Abstract
Introduction:
Hyperprolactinaemia is commonly observed in people with psychotic disorders due to D2 receptor blockade by antipsychotic drugs, although it may also exist in drug-naïve patients with first-episode psychosis. Recent studies suggest that hyperprolactinaemia may have a negative impact on cognitive function in people with early psychosis. We aimed to explore whether there are sex differences in the association between prolactin levels and cognitive performance in early psychosis patients.
Methods:
We studied 60 young patients with early psychosis (aged 18–35 years, 35% females) and a sex- and age-matched control group of 50 healthy subjects. Cognitive assessment was performed with the MATRICS Consensus Cognitive Battery. Prolactin, total cortisol, follicular-stimulating hormone, luteal hormone and sex steroids (testosterone in men, oestradiol and progesterone in women) were measured in plasma. Salivary cortisol was measured at different sampling times (awakening response, 10:00 and 23:00). Psychopathological status was assessed, and antipsychotic treatment was registered. Multiple linear regression analyses were used to explore the relationship between prolactin and cognitive tasks while adjusting for covariates.
Results:
Prolactin levels were associated with impaired processing speed in men, and this association was independent of cortisol and testosterone. In women, prolactin levels were not associated with processing speed tasks, although we observed a negative effect of prolactin on verbal learning and spatial working memory in female healthy subjects. The male-dependent effect maintained its significance after adjusting for education status, antipsychotic treatment and negative symptoms.
Conclusion:
Our study demonstrates that the previously reported association between high prolactin levels and impaired cognitive processes in early psychosis is restricted to men.
Introduction
Hyperprolactinaemia is a common condition in subjects with psychotic disorders. As dopamine (DA) released by the tuberoinfundibular dopaminergic pathway into the pituitary portal blood is the main prolactin-inhibiting factor, hyperprolactinaemia is a common consequence of D2 receptor blockade by antipsychotic drugs (Cookson et al., 2012; Horseman and Gregerson, 2013). However, increased prolactin levels have also been reported in drug-naïve patients with first-episode psychosis (FEP; Aston et al., 2010; Garcia-Rizo et al., 2012; Riecher-Rössler et al., 2013).
Although the most studied consequences of hyperprolactinaemia in psychotic subjects are amenorrhoea, galactorrhoea, sexual impairment and infertility, previous studies conducted in non-psychiatric populations (Henry and Sherwin, 2011) and animal models (Torner et al., 2013) suggest that hyperprolactinaemia may also have negative effects on cognition. In this regard, our group has demonstrated (Montalvo et al., 2014) that increased prolactin levels are associated with impaired processing speed in subjects with early psychosis (EP) and mediate the negative effects of antipsychotic-induced prolactin increase on processing speed.
In the study of the relation between prolactin levels and cognition, it is also important to explore whether there are sex differences because prolactin levels are higher in women (Horseman and Gregerson, 2013) and because cognitive effects may be modulated by specific sex steroids (e.g. 17-β-oestradiol or progesterone in women, testosterone in men; Castanho et al., 2014). It is also important to know whether the effects of prolactin on cognition are moderated by other hormones that might affect cognitive processes in patients with psychotic disorders, including sex steroids (Huerta-Ramos et al., 2014; Moore et al., 2013) and hypothalamic–pituitary–adrenal (HPA) axis hormones (Aas et al., 2011; Halari et al., 2004). In previous studies by our group, we have reported a sex difference in the association between cortisol awakening response (CAR) and cognitive tasks in individuals with EP (Labad et al., 2016): An increased CAR was associated with poorer processing speed and verbal memory in female patients only. However, another study reported that a blunted CAR, but not cortisol levels during the day, was associated with poorer verbal memory and processing speed in individuals with FEP (Aas et al., 2011).
In sum, several studies have addressed the relationship among prolactin, HPA axis measures or sex steroids and cognition in subjects with psychotic disorders; however, there are no studies that have considered all these hormones together with a particular focus on prolactin levels and sex differences. Thus, we conducted an exploratory analysis that might generate hypotheses for future studies on the topic. The main aim of our work was to explore whether there are sex differences in the association between prolactin levels and cognitive performance in people with EP. We also wanted to determine whether prolactin’s effects on cognition are direct or are explained by HPA axis measures or plasma concentrations of follicular-stimulating hormone (FSH), luteal hormone (LH) or sex steroids (oestradiol and progesterone in women; testosterone in men).
Methods
Participants
We studied 60 outpatients (39 men, 21 women) with EP, aged between 18 and 35 years, from the Early Intervention Service of the Hospital Universitari Institut Pere Mata (Reus, Spain). Each patient had a psychotic disorder (fulfilling Diagnostic and Statistical Manual of Mental Disorders [4th ed.; DSM-IV] criteria for a schizophreniform disorder [N = 14], schizophrenia [N = 10], schizoaffective disorder [N = 8] or psychotic disorder not otherwise specified [N = 28]) with a duration of illness of <3 years (65% had FEP). We used a control population of 50 healthy subjects (HS), matched by sex and age, who were recruited from the community using advertisements. The exclusion criteria were severe neurological disease or mental retardation; pregnancy; language difficulties; visual impairment; alcohol, heroin or cocaine dependence; and treatment with glucocorticoids or oral contraceptive pill use.
Clinical and cognitive assessment
All the patients were interviewed by a psychiatrist using the Schedules for Clinical Assessment in Neuropsychiatry (Wing et al., 1990). The ‘Operational Criteria’ (OPCRIT) checklist version 4.0 (available at http://sgdp.iop.kcl.ac.uk/opcrit/) was used to obtain DSM-IV diagnoses. The Positive and Negative Syndrome Scale (PANSS; Kay et al., 1990) was used to assess the severity of psychotic symptoms. We considered the five dimensions suggested by a consensus (Wallwork et al., 2012): positive, negative, disorganized, cognitive and depressive. The Calgary Depression Scale for Schizophrenia (CDSS; Addington et al., 1990) was administered to assess depressive symptoms.
The Spanish version of the MATRICS Consensus Cognitive Battery (MCCB) was used to assess neurocognitive functioning (Nuechterlein et al., 2008). The MCCB contains 10 tests within 7 domains to measure cognitive functioning (Table 1S).
Sociodemographic and clinical variables were obtained in a semi-structured interview. Consumption of alcohol was measured in standard units/day, tobacco in cigarettes/day and cannabis in joints/day. Psychopharmacological treatment was recorded during the neuropsychological assessment. Each antipsychotic dosage was transformed into chlorpromazine equivalents in milligram/day (Gardner et al., 2010). However, for descriptive information in tables, antipsychotic doses will be represented in risperidone equivalents taking into account the clinically equivalent doses suggested by Gardner et al. (2010). Of all 60 patients, 11 (18.3%) did not receive antipsychotic treatment, 41 (68.3%) received antipsychotic monotherapy (risperidone [n = 15], paliperidone [n = 5], olanzapine [n = 14], aripiprazole [n = 5], quetiapine [n = 1], perphenazine [n = 1]) and 8 (13.3%) received polytherapy. There were no sex differences regarding antipsychotic treatment.
Hormonal measures
Plasma
A fasting blood sample was obtained in the morning between 8:30 and 9:30 hours in resting conditions to measure unstimulated plasma prolactin, total cortisol levels and sex hormones. The participants were told to avoid stressful and physical activities or breast stimulation in the 12 hours prior to blood sampling. Prolactin, cortisol, FSH, LH, oestradiol, progesterone and testosterone concentrations were measured using the Maglumi 2000 Analyser chemiluminescence immunoassay system (SNIBE Co, Ltd, Guandong, China). The sensitivity for each assay was as follows: prolactin 77 pmol/L, cortisol 69 nmol/L, testosterone 0.5 nmol/L, oestradiol 29.7 pmol/L, progesterone 0.4 nmol/L, FSH 0.5 UI/L and LH 0.5 UI/L. The intra-assay and inter-assay coefficients of variation for all the serum assays were under 9%.
Saliva
We also obtained saliva samples with Salivette® tubes (Sarstedt AG & Co, Nümbrecht, Germany) at different times for cortisol determination: (1) one sample before the fasting blood sample, (2) one sample during the neuropsychological assessment (before and after the MCCB administration, which was conducted in the morning, with initial assessment times ranging between 9:00 and 12:00 hours) and (3) five home-collected saliva samples (another day of the fasting blood analysis and cognitive assessment) for calculating the CAR and cortisol levels over the day, using the following sampling times: awakening (T1), 30′ post-awakening (T2), 60′ post-awakening (T3), 10:00 hours (T4) and 23:00 hours (T5).
The saliva samples were processed at the Biobank at the Institut d’Investigació Sanitària Pere Virgili (IISPV). After centrifugation of the Salivette tubes at 3000 r/min for 5 minutes, the saliva was aliquoted and frozen at −20°C until the assay. Salivary cortisol was determined by a commercial chemiluminescence immunoassay (IBL, Hamburg, Germany). The sensitivity of the salivary cortisol assay was 0.08 nmol/L. The intra-assay and inter-assay coefficients of variation for this assay were under 8%.
Statistical analysis
SPSS version 19.0 (SPSS, Inc., Chicago, IL) was used for the statistical analyses.
Transformation of variables
We explored the distribution of plasma hormone concentrations and performed a log transformation (ln) for those hormones that had a skewed distribution. For salivary cortisol values, as suggested by recent expert consensus guidelines (Stalder et al., 2016), we transformed all the values using the following power transformation: X′ = (X 26 − 1)/0.26 (Miller and Plessow, 2013). The CAR was calculated using the area under the curve with respect to the increase (Pruessner et al., 2003) and the cortisol levels over the day using the area under the curve with respect to the ground (AUCg). Cortisol levels during the neuropsychological assessment were calculated as the mean value of the pre-MCCB and post-MCCB samples.
Although we transformed the cortisol values with a power transformation, we did not use transformed values in the calculation of cortisol levels throughout the day. This approach was preferred because the cited power transformation sometimes yields negative values for very low cortisol values (that would be the case for most evening cortisol levels), which could affect the interpretation of cortisol levels using the trapezoid formula.
Univariate and general lineal model
Pearson’s correlation was used to analyse the association between continuous variables. Partial correlation analyses were used when needed to adjust for potential confounders. A general lineal model (GLM) was performed to study the effect of sex and diagnostic groups (HS vs EP) on continuous data and the possible interaction of those factors. To compare female and male EP patients, a t-test was performed. Significance was set at p < 0.05 (two tailed).
Multiple linear regression analyses
To explore the association between prolactin and cognitive tasks, testing interactions among prolactin, sex and diagnosis as well as adjusting for potential confounders, we conducted a series of multiple linear regression analyses. Cognitive performance was considered the dependent variable, and an independent equation was conducted for each cognitive task. Prolactin was considered an independent variable, and we included sex, diagnosis, education level and antipsychotic treatment as the other independent variables. The three-way and two-way interactions among prolactin levels, sex and diagnosis were tested. Following hierarchical modelling, both significant interactions and lower level non-significant interactions were included in the final model.
Multiple testing
Many exploratory analyses were addressed in our study. We did not correct for multiple testing because the correction for multiple testing is not strictly necessary in analyses that are exploratory in nature (Bender and Lange, 2001).
Results
Univariate analysis and GLM
Clinically relevant variables of the two populations (HS and EP) and statistics corresponding to these variables can be seen in Table 1. As shown, EP patients had lower education, smoked more cigarettes and showed poorer cognitive performance in all tasks. No sex differences on the psychometric PANSS or CDSS scales were found among the EP subjects. Sex differences in the cognitive MCCB tests were small, with males performing worse than females in managing emotions and better in neuropsychological assessment battery (NAB) mazes, regardless of diagnosis. The only significant interaction between sex and diagnosis was found in continuous performance test–identical pairs (CPT-IP): healthy men outscored healthy women in this attention and vigilance task, whereas female patients outperformed male patients. We repeated the sex comparison on cognitive performance in the subgroup of patients receiving risperidone or paliperidone in monotherapy (Table 3S). Female patients receiving these prolactin-elevating antipsychotics showed better social cognition.
Clinical variables of the sample.
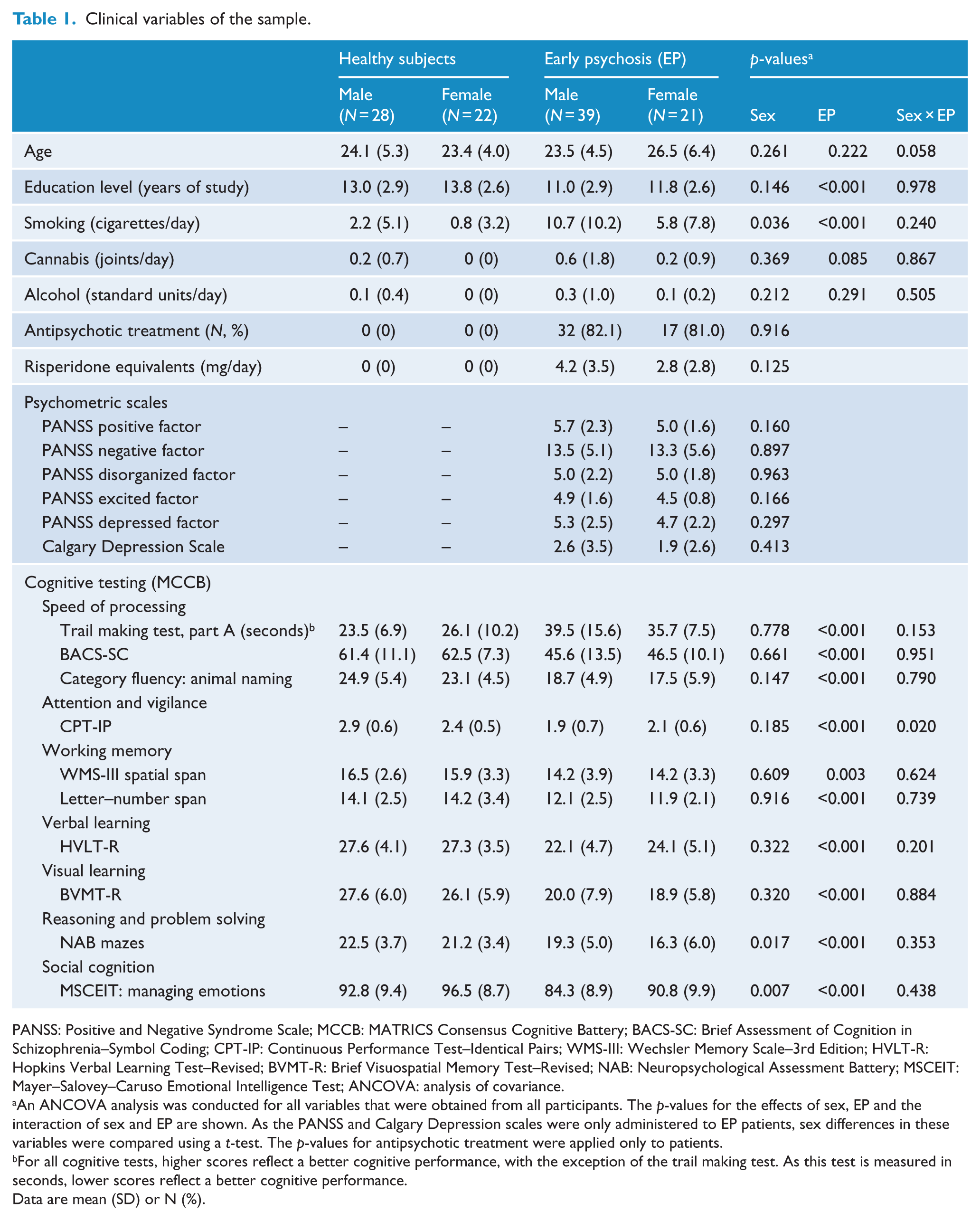
PANSS: Positive and Negative Syndrome Scale; MCCB: MATRICS Consensus Cognitive Battery; BACS-SC: Brief Assessment of Cognition in Schizophrenia–Symbol Coding; CPT-IP: Continuous Performance Test–Identical Pairs; WMS-III: Wechsler Memory Scale–3rd Edition; HVLT-R: Hopkins Verbal Learning Test–Revised; BVMT-R: Brief Visuospatial Memory Test–Revised; NAB: Neuropsychological Assessment Battery; MSCEIT: Mayer–Salovey–Caruso Emotional Intelligence Test; ANCOVA: analysis of covariance.
An ANCOVA analysis was conducted for all variables that were obtained from all participants. The p-values for the effects of sex, EP and the interaction of sex and EP are shown. As the PANSS and Calgary Depression scales were only administered to EP patients, sex differences in these variables were compared using a t-test. The p-values for antipsychotic treatment were applied only to patients.
For all cognitive tests, higher scores reflect a better cognitive performance, with the exception of the trail making test. As this test is measured in seconds, lower scores reflect a better cognitive performance.
Data are mean (SD) or N (%).
Endocrine variables in plasma and saliva are indicated in Table 2. Early morning fasting plasma levels of prolactin were higher in females than in males and higher in EP than in HS, with no interaction between the two factors. Prolactin levels by antipsychotic treatment group are presented in Table 2S. No significant influence of either sex or diagnosis was found for plasma cortisol, and female sex hormones (17-β-oestradiol and progesterone) were not affected by diagnosis. Regarding saliva, cortisol concentration in samples taken at the same time as plasma in the early morning was affected by sex (higher levels in females) and diagnosis (lower levels in EP), with no interaction. Neither CAR nor AUCg was significantly affected by sex or diagnosis.
Hormonal measures of the sample.
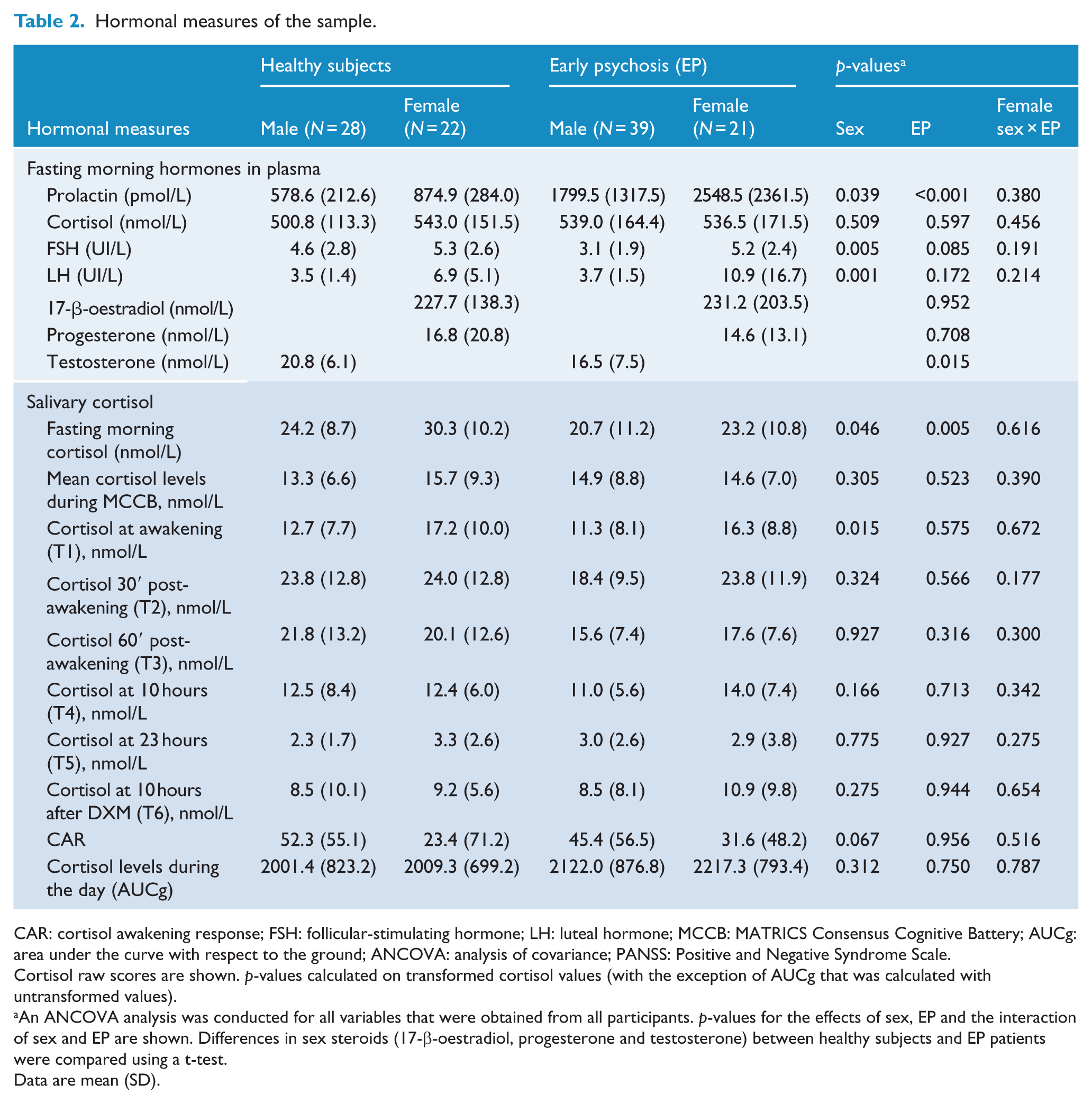
CAR: cortisol awakening response; FSH: follicular-stimulating hormone; LH: luteal hormone; MCCB: MATRICS Consensus Cognitive Battery; AUCg: area under the curve with respect to the ground; ANCOVA: analysis of covariance; PANSS: Positive and Negative Syndrome Scale.
Cortisol raw scores are shown. p-values calculated on transformed cortisol values (with the exception of AUCg that was calculated with untransformed values).
An ANCOVA analysis was conducted for all variables that were obtained from all participants. p-values for the effects of sex, EP and the interaction of sex and EP are shown. Differences in sex steroids (17-β-oestradiol, progesterone and testosterone) between healthy subjects and EP patients were compared using a t-test.
Data are mean (SD).
Correlation of prolactin and sex hormones with cognitive tasks
When all the subjects were studied together (HS and EP, Table 3), the correlational analyses indicated that in men, prolactin levels correlated negatively with testosterone, whereas in women, prolactin correlated negatively with plasma and saliva cortisol, and progesterone correlated positively with oestradiol.
Correlation analysis between hormones and cognitive tasks: sex-stratified analysis.
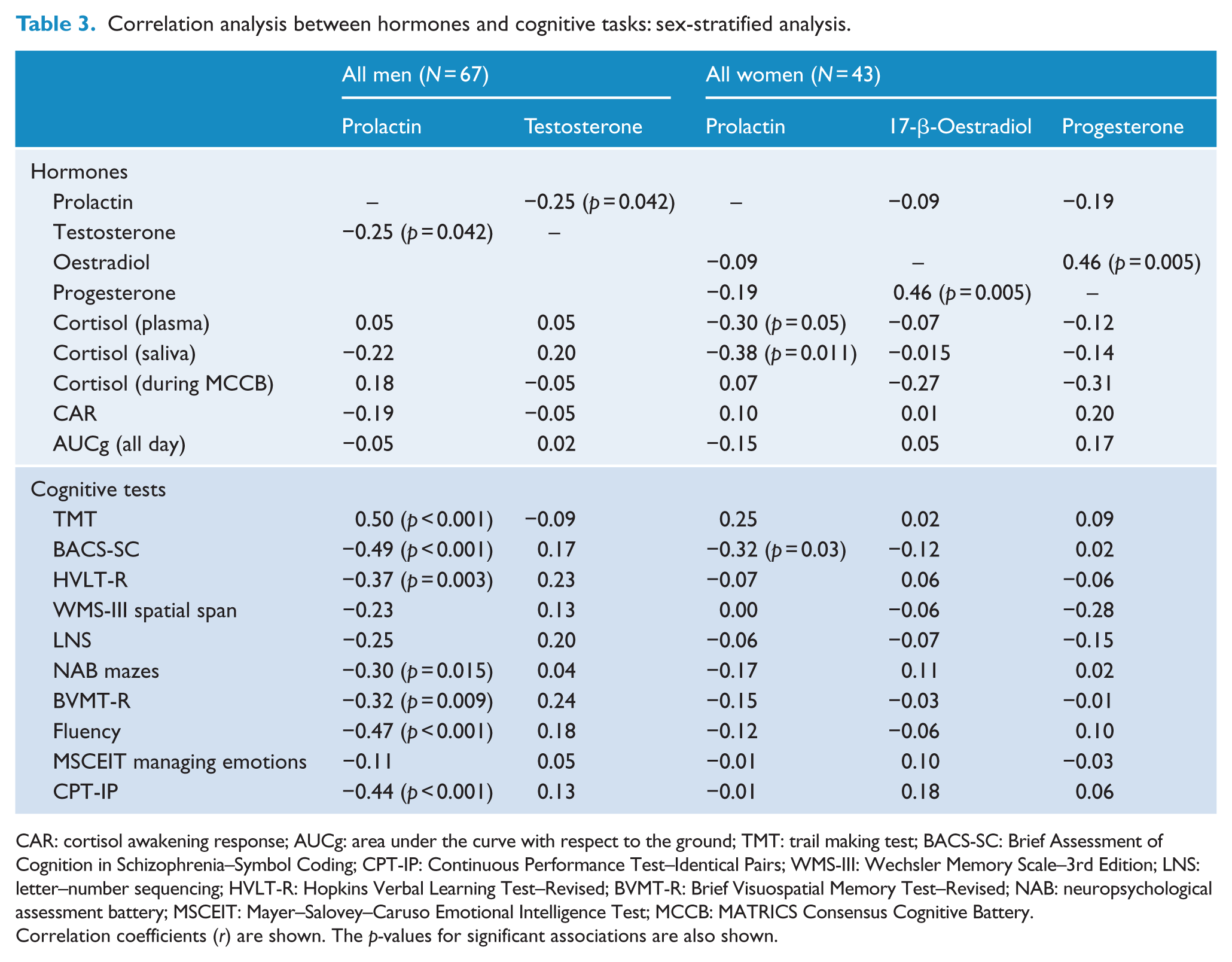
CAR: cortisol awakening response; AUCg: area under the curve with respect to the ground; TMT: trail making test; BACS-SC: Brief Assessment of Cognition in Schizophrenia–Symbol Coding; CPT-IP: Continuous Performance Test–Identical Pairs; WMS-III: Wechsler Memory Scale–3rd Edition; LNS: letter–number sequencing; HVLT-R: Hopkins Verbal Learning Test–Revised; BVMT-R: Brief Visuospatial Memory Test–Revised; NAB: neuropsychological assessment battery; MSCEIT: Mayer–Salovey–Caruso Emotional Intelligence Test; MCCB: MATRICS Consensus Cognitive Battery.
Correlation coefficients (r) are shown. The p-values for significant associations are also shown.
Regarding cognitive tasks, no statistically significant correlations were found with sex steroids. In women, FSH and LH levels did not correlate with cognitive functioning (data not shown). In men, LH levels were not associated with cognitive performance in any task; however, FSH levels were positively associated with CPT-IP scores (r = 0.329, p = 0.007). Prolactin levels correlated with poorer cognitive performance in most cognitive tasks in men and only one cognitive task in women (Brief Assessment of Cognition in Schizophrenia–Symbol Coding [BACS-SC]).
As correlation analyses included both HS and EP patients, we conducted an additional partial correlation analysis adjusting for education status and antipsychotic treatment. In these adjusted correlations, in men, prolactin was significantly associated with trail making test (TMT; r = 0.41, p = 0.001), BACS-SC (r = −0.35, p = 0.005) and CPT-IP (r = −0.30, p = 0.020). In women, prolactin was not associated with any cognitive measure when adjusting for education and antipsychotic treatment. FSH was associated with improved cognition in CPT-IP (r = 0.25, p = 0.050) in men only.
Multiple linear regression analyses
Multiple linear regression analyses in all participants
Multiple linear regression analyses of the association between prolactin and cognitive tasks are presented in Table 4.
The results of the multiple linear regression analyses exploring the association between prolactin and cognitive tasks in all participants.
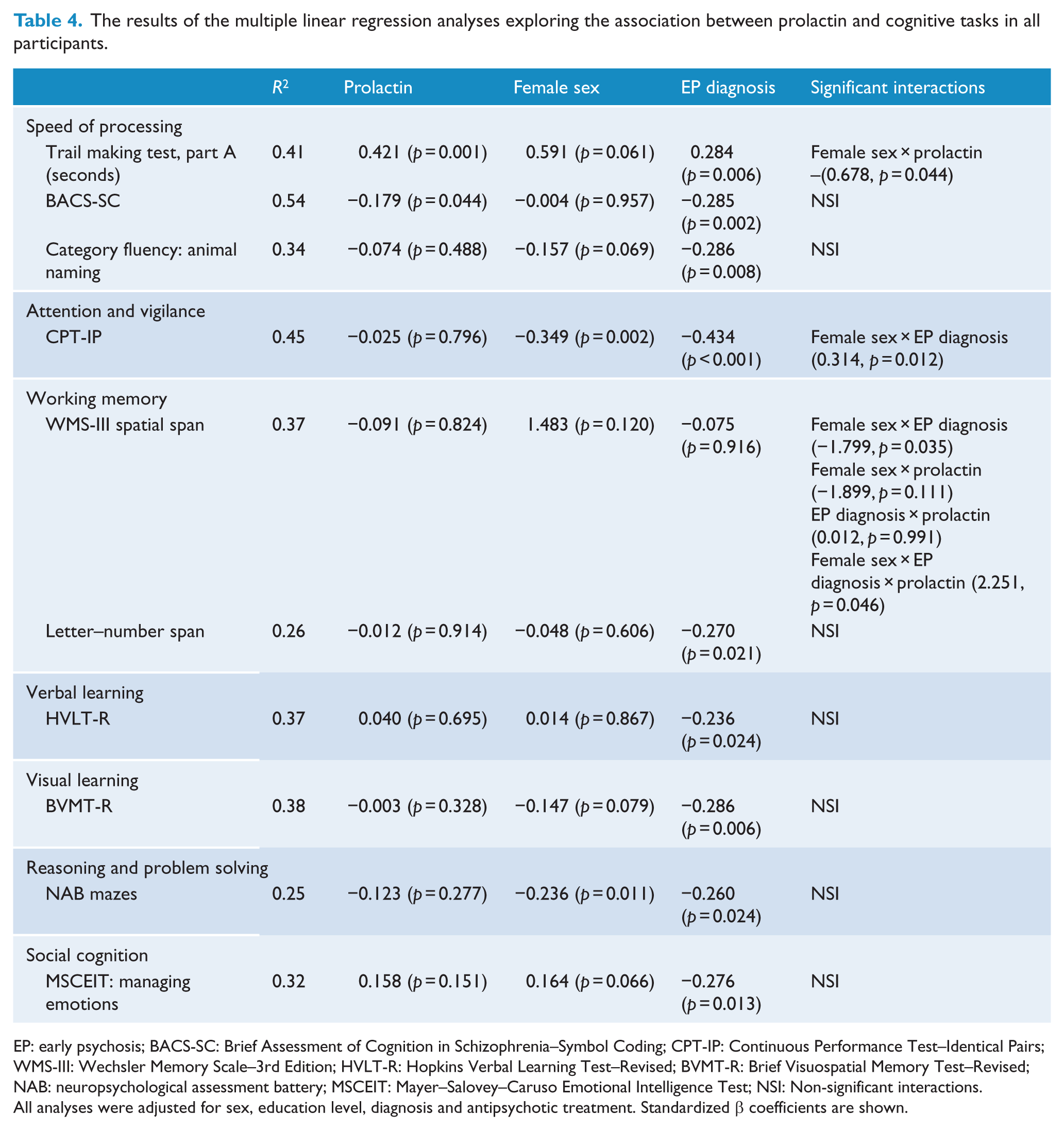
EP: early psychosis; BACS-SC: Brief Assessment of Cognition in Schizophrenia–Symbol Coding; CPT-IP: Continuous Performance Test–Identical Pairs; WMS-III: Wechsler Memory Scale–3rd Edition; HVLT-R: Hopkins Verbal Learning Test–Revised; BVMT-R: Brief Visuospatial Memory Test–Revised; NAB: neuropsychological assessment battery; MSCEIT: Mayer–Salovey–Caruso Emotional Intelligence Test; NSI: Non-significant interactions.
All analyses were adjusted for sex, education level, diagnosis and antipsychotic treatment. Standardized β coefficients are shown.
Processing speed, as measured by the TMT (higher scores reflected lower cognitive performance), was associated positively with prolactin levels and negatively with EP diagnosis, although the sex × prolactin interaction was also statistically significant, indicating that the effects of prolactin on this cognitive task differed between men and women. Another measure of processing speed (BACS-SC) was also affected by prolactin and EP diagnosis: A poorer processing speed was associated with increased levels of prolactin and an EP diagnosis, with no statistical interactions.
For working memory, as measured by the Wechsler Memory Scale–3rd Edition (WMS-III) spatial span task, there were two significant interaction terms in the final equation (a negative female sex × EP diagnosis interaction and a positive female sex × EP diagnosis × prolactin three-way interaction). These two interaction terms mean that female EP patients showed poorer performance in this cognitive task but that this subgroup (female EP) performed better as prolactin levels increased. To better understand these interaction terms, we have included a scatterplot figure as supplementary material (Figure 1S). As shown in this figure, female EP patients scored lower on the WMS-III spatial span task. However, female EP patients with higher prolactin levels showed better performance on this task.
Prolactin levels were not associated with other cognitive tasks, including attention and vigilance, working memory, verbal learning, visual learning, reasoning and problem solving and social cognition.
Multiple linear regression analyses in EP patients: adjusting for negative symptoms
Additional regression analyses were performed to further characterize the relation between prolactin and cognitive impairment in EP patients while adjusting for education status, negative symptoms and antipsychotic treatment. As shown in Table 5, negative symptoms were associated with impaired cognition in all domains but working memory. A negative relationship between prolactin and cognitive performance was observed in all the tasks measuring processing speed. In two of these three tasks (TMT and fluency), a sex × prolactin interaction was observed (the relation was found only in males), whereas on the BACS-SC, no sex-specific effects were found.
Results of the multiple linear regression analyses exploring the association between prolactin and cognitive tasks in patients with an early psychosis.
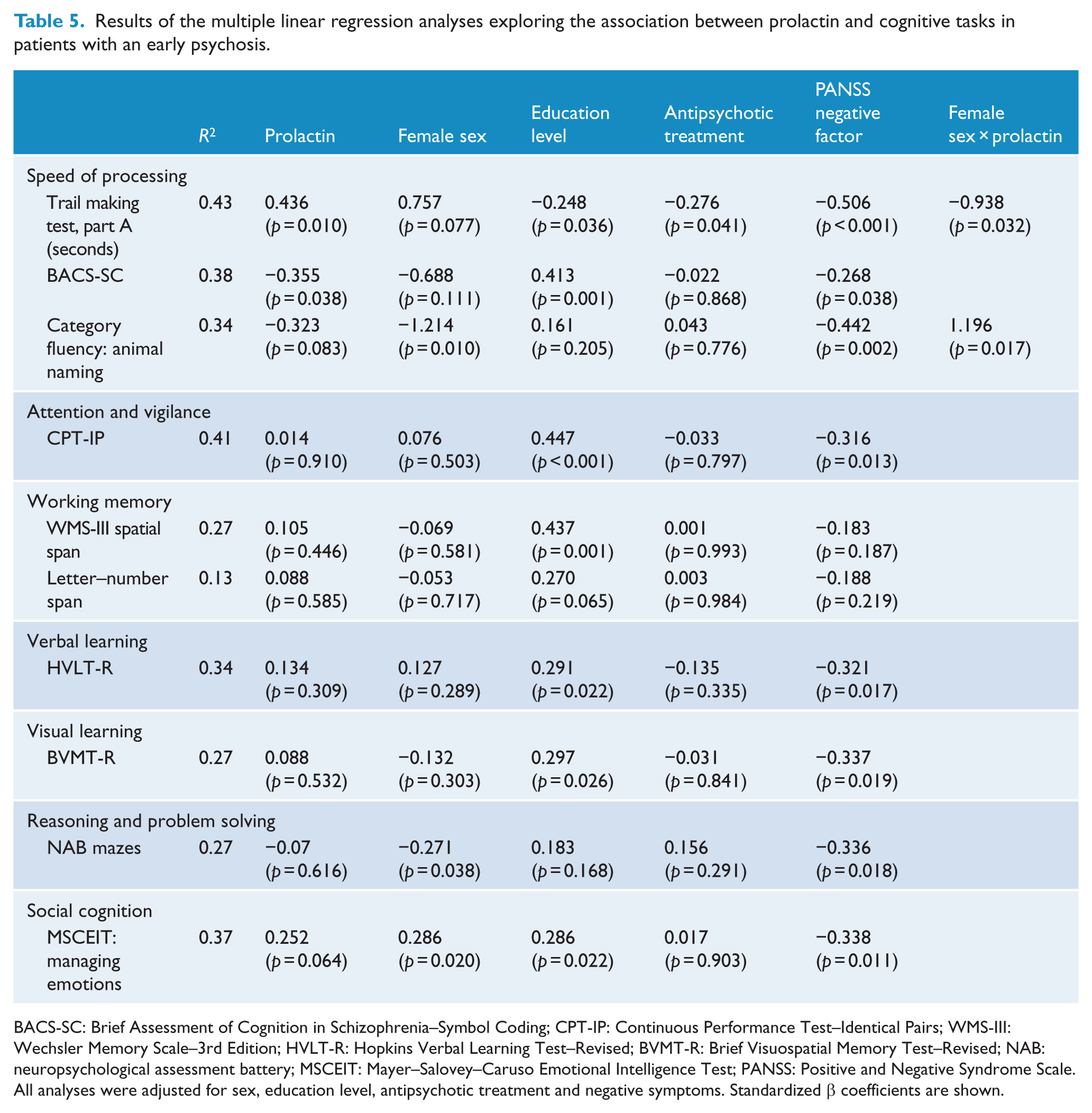
BACS-SC: Brief Assessment of Cognition in Schizophrenia–Symbol Coding; CPT-IP: Continuous Performance Test–Identical Pairs; WMS-III: Wechsler Memory Scale–3rd Edition; HVLT-R: Hopkins Verbal Learning Test–Revised; BVMT-R: Brief Visuospatial Memory Test–Revised; NAB: neuropsychological assessment battery; MSCEIT: Mayer–Salovey–Caruso Emotional Intelligence Test; PANSS: Positive and Negative Syndrome Scale.
All analyses were adjusted for sex, education level, antipsychotic treatment and negative symptoms. Standardized β coefficients are shown.
Multiple linear regression analyses by sex: adjusting for cortisol and sex steroids
We conducted additional multiple linear regression analyses in men and women separately, including sex steroids and cortisol measures (CAR, cortisol levels over the day) as covariates. All these analyses were also adjusted for education level. In men, prolactin levels were negatively associated with impaired performance in all three tasks measuring processing speed: TMT (β = 0.364, p = 0.002), BACS-SC (β = −0.313, p = 0.006) and fluency (β = −0.279, p = 0.027). These significant associations were independent of testosterone levels and cortisol measures. Testosterone levels or HPA axis measures were not related to cognitive performance in any cognitive task.
In women, prolactin levels were associated with poorer performance in two cognitive tasks, along with prolactin by diagnosis interactions: (1) WMS-III spatial span (prolactin effect: β = −1.132, p = 0.009; prolactin × early psychosis interaction: β = 3.406, p = 0.006) and (2) Brief Visuospatial Memory Test–Revised (BVMT-R; prolactin effect: β = −1.065, p = 0.024; prolactin × early psychosis interaction: β = 2.776, p = 0.016). These findings suggest that the pattern in the relationship between prolactin and these two cognitive tasks differs in EP and HS (interactions suggest that higher prolactin levels are associated with poorer performance in HS and better performance in EP; Figures 1S and 2S). These results complement the results of the analyses including all the participants (section ‘Multiple linear regression analyses in all participants’) once they are adjusted for HPA axis measures and sex hormones. Regarding HPA axis measures, cortisol levels during the day were associated with poorer performance in two cognitive tasks: TMT (β = 0.412, p = 0.022) and Hopkins Verbal Learning Test–Revised (HVLT-R; β = −0.345, p = 0.030). Neither oestradiol nor progesterone was associated with cognitive performance in any task.
Discussion
This work demonstrates that the previously reported association between high prolactin levels and impaired cognitive processes in EP is restricted to men. These sex differences were not driven by a better cognitive profile or lower prolactin levels in women. In men, neither testosterone levels nor cortisol levels moderated the relationship between prolactin and cognitive performance in processing speed tasks. In women, prolactin levels were not associated with processing speed tasks, although we observed a negative effect of prolactin on verbal learning and spatial working memory in female HS.
Given our previous results showing an association of high prolactin levels with impaired cognition in men and women with EP (Montalvo et al., 2014), we explored whether sex can affect such a relationship. As expected (Cookson et al., 2012; Horseman and Gregerson, 2013), we observed higher levels of prolactin in EP. Although prolactin levels in the controls were higher in women than in men, the specific effect of the diagnosis of a psychotic disorder was similar in both sexes. However, when the correlation between plasma prolactin and cognitive performance was studied in all the subjects, marked sex differences were observed: In men, prolactin was associated with an impairment in most cognitive domains, whereas in women, only a component of processing speed (BACS-SC) was negatively affected. The reason for the observed sex differences in the relationship between prolactin and cognition is not clear. Although EP men showed lower levels of testosterone than HS, testosterone levels were not associated with cognitive performance in any cognitive task. Some studies in the literature have reported that testosterone levels may modulate cognitive functioning in people with schizophrenia (Moore et al., 2013), although other studies have not reported such an association (Halari et al., 2004). In relation to female sex steroids, we did not find an association between oestradiol or progesterone and cognitive performance in women. These results are in accordance with previous studies of female patients with schizophrenia (Rubin et al., 2015), although other studies have reported that higher progesterone levels are associated with poorer performance in spatial memory tasks (Halari et al., 2004). We would like to clarify that markers of hypothalamic–pituitary–gonadal axis function in our study are peripheral and that central nervous system markers were not assessed. The reproductive age of women in our sample could bring some neuroprotection from oestradiol at the central nervous system that could partially explain some of the sex differences reported in our study.
In a previous study by our group of HPA axis measures of cognition in EP patients (Labad et al., 2016), we did report sex differences in the relationship between HPA axis measures and cognitive tasks. In our current study, the addition of two cortisol measures (CAR and cortisol levels over the day) or sex steroids in the linear regression analyses did not modify the relationship between prolactin and cognitive functioning in men or women, suggesting that prolactin may have a direct effect on cognition that is not moderated by the HPA or hypothalamic–pituitary–gonadal axes. However, in women, we found that higher cortisol levels during the day were associated with poorer processing speed and verbal learning, pointing to the sex-specific relationship between HPA axis measures and cognitive abilities that was found in our previous study (Labad et al., 2016).
Sex differences in the relationship between prolactin and cognition may be explained by sex-specific effects at the brain level. A sexual dimorphism has been described in patients with schizophrenia, particularly in the cortex, with men showing smaller cortex volumes, relative to cerebrum size, compared to women (Goldstein et al., 2002). Another study reporting reduced grey matter volume in the prefrontal cortex of patients with schizophrenia has shown similar reductions in dorsolateral prefrontal regions in men and women but larger reductions in men, compared to women, in the dorsomedial cortex (Gur et al., 2000). Prefrontal cortex volumes correlate with executive function tasks in patients with schizophrenia (Antonova et al., 2004). Although speculative, it is plausible that the sex-specific negative association of prolactin with the speed of processing tasks might rely on sex-specific volumetric brain abnormalities in the prefrontal cortex.
When adjusting for covariates including antipsychotic treatment and negative symptoms – two known variables that affect cognition (Bora et al., 2009; Sakurai et al., 2013) – of all cognitive tasks, poorer performance in those tasks related to processing speed was significantly associated with prolactin levels. These results are consistent with previous reports that have found an association between prolactin levels and cognitive tasks involving processing speed (Bratek et al., 2015; Montalvo et al., 2014). Antipsychotic-induced hyperprolactinaemia, which is caused by tuberoinfundibular blockade of D2 receptors (Horseman and Gregerson, 2013), may reflect the blockade of D2 receptors in other dopaminergic pathways including the striatum, which causes extrapyramidal symptoms, or the mesocortical pathway and may be related to worsening of cognitive and negative symptoms. The multivariate analyses suggest that there is an independent effect of prolactin, as all the analyses were adjusted for sex, education level, antipsychotic treatment and negative symptoms.
The possible mechanisms linking higher levels of prolactin to impaired cognitive function are not known; however, a direct action on the brain is quite possible. Only one study has investigated the influence of prolactin on cognitive functions in rats (Torner et al., 2013), showing impaired object recognition but not spatial memory deficit in hyperprolactinaemic rats; nonetheless, a wider effect of prolactin on brain function is plausible. It is well known that prolactin has access to the brain, although the precise mechanisms are still unclear. While the hypothesis has been accepted for years that the hormone accesses the brain through prolactin receptors located in the choroid plexus, recent evidence in mice with deletion of the receptor has demonstrated that it is not necessary to the mechanism and that endothelial cells of brain capillaries, rather than the choroid plexus, might be the route of transport (Brown et al., 2016). Regardless of the mechanisms, prolactin accesses the brain, where it can exert important behavioural effects, most of them related to reproductive and maternal behaviour. Importantly, for the relationship between prolactin and EP, there is evidence in animals that prolactin might potentiate striatal DA release in vitro and in vivo (Chen and Ramirez, 1988; Perkins and Westfall, 1978; Ramirez, 1983), suggesting a direct effect on the striatum. In fact, the presence of the receptor has been detected by immunohistochemistry in the cortex and striatum of the rat (Roky et al., 1996). However, in the mouse, there is no evidence of the expression of the prolactin receptor in either the prefrontal cortex or the dorsal or ventral striatum (Brown et al., 2010), suggesting that the possible influence of prolactin on cortical or striatal function might involve terminals arising from other brain areas where the receptor is expressed. Nothing is known in humans about the possible influence of hyperprolactinaemia on brain function.
The main limitation of our study is the cross-sectional design, which does not allow us to infer causality. Further prospective studies may overcome this limitation by repeatedly assessing prolactin levels and cognitive performance over time to explore whether persistent hyperprolactinaemia is a risk factor for cognitive decline in subjects with psychoses, particularly in men. Most of the patients in our study were receiving antipsychotic drugs, which may affect both prolactin and cognitive performance. Antipsychotic-induced extrapyramidal symptoms, which could affect processing speed, were not assessed. As our Early Intervention Service is an outpatient service, some patients who have previously been admitted to the referral Acute Psychiatric Unit are stabilized before attending our service. Moreover, patients must be informed of the research project and must sign the informed consent form; thus, in most cases, EP patients are clinically stable at recruitment. For all these reasons, most of the patients were taking antipsychotic drugs. We tried to address this limitation by adjusting most of the multivariate analyses for antipsychotic treatment. The fasting morning samples in plasma and saliva were obtained in a clinical setting, which may be considered a stressful environment for some participants. However, as EP patients had frequent appointments in this clinical unit, it is plausible that environmental novelty was lower for EP than for HS, which could partially explain the increased fasting morning salivary cortisol levels in this latter group. Finally, menstrual cycle status was not ascertained clinically because we preferred to adjust for sex steroids (oestradiol and progesterone) in the multivariate analyses. Up to 45% of women with psychotic disorders receiving antipsychotic treatment have irregular menses (Gleeson et al., 2016), which makes assessment of female psychotic patients with the same menstrual status difficult. Prolactin concentrations are influenced by oestradiol levels, with significantly higher levels during the ovulatory and luteal phases than during the follicular phase (Franchimont et al., 1976). For this reason, the multiple linear regression analyses were also adjusted for oestradiol concentrations.
If our findings are replicated in further studies, they may have potential clinical implications. Currently, the monitoring of hyperprolactinaemia side effects is focused on sexual and reproductive functions. However, our study suggests that prolactin may also have brain effects that could influence cognitive processes, which needs to be considered when treating patients with psychotic disorders and antipsychotic-induced hyperprolactinaemia.
In conclusion, our study suggests that there are sex differences in the relationship between prolactin levels and impairment of cognitive tasks involving processing speed in the early stages of a psychotic disorder. The detrimental effects of prolactin on cognition are not moderated by the HPA or hypothalamic–pituitary–gonadal axes.
Footnotes
Declaration of Conflicting Interests
J.L., I.M. and V.S.-G. have received honoraria for lectures or advisory boards from Janssen-Cilag, Otsuka or Lundbeck. The rest of the authors have no biomedical financial interests or potential conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the Instituto de Salud Carlos III (FIS, PI10/01607).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
