Abstract
Objectives:
Inter-episode mood instability has increasingly been considered in bipolar disorder. This study aimed to investigate emotional reactivity as a major dimension for better characterizing remitted bipolar patients with subthreshold mood symptoms and functional status. This study also aimed to investigate whether high-sensitivity C-reactive protein, a marker of low-grade inflammation, could be a biological marker of emotional dysregulation in bipolar disorder (BD).
Methods:
Cross-sectional study of 613 subjects who met
Results:
In total, 415 (68%) patients had abnormal emotional reactivity. Independent of potential confounders, including age, gender and subthreshold mood symptoms, serum levels of high-sensitivity C-reactive protein were significantly higher in patients with emotional hyper-reactivity (median = 4.0 mg/L, interquartile range = 2.7–5.6), and with emotional hypo-reactivity (median = 3.0 mg/L, interquartile range = 1–4) compared with patients with normal emotional reactivity (median = 0.95 mg/L, interquartile range = 0.4–1.9,
Conclusions:
Emotional reactivity appears to be a relevant dimension for better characterizing remitted bipolar patients with subthreshold mood symptoms. Levels of high-sensitivity C-reactive protein may be an objective marker of emotional dysregulation in BD. Further studies are needed to confirm our findings.
Keywords
Introduction
Bipolar disorder (BD) affects more than 1% of the world’s population and is a chronic illness characterized by acute manic and depressive episodes (Grande et al., 2016). Currently, inter-episode mood instability has increasingly been considered in BD (Broome et al., 2015b; Malhi et al., 2015; Marwaha et al., 2014). A growing body of evidence indicates that during remission, bipolar patients often present subsyndromal mood symptoms, which are associated with poor psychosocial functioning, cognitive impairment and reduced quality of life (Bonnín et al., 2012; Rosa et al., 2008; Strejilevich et al., 2013). Current diagnostic approaches used to assess mood symptoms essentially rely on symptom-based categories (Phillips and Kupfer, 2013). However, these approaches lack the sensitivity to detect subtle mood and emotion alterations in bipolar patients, particularly during the remission phase (Henry and Etain, 2010; Malhi et al., 2015; Malhi and Porter, 2016).
Persistent deficits in the emotion regulation process as well as abnormal emotional reactivity are frequently observed in individuals with BD (Gruber et al., 2011; Phillips et al., 2008). Emotional reactivity is defined by the magnitude of change from an emotional baseline state in response to emotion-eliciting stimuli (Gross, 1998). A multidimensional approach to assess bipolar patients has previously demonstrated a continuum of mood symptoms, ranging from global behavioral inhibition to a global activation (from major depressive episodes without manic symptoms, through depression with (hypo)manic symptoms and mixed states, to full manic states, respectively) (Henry et al., 2008, 2010). This multidimensional approach has also previously allowed the distinction between two subforms of depression in bipolar patients: one characterized by overall behavioral inhibition and emotional hypo-reactivity, and the other defined by mild behavioral activation associated with emotional hyper-reactivity (Henry et al., 2007). A dimensional approach may be useful to discriminate the broad spectrum of mood disturbances as well as provide a better understanding of the pathophysiology of mood and bipolar disorders (Henry et al., 2015; Malhi et al., 2015; Malhi and Porter, 2016).
In BD, dysregulated and chronic inflammatory responses have been considered to be one of the main underlying pathophysiological mechanisms (Berk et al., 2011; Goldstein et al., 2009; Padmos et al., 2008). In addition, studies have reported elevated levels of pro-inflammatory markers (e.g. cytokines and acute-phase proteins) in bipolar patients (Dargél et al., 2015; Modabbernia et al., 2013). C-reactive protein (CRP) is an acute-phase protein, produced by the liver, and it is a marker of low-grade inflammation (Gabay and Kushner, 1999). It is also the most commonly used laboratory test in clinical settings worldwide to assess systemic, peripheral inflammatory status (Emerging Risk Factors Collaboration et al., 2010; Pepys and Hirschfield, 2003). In apparently healthy individuals, serum levels of CRP are usually below 3 mg/L but may be up to 10 mg/L (Pepys and Hirschfield, 2003). Recently, a meta-analysis found significantly higher CRP levels in bipolar patients across mood phases (mania, depression and euthymia) compared to healthy controls (Dargél et al., 2015). A large prospective study from the general population has demonstrated that elevated levels of CRP were associated with late-onset BD (Wium-Andersen et al., 2016). Additionally, a recent study has found that healthy individuals showing difficulties in regulating their emotions in daily life had increased levels of CRP, suggesting that non-immunological behavioral and emotional stimuli could also be implicated in systemic inflammation (Appleton et al., 2013). To date, no studies have investigated the association between emotional reactivity, functioning and CRP levels in bipolar patients.
The aim of this study is to examine emotional reactivity as a major dimension that may contribute for better characterizing remitted bipolar patients with subthreshold mood symptoms and functional status. This study also aims to explore whether CRP could be a marker of emotional dysregulation in BD.
Patients and methods
Participants
A total of 613 bipolar outpatients aged over 18 years were recruited from 1300 outpatients evaluated in the French Network of Bipolar Expert Centers (FondaMental Advanced Centers of Expertise in Bipolar Disorders [FACE-BD]) from January 2009 to June 2014. This Network integrates research and clinical practice into community-based outpatient psychiatric care centers in France (Henry et al., 2011). Primary diagnosis was confirmed by psychiatrists using the Structured Interview for
Assessments
Patient’s symptoms were assessed using the following mood ratings: MADRS for depressive symptoms, YMRS for manic symptoms and the State-Trait Anxiety Inventory (STAI) for anxiety symptoms. The STAI is a 20-item instrument rated on a 4-point scale, ranging from almost never to almost always, with higher scores indicating greater anxiety. A questionnaire detailing self-reported comorbidities, lifestyle and medication use was also administered to all participants.
Emotional reactivity
The Multidimensional Assessment of Thymic States (MAThyS) was used to assess mood variations during the preceding week (Henry et al., 2008). The MAThyS is a 20-item, self-rated scale that evaluates quantitatively five dimensions, including emotional reactivity, cognition speed, psychomotor activation, motivation and sensory perception. The MAThyS total score ranges from 0 to 200; scores < 91 indicate global behavioral inhibition, while scores > 108 indicate global activation, across mood states (Henry et al., 2008). The emotional reactivity sub-score ranges from 0 to 40. Participants were classified into three groups according to validated cutoffs for different levels of emotional reactivity: 0 to <16 = emotional hypo-reactivity; 16–24 = normal emotional reactivity; and >24 to 40 = emotional hyper-reactivity (Atzeni et al., 2013). The MAThyS has previously been shown to have validity and internal consistency (Cronbach’s alpha coefficient = 0.95) (Henry et al., 2008).
Functional status
Psychosocial functional impairment was assessed using the Functioning Assessment Short Test (FAST) (Rosa et al., 2008). This a 24-item scale that covers six specific areas of functioning namely, autonomy, occupational functioning, cognitive functioning, financial issues, interpersonal relationships and leisure time. Items are rated using a 4-point Likert scale, where 0 = no difficulty, 1 = mild difficulty, 2 = moderate difficulty and 3 = severe difficulty. Overall FAST scores range from 0 to 72, with higher scores indicating greater disability and a threshold score of 11 indicating significant disability (Rosa et al., 2008).
Biological assessment
Fasting blood samples were collected from patients between 7:00 and 9:00 a.m. and were used to measure high-sensitivity CRP (hsCRP), which is the most sensitive assay for assessing CRP levels. Samples were collected in 5-mL ethylenediamine tetraacetic acid (EDTA) coated tubes (BD Vacutainer, Franklin Lakes, NJ). The immunoturbidimetric method was used for quantifying serum concentration of hsCRP via the Cobas8000 biochemistry analyzer (Roche Diagnostic, Meylan, France). Reagents and calibrators were used according to manufacturer guidelines with analytic measuring ranges set at 0.3–350 mg/L.
Statistical analysis
Sociodemographic and clinical data were analyzed with descriptive statistics. Frequencies and percentages were presented for categorical data. Measures of central tendency (means and medians) and of dispersion (variance, standard deviation and interquartile range [IQR]) were calculated for continuous data. The three groups with different levels of emotional reactivity were compared for the aforementioned variables by chi-square tests (
The differences in the functioning level between the three groups (emotional hypo-reactivity, normal emotional reactivity and emotional hyper-reactivity) were also assessed. Cohen’s
Results
Of the 613 remitted bipolar patients, 357 (58.2%) were female and the mean age was 41.2 (±12.4) years. The mean age of onset of the BD was 25 years. In all, 300 (49%) patients were diagnosed with BD type I, and 11% were classified as rapid cycling. Table 1 shows the sociodemographic and clinical characteristics of the 613 remitted bipolar patients, comparing those with emotional hypo-reactivity, normal emotional reactivity and emotional hyper-reactivity.
Sociodemographic and clinical characteristics of 613 remitted bipolar patients with different levels of emotional reactivity.
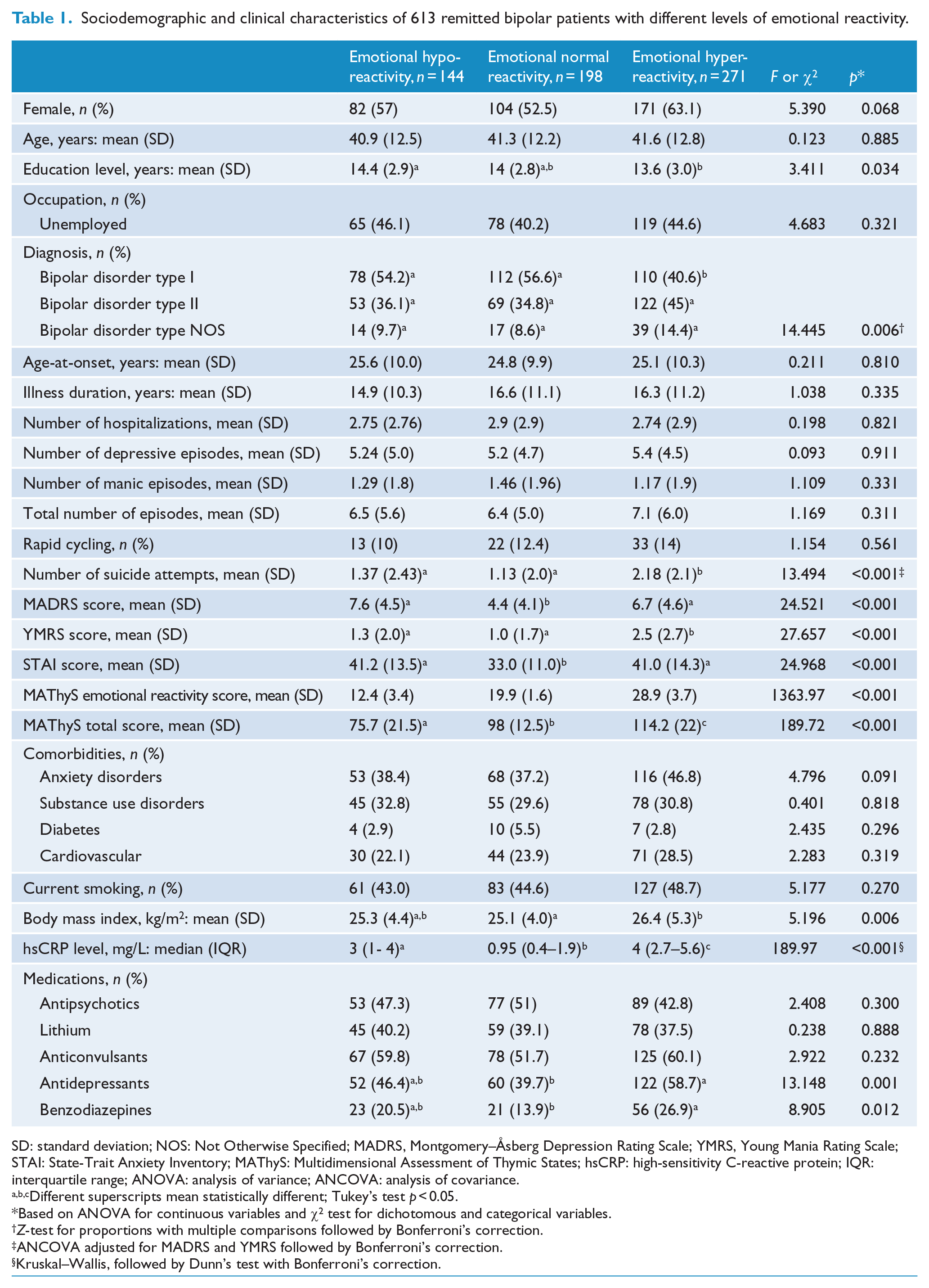
SD: standard deviation; NOS: Not Otherwise Specified; MADRS, Montgomery–Åsberg Depression Rating Scale; YMRS, Young Mania Rating Scale; STAI: State-Trait Anxiety Inventory; MAThyS: Multidimensional Assessment of Thymic States; hsCRP: high-sensitivity C-reactive protein; IQR: interquartile range; ANOVA: analysis of variance; ANCOVA: analysis of covariance.
Different superscripts mean statistically different; Tukey’s test
Based on ANOVA for continuous variables and χ2 test for dichotomous and categorical variables.
ANCOVA adjusted for MADRS and YMRS followed by Bonferroni’s correction.
Kruskal–Wallis, followed by Dunn’s test with Bonferroni’s correction.
Patients with emotional hyper-reactivity were more likely to be female (63%),
There were no significant differences between the three groups with respect to age, age-at-onset, illness duration, and total number of previous mood episodes, manic/depressive episodes and comorbidities. Both groups with abnormal emotional reactivity (hypo or hyper) had similar levels of depressive subthreshold symptomatology, which was higher than in the group with normal emotional reactivity,
Emotional reactivity and hsCRP levels
Figure 1 shows the distribution (median and IQRs) of hsCRP serum levels in the 613 remitted bipolar patients, comparing the three groups based on the different levels of emotional reactivity. Levels of hsCRP were statistically different between the three groups, χ2(2) = 189.97,
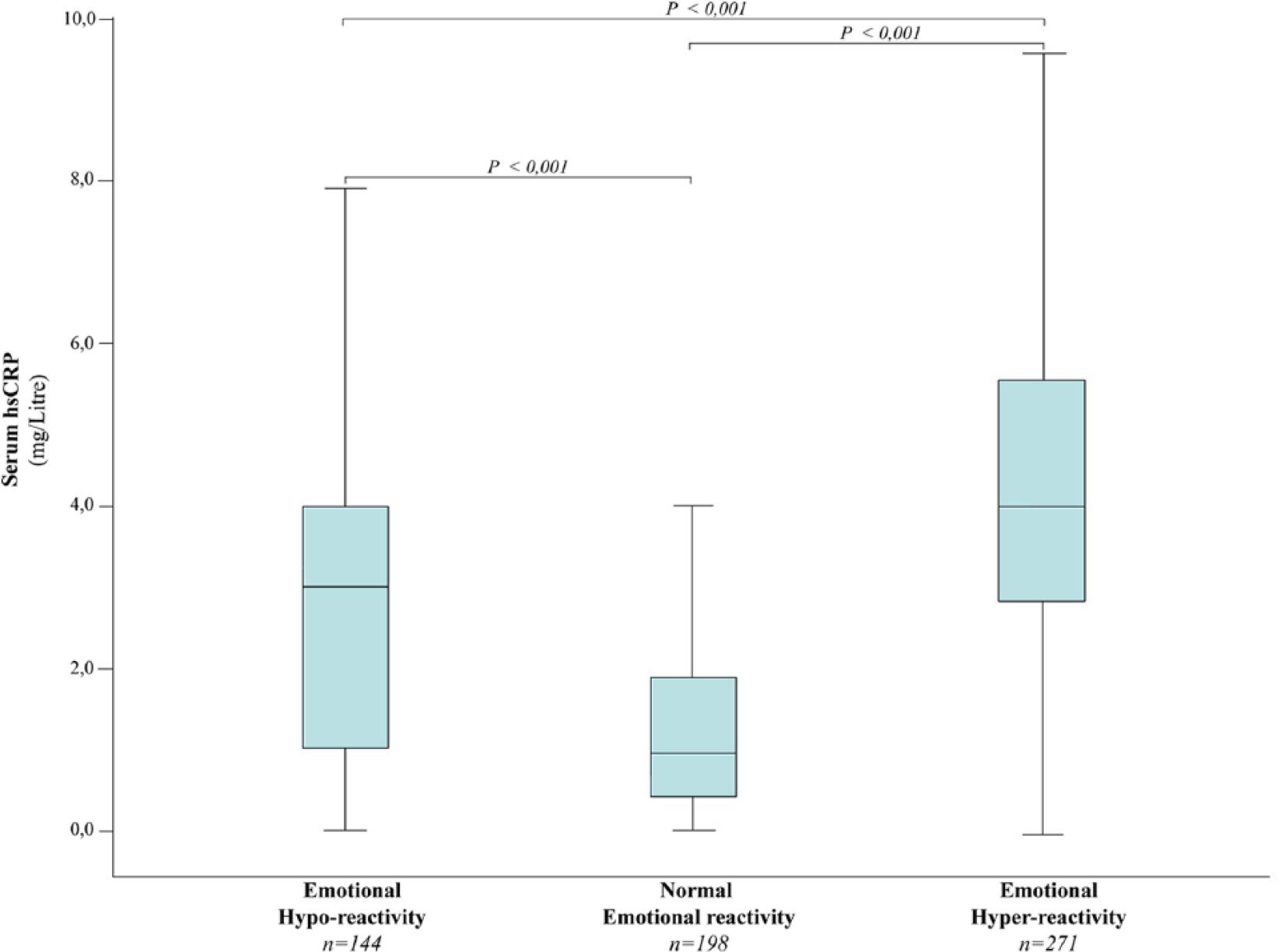
Serum levels of high-sensitivity C-reactive protein of 613 remitted bipolar patients with different levels of emotional reactivity.
Emotional reactivity and functioning status
Patients with abnormal emotional reactivity differed significantly from those with normal emotional reactivity with regard to global,
Psychosocial functioning of 613 remitted bipolar patients with different levels of emotional reactivity.
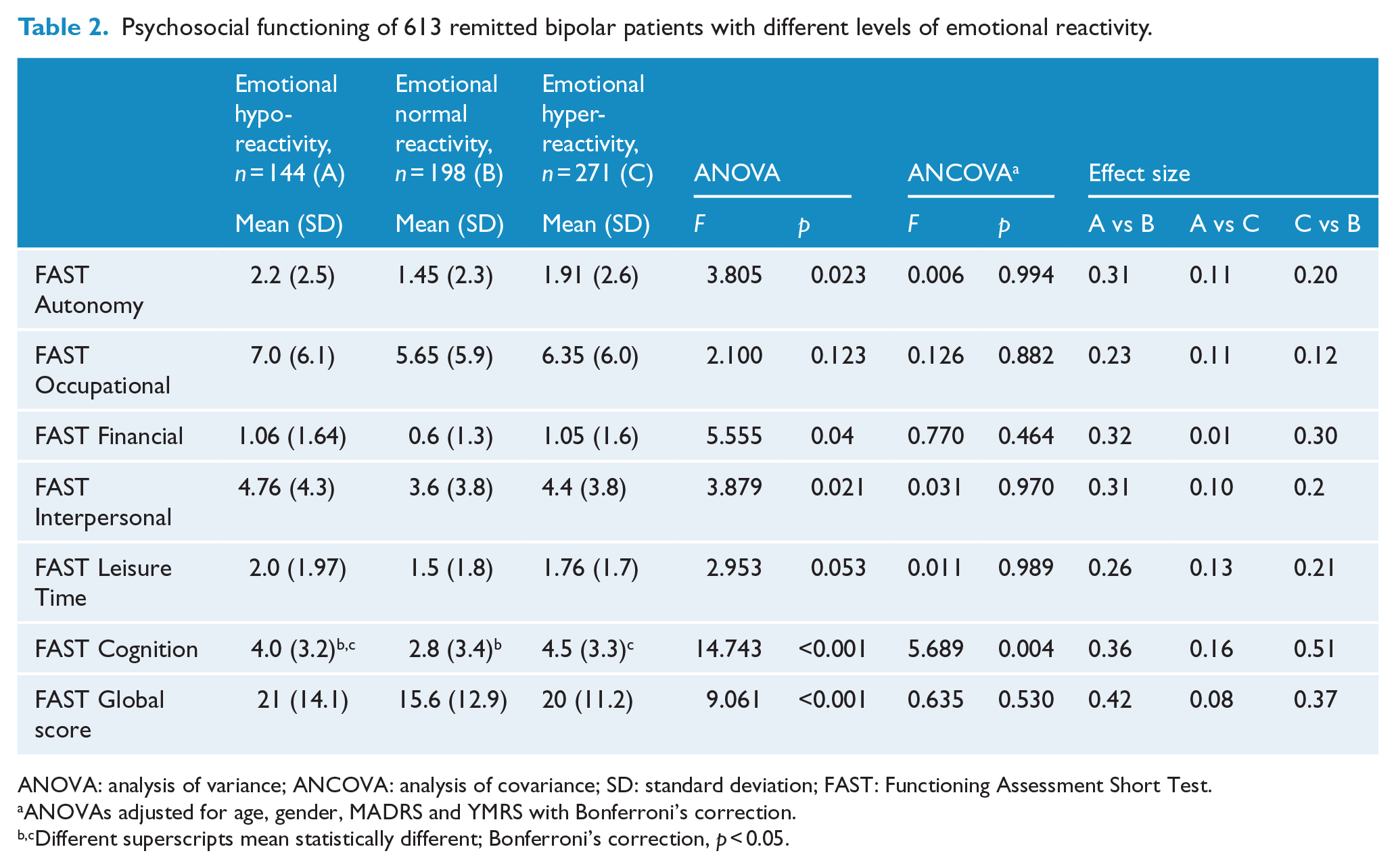
ANOVA: analysis of variance; ANCOVA: analysis of covariance; SD: standard deviation; FAST: Functioning Assessment Short Test.
ANOVAs adjusted for age, gender, MADRS and YMRS with Bonferroni’s correction.
Different superscripts mean statistically different; Bonferroni’s correction,
Discussion
The main finding of this study is that emotional reactivity appears to be a clinically relevant dimension for better characterizing bipolar patients with subthreshold mood symptoms during inter-episode periods. Although all bipolar patients were in remission—as defined by the categorical, symptom-based approach for criteria of remission (Tohen et al., 2009), they presented with significantly different levels of emotional reactivity after adjusting for MADRS and YMRS scores. By assessing these patients using a dimensional approach (Henry et al., 2008), we observed that patients with abnormal emotional reactivity (hypo or hyper) presented higher levels of subthreshold mood symptoms. These subtle thymic elements commonly found in remitted bipolar patients may result from emotional dysregulation, leading to emotional hypo- or hyper-reactivity that becomes chronic and corresponds to subsyndromal mood symptoms. This reinforces the idea that emotional dysregulation in BD may exist beyond acute mood episodes. Therefore, a dimensional approach may be more suitable to place bipolar patients along a gradient of symptom intensity rather than artificially categorize patients based on arbitrary cutoffs (Henry and Etain, 2010; Malhi and Porter, 2016).
Growing evidence has described affective dysregulation during the inter-episode period in BD as well as in some personality disorders such as mood instability—rapid oscillations of intense affect, with a difficulty in regulating these oscillations or their behavioral consequences (Broome et al., 2015a; Marwaha et al., 2014). This definition however does not take into account the emotional hypo-reactivity (low affect), which is also an important feature of mood instability. Thus, using the emotional reactivity as a dimension, ranging from hypo- to hyper-reactivity, could be more appropriate in BD clinical practice.
The complex patterns of affective instability appear to be the rule in the dynamics of BD (Bonsall et al., 2012). In our study, patients presenting emotional hyper-reactivity had significantly higher number of suicide attempts and were more likely to present subtle mixed features. Additionally, both groups with abnormal emotional reactivity (hypo or hyper) had similar levels of anxiety, indicating that emotional hyper-reactivity state may also be independent of anxiety levels. Moreover, the proportion of patients using antidepressants was significantly higher in the group with emotional hyper-reactivity. This group appears to belong to a broad spectrum of mixed states, where patients may respond better to mood stabilizers or antipsychotics than to antidepressants (Malhi, 2015). Together, these findings reflect the mood instability features reported in bipolar patients (Bonsall et al., 2012; Marwaha et al., 2014), suggesting a global mood instability pattern, particularly, in patients with emotional hyper-reactivity. Thus, rather than being considered the recurrence of mood episodes, BD could be viewed as a global level of behavioral alteration, fluctuating from inhibition to excitation, from subtle mood variations to full-blown episodes (Henry and Etain, 2010). In BD, a dimensional approach may provide not only relevant clinical information, such as the severity of impairment, but may also lead to diagnoses that are closer to the pathophysiology of the disorder, making it more indicative of the treatments required.
There is now compelling evidence to suggest that systemic low-grade inflammation may be implicated in the pathophysiology of mood and bipolar disorders (Berk et al., 2011; Dargél et al., 2015; Goldstein et al., 2009; Wium-Andersen et al., 2016). Recently, a study has found significantly higher serum levels of CRP in bipolar patients across mood states compared to healthy subjects (Dargél et al., 2015). In the present study, we explored hsCRP as a potential biological marker of emotional reactivity as a major dimension in BD. Patients with abnormal emotional reactivity had significantly increased hsCRP levels than patients with normal emotional reactivity. Considering only patients with abnormal emotional reactivity, hsCRP levels were significantly higher in the group with emotional hyper-reactivity than in those with emotional hypo-reactivity. These associations were maintained after controlling for factors known to alter levels of CRP, including age, gender, mood symptoms, BMI, cardiometabolic disorders and smoking status (O’Connor et al., 2009). These findings contribute to the evidence linking emotion dysregulation and low-grade inflammation (Appleton et al., 2013), supporting the hypothesis that CRP may be a biological marker of emotional dysregulation in BD. Clinically, using a dimensional approach such as emotional reactivity associated with such a biomarker (e.g. CRP) may allow the traditional categorical diagnosis of BD to be more flexible, facilitating a clinical approach that is closer to the BD pathophysiology, helping psychiatrists to provide more individualized interventions to bipolar patients.
Although all patients were in clinical remission, we found that most of the patients had functional impairment. These findings add to the evidence to the fact that functional impairment persists during the inter-episode period of BD (Bonnin et al., 2016; Rosa et al., 2008). Additionally, patients with abnormal emotional reactivity showed higher functional impairment than those with normal emotional reactivity. After adjusting for potential confounding factors such as age, gender and mood symptoms, patients with emotional hyper-reactivity presented with significantly lower cognitive functioning (
Our study showed that bipolar patients with poorer functioning and abnormal emotional reactivity had greater levels of CRP. Moreover, patients with marked impairment in cognitive functioning (those within the group of emotional hyper-reactivity) had the highest CRP levels. A recent study reported an association between increased levels of CRP and cognitive impairment in individuals with BD (Dickerson et al., 2013). However, this study included a heterogeneous sample, comprising inpatients and outpatients, most of them within acute mood episode (Dickerson et al., 2013). The biological mechanism linking low-grade inflammation, cognitive functioning and BD is not fully understood and it is likely to be related to inflammatory processes within the cerebral vasculature, especially the blood-brain barrier (Hsuchou et al., 2012). CRP does not cross the blood-brain barrier and thus it does not affect the brain directly. Nonetheless, CRP appears to increase the permeability of the blood-brain barrier to small molecules such as pro-inflammatory cytokines. As such, CRP serum levels may indicate indirectly an inflammatory status in the brain (Hsuchou et al., 2012).
Our results must be interpreted in light of some limitations. First, this is a cross-sectional investigation of emotional reactivity as a major dimension to assess remitted bipolar patients with subthreshold symptoms. Therefore, prospective studies are needed to replicate these results in larger samples. Second, hsCRP levels were used as an objective marker of abnormal emotional reactivity, and no conclusions can be substantiated regarding the directionality of either this association or the difference in hsCRP levels between patients with emotional hypo-reactivity and emotional hypo-reactivity. The issue of directionality is particularly relevant given the possibility that low-grade inflammation may precede the presence of emotional dysregulation (Appleton et al., 2013).
As many studies have observed associations between mood symptoms and CRP (Berk et al., 2011; Dargél et al., 2015; Goldstein et al., 2009) and as emotional dysregulation is associated with BD (Gross, 1998, Gruber et al., 2011), we expected that subthreshold mood symptoms would be on the pathway between emotional hypo/hyper-reactivity and CRP. Instead, after adjusting for mood symptoms, our findings suggest that abnormal emotional reactivity might influence CRP through pathways other than mood symptoms. A number of other plausible psychophysiological mechanisms may help to explain how abnormal emotional reactivity could influence inflammation, including heightened hypothalamic–pituitary–adrenal axis and sympathetic nervous system activity and resultant stress hormones (e.g. cortisol, catecholamines) that could initiate an inflammatory response characterized by the production of a number of inflammatory markers, including CRP (Miller et al., 2002). Moreover, recent studies suggest that the final common pathway disrupted by low-grade inflammation may be neuroplasticity subserving emotional regulation and cognitive/behavior flexibility (Szabó et al., 2014), which are often impaired in BD (Phillips et al., 2008). We recommend that future studies obtain temporal and objective measures of emotional reactivity and low-grade inflammation to test such potential pathways. Third, while we controlled for a well-characterized set of potential confounders, it is possible that unmeasured variables such as genetic factors and adverse childhood experiences (Danese et al., 2008) could be influencing the observed associations. Childhood maltreatment predisposes emotion dysregulation and may play a role in increasing the risk for chronic inflammation in adulthood (Danese et al., 2008). It is still unclear whether emotional reactivity is likely to have independent, synergistic or overlapping effects on health and biology. Fourth, although higher levels of CRP were found in the groups of patients with higher functional and cognitive impairment, other inflammatory markers (e.g. homocysteine) have also been related to cognitive impairment in BD (Dittmann et al., 2008), thus caution should be taken in the interpretation of the data presented. However, we could speculate that frequent acute endocrine and immune systems activation, which could be induced by many lifestyle and environmental factors, may over time lead to wear-and-tear (i.e. allostatic load) and interfere with these processes (McEwen, 2003). A decrease in psychosocial functioning may in turn be exacerbated by emotional disturbances and result in increased chronic activation of endocrine and immune systems, creating a vicious cycle of cumulative functional impairment over time (Vieta et al., 2013). As such, CRP may be a proxy of an allostatic environmental load as well as a marker of non-communicable disorders, many of which share CRP as a biomarker (O’Neil et al., 2015). Fifth, we assessed emotional reactivity using a self-reported questionnaire (MAThyS) (Henry et al., 2008) that could be a critical issue for studies of emotional dysregulation and mood instability given their dynamic nature (Bonsall et al., 2012; Broome et al., 2015a). The increasing possibilities to use mobile technologies (such as smartphones and wearable sensors) to capture the behavioral, physiological and environmental correlates of emotional reactivity (Glenn and Monteith, 2014) might provide a much richer and deeper understanding of emotional reactivity in mood and bipolar disorders (Ayub Dargél et al., 2016; Malhi et al., 2015). Sixth, although the FAST is an interviewer-administered instrument and provides a clinical evaluation of the functional impairment (Rosa et al., 2008), we cannot rule out that the reported subjective complaints of our patients, made worse by subthreshold mood symptoms and abnormal emotional reactivity, may have possibly contributed to a lower psychosocial functioning. A neurocognitive assessment would have also strengthened our results; however, this was beyond the scope of this study.
Finally, this cross-sectional study assessed bipolar patients under various therapeutic regimens and, thus, changes in the hsCRP levels related to medication use. However, after controlling for potential mediator factors that may have influenced hsCRP levels, including MADRS and YMRS scores and medication use, the levels of hsCRP remained significantly higher in these patients. Recent meta-analysis have shown that CRP levels were significantly elevated in euthymic BD patients compared to controls independently of use of antipsychotic or lithium (Dargél et al., 2015), reinforcing the hypothesis that there may exist a component of chronic inflammation in non-acutely ill BD patients (Padmos et al., 2008). Furthermore, antidepressants as well as mood stabilizers (e.g. lithium, antipsychotics and anticonvulsants) appear to act in varying capacities to down-regulate the production of pro-inflammatory messenger RNA (mRNA) and protein expression that might alter levels of CRP (Haarman et al., 2014). Future studies including longitudinal analysis stratified by pharmacological treatment should more rigorously evaluate this hypothesis.
This study has a number of strengths. The current findings highlight the clinical relevance of emotional reactivity as a dimensional approach for better characterizing bipolar patients with subthreshold mood symptoms during inter-episode periods. To our knowledge, this is the first study taking into account emotional reactivity, hsCRP levels and psychosocial functioning in a large, representative sample of bipolar patients with subthreshold symptomatology. We considered CRP, a widely used marker of low-grade inflammation (Emerging Risk Factors Collaboration et al., 2010; Pepys and Hirschfield, 2003), as an objective validator of emotional reactivity, which we considered a relevant dimension in BD. Biomarkers are not subject to reporting biases and can provide insight into physiologic mechanisms through which emotional reactivity may influence health, including psychosocial functioning (Appleton et al., 2013). Emotional reactivity was evaluated using a validated tool with good psychometric properties for dimensional mood assessment (Henry et al., 2008). In addition, we adjusted for many potential confounders that could potentially explain associations between emotional reactivity and CRP (Appleton et al., 2013; O’Connor et al., 2009), thus improving confidence that observed relationships are not spurious.
In conclusion, emotional reactivity appears to not only be a pertinent dimension for better characterizing bipolar patients with subsyndromal mood symptoms but also a clinically relevant phenomenon, with functional consequences. Further precision in its definition and measurement in different disorders, in combination with objective and continuous assessment of physiology, behavior and environment correlates, would shed light on its transdiagnostic features. In this regard, emotional reactivity could be viewed as a construct of the kind envisaged in the Research Domain Criteria—RDoC (Insel et al., 2010): it is dimensional and is likely to reflect dysfunction in a core behavioral function of the brain. However, as this is the first study to explore emotional reactivity, functional status and hsCRP levels in remitted bipolar patients, further studies are needed to confirm our findings.
Footnotes
Acknowledgements
We thank participants and staff of the Bipolar Expert Centers for their important contributions. We also thank Ms Rebecca Grant, MSc at Pasteur Institute, Paris, for her review of the manuscript; and Ms Luísia Alves, BA at Federal University of Rio Grande do Sul, Porto Alegre, Brazil, for her technical support in formatting the manuscript. Ms Grant and Ms Alves reported no conflict of interest. We are also indebted to the reviewers for their comments. Following is the list of collaborators: Henry C (Unité Perception et Mémoire, Institut Pasteur, Paris, France; INSERM, U955, Equipe 15 Genetic Psychiatry, Créteil, France; Université Paris-Est, UMR_S955, UPEC, Créteil, France; Pôle de Psychiatrie, Hôpital H. Mondor—A. Chenevier, AP-HP, Créteil, France), Leboyer M and Etain B (Fondation FondaMental, Fondation de cooperation scientifique, Créteil, France; INSERM, U955, Equipe 15 Genetic Psychiatry, Créteil, France; Université Paris-Est, UMR_S955, UPEC, Créteil, France; Pôle de Psychiatrie, Hôpital H. Mondor—A. Chenevier, AP-HP, Créteil, France), Laouamri H (Fondation FondaMental, Fondation de cooperation scientifique, Créteil, France), Raust A (Fondation FondaMental, Fondation de cooperation scientifique, Créteil, France; INSERM, U955, Equipe 15 Genetic Psychiatry, Créteil, France; Université Paris-Est, UMR_S955, UPEC, Créteil, France; Pôle de Psychiatrie, Hôpital H. Mondor—A. Chenevier, AP-HP, Créteil, France), Loftus Josephine (Fondation FondaMental, Fondation de cooperation scientifique, Créteil, France; Pôle de Psychiatrie, Centre Hospitalier Princesse Grace, Monaco, France), Bellivier F and Sportiche S (Fondation FondaMental, Fondation de cooperation scientifique, Créteil, France; AP-HP, GH Saint-Louis—Lariboisière—Fernand Widal, Pôle Neurosciences, Paris, France), Aouizerate B (Fondation FondaMental, Fondation de cooperation scientifique, Créteil, France; Service de psychiatrie adulte, Centre Expert Trouble Bipolaire, Hôpital Charles Perrens, Bordeaux, France), Olié E and Ducasse DMD (Fondation FondaMental, Fondation de cooperation scientifique, Créteil, France; Psychiatric Emergency Department, CHRU Montpellier, France; INSERM U1061, Montpellier University, Montpellier, France), Viglianaise N and Lescalier L (Fondation FondaMental, Fondation de cooperation scientifique, Créteil, France, Département de Psychiatrie, Hôpital Sainte-Marguerite, Marseille, France), Cohen RF and Wajsbrot-Elgrabli O (Fondation FondaMental, Fondation de cooperation scientifique, Créteil, France; Centre Hospitalier Universitaire de Nancy, Hôpitaux de Brabois, Université de Lorraine, Vandoeuvre les Nancy, France), Polosan M and Garçon S (Fondation FondaMental, Fondation de cooperation scientifique, Créteil, France; University Joseph Fourier, Grenoble, France; Centre Hospitalier Universitaire de Grenoble, Grenoble, France; Grenoble Institut des Neurosciences (GIN), Inserm U 836, Chemin Fortuné Ferrini, La Tronche, France), Hardy-Bayle MC and Kayser N (Fondation FondaMental, Fondation de cooperation scientifique, Créteil, France; Centre Hospitalier de Versailles, Le Chesnay, France; Université de Versailles Saint-Quentin-en-Yvelines, Versailles, France).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was supported by the Institut National de la Santé et de la Recherche Médicale (INSERM), Assistance Publique des Hôpitaux de Paris (AP-HP), Fondation FondaMental (RTRS Santé Mentale), and by the Investissements d’Avenir Program of the Agence Nationale pour la Recherche (ANR-11-IDEX-0004-02 and ANR-10-COHO-10-01). A.A.D. is supported by a postdoctoral grant from the ANR–SAMENTA and the Laboratory of Excellence Program ‘Revive’. The funding agencies had no role in the conduct or publication of the study.
