Abstract
Objective:
To examine the provenance and implications of seizure threshold titration in electroconvulsive therapy.
Background:
Titration of seizure threshold has become a virtual standard for electroconvulsive therapy. It is justified as individualisation and optimisation of the balance between efficacy and unwanted effects.
Result:
Present day threshold estimation is significantly different from the 1960 studies of Cronholm and Ottosson that are its usual justification. The present form of threshold estimation is unstable and too uncertain for valid optimisation or individualisation of dose. Threshold stimulation (lowest dose that produces a seizure) has proven therapeutically ineffective, and the multiples applied to threshold to attain efficacy have never been properly investigated or standardised. The therapeutic outcomes of threshold estimation (or its multiples) have not been separated from simple dose effects. Threshold estimation does not optimise dose due to its own uncertainties and the different short-term and long-term cognitive and memory effects. Potential harms of titration have not been examined.
Conclusion:
Seizure threshold titration in electroconvulsive therapy is not a proven technique of dose optimisation. It is widely held and practiced; its benefit and harmlessness assumed but unproven. It is a prematurely settled answer to an unsettled question that discourages further enquiry. It is an example of how practices, assumed scientific, enter medicine by obscure paths.
Electroconvulsive therapy (ECT) stimulus titration has become a treatment standard. The most recent draft RANZCP Professional Practice Guideline for ECT says ‘relative efficacy … is dependent on dose relative to the individual’s seizure threshold’ and assumes threshold titration in its dosing recommendations (RANZCP, 2017). Two state Health Departments now virtually endorse it as the expected process (South Australian Department of Health, 2016; Victorian Department of Health, 2015). In the United Kingdom, the Royal College of Psychiatrists repeats the recommendations for stimulus titration (Waite and Easton, 2013). It is implicitly assumed in research. Much of the recent literature repeats that titration is now the best practice. This is typical: ‘in modern ECT research, stimulus titration has clearly emerged as the most commonly utilized electrical dosage method that balances efficacy and cognitive side effects’ (Rasmussen, 2015) (this conclusion quotes research that does not actually confirm that titration ‘balances efficacy and cognitive effects’). Richard Abrams (2002b), 15 years ago, pointed out that no relationship had been demonstrated between the therapeutic effect and either the threshold or seizure duration. He argued for age-based dosing.
Whence comes threshold stimulus titration?
The accepted wisdom that the seizure had the therapeutic effect goes back to Meduna (Fink, 1984) but the idea that electricity above threshold added to cognitive deficit without adding benefit was largely drawn from Cronholm and Ottosson’s (1960) studies of bilateral ECT. Cronholm and Ottosson talked about ‘threshold’ and their studies looked at ‘moderately above’ and ‘considerably above’ threshold stimulation. They used a Siemens Konvulsator, a ‘quarter sine wave’ pulsed constant current machine and later reported that ‘moderately above threshold’ in the 1960 study meant holding down the button until the tonic phase developed (D’Elia et al., 1983). Although their measures were incommensurate with our current measures, the ‘moderately above threshold’ dose was approximately equivalent to 200 mC (Cronholm and Ottosson, 1963). This was not a large dose by present standards, yet clearly above threshold, given that they maintained the stimulus until the tonic phase seizure was established (and they restimulated if not happy with the quality of the seizure). This was Cronholm and Ottosson’s idea of threshold, used in their 1960 study, and still used in their 1983 studies (D’Elia et al., 1983). Their idea of threshold was a stimulus that lasted long enough to push the evolving seizure far enough along its path that it reliably continued to completion. That dynamic was duration of stimulus. The first clear statement of the later (and current) idea of threshold is by Sackeim et al. (1986). He refers back to Cronholm and Ottosson but his approach was to find the level of stimulus that would trigger the seizure by repeated stimuli that increased the frequency of the stimulus from 20 to 140 pulses per second (Sackeim et al., 1993). This was a dynamic of intensity. It is difficult to trace how the idea of threshold developed from Cronholm and Ottosson’s duration of stimulus in 1960 to the intensity of stimulus described by Sackeim in 1986 (Abrams, 2002a).
A justification for stimulus titration is the hope that threshold points to the point of optimum balance between efficacy and unwanted effects. Writing about threshold in 1987, Sackeim et al. (1987a) references Cronholm and Ottosson’s practice (D’Elia et al., 1983) and implies that the balance of efficacy and side effects turns on threshold (while pointing out that had not been substantiated). Sackeim’s (1987a) study confirmed that, for unilateral treatments, doses at the threshold were ineffective and that higher doses were needed. Sackeim et al.’s (1993) paper showed again that unilateral treatment at threshold had little therapeutic effect but claimed that the effect of high-dose unilateral treatment was related to the multiple of threshold, not dose, because ‘the absolute electrical dose had no relation with clinical outcome’(p. 842) (a close reading of that paper suggests that conclusion comes from multivariate analysis that entered intercorrelated independent variables; the significance of threshold and the negation of dose may reflect the statistical method).
The vicissitudes of titration
When threshold dosing proved ineffective, electrical doses were increased by multiplying the threshold. Up to 12 times threshold was used (McCall et al., 2000). There was no way to disentangle threshold from absolute dose effects in the reports of the studies, yet threshold titration was increasingly assumed.
There was a thread of comment and research that continued to raise problems about the titration assumption. European and American studies found no advantage from titration and commented that it led only to more treatments without corresponding therapeutic improvement (Aten et al., 2015; Bennett et al., 2012; Shapira et al., 1996).
Thoughtful commentators have pointed out that threshold is not clearly fixed (Abrams, 2002b). A threshold seizure was arbitrarily defined as at least 25 seconds in the research literature (Sackeim et al., 1987b). That 25-second criterion is not followed in many clinics or in the proposed RANZCP guidelines (RANZCP, 2017) where any seizure activity counts as threshold. The threshold is affected by the wave form, that is, pulse width (Swartz and Manly, 2000), frequency, current, stimulus duration (Andrade et al., 2002), the position and the size of the electrode (Peterchev et al., 2010) and the starting dose and number of titration stimuli (Swartz, 1990). It is affected very significantly by the anaesthetic (Gálvez et al., 2015; Lyndon and Wilson, 2015; Patel et al., 2006).
It has been argued that ‘charge’ millicoulomb (mC) is the best measure of dose (Sackeim et al., 1987b). The mC is not a measure that indicates many of the important characteristics of the stimulus. A coulomb is a measure like a litre of water. A litre delivered through a hose at high pressure has a very different effect from a litre poured slowly out of a jug. A millicoulomb delivered at high current is different from a millicoulomb at low current. Most of the electrode current passes through the scalp (Sackeim et al., 1987b). It is the electrical field that induces current in the brain and hence variations in the energy or the current pressure are determined by the amperage, frequency, duration and pulse width settings of the machine that are not indicated by the millicoulomb measure (Peterchev et al., 2010). Presuming operators accept the settings of the manufacturer that might be of little consequence. However, sophisticated machines have proprietary dosage adjustment algorithms that are opaque to the unsophisticated user and different between manufacturers (Mecta Corporation, 2013; Somatics LLC, 2015). The inconsistency undermines the precision of threshold measurement that seems to be part of the rationale for titration (Chanpattana, 2001).
In its history, derivation, justification and practice the argument for threshold titration is incoherent. So what? If it can be shown to work, who cares about coherence. Much of medicine is composed of incoherent practices that have been shown to work. The science (in the sense of knowing) is in demonstrating that it works, not explaining how it works. But stimulus titration has not been shown to work.
What is at stake?
In the argument about the validity of stimulus titration, we can lose sight of what is at stake. First is optimising the balance between benefits and harms. The second is the individualisation of treatment to the patient. Both of these are important, and titration seems to offer something.
Balancing benefits and harms
We are trying to find an optimum balance between two probably monotonic relationships: between dose and mood and between dose and memory and cognitive impairment (see Figure 1).
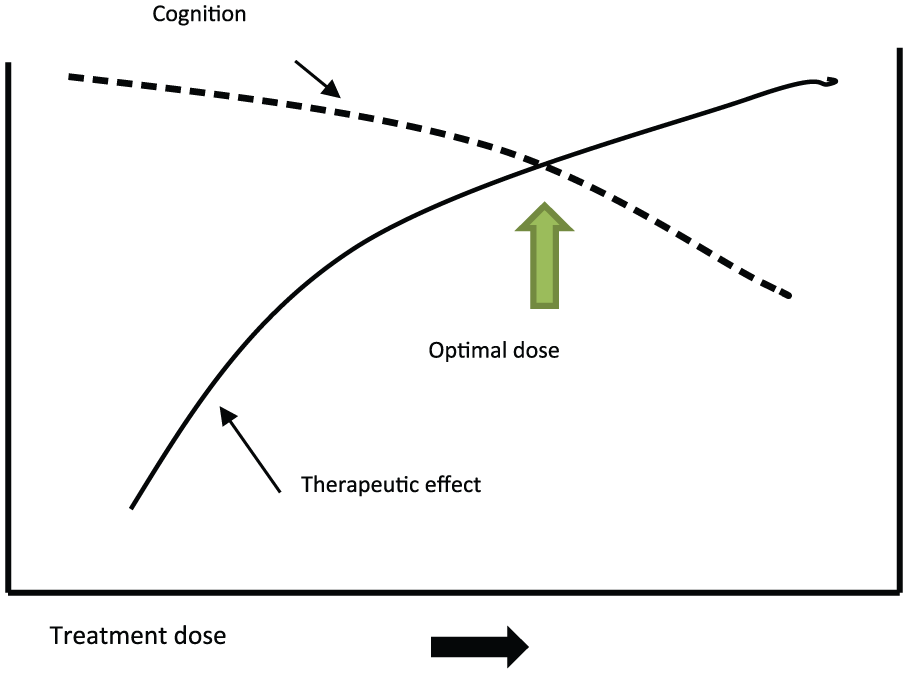
The balance point.
In 1960, following Cronholm and Ottosson, the optimum balance point seemed (only seemed) to be at the seizure threshold. That turns out not to be the balance point, yet we have imagined that we have evidence that some multiple of the threshold gives us that balance point.
The wisdom descending from Cronholm and Ottosson (1960) was that doses exceeding what is required for seizure add nothing except unwanted effects. After 50 years, research seems to point to a straightforward relationship between dose and efficacy up to doses many times above what is required for a seizure (Abrams, 2002b). What limits doses are the memory and cognitive effects.
Cognitive (memory) effects
There appears to be little doubt of the relation between dose and cognitive and memory effect. However, the measurement of cognitive and memory effects turns out to be very difficult because of problems not only with measurement but also problems with description of what we want to measure.
Cronholm and Ottosson (1961) showed ECT caused a short-term (1 week) impairment of ‘retention’ but, simultaneously, ‘learning’ improved proportionally with the improvement in mood with ECT. This confirmed the findings of at least eight studies before 1960.
In Sackeim et al.’s (1993) studies, the immediate cognitive effects of high- and low-dose ECT were very varied (i.e. showed standard deviations much larger than differences of mean scores). After 2 months of treatment, there was little difference in the cognitive effects between treatment forms, unilateral or bilateral, high or low dose. Sackeim’s later studies (Sackeim et al., 2000, 2008) found a statistically significant but very small cognitive effect in only one test of autobiographical memory 2 months after treatment. His multi-site study (Sackeim et al., 2007) showed many deficits in the days after ECT but improvement above baseline in nine of eleven tests at 6 months.
McCall et al. (2000) compared cognitive effects of titrated versus fixed high-dose ECT within 3 days of treatment. They confirmed that higher doses increased both antidepressant effect and short-term (same day and two day) cognitive impairment. This study has been cited repeatedly to show a benefit for threshold titration and so it deserves a close reading. It shows a relationship between dose and both positive antidepressant effect and negative cognitive effect, a trade-off that is seen repeatedly. Between fixed high dose and titrated dose there were observed differences in post-treatment reorientation time, and the Mini-Mental State Exam (MMSE) and autobiographical memory, only two of the nine cognitive tests administered. The dose was translated into three levels of threshold multiples to reinforce that finding of dose on therapeutic effect and for reorientation time and autobiographical memory (and not for any other cognitive tests).That regression showed that when the threshold multiple was entered in the equation, all other independent variables, including absolute dose, became non-significant. Both the threshold multiple and absolute dose were entered into a logistic regression (an unusual choice for continuous dependent data). Threshold multiple and absolute dose are highly correlated, and one variable attracts most of the variance by being a proxy for the other. This does not indicate that absolute dose is irrelevant, only that statistical technique is not trouble-free and interpretation is slippery.
One must remember that the finding that dosing by multiple of threshold had less cognitive effect applied to only two of nine tests at 1 and 3 days. There was no follow-up beyond 3 days. Dosing by threshold multiple had less therapeutic effect and the threshold-dosed subjects had on average 1.5 more treatments. This study does not prove the advantage of stimulus titration in the way the study is repeatedly cited. The study does underline the well-established short-term cognitive impact and therapeutic effect of higher dose as well as the trade-off between therapeutic effect and cognition.
Notwithstanding, it is safe to say that recovery of orientation, new learning and recent recall are measurably impaired immediately after ECT, most obviously by bilateral ECT. As soon as 3 weeks after ECT, there is no reliably measurable defect in most cases and, most commonly, there is a measurable improvement from the pre-treatment performance in depressed subjects (Abrams, 2002a; Sackeim et al., 2007). Reported individual cases of long-term impaired memory seem to be related to bilateral treatment. A meta-analysis by Semkovska and McLoughlin (2010) concluded, ‘Cognitive abnormalities associated with ECT are mainly limited to the first 3 days posttreatment. Pretreatment functioning levels are subsequently recovered. After 15 days, processing speed, working memory, anterograde memory, and some aspects of executive function improve beyond baseline levels’. Retrograde autobiographical amnesia has turned out to be much harder to grasp and measure (Semkovska and McLoughlin, 2013) despite the frequent reports of this memory impairment. The jury remains ‘out’ about the findings (Semkovska and McLoughlin, 2014).
Autobiographical memory overlaps with subjective memory which is the patient’s report of the feeling of altered memory but one that is not measurable by objective tests. It is difficult to know how to describe a complaint of memory disturbance that cannot be measured even by subtle tests. The nature of subjective memory impairments is not agreed and difficult to answer dogmatically but patient’s complaints seem indubitable (Semkovska and McLoughlin, 2014). The rates of persisting subjective memory impairment beyond 6 months can be alarmingly high (Rose et al., 2003) but there is little evidence about how that relates to ECT dose and no evidence that titrated versus non-titrated dosing yields different long-term subjective memory effects.
If short-term cognitive effects predict long-term effects, it is important to measure and minimise short-term effects (Donel Martin, personal communication, 8 May 2017). We do not know if short-term effects predict long-term effects nor is there any way to reduce possible cognitive effects other than reducing dose and trading away efficacy. Researchers face an apparent contradiction that short-term cognitive loss is related directly to dose while long-term cognitive gain is related to efficacy which is related to dose. Which should get priority? The transience of the immediate cognitive effect confounds the calculation of the balance of therapeutic effect and harms.
Individualisation of treatment
Individualisation of treatment is a worthwhile goal and is a primary justification for stimulus titration. However, individualisation needs an identifiable and safe criterion. Adjusting the dose of a medicine to body weight is a sensible therapeutic individualisation but you need to demonstrate that it is useful and safe. If you adjusted lithium dose by body weight you would soon be in real trouble. For lithium, we have, in serum level, an individualisation for safety and possibly efficacy (Severus et al., 2009).
ECT stimulus threshold has not been demonstrated to usefully individualise ECT dose. In fact, the first attempt to individualise by threshold demonstrated its lack of effect. The multiplication of threshold dose proceeded without any proof that threshold was useful. What looks like a good idea has its origin in a misreading of older research (Cronholm and Ottosson, 1960). While that old research was of good quality, it reflects the technology and discriminations of a past time and has required reinterpretation.
Could titration harm?
If stimulus titration turns out to be a non-scientific practice, so what? But could titration be harmful? It will not maim people like a wrongly judged lithium dose might. But it is not proven that it is harmless. Titration usually adds at least one session of treatment (Bennett et al., 2012). We simply do not know if the number of treatment sessions (and hence anaesthetics, induced paralysis and ventilation) contribute to cognitive and memory effects. The multipliers applied to a titrated threshold remain a wild card and in some cases can dictate worryingly high dosages which newer machines have been designed to apply.
We do not currently have the means to answer this reasonable question.
Conclusion
We have taken on a practice of stimulus titration believing that it is based on scientific evidence but find on closer examination that it might not be so. Stimulus titration does not provide the hoped-for answers to questions about ECT efficacy and harms.
If there are differences between threshold titration and formula dosing (such as dosing by age and gender), they remain too subtle to be obvious in research, let alone in clinical practice where differences in technique are swamped by individual patient differences.
If the task is to find the best balance between efficacy and unwanted effects, then our belief that we have found it in titration distracts us from more subtle adjustments that we might make on careful observation of the patient. Richard Abrams’ (2002c) provocation is still unanswerable:
‘… the seizure threshold is a “moving target” that varies with the stimulus parameters used to elicit it, … an infinite variety of seizure thresholds exist in the same patient, with no rationale provided for choosing among them. Far better to take the bull by the horns and give every patient a treatment that has the best chance of success …. The best way to determine the ECT dose would be to measure the patient’s clinical antidepressant response to each treatment and adjust the stimulus dose accordingly’.
What form of ECT has the best chance of success is still a question, 80 years after its first use. Threshold titration does not provide the answer to that question, and our commitment to titration may only halt a curiosity which might lead to practical improvement. It should remind us that practices come into medicine along paths of history and influence that are not always the paths of scientific ‘truth’.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
