Abstract

To the Editor
There are few reports on the idiosyncratic drug reactions induced by olanzapine. Various types of such reactions ranging from benign drug eruptions like fixed drug eruptions, photo-onycholysis to severe hypersensitive vasculitis have been reported (Chawla et al., 2017). Here, we present a case of subacute photo-allergic reactions possibly induced by olanzapine.
A 34-year-old woman, Mrs A, who suffered from schizophrenia, was started on olanzapine 5 mg/day. She was neither on any other medications nor had any comorbid general medical or psychiatric diagnoses at that time. As she had shown a partial response with the 5 mg/day of olanzapine, the dose was up-titrated to 10 mg/day on the subsequent follow-up after 2 weeks.
Within a week, she represented with the complaint of bilateral symmetrical skin-lesions over the dorsum of her feet and hands, exclusively involving the sun-exposed parts. She reported that these lesions started appearing 10 days earlier, since then gradually progressing over the past week. Dermatology consultation described multiple ill-defined plaques with surface oozing, crusting and bleeding points over the dorsal aspect of bilateral hands and the feet (Figure 1). This was considered consistent with a picture of subacute eczema. She was started on topical corticosteroids and advised to reduce or cease the olanzapine. Hence, olanzapine was cross tapered with risperidone. Within a week after stopping the medication, the lesions had resolved and no worsening of the psychosis noted. On the adverse drug reaction probability scale (Naranjo), we calculated a score of 7 for her skin lesions, which indicates a probable association (Naranjo et al., 1981).
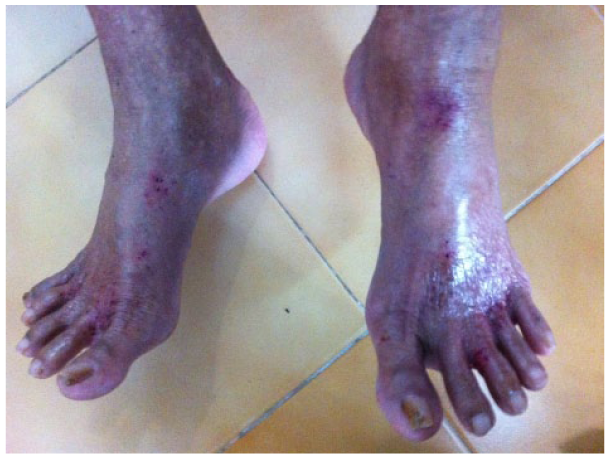
Showing the photo-allergic subacute eczematous lesions over the feet.
N-oxide metabolites are frequently associated with the idiosyncratic drug reactions (Uetrecht, 2002). Olanzapine can form N-oxide metabolites through a minor pharmacokinetic pathway. We hypothesize this mechanism might explain the rare occurrence of idiosyncratic immune reactions.
We would suggest that clinicians be aware of this rare occurrence and recommend suspicion early in the course of treatment, as the treatment may warrant dose reduction or discontinuation.
Footnotes
Acknowledgements
We would like to thank our patient and her family members for permitting this report to be published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
