitoring for adverse effects is needed, and a washout period is necessary if switching from an SSRI/SNRI.
Dose–response relationship with antidepressants
A meta-analysis of placebo-controlled RCTs assessing the efficacy of SSRIs in SAD (Hedges et al., 2007) reported that there was no evidence for a dose–response relationship in studies that compared different doses, but the authors concluded that the answer was not yet clear. Guidelines by the British Association for Psychopharmacology (Baldwin et al., 2014) advised that fixed-dose RCTs did not provide convincing evidence for a dose–response relationship in treating SAD.
Duration of treatment with antidepressants
A large body of evidence from RCTs suggests that non-responders at 8–12 weeks may become responders with continuation of the same medication over 6–12 months (Ipser et al., 2008; Lader et al., 2004; Stein et al., 2003, 2005; Versiani et al., 1997).
A systematic review and meta-analysis (Canton et al., 2012) concluded that the effect of SSRIs generally separated from placebo by weeks 4–6 on a number of response or other outcome measures and that SSRI-placebo differences tended to increase out to 12 weeks of treatment (the limit of most studies).
European College of Neuropsychopharmacology guidelines for the investigation of efficacy in SAD (Montgomery et al., 2004) recommended a 12-week trial of medication, on the basis that drug–placebo difference is most likely to be seen at this point and a number of patients who are non-responders at 8 weeks become responders at 12 weeks. A systematic review of pharmacotherapy for SAD (Ipser et al., 2008) and the updated British Association of Pharmacotherapy guidelines (Baldwin et al., 2014) also recommended 12 weeks as representing a trial of treatment.
There are no studies that provide direct evidence to guide the duration of maintenance treatment or to demonstrate whether long-term treatment (e.g. greater than 6 months) reduces the relapse rate. However, a long-term trial of moclobemide, in which there was a good response rate, demonstrated high rates of symptom recurrence when the medication was discontinued after 12–20 months of treatment (Versiani et al., 1997).
Few guidelines have addressed the question of treatment duration specific to SAD; where this has been done, an advantage for staying on medication for up to 6 months has been reported (Baldwin et al., 2014) and a treatment period of 12–24 months has been recommended (Canadian Psychiatric Association, 2006). A systematic review of pharmacotherapy for SAD (Ipser et al., 2008) reported studies showing continued improvements over 12 months and recommended that treatment continue for this period.
Other medications
Moclobemide, a RIMA, appears to be only modestly efficacious in the treatment of SAD. It has been reported to be less effective than venlafaxine (effect size = 0.23, NNT = 7.7), phenelzine and paroxetine (Davis et al., 2014). Moclobemide was not more effective than placebo in one meta-analysis (Bandelow et al., 2015). Two meta-analyses – Stein et al. (2000) and Blanco et al. (2003) – found that SSRIs were significantly more effective than RIMAs, including moclobemide and brofaromine. Another meta-analysis (Canton et al., 2012) reported combined ORs for studies of moclobemide and brofaromine of 2.96 (95% CI = [1.78, 4.91]); for moclobemide alone it was 1.95 (95% CI = [1.37, 2.79]), with significant heterogeneity, and the authors conclude that moclobemide appears to be only modestly effective compared with the SSRIs and SNRIs. Corresponding NNTs ranged from 1.9 to 41.6 for moclobemide (six studies) and 2.4–5.8 for brofaromine (three studies). Brofaromine is not available in Australia or New Zealand. The benzodiazepines clonazepam, bromazepam and alprazolam were all reported to show superiority over placebo in the treatment of SAD in a meta-analysis that included one trial of each agent (Canton et al., 2012). One study of clonazepam estimated an effect size of 0.97 (95% CI = [0.49, 1.44]; NNT = 1.97; Davidson et al., 1993). A meta-analysis that included five trials (one with alprazolam and five with clonazepam) estimated a combined SMD of –0.96 (–1.56 to –0.36; Mayo-Wilson et al., 2014). However, the potential for cognitive impairment, for falls in older people, additive central nervous system depressant effects, and dependence, together with a lack of efficacy against depression, a commonly comorbid condition, place benzodiazepines as medications of last resort.
Mirtazapine (a tetracyclic antidepressant) for the treatment of SAD has been evaluated in three studies. One reported an estimated effect size against placebo of 0.13 (NNT = 13.5; Davis et al., 2014) and another reported an SMD of –0.41 (NNT = 4.3; de Menezes et al., 2011). The third study reported no benefit versus placebo (Canton et al., 2012). A systematic review and meta-analysis (Mayo-Wilson et al., 2014) concluded that mirtazapine was not superior to wait-list.
Anticonvulsants were superior to placebo in the treatment of SAD in a small number of studies. One meta-analysis reported that pregabalin was not superior to placebo (Davis et al., 2014). Another systematic review (Canton et al., 2012) reported three positive RCTs of alpha-2 delta ligands (pregabalin and gabapentin), with a combined OR of 3.11 [1.92, 5.04]; NNT = 6.8–8.3. The onset of anxiolytic effects was reported to occur within the first week of treatment. Dose–response has only been formally assessed for pregabalin, and efficacy is only evident at the maximum dose (600 mg/day), in contrast to its use in GAD. Another meta-analysis (Mayo-Wilson et al., 2014) estimated the combined SMD as –0.81 (–1.36 to –0.28) for five trials of anticonvulsants (one each for gabapentin and levetiracetam, and three studies of pregabalin).
It is not possible to draw conclusions about the efficacy of antipsychotics in the treatment of SAD. One RCT each for olanzapine and quetiapine were reported in a systematic review (Canton et al., 2012), but participant numbers were very small.
Beta blockers, such as atenolol and propranolol, are widely prescribed for social anxiety. They are not anxiolytic but decrease tremor and heart rate through reduction of sympathetic tone. It was originally hoped that this would reduce social anxiety. However, studies have failed to show superiority over placebo (Steenen et al., 2016). This should in fact not be surprising, since fears in SAD are usually more general than a focus on tremor or heart rate. Beta blocker medications should not be prescribed in SAD.
Combination of CBT and antidepressant medication
Some studies have reported that the combination of CBT plus medication was superior to either medication alone or CBT alone in the treatment of SAD (Bandelow et al., 2007; Canton et al., 2012). However, one study reported that the combination of CBT plus medication for SAD was superior to CBT alone in the short term, but equivalent in the long term (Würz and Sungur, 2009).
Five trials comparing medication plus CBT with medication alone, or with CBT alone, were reported in a systematic review (Canton et al., 2012). The combination treatment was usually superior to the single treatment, but the difference was only significant in a quarter of the studies (Canton et al., 2012).
A meta-analysis (Bandelow et al., 2015) found that combined CBT and medication was more effective than CBT alone and that pharmacotherapy was associated with higher pre–post effect sizes than CBT. Another systematic review and meta-analysis (Mayo-Wilson et al., 2014) identified only five trials evaluating the combination of CBT and medication. It reported a similar effect size across these trials (SMD = –1.30) compared with individual CBT (SMD = –1.19), which was slightly greater than the effects of SSRIs or SNRIs (SMD = –0.91).
Treatments under investigation
Anticonvulsants,
Repetitive transcranial magnetic stimulation has been trialled in OCD and PTSD, and more recently, there is a case report of its use in SAD, with the authors reporting symptomatic improvement in two patients and suggesting further study could be warranted (Paes et al., 2013).
Other treatment guidelines for SAD
NICE guidelines (National Institute for Health and Care Excellence, 2013) recommend CBT as first-line, and SSRIs (escitalopram or sertraline) if CBT is ineffective or declined. Canadian Psychiatric Association clinical practice guidelines for anxiety disorders (Canadian Psychiatric Association, 2006) list CBT, SSRIs and venlafaxine equally as first-line treatments. The British Association of Psychopharmacology guidelines (Baldwin et al., 2014) recommended SSRIs as first-line agents, advising that 12 weeks should be allowed as a trial period, and recommending against routine use of higher doses. The World Federation of Societies of Biological Psychiatry guidelines for the pharmacological treatment of anxiety, obsessive-compulsive and post-traumatic stress disorders, first revision (Bandelow et al., 2008) recommend SSRIs (escitalopram, fluvoxamine, paroxetine and sertraline) and the SNRI venlafaxine and suggest buspirone augmentation in the case of treatment resistance.
Generalised anxiety disorder
Diagnosis
All people worry about things that have a possibility of going wrong, but can temporarily dismiss their worries so that they can get on with the task in hand. People with GAD (Table 12):
Worry excessively over a succession of everyday things – some within their control and some not (e.g. relationships, family, finances, work, study, illness, community and world affairs), and worry excessively about things within their control going wrong in catastrophic and improbable ways.
Are troubled by persistent restlessness, a feeling of being on edge or muscle tension.
Essential features of generalised anxiety disorder.

For diagnostic criteria for GAD, refer to DSM-5 page 222 (American Psychiatric Association, 2013) and ICD-10 diagnostic criteria for research page 95 (World Health Organization, 1993).
GAD: generalised anxiety disorder; DSM-5: Diagnostic and Statistical Manual of Mental Disorders, 5th edition; ICD-10: International Statistical Classification of Diseases and Related Health Problems, 10th revision.
People with GAD seek reassurance to reduce immediate worry, avoid events that could have negative consequences and spend considerable time preparing for events to minimise worry (Andrews et al., 2010b, 2016; Beesdo-Baum et al., 2012).
The art in making the diagnosis of GAD is to shift the focus from the problem being worried about today, to the act of worrying. The question ‘Do you think you are a worrier by nature?’ often leads to a discussion of how worrying – not today’s target for worry – is the core issue.
Natural history
Prevalence
Within Australia and New Zealand, around 1 in 20 adults will experience GAD in their lifetime, with an estimated lifetime prevalence of 6% (Andrews et al., 2001; Grant et al., 2005; Hunt et al., 2002; McEvoy et al., 2011; Oakley Browne et al., 2006). Approximately half of these adults report that they have experienced GAD in the past year, an indication of how chronic the condition is (Andrews et al., 2001; Hunt et al., 2002; Oakley Browne et al., 2006; Slade et al., 2009a).
GAD, like the other anxiety disorders, is two to three times more likely in women than men (Carter et al., 2001; Comer et al., 2011; Grant et al., 2005; Maier et al., 2000; Oakley Browne et al., 2006; Teesson et al., 2011; Wells, 2006) and more common in people who are separated, divorced or widowed, less educated or unemployed (Hunt et al., 2002).
Course and prognosis
The prevalence of GAD as currently defined peaks in middle age, after a relatively late mean age of first onset in the early thirties (Byers et al., 2010; Carter et al., 2001; Grant et al., 2005; Hobbs et al., 2014; Hunt et al., 2002; Kessler et al., 2005a; McEvoy et al., 2011; Mackenzie et al., 2011). Most people who become disabled and distressed by their chronic worrying, and so meet criteria for GAD for the first time, have always been nervous and anxious about their immediate situation.
GAD is a chronic disorder which can be exacerbated by stressful life events. Most patients are still affected after 10 years and half of those who remit will relapse (Holaway et al., 2006; Yonkers et al., 2000).
Distress, disability and impairment
GAD is associated with functional, occupational and quality of life deficits, and substantial economic costs due to decreased work productivity and increased health care utilisation costs (National Institute for Health and Care Excellence, 2011a; Olatunji et al., 2007; Revicki et al., 2012; Sherbourne et al., 2010; Wittchen, 2002; Zhu et al., 2009).
The threshold for the diagnosis in terms of scores on the World Health Organization’s Disability Assessment Schedule indicates that the disability associated with GAD is comparable to that associated with major depressive disorder (Andrews et al., 2010c).
Service use
GAD is frequently under-recognised, with less than one-third of patients receiving adequate treatment (Baldwin et al., 2012; Hunt et al., 2002; Revicki et al., 2012). Less than half of the people who experience GAD seek treatment. When they do, it is mostly through primary care rather than specialised mental health services (Andrews et al., 2001, 2016; Hunt et al., 2002).
While GAD is the most common anxiety disorder in primary care (Maier et al., 2000; Üstün and Sartorius, 1995; Wittchen, 2002), those who seek treatment only do so an average of 10 years after the onset (Wang et al., 2005). Even then, treatment tends to be sought for comorbid somatic, panic, or depressive disorders or painful physical symptoms (Campayo et al., 2012; Judd et al., 1998; Kessler and Wittchen, 2002). For these reasons, systematic assessment of GAD in primary care is important.
Assessment
The aims of assessment are as follows:
To establish a good therapeutic relationship;
To establish a primary diagnosis of GAD;
To rule out differential diagnoses;
To identify comorbid disorders that may affect treatment and outcome;
To provide a foundation for treatment planning.
Comprehensive assessment includes obtaining information about all of the following:
The severity and duration of worry;
The degree of distress and functional impairment;
Substance use disorders and medical conditions;
Comorbid depressive or anxiety disorders;
Personal and family history of mental disorders;
Experience of, and response to, past treatments;
The quality of interpersonal relationships, living conditions and employment;
The patient’s goals and expectations of treatment.
Differential diagnosis
Distinguishing GAD from non-pathological worry
Several features of the worries distinguish GAD:
The amount of worry is more frequent, more extreme and out of keeping with the threat posed by the adverse event, should it occur. People with GAD report spending much more time per day worrying than non-clinical populations (Andrews et al., 2016) and, although many people worry when there is a problem, people with GAD worry about the future even when things are going well (e.g. worrying about the health of children even when they are not sick).
The worry is highly pervasive, pronounced, difficult to control and frequently occurs without precipitant (e.g. patients’ report that the worry is intrusive, hard to stop, comes into their minds when they want to concentrate on other things).
The history of excessive worry has a long duration (years rather than hours or days).
The worrying impacts on the patient’s quality of life; it is distressing or disabling (e.g. it is associated with muscle tension, impaired sleep, relationship difficulties and reduced work productivity).
Distinguishing GAD from other mental disorders
Excessive substance use or withdrawal (including caffeine) and medical conditions (such as hyperthyroidism) should be excluded. If a patient’s worry is focused on a single concern, then other diagnoses may be more appropriate.
Patients with OCD describe repetitive, unwanted intrusive thoughts, images and impulses that tend to have a circumscribed and consistent focus on harm, and/or contamination (obsessions), as well as repetitive ritualistic behaviours (compulsions). In contrast, GAD worries are typically internally congruent, self-initiated, relate to everyday events and are predominantly verbal/linguistic (American Psychiatric Association, 2013).
Patients with major depressive disorder and GAD often report low mood as primary, and worry as secondary, whereas patients with GAD and no comorbid major depressive disorder are more likely to feel predominantly anxious, with low mood as a secondary feature. They report future-oriented cognitions (i.e. ‘What if …’) and endorse GAD symptoms (e.g. muscle tension), whereas depressed patients report persistent low mood and anhedonia as primary difficulties, with past-oriented rumination (e.g. ‘Why did I allow that to happen?’) and major depressive disorder symptoms such as sadness, loss of interest and worthlessness (Andrews et al., 2016; Papageorgiou and Wells, 1999; Watkins et al., 2005).
Key cognitive symptoms and measures to aid in diagnosis are listed in Appendix 1.
Screening questions
General screening questions for GAD include the following:
Do you think you are a worrier by nature?
If yes, ask: When you do worry, what is the worst thing that can happen?
Self-report questionnaires
Two free self-report symptom measures can assist with immediate and accurate assessment and diagnosis (Table 13). The Penn State Worry Questionnaire-3 (PSWQ-3; Berle et al., 2011) is a 3-item measure of worry severity and the Generalized Anxiety Disorder-7 (GAD-7; Spitzer et al., 2006) is a 7-item screener for GAD symptoms.
Self-report measures for generalised anxiety disorder.
Sources: Meyer et al. (1990), Spitzer et al. (2006), Zimmerman (2016) and SAMHSA-HRSA Center for Integrated Health Solutions.
PSWQ-3: Penn State Worry Questionnaire-3; GAD-7: Generalized Anxiety Disorder-7.
Due to the high comorbidity of GAD and depression and the negative outcomes associated with having a comorbid depression diagnosis, all patients with suspected GAD diagnosis should also be screened for depression; the Patient Health Questionnaire-9 (PHQ-9) is useful (Kroenke et al., 2001). A patient’s response to item 9 on the PHQ-9 indicates suicide risk.
Comorbidity
GAD is comorbid with other disorders more frequently than not (Brown and Barlow, 1992; Brown et al., 2001; Hunt et al., 2002). There are high rates of comorbidity between GAD and other anxiety and depressive disorders (Andrews et al., 2002; Brown et al., 2001; Comer et al., 2011).
The presence of comorbid anxiety and depressive disorders should be routinely assessed as comorbidity increases the severity of the illness, functional impairment and economic costs (Andrews et al., 2002; Moffitt et al., 2007; Tyrer et al., 2004; Zhu et al., 2009). The risk of comorbid medical conditions in GAD is also elevated including pain syndromes, hypertension, cardiovascular and gastric conditions (Comer et al., 2011; Teesson et al., 2011). While medical comorbidity is associated with more severe anxiety symptoms and anxiety-related disability, the effectiveness of anxiety interventions is not significantly affected by the presence of multiple medical comorbidities, with the exception of migraine sufferers who display less improvement at long-term follow-up (Campbell-Sills et al., 2013).
Overall, medical comorbidity across anxiety disorders is not associated with poorer outcomes (Olatunji et al., 2010), and there is evidence that people with and without comorbid disorders respond similarly to face-to-face CBT and dCBT (Johnston et al., 2013; McEvoy and Nathan, 2007; Norton et al., 2008).
Treatment
Overview
Education about the nature and treatment of GAD is advised for all presentations of GAD. Patients should take an active interest in monitoring their improvement using a questionnaire-based assessment instrument (e.g. GAD-7 or PSWQ-3) and should be involved in the development of a treatment plan.
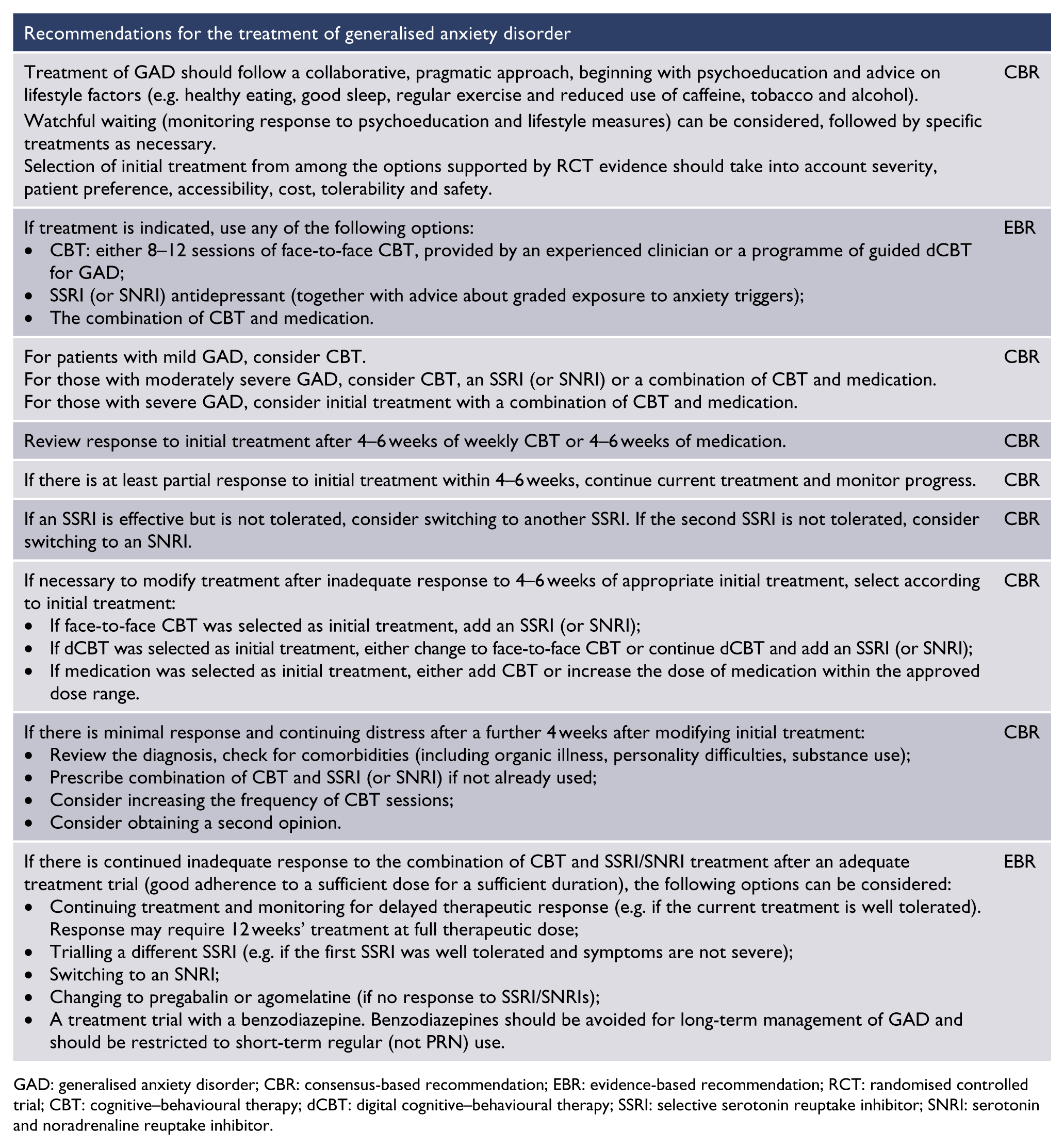
GAD: generalised anxiety disorder; CBR: consensus-based recommendation; EBR: evidence-based recommendation; RCT: randomised controlled trial; CBT: cognitive–behavioural therapy; dCBT: digital cognitive–behavioural therapy; SSRI: selective serotonin reuptake inhibitor; SNRI: serotonin and noradrenaline reuptake inhibitor.
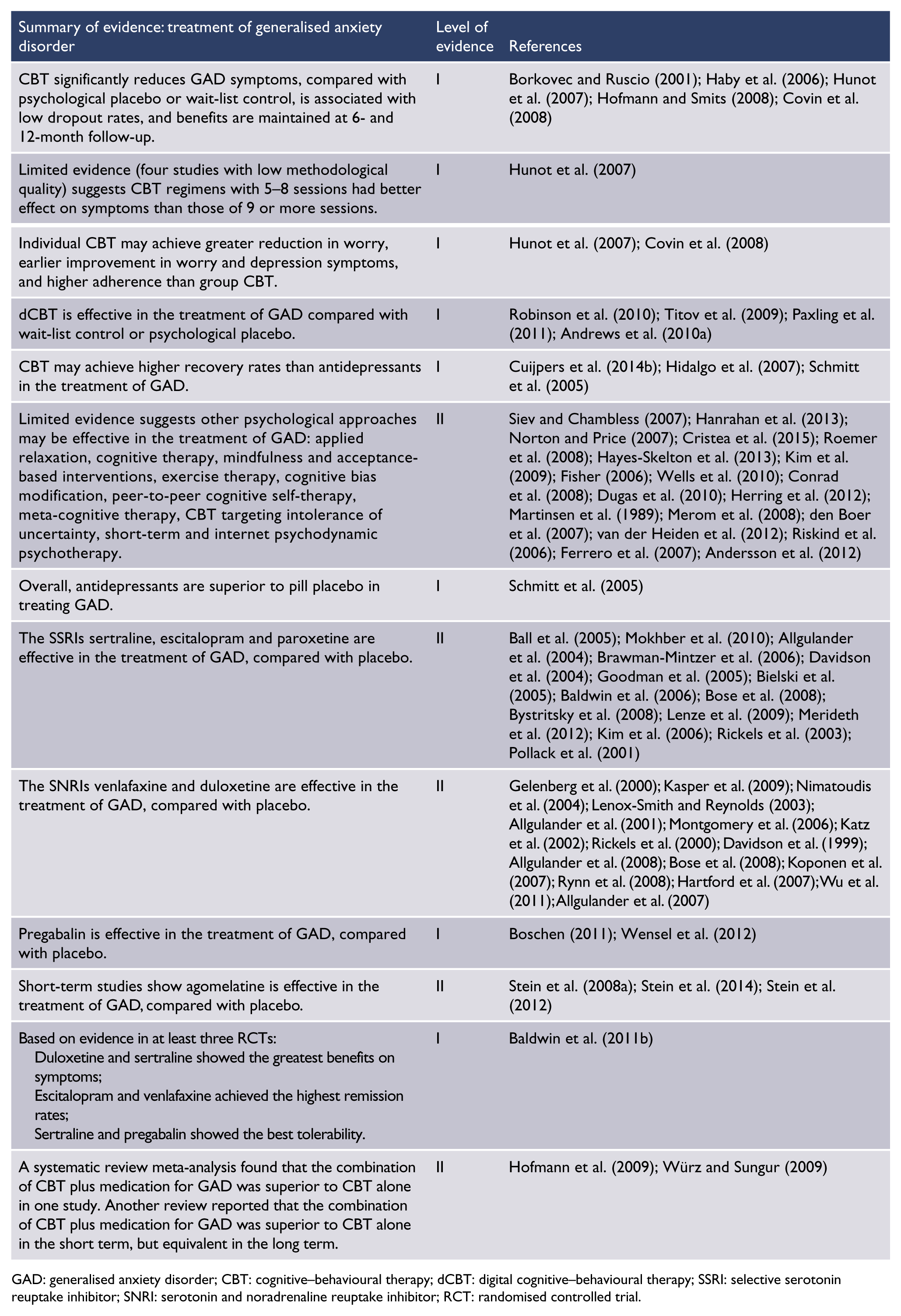
GAD: generalised anxiety disorder; CBT: cognitive–behavioural therapy; dCBT: digital cognitive–behavioural therapy; SSRI: selective serotonin reuptake inhibitor; SNRI: serotonin and noradrenaline reuptake inhibitor; RCT: randomised controlled trial.
CBT (including face-to-face CBT and dCBT) and antidepressant pharmacotherapy with SSRIs or SNRIs have been demonstrated to be effective by a large body of evidence. There is limited or low-quality evidence for other psychological therapies, other antidepressant classes and other medication classes.
A collaborative, pragmatic approach is recommended, beginning with psychoeducation and advice on lifestyle factors (see section ‘General issues in the recognition and management of anxiety disorders’), followed by specific treatment. Initial treatment options are CBT (face-to-face CBT with an expert clinician, usually a clinical psychologist; or guided dCBT), medication with an SSRI (or an SNRI if SSRIs are ineffective or are not tolerated) in combination with graded exposure to anxiety triggers, or a combination of CBT plus medication. Selection of initial treatment should take into account severity, patient preference, accessibility, cost, tolerability and safety.
An SSRI may be used instead of CBT or added to CBT for patients who have not engaged in CBT. The combination of an SSRI and CBT (face-to-face or dCBT) should be considered for patients with severe GAD (i.e. patients with a score of 15–21 on GAD-7 or patients that report extreme distress and/or functional impairment due to worrying).
SSRIs, specifically sertraline, are recommended as the first-choice pharmacological treatment and third-line treatment after dCBT and face-to-face CBT (National Institute for Health and Care Excellence, 2011a). Alternatively, or in the case of inadequate response, venlafaxine or duloxetine can be used.
Agomelatine and pregabalin have efficacy in GAD and can be considered if SSRIs or SNRIs are not tolerated or are ineffective. Agomelatine is not registered for use in Australia for the treatment of GAD. Agomelatine is not available in New Zealand. Benzodiazepines and atypical antipsychotics are not recommended in the treatment of GAD (National Institute for Health and Care Excellence, 2014). Buspirone and imipramine are effective, but have significant side effects and should be reserved for situations where standard care is ineffective. Beta blockers should not be used.
Response to antidepressant treatment for GAD is relatively slow, as indicated by findings that antidepressants separate from placebo most convincingly at 12 weeks and that response rates continue to increase over follow-up periods extending to 6 months.
It is recommended that antidepressant medication be started at a low dose (half the normal daily dose), as patients may be overly sensitive to adverse effects or experience an initial increase in anxiety/agitation, which could compromise adherence. The dose can then be slowly increased. Given the absence of a clear dose–response relationship, pharmacotherapy should be given at the minimum therapeutic dose for an initial period of 4 weeks before assessing response.
Overall efficacy of psychological treatments
A meta-analysis of 41 studies (total n = 2132) that examined the efficacy of various types of psychological therapies (mostly CBT) for GAD (Cuijpers et al., 2014b) reported a large pooled effect size when comparing all psychotherapy studies for GAD versus control condition (e.g. wait-list or active control) for symptoms of anxiety (effect size = 0.84, NNT = 2.23), worry (effect size = 0.95, NNT = 2.01) and depression (effect size = 0.71, NNT = 2.60). Overall, the effect size for psychological treatment corresponded to an NNT of approximately 2 (Cuijpers et al., 2014b).
Large effects were found for the individual variants of psychotherapy: dCBT (effect size = 1.05, NNT = 1.85), face-to-face CBT (effect size = 0.90, NNT = 2.10) and applied relaxation (effect size = 0.86, NNT = 2.19), with a medium effect size for behavioural only psychotherapy (effect size = 0.57, NNT = 3.18; Cuijpers et al., 2014b).
The number of studies comparing CBT with other psychotherapies (e.g. applied relaxation) was too small to draw conclusions about comparative effectiveness. However, there was initial evidence (N = 4; OR = 3.00; I2 = 0) that CBT was more effective than applied relaxation in the longer term (12-month follow-up) thus is preferable over applied relaxation as the first-line treatment of GAD (Cuijpers et al., 2014b).
CBT was more efficacious than other psychotherapies (behaviour therapy, cognitive therapy, psychodynamic therapy, supportive therapy). However, confidence in these psychotherapy comparisons is limited due to a lack of replicated RCTs and substantial heterogeneity in studies (Borkovec and Ruscio, 2001; Cuijpers et al., 2014b; Hunot et al., 2007).
CBT for GAD
Structure and components
CBT programmes address the physical, cognitive and behavioural symptoms of GAD and aim to prevent relapse. Typical CBT programmes (face-to-face or dCBT) include three stages (Andrews et al., 2014, 2016) and a range of components (Table 14).
Components of CBT for generalised anxiety disorder.

CBT: cognitive–behavioural therapy.
The first stage includes assisting the patient to notice their habitual worrying (e.g. ‘Last week you were worrying about that, and this week you are worrying about this – do you think you are a worrier by nature?’), formulation (or case conceptualisation), educating about the nature of GAD with treatment rationale, symptom monitoring (using GAD-7) and addressing factors that facilitate or hinder therapy (motivational interviewing and education of family members/spouses may be helpful).
The second stage includes reducing:
Physical symptoms – arousal reduction such as progressive muscle relaxation, breathing training and cardio exercise (30 minutes, three times a week) and management of misuse of alcohol and other substances (as with other anxiety disorders).
Cognitive symptoms – challenge unhelpful thinking such as overestimation of the likelihood of something bad happening (mistaking possibility with probability), underestimation of ability to cope, intolerance of uncertainty, beliefs around the benefit of worrying and the process of worrying about worrying. Use structured problem-solving to convert worries about stressors into identifiable, quantifiable and solvable problems with action plans.
Behavioural avoidance – encourage patients to gradually confront situations and activities they avoid by using graded exposure (e.g. completing activities without over-preparing and reducing reassurance-seeking) and increase engagement in activities that are unplanned.
The final stage targets relapse prevention. It includes identifying health maintenance strategies and early warning signs, making a plan to follow if warning signs appear in the future, and identifying future goals.
CBT can be delivered digitally (accessed by computer, tablet or smartphone application) or by a trained professional. It can be adapted to individual considerations of the patient (e.g. culture and age) with appropriate supervision as needed.
Efficacy
Several systematic reviews and meta-analyses have demonstrated that CBT significantly reduces GAD symptoms, is markedly more effective than placebo or wait-list control, is associated with low dropout rates and with benefits maintained at 6- and 12-month follow-up (Borkovec and Ruscio, 2001; Covin et al., 2008; Haby et al., 2006; Hofmann and Smits, 2008; Hunot et al., 2007).
A meta-analysis of 13 studies found that the magnitude of effect on symptoms was better for CBT regimens with five to eight sessions, versus those of nine or more sessions. However, this finding was based on a small number of studies (n = 4) with low methodological quality limiting confidence in this finding (Hunot et al., 2007).
Individual and group therapy are equally effective in terms of anxiety symptom reduction and long-term effects. However, individual therapy may achieve larger gains in worry reduction (pre–post treatment effect sizes reported as –1.72 for individual therapy vs –0.91 for group therapy), earlier improvement in worry and depression symptoms, and higher adherence (9% attrition vs 24% attrition for group; Covin et al., 2008; Hunot et al., 2007).
Mode of delivery
The efficacy of dCBT for GAD has been demonstrated by RCTs conducted in volunteers from the community who met diagnostic criteria for GAD (total N = 287), with large effect size superiority over the control group for reductions in GAD symptoms (range = 1.05–1.24, NNT = 2) and depression (range = 0.85–1.02, NNT = 2; Paxling et al., 2011; Robinson et al., 2010; Titov et al., 2009). Recovery rates were good (70%), clinician time was reduced and benefits were maintained at 3 months, 1 year and 3 years post-treatment (Paxling et al., 2011; Robinson et al., 2010; Titov et al., 2009).
The effectiveness of dCBT for GAD has also been demonstrated in trials conducted in primary care (n = 588) with large effect sizes for GAD symptoms (effect size = 0.91) and a recovery rate of 62% at post treatment (Mewton et al., 2012). A meta-analysis of nine studies of dCBT versus wait-list control or psychological placebo showed an effect size superiority of 0.70 (95% CI = [0.39, 1.01]; NNT = 2.63; Andrews et al., 2018).
In the research trials of dCBT for GAD included in Andrews et al. (2018), adherence ranged from 74% to 80% for a technician- and clinician-assisted animated programme (75–130 minutes of clinician time). Adherence was lower in effectiveness studies: 55% in primary care and estimated at 26% for unguided self-help (Karyotaki et al., 2015).
The effectiveness of dCBT for GAD has been demonstrated in a number of transdiagnostic examinations for anxiety disorders in primary and secondary care (Adelman et al., 2014; Bell et al., 2012; Johnston et al., 2011; Mayo-Wilson and Montgomery, 2013; Titov et al., 2010). Meta-analyses of dCBT for anxiety disorders (not specific to GAD) have also demonstrated that dCBT and face-to-face therapy have comparable outcomes (Adelman et al., 2010a, 2014; Cuijpers et al., 2009; Mayo-Wilson and Montgomery, 2013).
dCBT for mixed anxiety and depression has demonstrated efficacy (effect size = 0.85) and effectiveness (effect size = 1.20) in reducing GAD symptoms (Newby et al., 2014). In the light of these effectiveness data (n = 1681 patients), it appears that the effect size superiority of dCBT for GAD over control groups is comparable to that of face-to-face CBT. We are unaware of any direct comparisons between dCBT and face-to-face CBT.
Other psychological treatments
RCTs with and without replication provide initial evidence for the effectiveness of other psychotherapy variants, including:
Applied relaxation (Conrad et al., 2008; Dugas et al., 2010; Fisher, 2006; Siev and Chambless, 2007; Wells et al., 2010);
Cognitive therapy (effect size = 1.81; Hanrahan et al., 2013; Norton and Price, 2007);
Mindfulness and acceptance-based interventions (Hayes-Skelton et al., 2013; Kim et al., 2009; Roemer et al., 2008);
Exercise as a standalone intervention and to augment CBT (Herring et al., 2012; Martinsen et al., 1989; Merom et al., 2008);
Cognitive bias modification (Cristea et al., 2015);
Peer-to-peer cognitive self-therapy (den Boer et al., 2007);
Meta-cognitive therapy (van der Heiden et al., 2012; Wells et al., 2010);
CBT targeting intolerance of uncertainty (Dugas et al., 2010);
Short-term and internet psychodynamic psychotherapy (Andersson et al., 2012; Ferrero et al., 2007; Riskind et al., 2006);
Alternative therapies (Kasper et al., 2010; Lakhan and Vieira, 2010; Sarris et al., 2011; Sarris and Kavanagh, 2009; Woelk and Schlafke, 2010).
However, additional replicated, high-quality, direct comparison and disorder-specific research is needed in order to make conclusions specific to the treatment of GAD.
Pharmacological treatment
Pharmacotherapy has been extensively studied and shown to be efficacious in the treatment of GAD.
Before starting any medication, psychoeducation should be provided about anxiety and GAD symptoms, and advice given about lifestyle factors that may be contributing or perpetuating symptoms. The timeframe for change should also be discussed as response is relatively slow, as indicated by findings that antidepressants separate from placebo most convincingly at 12 weeks and that response rates continue to increase over follow-up periods extending to 6 months. Potential adverse effects of medication should also be discussed carefully.
Throughout treatment, the patient should be encouraged to gradually increase exposure to feared situations, for example, situations with uncertain outcomes.
Efficacy of SSRIs and SNRIs
Antidepressants have been shown to be superior to pill placebo in treating GAD (effect size = 0.35, NNT = 5.15; Schmitt et al., 2005).
Evidence from RCTs supports the use of SSRIs as pharmacotherapy for GAD. RCTs have shown benefits with sertraline (Allgulander et al., 2004; Ball et al., 2005; Brawman-Mintzer et al., 2006; Mokhber et al., 2010), escitalopram (Baldwin et al., 2006; Bielski et al., 2005; Bose et al., 2008; Bystritsky et al., 2008; Davidson et al., 2004; Goodman et al., 2005; Lenze et al., 2009; Merideth et al., 2012) and paroxetine (Baldwin et al., 2006; Ball et al., 2005; Bielski et al., 2005; Kim et al., 2006; Pollack et al., 2001; Rickels et al., 2003).
Sertraline has highest acceptance, risk-to-benefit ratio and cost-effectiveness profile of pharmacological treatment options for GAD (National Institute for Health and Care Excellence, 2011a). It is therefore recommended as the first-line therapy of choice but the decision remains contentious (Gale and Millichamp, 2011).
Research also supports the use of the SNRIs, venlafaxine (Allgulander et al., 2001, 2008; Bose et al., 2008; Davidson et al., 1999; Gelenberg et al., 2000; Kasper et al., 2009; Katz et al., 2002; Lenox-Smith and Reynolds, 2003; Montgomery et al., 2006; Nimatoudis et al., 2004; Rickels et al., 2000) and duloxetine (Allgulander et al., 2007, 2008; Hartford et al., 2007; Koponen et al., 2007; Rynn et al., 2008; Wu et al., 2011).
Pregabalin
Pregabalin, a gamma-aminobutyric acid (GABA) analogue, has been demonstrated to be more effective than pill placebo in patients with GAD in meta-analyses (Boschen, 2011; Wensel et al., 2012) and clinical trials (Feltner et al., 2003; Kasper et al., 2009; Lydiard et al., 2010; Montgomery et al., 2006; Pohl et al., 2005; Rickels et al., 2005). Meta-analyses have estimated the overall effect size at 0.35 (Boschen, 2011; Wensel et al., 2012). It may also reduce depressive symptoms (Stein et al., 2008b).
There is increasing therapeutic effect on somatic symptoms up to the maximum dose of 400–600 mg/day (Boschen, 2012). However, dropout rates are high due to common adverse events including somnolence, dizziness, headache and dry mouth (which are particularly detrimental in the elderly; Wensel et al., 2012). In addition, the optimal duration of treatment and the risk of relapse and withdrawal remain unclear.
Pregabalin is not registered in Australia or New Zealand for the treatment of GAD.
Agomelatine
Agomelatine, a melatonergic antidepressant, has been found to be more effective than placebo in the short term and may be as effective as escitalopram but better tolerated (Stein et al., 2008a, 2014), including minimal sexual side effects (Gale and Millichamp, 2011).
Agomelatine is not registered in Australia for the treatment of GAD. It is not available in New Zealand.
Benzodiazepines
There is evidence from meta-analyses that benzodiazepines are efficacious in the short-term reduction of anxiety symptoms (Martin et al., 2007; Offidani et al., 2013). The efficacy of benzodiazepines as a long-term pharmacotherapy strategy in GAD has not been examined.
Benzodiazepines are relatively ineffective against cognitive anxiety symptoms and ineffective against depression (Starcevic, 2015) and are associated with extensive adverse effects including risk of falls, cognitive impairment, impaired driving and physical dependence and withdrawal (Hollister et al., 1993; Mitte, 2005a; Woods et al., 1992). For these reasons, benzodiazepines are not recommended for the treatment of GAD in primary or secondary care (National Institute for Health and Care Excellence, 2011b).
Quetiapine
Quetiapine, an atypical antipsychotic agent, is moderately effective for the treatment of GAD. A meta-analysis of four RCTs (n = 2265) reported an OR of 2.21 (95% CI = [1.10, 4.45]; Depping et al., 2010), while another meta-analysis of four RCTs (n = 1383) reported an RR of 1.31 (95% CI = [1.20, 1.44]; LaLonde and Van Lieshout, 2011).
The risk for discontinuation due to adverse events during quetiapine treatment (extended-release formulation) appears to be associated with dose and diagnosis. Patients with GAD appear more likely than patients with other diagnoses to experience adverse effects when taking quetiapine extended release 300 mg per day.
The use of quetiapine must be balanced against the risk of metabolic syndrome (weight gain), prolonged QTc syndrome, high dropout due to adverse effects (sedation, extrapyramidal side effects) and is not recommended for patients with GAD until more rigorous studies on the long-term safety and efficacy of this agent are available (Depping et al., 2010; LaLonde and Van Lieshout, 2011; Lorenz et al., 2010; National Institute for Health and Care Excellence, 2011b).
Imipramine and buspirone
Buspirone and imipramine have each demonstrated superiority to placebo in the treatment of GAD (Chessick et al., 2006; Davidson et al., 1999; Hoehn-Saric et al., 1988; Laakmann et al., 1998; Lader and Scotto, 1998; Mitte et al., 2005; Mokhber et al., 2010; Pollack et al., 1996b; Rickels et al., 1993; Rocca et al., 1997).
However, imipramine is associated with serious adverse effects and potential toxicity in overdose and buspirone has shown limited effectiveness in clinical practice (Chessick et al., 2006). Therefore, these medications should only be considered in patients who fail to respond to first-line agents.
Hydroxyzine
The antihistamine, hydroxyzine, has demonstrated effectiveness superior to placebo and similar to benzodiazepines and buspirone in patients with GAD (Guaiana et al., 2010; Lader and Scotto, 1998; Llorca et al., 2002). However, it is currently unavailable in Australia and New Zealand and is not recommended for physicians practising in these countries.
Pharmacotherapy comparisons
A meta-analysis of 48 studies found superiority of medication over pill placebo control (effect size = 0.33, NNT = 5.43) but no differences in efficacy between benzodiazepines, azapirones (e.g. buspirone) and SSRI/SNRIs (Mitte et al., 2005).
In a review of RCTs (Baldwin et al., 2011a), the authors concluded that there is evidence for the efficacy of certain SSRIs, SNRIs, pregabalin and quetiapine, but in wider clinical practice overall outcomes can be poor in many patients. A review focusing on agents evaluated in three or more studies (Baldwin et al., 2011b) found that duloxetine produced the greatest response (followed by sertraline), escitalopram produced the greatest remission rates (followed by venlafaxine) and sertraline showed the best tolerability (followed by pregabalin).
Duration of treatment
The optimal duration of treatment has not been determined. Some authors recommend a minimum of 1 year (Davidson, 2008).
Combination of medication and CBT
Despite common use in clinical practice, particularly with severe GAD, there is currently a lack of high-quality, replicated, conclusive evidence to support the routine combination of CBT and pharmacotherapy for GAD. While one meta-analysis reported moderate to large pre–post treatment effect sizes for combined pharmacotherapy and CBT for GAD (0.45 and 1.10) (Hofmann et al., 2009), this was calculated on the basis of only two studies and gains were not maintained at 6-month follow-up. In a recent meta-analysis of 52 RCTs (3623 patients), in which the effects of treatment with antidepressant medication were compared with the effects of combined pharmacotherapy and psychotherapy in adults with anxiety and depressive disorders (not specifically GAD), combined treatment was superior to pharmacotherapy alone (g = 0.43; 95% CI = [0.31, 0.56]; NNT = 4.20; Cuijpers et al., 2014b). However, this meta-analysis included only one study specific to GAD, which found no differences in outcome between patients with GAD offered venlafaxine with 12 sessions of CBT versus venlafaxine alone (p = 0.17; Crits-Christoph et al., 2011). Thus, there remains a need for direct comparison of replicated RCTs.
Questions also remain about whether there are additional benefits for combining CBT and pharmacotherapy when either one has not worked or when there is comorbidity or extremely severe symptoms.
Other treatment guidelines for GAD
NICE guidelines on the management of GAD in adults (National Institute for Health and Care Excellence, 2011a) recommend that pharmacological interventions should only be routinely offered to people who have not benefitted from psychological interventions. SSRIs, specifically sertraline, are recommended as the first-choice pharmacological treatment and third-line treatment after psychological interventions (National Institute for Health and Care Excellence, 2011a).
The Canadian clinical practice guidelines for the management of anxiety disorders (Katzman et al., 2014) recommend CBT as an effective first-line treatment for GAD including dCBT. The addition of an SSRI or SNRI is recommended for patients who do not benefit from initial treatment with CBT.
Treatment-refractory anxiety disorders
Around 5% of people with panic disorder will have some form of treatment-refractory state (Andrews and World Health Organization Collaborating Centre for Classification in Mental Health, 2006). There is little data available on the rate of treatment resistance in panic disorder, SAD and GAD.
Assessment
First, consider if the person has had an adequate trial of treatment. There may be issues with adherence to medication, or the medication dose may be inadequate. Similarly, the person may have not had structured CBT which includes all the effective components, may not have had frequent enough sessions (weekly sessions are recommended for the first 4–6 weeks), or may not have completed the tasks or attempted exposure to anxiety-triggering stimuli. To assess this, it can be useful to consult with the patient’s therapist and may also be useful to ask the patient what happened in the treatment.
Questions to ask to assess therapy
What strategies have you been shown for anxiety management?
Can I look at your homework diary?
Can you tell me in detail about your last exposure task?
Second, the formulation should be reviewed, noting the social, environmental and cultural factors limiting improvement. A second opinion should be obtained from a tertiary referral centre (or, if not possible, from a consultant psychiatrist or clinical psychologist).
It is important to consider the social situation, level of stress, loss of function, quality of life and secondary gains (if any) that the patient has experienced.
Third, noting what treatments have already been given, you should consider if there are second-line treatments with evidence that have not been trialled, and recommend these before moving beyond those for which there is robust evidence of efficacy or effectiveness.
Management
Caveat
The following advice is our expert consensus. There is limited to no trial data to support some of these approaches. The evidence for each condition has been discussed in the previous sections by diagnoses.
General principles of treatment
Only one treatment modality should be trialled at a time. Each treatment should be approached as an ‘n of 1’ treatment trial. Before and after each new treatment, it is useful to measure symptom level and general distress. The person’s level of function should be monitored continually. If a modality has not worked after an adequate trial (good adherence to an adequate dose or treatment intensity), it should be stopped and another modality trialled. Combination pharmacotherapy or augmentation should be avoided. Doses of medication or therapy should be at a minimally effective level.
The general principles of management include the following:
Continuity of care, regular (but not frequent) review;
Encouraging the patient to continue an active life and avoid the sick role;
Setting incremental goals for functional improvement;
Encouraging hope for the future. Remember that even if therapies have not been effective, the patient’s situation may change and allow them to revisit previous treatments;
Continually seeking peer review.
Shifting the focus of treatment
At this time, the goal of treatment should shift from symptom remission to symptom management and functional improvement while living with a chronic disorder. Improving a person’s level of function may improve quality of life more than further attempts to reduce symptoms.

CBR: consensus-based recommendation.
A careful approach is needed, primarily aiming to improve quality of life. The clinician should arrange to see the patient regularly, but not too often.
At this time, one should discuss goals with the patient and provide psychoeducation around a recovery model. Reframe the goals of treatment to focus more directly on maximising functioning and less on symptom remission.
At this stage, we suggest seeing the patient regularly (e.g. once a fortnight), providing containment and support, and increasing resilience. The time interval between appointments can gradually be increased.
Psychological therapies
The focus of psychological therapy shifts to support and adaptation to chronic illness. The patient can be introduced to the recovery model and encouraged to review their goals.
There is a place for dynamically based supportive psychotherapy and for interpretive or exploratory therapy.
In line with a goal of maximised functioning, consider using structured treatments, including ACT or mindfulness-based therapies, which have therapeutic goals that include accepting the current lived experience and minimising distress. Such therapies should be provided by experienced clinicians trained in these techniques. An adequate number of sessions should occur before each modality is abandoned. If a modality has some efficacy, then one should consider continuation or ‘booster’ sessions.
Medications
Medications should be used to reduce symptoms and improve level of function. In general, use one medication at a time, measure outcomes using both symptoms scales and level of function, monitor for side effects, and withdraw a medication that has failed before starting a new medication.
There is no systematic evidence that combining or augmenting medications has any additional benefit. We do not recommend psychotropic polypharmacy.
The aim is to achieve the maximal therapeutic benefit at the minimal dose.
There is an evidence base to support the use of TCAs, moclobemide, MAOIs, mirtazapine, pregabalin and agomelatine (see sections on specific diagnoses). If these are ineffective, then maintenance with benzodiazepines could be considered. A benzodiazepine without a short half-life should be selected. The dose should be kept within the lower end of the therapeutic range and not increased once the patient has responded. Risks should be carefully discussed with the patient.
The atypical antipsychotics may lead to some symptomatic relief, but the risk/benefit ratio is such that they are less likely to lead to gains than the options listed above. Their use is not supported by evidence from RCTs. If used, the starting dose should be very low and the patient should be monitored carefully for extrapyramidal side effects, particularly akathisia. It is important to monitor for metabolic side effects and to monitor QTc length by regular ECGs.
Anxiety disorders in special populations
Pregnancy and postnatal presentations of anxiety
Anxiety symptoms and disorders are common during pregnancy, with estimated prevalence of approximately 18–25% for self-reported anxiety symptoms, approximately 15% for clinical diagnosis of any anxiety disorder and approximately 4% for GAD (Dennis et al., 2017). In the postnatal period, the estimated prevalence of any anxiety disorder is approximately 10% (Dennis et al., 2017).
During pregnancy
The goal of treatment during pregnancy and lactation is syndrome remission. Non-pharmacological treatment such as CBT (face-to-face or dCBT) is recommended as first-line treatment in pregnant women with anxiety disorders.

CBT: cognitive–behavioural therapy; dCBT: digital cognitive–behavioural therapy; CBR: consensus-based recommendation; EBR: evidence-based recommendation.
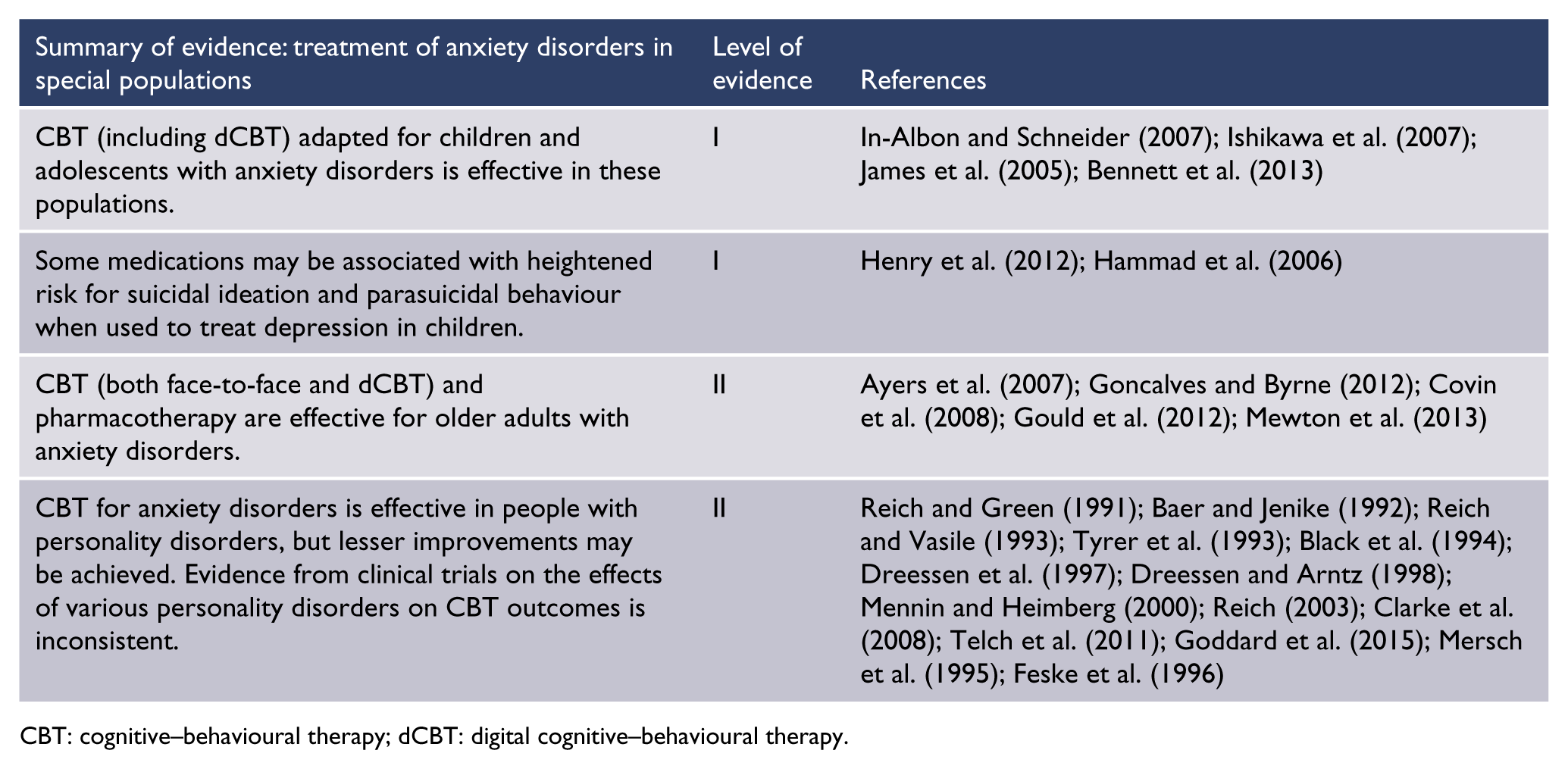
CBT: cognitive–behavioural therapy; dCBT: digital cognitive–behavioural therapy.
Medication should be considered for women with severe or disabling anxiety disorders. If medication is required, an SSRI or SNRI are first-line; to minimise the potential for adverse effects on the foetus and neonate, up-to-date information about potential agents should be sought. Vigilant monitoring is indicated (Rubinchik et al., 2005).
Women with anxiety disorders who are taking medication for anxiety and who wish to become pregnant will need up-to-date information about the potential risks and benefits of continuing on the medication and information about alternative management strategies.
Post partum
Increased support should be offered in the postnatal period. Anxiety disorders can have an impact on mother–infant relationship and attachment (Campbell et al., 2004; Nicol-Harper et al., 2007).
Nursing mothers can find CBT difficult due to time pressures, although this is less of a problem with dCBT. SSRIs are of benefit in moderate-to-severe anxiety disorders, but clinicians must take into account safety with breastfeeding before prescribing any medication.
Older adults
CBT (face-to-face or dCBT) and pharmacotherapy have been shown to be effective for older adults with anxiety disorders (Ayers et al., 2007; Covin et al., 2008; Goncalves and Byrne, 2012; Gould et al., 2012; Mewton et al., 2013).
In a large effectiveness study, older adults did well with dCBT (Mewton et al., 2013). The general treatment recommendations stand and the indication for CBT (face-to-face or dCBT) is emphasised because many older people are taking a range of other medications that may present a risk of interaction or additive adverse effects with medications for anxiety disorders. Even SSRIs, which are better tolerated than other medications (Mottram et al., 2006), can cause the Syndrome of Inappropriate Diuretic Hormone Secretion (SIADH) increasing the possibility of delirium and seizures secondary to hyponatraemia, and are associated with an increased risk of gastrointestinal (GI) and other bleeds for those with established risk factors such as bleeding history, treatment with nonsteroidal anti-inflammatory drugs (NSAIDs), steroids and anticoagulants (Taylor et al., 2015). Older people are also more susceptible to antidepressant-induced hyponatraemia and other adverse events.
In view of these safety issues, CBT (face-to-face or dCBT) should be considered as first-line treatment, even in those with mild cognitive impairment (Orgeta et al., 2015). CBT may need to be augmented with motivational interviewing to increase adherence if ambivalence towards attending psychological therapy is present (Gould et al., 2012; Hunot et al., 2007).
Children and adolescents
Diagnostic issues
For a diagnosis of SAD, the anxiety must occur in peer settings (not just during interactions with adults) and may be expressed by crying, tantrums, freezing, clinging, shrinking or failure to speak in social situations (American Psychiatric Association, 2013).
For a diagnosis of GAD, children only need to present with one (rather than three) out of six associated physical symptoms of anxiety with consideration that the worries tends to be age appropriate, that is, focused on school or sporting performance (American Psychiatric Association, 2013).
There are no paediatric-specific criteria for panic disorder, and full-blown panic disorder is relatively rare prior to late adolescence.
Treatment issues
CBT (including dCBT) adapted for children and adolescents with anxiety disorders is efficacious and the treatment of choice (Bennett et al., 2013; In-Albon and Schneider, 2007; Ishikawa et al., 2007; James et al., 2005).
A review of medication for anxiety disorders in children and adolescents concluded there was evidence of efficacy for the SSRIs (Reinblatt and Riddle, 2007). Advice should be sought before prescribing medication for children and adolescents, due to the lack of high-quality, replicated research evidence on side effects and withdrawal risks in this area, and as there is evidence that some medications may be associated with heightened risk for suicidal ideation and parasuicidal behaviour when used to treat depression in children (Hammad et al., 2006; Henry et al., 2012), although recent evidence suggests that the risks may have been overstated (Friedman, 2014).
When children or teenagers are not engaging in, or are resistant to, treatment with CBT it can be helpful to talk to parents about finding motivators in the child or teenager’s life.
Some internet-delivered courses have been shown to reduce anxiety in children and adolescents. A review of 27 studies of computerised CBT for young people aged 12–25 (Pennant et al., 2015) reported a reduction in anxiety symptoms versus control (SMD = 0.77; 95% CI = [–1.45, –0.09]). dCBT programmes are available in Australia and New Zealand. These include The Brave Program available at https://brave4you.psy.uq.edu.au (Spence et al., 2016) and This Way Up TeenSTRONG Program available at https://thiswayup.org.au.
Māori
Māori are about one and a half times more likely to have anxiety and mood disorder symptoms compared with non-Māori New Zealanders in community surveys (Ministry of Health, 2014). The most common lifetime disorders for Māori were anxiety, with a lifetime prevalence of 31.3% (Baxter et al., 2006). The high prevalence of mood and anxiety disorders among Māori is associated with higher rates of serious outcomes including suicide (almost twice that of non-Māori New Zealanders) and elevated rates of hospitalisation for intentional self-harm (Ministry of Health, 2010).
Although similar levels of presentation for anxiety disorders in primary care have been reported for Māori and non-Māori (Arroll et al., 2009), admission rates for Māori are higher in acute services, with Māori presenting to secondary mental health services with more severe symptoms (Baxter, 2008). One report has suggested that Māori may be less likely to be referred for psychotherapy (Kumar et al., 2008).
When treating Māori with anxiety disorders, cultural beliefs about anxiety and mental illness should be explored, as well as the language used to describe anxiety in the person’s community. Interventions offered should follow accepted guidelines and be tailored to the individual’s goals and preferences.
Aboriginal and Torres Strait Islander people
The Australian National Strategic Framework for Aboriginal and Torres Strait Islander Peoples’ Mental Health and Social and Emotional Wellbeing 2014–2019 (Department of the Prime Minister and Cabinet, 2017) recognises the complex interplay of cultural, historical, social, personal and spiritual factors determining health and mental health outcomes while acknowledging and foregrounding resilience and strength in the face of adversity.
Aboriginal and Torres Strait Islander people experience more than twice the rate of psychological distress as the non-Indigenous Australian population (Australian Bureau of Statistics, 2010) and high levels of exposure to stressors. A history of trauma in general is associated with increased risk of anxiety disorders. Many Aboriginal and Torres Strait Islander people have experienced chronic, inter-generational trauma.
Surveys among Aboriginal and Torres Strait Islander communities report anxiety prevalence rates of 1.4–5% of community members (Parker, 2010). More focused surveys, such as the Western Australian Aboriginal Child Health Survey, showed that up to one-quarter of surveyed children aged 4–17 years may have been at risk of developing emotional and behavioural disorders that may be associated with anxiety conditions (Zubrick et al., 2005). When treating Aboriginal and Torres Strait Islander people with anxiety disorders, cultural beliefs about anxiety and mental illness should be explored, as well as the language used to describe anxiety in that individual’s community. Interventions offered should follow accepted guidelines and be tailored to the individual’s goals and preferences.
People with comorbid mental disorders
Comorbidity with other mental disorders is notably high among people with anxiety disorders. Mood disorders, substance use disorders and personality disorders are the most common comorbid disorders (Ansell et al., 2011; Barlow et al., 1986; Brown and Barlow, 1992; Sanderson and Barlow, 1990).
Comorbid substance use disorders
There is a paucity of good-quality evidence on the optimal management of anxiety disorders with comorbid substance use disorders to guide practice (Baillie et al., 2013). Prioritising treatment of the substance use has been recommended, as well as integrated treatment of the anxiety disorder and alcohol use (Stapinski et al., 2015).
Comorbid personality difficulties
Prevalence
In a recent meta-analysis of approximately 14,000 participants, the rate of any comorbid personality disorder was high across all anxiety disorders, with proportions ranging from 0.35 for PTSD to 0.52 for OCD (Friborg et al., 2013). The anxious, fearful personality disorders (avoidant, dependent and obsessive-compulsive personality disorders) were most commonly co-occurring.
Impact of personality disorders on anxiety disorders
Personality disorders are characterised by relatively stable impairments in personality functioning and the presence of pathological personality traits (American Psychiatric Association, 2013). Common impairments across personality disorders involve chronic and marked difficulties with identity and self-esteem, effective self-direction and goal-setting, empathy and intimacy, emotion regulation, distress tolerance and impulse control (American Psychiatric Association, 2013).
Although there are some conflicting reports, studies generally find that, compared with patients with anxiety disorders who do not have a personality disorder, those with personality disorders report more severe anxiety disorder symptoms, more severe concurrent depressive and anxious symptoms, lower levels of functioning and higher general distress (Heffernan and Cloitre, 2000; Herbert et al., 1992; Matsunaga et al., 1998; Mellman et al., 1992; Mersch et al., 1995; Ozkan and Altindag, 2005; Reich et al., 1994; Tenney et al., 2003). Some studies also find that the presence of a comorbid personality disorder is associated with an earlier age of onset, greater chronicity and an increased likelihood of relapse of the anxiety disorder (Ansell et al., 2011; Ozkan and Altindag, 2005; Pollack et al., 1992).
The impact of comorbid personality disorders on anxiety disorders may vary with the type of anxiety and personality disorder (Ansell et al., 2011). Various personality disorders may influence the symptoms of panic disorder with agoraphobia; comorbid borderline personality disorder may intensify and prolong panic attacks, while an avoidant personality disorder may exacerbate social withdrawal and agoraphobic avoidance. A dependent personality disorder, on the other hand, may aggravate the patient’s reliance on others to accompany them when they are anxious or expect to be so (Friborg et al., 2013).
Effect of comorbid personality disorders on CBT outcomes
Clinicians may often assume that anxious patients with comorbid personality difficulties will not benefit from CBT.
Some studies find that comorbid personality disorders have a negative effect on CBT outcomes (Tyrer et al., 1993), whereas others report no significant impact (Dreessen et al., 1997; Mersch et al., 1995). Other studies find that while patients with anxiety disorders and comorbid personality disorders benefit as much as those without comorbid personality disorders, they are more likely to drop out of treatment (Clarke et al., 2008).
Several reviews have been undertaken to synthesise the empirical literature in this area, although meta-analyses and systematic reviews are yet to be conducted. Reviews of CBT outcomes for patients with panic disorder or OCD suggest that comorbid personality disorders have a negative impact on treatment outcomes (Mennin and Heimberg, 2000; Reich and Green, 1991; Reich and Vasile, 1993), although one review (Baer and Jenike, 1992) found that only schizotypal personality disorder predicted poor treatment outcome for OCD. The authors of another review (Dreessen and Arntz, 1998) examined a broader array of anxiety disorders and limited their review to studies chosen via the ‘best evidence’ procedure. This review did not find that personality disorders in general had a negative impact on CBT outcome.
Disparate findings may partly relate to methodological issues (Reich, 2003). For example, in one study (Black et al., 1994), when personality disorders were assessed categorically (via structured diagnostic interview), they were not significant predictors of cognitive therapy outcome, but when assessed dimensionally (via self-report questionnaire), the presence of personality disorders was a negative predictor of treatment outcome.
Results may also vary depending on which outcome variables are assessed. One study found that socially phobic patients with or without comorbid avoidant personality disorder improved similarly on measures of social anxiety, depression and social adjustment, but patients with a comorbid avoidant personality disorder improved less on measures of trait anxiety and self-esteem (Feske et al., 1996).
The most recent studies in this area have examined the impact of comorbid personality disorders on treatment, while controlling for baseline symptom severity. In a study of patients with panic disorder undertaking a group CBT programme (Telch et al., 2011), patients with personality disorders reported higher baseline and post-treatment symptom scores. Although the presence of personality disorder pathology did predict poorer treatment outcome, its predictive utility was modest compared with baseline symptom severity.
The effect of comorbid personality disorders was examined in a study of over 1200 patients undergoing evidence-based psychological therapy for mild-to-moderate anxiety and depression (Goddard et al., 2015). It found that greater degrees of self-reported comorbid personality difficulties independently predicted poorer treatment outcome (including reduced absolute change on all outcome measures), after controlling for baseline symptoms of depression, anxiety, functional impairment, demographic status and the number of treatment sessions attended. Interestingly, personality difficulties did not predict higher rates of treatment dropout. Consistent with the panic disorder study (Telch et al., 2011), the predictive utility of personality difficulties in determining treatment outcome was significant but modest.
Despite complexities in the literature, it is clear that individuals with significant personality difficulties seek CBT for their anxiety disorders. These individuals do respond to treatment, but it is likely that they respond with smaller effects that those without comorbid personality concerns. However, it is also likely that the severity of baseline symptoms is a stronger predictor of poor treatment outcome than the presence of personality dysfunction. In short, the presence of comorbid personality dysfunction is not in itself a contraindication to CBT for anxiety disorders.
Clinical considerations
Clinicians should avoid overestimating the impact of personality pathology on the therapeutic outcome of CBT for the anxiety disorders. However, whenever a patient presents with multiple comorbid conditions, sound clinical judgment will be essential when determining treatment priorities. Potential barriers to treatment should be considered and a plan for managing these should be developed.
Footnotes
Appendix 1
Appendix 2
Acknowledgements
The following people and organisations assisted with the external review of the RANZCP clinical practice guidelines for the treatment of panic disorder, social anxiety disorder and generalised anxiety disorder:
Expert Contributors
Dr Chris Wever Associate Professor Rocco Crino Professor Sean Hood Professor David Baldwin Professor Paul Salkovskis Ms Ashna Basu Ms Siobhan Loughnan Dr Kathleen O’Moore
The following organisations provided a submission during the public consultation period which lasted from 30 June to 30 July 2017. During the consultation period, a draft of the guidelines was publically available on the RANZCP website.
Organisations that provided feedback during the public consultation period
Australian Clinical Psychology Association Australian College of Mental Health Nurses Australian Psychological Society beyondblue Dietitians Association of Australia National Mental Health Consumer and Carer Forum Private Mental Health Consumer Carer Network Servier Australia WA Primary Health Alliance
The RANZCP also acknowledges the input of the 21 individuals who provided feedback during the public consultation period.
Special acknowledgements
Ms Jennifer Harman, Medical Writer, Meducation
The RANZCP project team
Ms Rosie Forster, Executive Manager, Practice, Policy and Partnerships Ms Niamh Byrne, Project Officer Dr Huseyin Mustafa, Project Manager (November 2014–November 2017) Ms Heather Bird, Project Officer (July 2016–February 2017)
Declaration of Conflicting Interests
Members of the working group signed a deed of undertaking at the time of appointment in which they agreed to declare any conflict, whether actual, potential, perceived or likely to arise.
To manage conflicts of interest of the working group during the CPG development process:
As a part of the standing items of all meetings, all working group members were asked to declare their conflicts of interest during each teleconference meeting and these were recorded. If a conflict of interest (COI) was declared, the individual(s) concerned was excluded from the discussion and right to vote/contribute. The working group members signed an updated COI form at the time of submitting the clinical practice guidelines for the treatment of panic disorder, social anxiety disorder and generalised anxiety disorder for publication.
Disclaimer
Compiled for the Royal Australian and New Zealand College of Psychiatrists (RANZCP), this information and advice is based on current medical knowledge and practice as at the date of publication. It is intended as a general guide only, not as a substitute for individual medical advice. The RANZCP and its employees accept no responsibility for any consequences arising from relying upon the information contained in this publication.
Funding
The development of these guidelines was supported and funded by the RANZCP. The RANZCP acknowledges the significant pro bono input of the RANZCP Fellows and other expert contributors in the development of these guidelines. The RANZCP thanks those who have given of their time, experience and expertise.
