Abstract
Objective:
This review examines the existing evidence for the relationship between borderline personality disorder and polycystic ovary syndrome, and to identify commonalities in etiological mechanisms of borderline personality disorder and polycystic ovary syndrome that might explain the relationship between these seemingly disparate disorders.
Methods:
A search of Medline, EMBASE and Cochrane Central was undertaken on 5 December 2016 to identify studies investigating women with borderline personality disorder and polycystic ovary syndrome (or symptoms and markers specific to polycystic ovary syndrome).
Results:
Nine studies were identified, including three cross-sectional studies investigating symptoms of polycystic ovary syndrome in women with borderline personality disorder, two cross-sectional and one cohort study examining the prevalence of psychiatric diagnoses in women with polycystic ovary syndrome and three case reports of comorbid borderline personality disorder and polycystic ovary syndrome.
Conclusion:
Overall, the literature shows women with borderline personality disorder to have higher than expected serum androgen levels and incidence of polycystic ovaries, which can be key features of polycystic ovary syndrome. However, this research is still in its infancy, which limits our understanding of this potential comorbid phenomenon. Given the emerging anecdotal and empirical evidence to date, a theoretical discussion of the potential psychoneuroendocrinological mechanism underlying the borderline personality disorder and polycystic ovary syndrome comorbidity is provided. Further rigorous studies using standardized diagnostic criteria for polycystic ovary syndrome are warranted. Specifically, the use of prospective controlled cohort studies may be able to determine the causality and temporality of observed comorbid borderline personality disorder and polycystic ovary syndrome.
Keywords
Introduction
Borderline personality disorder (BPD) is a serious and highly prevalent psychiatric disorder characterized by emotional dysregulation, disturbed impulse control and recurrent self-harm, self-mutilation and suicidality (American Psychiatric Association, 2013). Individuals with BPD can present as volatile when symptoms of BPD fluctuate rapidly and are severe (e.g. rage and self-injurious behavior). Research has shown that 75% of diagnosed individuals have attempted suicide at least once, and 10% of them complete suicide (Black et al., 2004). BPD is the most common personality disorder within clinical populations (Leichsenring et al., 2011) and is estimated to have a prevalence of 2.7% to 5.9% among the general population (Grant et al., 2008; Quirk et al., 2016; Trull et al., 2010). BPD is associated with high healthcare usage (Leichsenring et al., 2011), and symptoms can impair the individual’s employment, life in the community and family interactions, with significant individual and social impact. Psychotherapy is the first-line treatment for BPD, followed by the use of adjunctive psychotropic medication such as mood stabilizers, antidepressants and antipsychotics to target specific symptoms of BPD (Choi-Kain et al., 2016; Paris, 2009).
Polycystic ovary syndrome (PCOS) is the most common endocrine disorder in women of reproductive age (Goodarzi et al., 2011). Prevalence figures vary according to the diagnostic criteria used, of which there are three current – the National Institutes of Health (NIH) criteria (Zawadski and Dunaif, 1992), the Rotterdam criteria (The Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group, 2004) and the criteria recommended by the Androgen Excess and PCOS Society (AE-PCOS) (Azziz et al., 2009). General estimates reveal that PCOS affects between 8.7% and 21% of women of reproductive age (March et al., 2010). The four key features of PCOS are hyperandrogenemia, clinical hyperandrogenism, oligo-anovulation and polycystic ovaries (Azziz et al., 2009). Over time and in accordance with stage of life, these symptoms of PCOS can result in infertility or pregnancy complications including gestational diabetes and pre-eclampsia (Fauser et al., 2012). PCOS has also been associated with increased risk of endometrial cancer of up to fourfold (Barry et al., 2014; Gottschau et al., 2015), as well as kidney, colon and brain cancers, although this requires further research (Gottschau et al., 2015). Unsurprisingly, it is estimated that PCOS and its associated health risk factors account for millions of healthcare costs per year (Teede et al., 2010). Treatment for PCOS is often targeted at specific symptoms or individual needs, and comprise pharmacologic interventions such as oral contraceptives and metformin, and lifestyle modifications, particularly for obese PCOS patients.
BPD and PCOS share a higher prevalence of several specific, serious health consequences. Women with PCOS are significantly more likely to develop metabolic complications such as obesity and type 2 diabetes, and cardiovascular disease (Fauser et al., 2012). Rates of metabolic disturbance are two to four times higher in women with PCOS compared to women without PCOS (Moran et al., 2010), with the highest rates of metabolic disturbance observed within the subset of PCOS patients with hyperandrogenemia and oligomenorrhea (Fauser et al., 2012). Likewise, emerging literature has shown that BPD is also associated with obesity, type 2 diabetes and cardiovascular disease (Dixon-Gordon et al., 2015; Frankenburg and Zanarini, 2011; Kahl et al., 2013). Recently, rates of metabolic syndrome have been shown to be up to twice as high in the BPD population compared to other adults in primary care (Kahl et al., 2013). BPD and PCOS are also both strongly associated with the psychological consequences of mood and anxiety disorders (Scaruffi et al., 2014; Tomko et al., 2014).
These commonalities as well as anecdotal clinical observations have sparked a small, growing body of new research that points to an increased risk of comorbidity between the severe psychiatric disorder of BPD and the physical, endocrinological disorder of PCOS. Individuals with comorbid conditions are often more complicated to treat and manage. Evaluating the evidence of an underlying relationship between BPD and PCOS, with focus on commonalities in etiological mechanisms, can potentially provide some insight into causal directions of these significant diseases and have important implications for healthcare providers regarding the complex treatment and management requirements of patients who suffer from these conditions. An improved understanding of BPD and PCOS comorbidity, as has been only recently recognized within the literature, will potentially serve to improve treatment approaches for both disorders.
Therefore, this review aims (1) to examine existing evidence for the relationship between BPD and PCOS and (2) to examine commonalities in etiological mechanisms of BPD and PCOS that might explain the relationship between these seemingly disparate disorders.
Materials and methods
Data sources and search methods
Electronic searches of Medline, EMBASE and Cochrane Central were performed on 5 December 2016 to identify all published studies. Search strategies were conducted using systematic methods and contained subject headings and key words for ‘borderline personality disorder’, ‘personality disorder’, ‘polycystic ovary syndrome’ and ‘hyperandrogenism’ (see Appendices A, B and C). The reference lists of all relevant articles were then examined to identify further relevant studies.
Study selection
Inclusion criteria
This review was of international scope and included published quantitative and qualitative studies of any methodological design (e.g. randomized controlled trials and case reports). Articles that comprised data on women with BPD as either the population or symptomology of focus, and which also comprised data on PCOS or symptoms and markers specific to PCOS, were included. Study selection also extended to articles reporting on Cluster B personality disorders without being specific to BPD due to the descriptive similarities of personality disorders within this diagnostic cluster (American Psychiatric Association, 2013). For example, personality disorders in Cluster A tend have more ‘odd’ and ‘eccentric’ personality traits, whereas Cluster B commonly have personality traits that are ‘dramatic’, ‘emotional’ and ‘erratic’ (American Psychiatric Association, 2013). All pertinent studies meeting these criteria, and published from electronic database inception to 5 December 2016, were included in this review.
Exclusion criteria
We excluded articles that studied endocrine features not well associated with symptoms of PCOS (e.g. genotypic distributions of PCOS). Given the more general endocrine foci of these records, some studies also evaluated contained male data. These were excluded from this review since evidence for a ‘PCOS equivalent’ in males remains controversial, and PCOS is currently recognized as a female endocrine disorder exclusively (Barthelmess and Naz, 2014). Cluster A and Cluster C personality disorders were excluded due to their divergence with Cluster B diagnoses. Studies evaluating specific, singular personality traits (e.g. impulsivity and dissociation) were also excluded.
Results
Our search of Medline, EMBASE and Cochrane Central retrieved 244 records, 38 of which were duplicates and were thus removed. Of the remaining 206 records, 195 were excluded based on inspection of the title or abstract. A total of 11 full-text records were assessed for eligibility, with a further 2 records excluded (Figure 1). Nine records were identified to contain information specific to BPD (or at least Cluster B personality disorders specifically) and PCOS (or at least symptoms and markers specific to PCOS), per our inclusion criteria. Three of these records were cross-sectional studies investigating the prevalence of PCOS or symptoms of PCOS in women with BPD (Dettenborn et al., 2016; Rausch et al., 2015; Roepke et al., 2010), while three records were cross-sectional and cohort studies examining the prevalence of psychiatric diagnoses in women with PCOS (Cesta et al., 2016; Sahingoz et al., 2013; Scaruffi et al., 2014). Finally, another three records were case reports of comorbid BPD and PCOS (Jain et al., 2015; Sharma, 2015; Trisno et al., 2016).
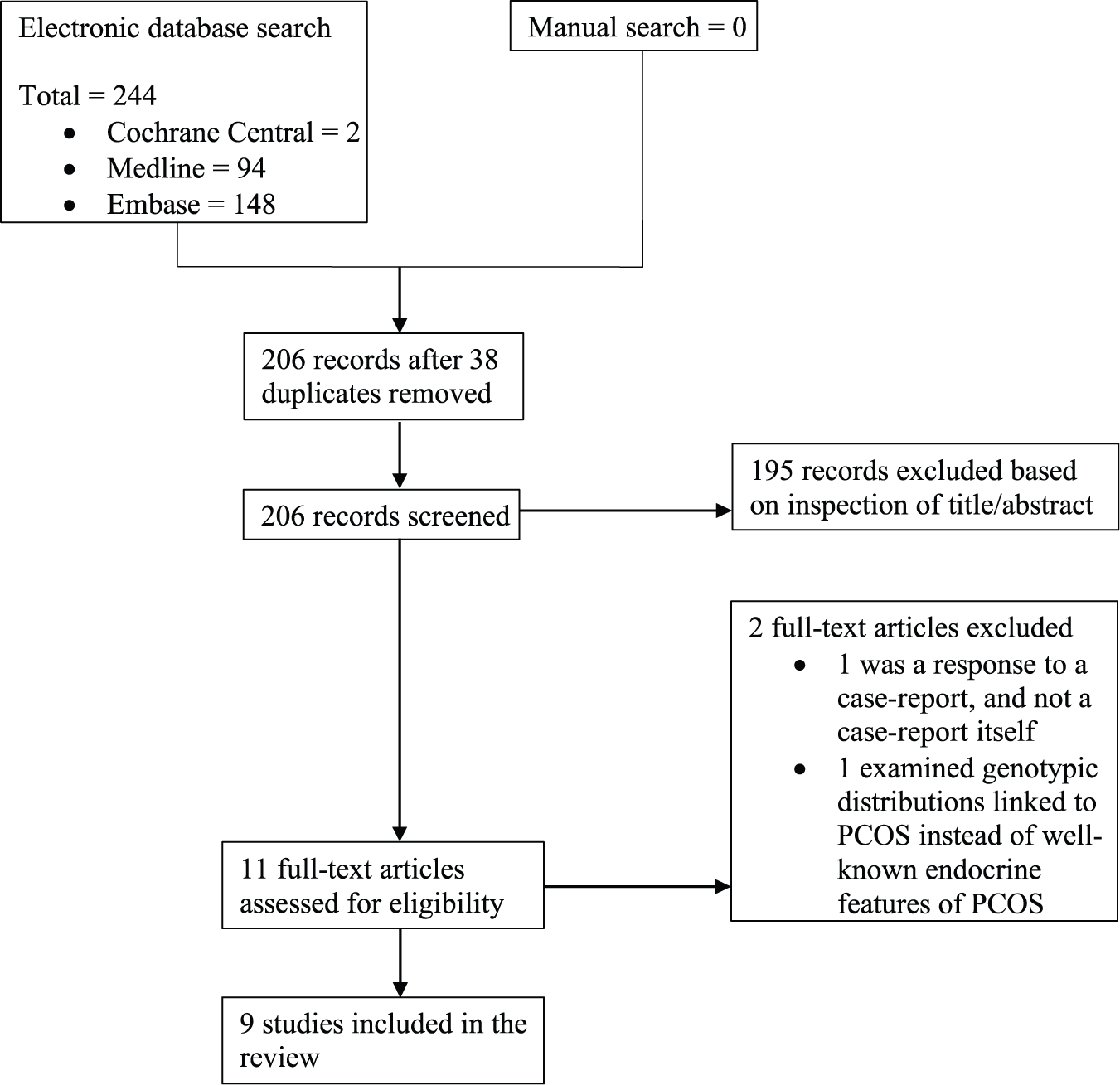
Flow diagram of the search and inclusion of studies regarding BPD and PCOS.
Studies examining the prevalence of PCOS in women with BPD
Of the nine studies included in this review, Roepke et al. (2010) was the first study, published only relatively recently in 2010, to demonstrate a link between PCOS symptoms and BPD. Within a patient group of 31 women with BPD who had been admitted to a specialized inpatient psychiatric treatment program, compared to 30 healthy controls, it was found that a significantly greater proportion of patients, 30.4%, had polycystic ovaries compared to 6.9% in the healthy control group (p = 0.03). Moreover, the patient group had significantly elevated serum androgen concentrations compared to the control group (free testosterone, p = 0.04; androstenedione, p = 0.02; 17 alpha-hydroxyprogesterone, p = 0.02), a finding that was independent of body mass index (BMI) and also, interestingly, polycystic ovary status. However, this study is limited in that it did not measure the presence of other clinical features of PCOS in line with any of the accepted criteria, and thus can only be interpreted with regard to polycystic ovaries and serum androgen levels specifically. Furthermore, some patients in this study reported prior use of valproate, a medication that has been shown to increase testosterone levels and symptoms of hyperandrogenism – and therefore PCOS – in women (Zhang et al., 2016), and so cannot be excluded as a confounding factor. Another confounding factor in this study was the presence of comorbid dysthymia within the sample, which was not controlled for and could have influenced the results.
Building on these initial research efforts, in 2015, Rausch et al. (2015) reached comparable conclusions in their study evaluating basal saliva testosterone levels in 35 medication-free female patients with BPD compared to 26 matched healthy volunteers. Independent of BMI, menstrual cycle and smoking status, BPD patients were found to have significantly raised testosterone levels compared to their healthy counterparts (p = 0.01) Notably, mean testosterone values for the BPD group remained in the normal range despite the significant increase, leading the authors to suggest that these results may be due to changes in the fine-tuning of the hypothalamic–pituitary–gonadal (HPG) axis in BPD patients, and not because of its complete malfunction. Nonetheless, many women with PCOS have normal total testosterone levels, but have elevated free testosterone levels that result in the male-like hormonal features seen in PCOS (Azziz et al., 2009). So, the sole measurement of total testosterone is considered not relevant to the diagnosis of PCOS (Azziz et al., 2009). Although this study lacked a clinical control group to control for the presence of comorbidities in the BPD group, it did explore the potential influence of comorbid major depression disorder (MDD) and post-traumatic stress disorder (PTSD) on testosterone levels within the BPD sample and found no significant differences between patients with and without a diagnosis of either MDD or PTSD. However, this study did not focus on PCOS specifically, and therefore, no other features of PCOS were investigated and compared between the two groups.
In 2016, Dettenborn et al. (2016) provided further evidence for an association between BPD and PCOS symptomology using hair analysis to demonstrate significantly increased hair testosterone levels in 18 female BPD patients compared to 17 matched healthy volunteers (p = 0.03). This finding remained even when factors such as medication, smoking, MDD and PTSD were controlled for. The use of hair analysis to measure testosterone levels in this study provides the first evidence of its type regarding the cumulative excretion of testosterone over several months in patients with BPD, and not just the immediate excretion rate as measured in blood and saliva measurements. However, this study is limited by its small sample size, and like the previous studies, by the lack of a clinical control group to control for the presence of other psychiatric comorbidities in the BPD group and assessment of the other diagnostic characteristics of PCOS.
Studies examining the prevalence of BPD in women with PCOS
In 2013, also taking a cross-sectional design approach, Sahingoz et al. (2013) were the first to describe the prevalence of personality disorders (and other axis-II psychiatric disorders) in women already diagnosed with PCOS. The outcome measure for ascertaining the presence of personality disorder in this cohort was the Structured Clinical Interview for DSM, Revised Third Edition Personality Disorders (SCID-II). These authors compared 73 patients with PCOS to 73 no-PCOS control subjects in this cross-sectional study and used the NIH criteria to independently diagnose PCOS. Even though axis-II psychiatric disorders (i.e. personality disorders and mental retardation) were significantly more common in PCOS patients (23.3%) compared to the healthy control group (9.6%), no significant difference in the prevalence of BPD, specifically, was observed between the two groups. This study was limited by a relatively small sample size of women recruited from a single gynecology outpatient clinic, and thus may not be representative of the general population. These authors also did not examine the effects that the clinical characteristics of PCOS (e.g. hirsutism and acne) might have on psychiatric disorders in these women. It is plausible that some of the physical symptoms of PCOS could cause substantial emotional distress to the individual (Benson et al., 2009b; Hahn et al., 2006). However, this area of research is still controversial (Annagür et al., 2013; Kerchner et al., 2009).
Scaruffi et al. (2014) also investigated the prevalence of personality disorders (and personality traits) in 60 women with PCOS compared to 45 matched healthy controls, undertaken in 2014. All the women with PCOS in this study met the Rotterdam criteria for PCOS. The Rorschach Test and the Million Clinical Multiaxial Inventory-III (MCMI-III) were used to diagnose the personality disorders. Overall, the results showed that women with PCOS have a three- to fivefold increased rate of mental illness generally (e.g. anxiety, depression, bipolar disorder and thought disorder) compared to women with no PCOS. BPD and paranoid personality disorder were examined together, with the rate of these diagnoses being found to be significantly higher in women with PCOS compared to women without PCOS (p < 0.05). A major limitation in interpreting these findings is that these two disorders were not reported separately, and thus this finding is not specific to BPD specifically. Furthermore, as reported by the authors, these women were young (mean age = 25.8 years, standard deviation (SD) = 4.27) and mostly still in university. As such, this group of patients may not be representative of the general population.
Published most recently in 2016, the largest relevant study to date is a Swedish matched cohort study conducted by Cesta et al. (2016) examining the prevalence of psychiatric disorders in women with PCOS. In this study, 24,385 women diagnosed with PCOS between 1990 and 2013 were identified using national patient registers and compared to matched individuals (1:10/100) from the general population. Women with PCOS were found to have significantly higher adjusted odds ratios (AORs) for Cluster B personality disorders (odds ratios were adjusted for the presence of any other psychiatric disorder aside from the index disorder, e.g., schizophrenia spectrum disorders, bipolar disorder, depressive or anxiety disorders, eating disorders, personality disorders, autism spectrum disorders and attention-deficit/hyperactive disorder) compared to the matched comparison individuals (AOR = 1.27; 95% CI = [1.15, 1.41]). Sub-analyses were also carried out to investigate whether the use of either the NIH or the Rotterdam diagnostic criteria affected the prevalence of psychiatric disorders in PCOS. These sub-analyses divided the patient cohort into two groups – women diagnosed with PCOS from 1990 to 2003 when the NIH criteria was in use, and women diagnosed from 2004 to 2013 when the Rotterdam criteria was in use. Interestingly, the authors found higher estimates for Cluster B personality disorders in women with PCOS when the NIH criteria was being used, compared to the main analysis. However, the estimated rates were slightly attenuated (although still significant) when the Rotterdam criteria was used. This demonstrates the variable estimates achieved between scales, and highlights a future important task of improving diagnostic parameters for PCOS. This study revealed a lower prevalence of PCOS (as stated by the authors; exact number not provided) compared to the prevalence expected in the general population (i.e. 8.7–21%) (March et al., 2010), most likely due to the fact that women with PCOS were identified through national patient registers, thus possibly introducing selection bias into the study. Hence, the women in this study may have had more severe cases of PCOS because PCOS does not generally require hospital-based medical attention unless the woman is experiencing severe comorbidities or sequelae of PCOS. Moreover, there was no data available regarding the other characteristics of PCOS (e.g. insulin resistance, BMI and hirsutism) and how they can contribute to psychiatric disorders in this population, as mentioned earlier.
Case reports of comorbid BPD and PCOS
The first case report of comorbid BPD and PCOS was from Jain et al. (2015). The patient was diagnosed with PCOS at the onset of puberty, and then with Tourette Syndrome at 19 years. At 22 years, upon developing BPD-like symptoms including self-mutilating behavior, she was diagnosed with BPD and was subsequently treated with behavior therapy, which she responded well to.
In another 2015 case study, Sharma (2015) conducted a retrospective chart review that identified three women with comorbid BPD and PCOS, noting that although there were other psychiatric diagnoses comorbid with PCOS, BPD was consistently comorbid with PCOS. Similar to the previous case report, all three women had been diagnosed with PCOS many years before they were diagnosed with BPD. Few details were provided regarding their treatment and follow-up, except that they had multiple psychiatric hospitalizations, poor quality of life and eventual functional disability.
The most recent case report by Trisno et al. (2016) published in 2016 describes the case of a 22-year-old female with a history of sexual abuse by her stepfather from 10 to 15 years of age. She experienced severe symptoms of BPD that worsened premenstrually, requiring multiple admissions to the hospital. Her symptoms did not improve with dialectical behavior therapy. However, after later being diagnosed with and receiving treatment for PCOS, she experienced improvement in her mood and self-harming thoughts and behaviors.
Discussion
Summary
Data from the literature thus far indicate that women with BPD have higher than expected serum androgen levels and incidence of polycystic ovaries. However, the evidence suggesting an increased prevalence of PCOS in women with BPD is still limited, and none of the full PCOS criteria have yet been used to compare the prevalence of PCOS between BPD patients and healthy control groups. There is also currently no clear evidence of whether BPD is more prevalent in women with PCOS than in women without PCOS, as only one study (Sahingoz et al., 2013) reported BPD-specific rates. Nevertheless, even if the comorbidity between PCOS and BPD hold true only within a subgroup of BPD patients, the implications for these patients’ clinical presentations, physical health and subsequent management is significant (Frankenburg and Zanarini, 2011; Roepke et al., 2010). Therefore, it is important to delineate the relationship between, and also the temporality of, these two disorders.
All of the studies included in this review were cross-sectional studies or case reports, with the exception of one cohort study. Although cross-sectional studies are suitable for estimating the prevalence of a disease in a population, and case reports are often employed to present rare or new observations to the medical community, these types of studies are not overly useful in determining the possible mechanisms behind the etiology of the disorders, whether as separate or comorbid disorders. Nonetheless, despite these research designs providing estimates of association only, and therefore low levels of evidence, these studies have been pivotal in generating hypotheses for future research in this new field (Levin, 2006; Nissen and Wynn, 2014). As such, these studies provide a platform for future research that allows the opportunity to build upon current observations.
Given the evidence of association between BPD and PCOS thus far, and based on the commonalities in etiological mechanisms of BPD and PCOS, we now propose a plausible biological mechanism that might help to explain the relationship between these seemingly disparate disorders.
Potential mechanism underlying the association between BPD and PCOS
Childhood trauma resulting in increased hypothalamic–pituitary–adrenal axis activity
A history of childhood trauma, defined as events of abuse (physical, emotional or sexual) or neglect (physical or emotional) occurring before the age of 18 years (Leeb et al., 2008), is overwhelmingly present in individuals with BPD. This has been demonstrated repeatedly within the research, with reported figures of up to 84% of BPD patients having experienced some form of abuse or neglect (Bandelow et al., 2005; Martín-Blanco et al., 2014; Waxman et al., 2014; Zanarini et al., 2000), and usually sexual and physical abuse (Zanarini, 2000; Zanarini et al., 1997). As such, childhood trauma is considered one of the most important factors in the etiology of BPD (Joyce et al., 2003; Martín-Blanco et al., 2014; Waxman et al., 2014). The experience of traumatic events or victimization in mentally unwell women has been suggested to rarely occur in isolation (Mcfarlane et al., 2006), and a large proportion of women with severe mental illness are victimized repeatedly during their lives (Goodman et al., 1997). This clustering of childhood abuse is important when considering the effects of cumulative stressors on later-life adjustment and psychiatric symptomology (Sundermann et al., 2013). Childhood maltreatment coupled with genetic vulnerability evokes a stress response that can promote pathophysiological processes (Lupien et al., 2009), thus predisposing an individual to BPD (Fonagy et al., 2011). Chronic stress results from prolonged early life trauma, and also when the stressor itself is short in duration but is perceived to be threatening for much longer. As such, individuals with a history of childhood trauma often have altered hypothalamic–pituitary–adrenal (HPA) axis activity.
The HPA axis is a dynamic, self-regulated feedback pathway that mediates stress responses in the body through a cascade of events that eventually result in the secretion of cortisol into the bloodstream. Cortisol then negatively feeds back to inactivate the HPA axis by binding with glucocorticoid receptors located in different parts of the brain including the hippocampus, medial prefrontal cortex, pituitary and hypothalamus. In the face of stress, the HPA axis results in behavioral and physiological processes that help to enable better coping mechanisms and to redirect energy needed to overcome stressors.
Compared to healthy controls, people with BPD have higher serum levels of cortisol, and release greater amounts of cortisol in response to stress (Carvalho Fernando et al., 2012; Heim et al., 2003; Lieb et al., 2004; Rinne et al., 2002), that is, they have a hyper-responsive HPA axis and lowered feedback sensitivity. This can cause some individuals to become very susceptible to stress. Indeed, patients with BPD tend to report more intense daily hassles (Jovev and Jackson, 2006) as well as increased inner tension (Kuo and Linehan, 2009), making them prone to symptoms such as rage, fluctuating mood and self-injurious behavior. These stresses are now recognized to result in structural changes of the adult brain such as cell atrophy and loss of neurons (Brambilla et al., 2004). Patients with BPD have been found to have reduced hippocampal volume (Brambilla et al., 2004; Schmahl et al., 2003), smaller amygdala volume (Schmahl et al., 2003), smaller anterior cingulate cortex volume and a unique, compromised frontolimbic network functioning (Goodman et al., 2011; Whittle et al., 2009). These areas of the brain are involved in memory, cognition and emotion regulation – all of which correspond with the severe, stress-induced symptoms observed in BPD. Nonetheless, there are contradictory results that show decreased HPA axis activity and hypersuppression from the feedback pathway in BPD (Carpenter et al., 2007; Carrasco et al., 2007; Heim et al., 2003). Heim et al. (2003) explain this discrepancy by positing that early life stress changes the set point of the HPA axis through the sensitization of the anterior pituitary to corticotrophin-releasing hormone (CRH), which subsequently increases the HPA axis activity. However, chronic hypothalamic CRH hypersecretion may down-regulate CRH receptors in the anterior pituitary, producing depression and anxiety symptoms because of the effects of CRH at other sites of the brain (CRH also coordinates behavioral, autonomic, endocrine and immune responses to stress) (Arborelius et al., 1999). Therefore, they hypothesize that the decreased HPA axis activity seen in some of these individuals may instead be a result of the depression and not the early life stress.
Studies that have investigated HPA axis reactivity in PCOS show similar results. Increased HPA reactivity and subsequent lowered feedback sensitivity have been demonstrated by a number of studies (Benson et al., 2009a; Ilic et al., 2013; Lanzone et al., 1996; Milutinovic et al., 2011). This may result in the maladaptive coping strategies (Benson et al., 2010) and reduced psychological quality of life (Li et al., 2011) that women with PCOS are known to often have. Furthermore, emerging evidence in the study of brain structure and function in women with PCOS show that women with PCOS tend to have altered white-matter microstructure (Rees et al., 2016) and poorer cognition compared to women without PCOS (Rees et al., 2016; Schattmann and Sherwin, 2007), although conflicting evidence exists (Barnard et al., 2007). However, there are also studies that indicate no significant difference between the HPA activity in women with PCOS and women with no PCOS (Azziz et al., 1998; Carmina and Lobo, 1990). It is important to note that all of these studies looking at the stress response and subsequent HPA activity in PCOS must be interpreted with caution, as psychopathology can significantly influence results, which has not been systematically assessed nor controlled for in the research to date.
Childhood sexual abuse has also been associated with gynecologic disorders such as chronic pelvic pain, vaginal infections, dysmenorrhea and menorrhagia has been associated with childhood sexual abuse (Kaliray and Drife, 2004), while the emotional abuse of women has been found to result in more consultations for cervical smear abnormalities (Johnson et al., 2007). Still, little is known about the effects of childhood trauma on these disorders. A recent study published in 2016 (Schliep et al., 2016) did not find a significant association between the presence of ovarian cysts and childhood physical abuse. However, this study was limited in that it only inquired about lifetime physical abuse and adult sexual abuse, which does not encompass the full spectrum of childhood trauma.
Increased HPA axis activity resulting in PCOS-like symptoms
When stimulated, aside from cortisol, the adrenal glands also produce androgens such as dehydroepiandrosterone (DHEA), dehydroepiandrosterone sulfate (DHEAS) and androstenedione (Berne, 2008). These androgens make up about 50% of androgens in females (Burger, 2002) and do not feedback to the hypothalamus or the pituitary glands. Albeit considered weak androgens, they are converted in the periphery into more potent androgens such as testosterone and dihydrotestosterone (DHT), which have direct androgenic activity (Enea et al., 2011). Hence, the hyperactivation of the HPA axis due to chronic stress can increase the amount of androgens in the body and result in clinical hyperandrogenism, as has been evidenced in both PCOS and BPD, and is a key feature in the diagnosis of PCOS. Indeed, an adrenal component has been observed in a substantial number of women with PCOS, with adrenocortical dysfunction being associated in about 30–50% of PCOS patients (Azziz, 2000; Kumar et al., 2005). This also corresponds to the observations of increased testosterone levels in women with BPD (Dettenborn et al., 2016; Rausch et al., 2015; Roepke et al., 2010) described earlier (Table 1).
Summary of included studies.
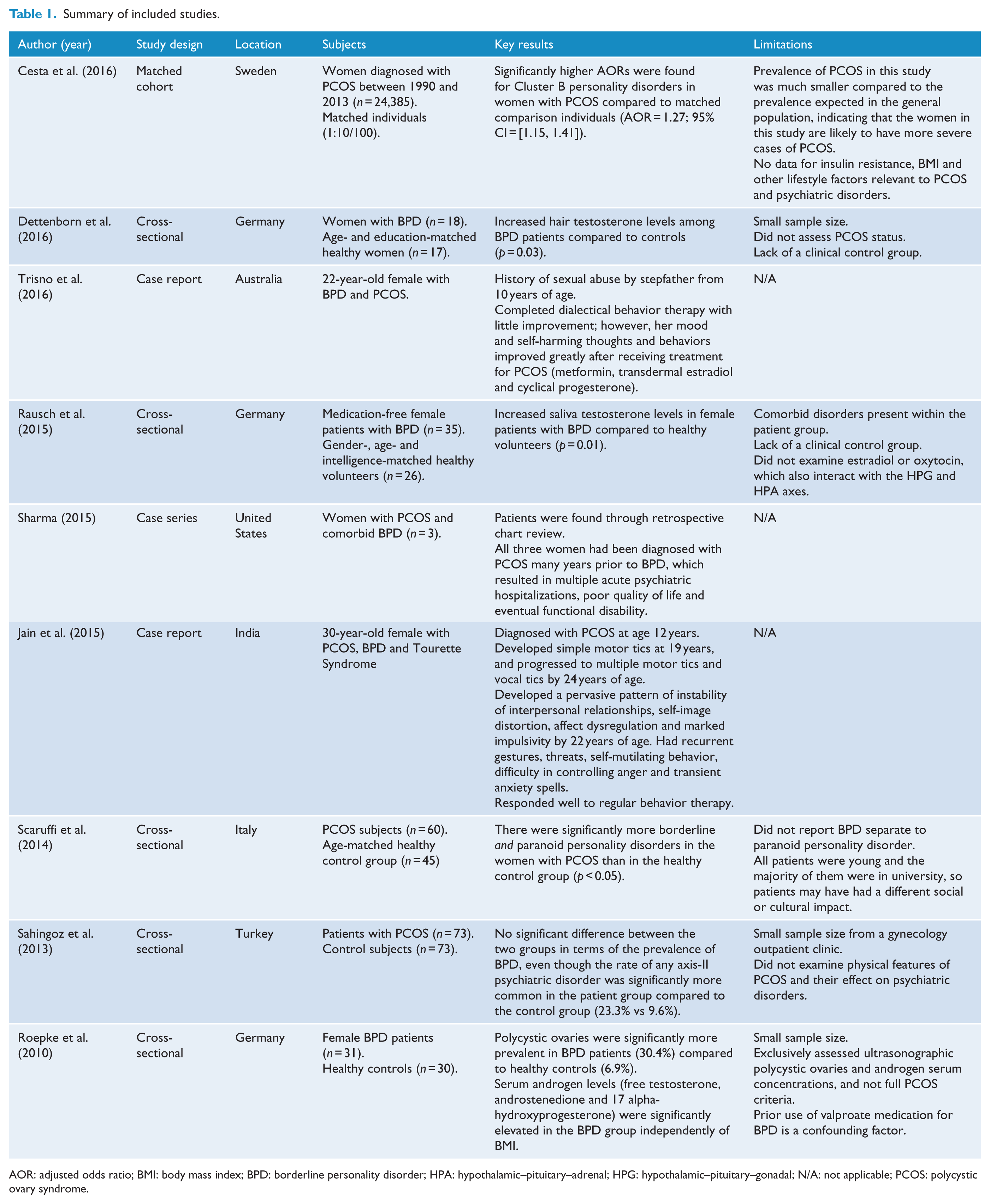
AOR: adjusted odds ratio; BMI: body mass index; BPD: borderline personality disorder; HPA: hypothalamic–pituitary–adrenal; HPG: hypothalamic–pituitary–gonadal; N/A: not applicable; PCOS: polycystic ovary syndrome.
Furthermore, the end product of the HPA axis, cortisol, has inhibitory effects on all three levels of the HPG axis (Johnson et al., 1992). Therefore, hyperactivation of the HPA axis can lead to the inhibition of the HPG axis, resulting in the menstrual dysfunction seen in PCOS. Cysts may also develop in the ovary as a result of stress activating a sympathetic neural pathway in the hypothalamus, which releases norepinephrine into the ovary (Toufexis et al., 2014), which may explain the finding of increased polycystic ovaries in women with BPD by Roepke et al. (2010).
Although neither an increase in the incidence of menstrual dysfunction nor polycystic ovaries have been demonstrated in women with BPD, a study conducted in 2003 (DeSoto et al., 2003) found that symptoms of BPD in women could be anticipated based on the variability and change in estrogen levels during their menstrual cycles – higher estrogen levels were associated with more severe BPD symptoms. This pattern held even when depression, hostility and anxiety were controlled for statistically. Hence, this shows that hormonal imbalance can also impact the severity of BPD, thus making the study of the relationship between PCOS and BPD even more crucial.
Conclusion
Collectively, the recent literature is showing increasing evidence for a psychoneuroendocrinological link between BPD and PCOS. Even though study methodologies designed to explore this phenomenon are still to reach their full potential, for example, using rigorous PCOS criteria rather than measuring key singular symptoms of PCOS (e.g. serum androgen levels) in women with BPD, such symptoms are key characteristics of PCOS, and thus, coupled with the strong theoretical rationale for the link between BPD and PCOS, points to the plausibility of increased likelihood of BPD and PCOS comorbidity, at least in clinical samples of patients with a history of childhood trauma. Nevertheless, further larger studies using standardized diagnostic criteria for both PCOS and BPD are needed, and confounding factors such as other psychiatric and endocrinological comorbidities need to be controlled for in future studies. Data on the prevalence of comorbid BPD and PCOS are also still very limited, and future research should strive to investigate rates of BPD/PCOS co-occurrence. The use of prospective studies may also be able to help determine causality and temporality behind not only the relationship between BPD and PCOS but also the etiology of both disorders. Given the far-reaching health effects of both BPD and PCOS, further research in this area may potentiate change in current clinical practice and lead to improvement of screening procedures and the development of new interventions with the aim of restoring the alterations in the HPA and HPG axes of these individuals, to ultimately add to the mental and physical health and quality of life in this patient population.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
