Abstract
Objective:
Immune alterations may play a role in bipolar disorder etiology; however, the relationship between overall immune system functioning and mood symptom severity is unknown.
Methods:
The two comparative effectiveness trials, the Clinical and Health Outcomes Initiatives in Comparative Effectiveness for Bipolar Disorder Study (Bipolar CHOICE) and the Lithium Treatment Moderate-Dose Use Study (LiTMUS), were similar trials among patients with bipolar disorder. At study entry, white blood cell count and bipolar mood symptom severity (via Montgomery-Aasberg Depression Rating Scale and Bipolar Inventory of Symptoms Scale) were assessed. We performed analysis of variance and linear regression analyses to investigate relationships between deviations from median white blood cell and multinomial regression analysis between higher and lower white blood cell levels. All analyses were adjusted for age, gender, body mass index, smoking, diabetes, hypertension and hyperlipidemia.
Results:
Among 482 Bipolar CHOICE participants, for each 1.0 × 109/L white blood cell deviation, the overall Bipolar Inventory of Symptoms Scale severity increased significantly among men (coefficient = 2.13; 95% confidence interval = [0.46, −3.79]; p = 0.013), but not among women (coefficient = 0.87; 95% confidence interval = [−0.87, −2.61]; p = 0.33). Interaction analyses showed a trend toward greater Bipolar Inventory of Symptoms Scale symptom severity among men (coefficient = 1.51; 95% confidence interval = [−0.81, −3.82]; p = 0.2). Among 283 LiTMUS participants, higher deviation from the median white blood cell showed a trend toward higher Montgomery-Aasberg Depression Rating Scale scores among men (coefficient = 1.33; 95% confidence interval = [−0.22, −2.89]; p = 0.09), but not among women (coefficient = 0.34; 95% confidence interval = [−0.64, −1.32]; p = 0.50). When combining LiTMUS and Bipolar CHOICE, Montgomery-Aasberg Depression Rating Scale scores increased significantly among men (coefficient = 1.09; 95% confidence interval = [0.31, −1.87]; p = 0.006) for each 1.0 × 109/L white blood cell deviation, whereas we found a weak association among women (coefficient = 0.55; 95% confidence interval = [−0.20, −1.29]; p = 0.14). Lower and higher white blood cell levels correlated with greater symptom severity and specific symptoms, varying according to gender.
Conclusion:
Deviations in an overall immune system marker, even within the normal white blood cell range, correlated with mood symptom severity in bipolar disorder, mostly among males. Studies are warranted investigating whether white blood cell count may predict response to mood-stabilizing treatment.
Introduction
Immune alterations including pro-inflammatory processes have been shown to be involved in the etiology of bipolar disorder (Bai et al., 2014; Dargel et al., 2015; Drexhage et al., 2010). These findings may have potential effects on clinical treatment decisions since markers of the immune system have been associated with differing treatment responses in bipolar disorder (Li et al., 2015). For example, high baseline levels of transforming growth factor beta-1 (TGF-β1) predicted better response to the combination treatment with quetiapine and lithium during acute manic episodes (Li et al., 2015). Furthermore, studies have indicated that anti-inflammatory drugs adjunctive to mood-stabilizers may have therapeutic effects (Arabzadeh et al., 2015; Ayorech et al., 2015), and pro-inflammatory markers to predict better response to anti-inflammatory treatment have been emphasized in patients with depression (Köhler et al., 2014; Raison et al., 2013).
Thus, the immune system may play an important role in the etiology of bipolar disorder and the development of personalized treatment options. However, although treatment regimens differ substantially depending on the clinical presentation of bipolar disorder patients (Bowden et al., 2010; Vieta et al., 2008), biomarkers have not yet been integrated in everyday clinical practice. In addition, it remains unclear whether findings of immune system alterations in bipolar disorder represent causal pathways or rather are confounded by other factors such as body mass index (BMI) or smoking. Nevertheless, due to bipolar disorder clinical heterogeneity, reviews have emphasized the need for biomarkers predicting symptom specificity, thereby possibly supporting prediction of treatment response including more personalized treatment (Goldstein and Young, 2013; Walker et al., 2014). Indeed, some studies have indicated that pro-inflammatory markers, such as C-reactive protein (CRP) or interleukin-1 receptor antagonist (IL-1ra), are associated with bipolar disorder severity (Hope et al., 2013) and symptom-clusters, including affective (Hope et al., 2011), psychotic (Hope et al., 2013) and cognitive (Hope et al., 2015; Lotrich et al., 2014) profiles. Nevertheless, to date, clinical trials have included few patients and largely limited investigation to pro-inflammatory markers. Furthermore, no trial has investigated associations between an overall immune system marker and the broad spectrum of bipolar disorder mood symptoms, covering both mood symptom severity and specific groups of mood symptoms. Finally, knowledge is lacking whether potential immune system biomarkers may differ depending on baseline demographics, such as gender, as well as other illness characteristics.
The white blood cell (WBC) count is a measure of the overall immune system activity, with particularly high levels (i.e. leukocytosis), indicating robust inflammatory response, and particularly low levels (i.e. leucopenia), indicating insufficient immune activity. In studies investigating immune alterations in somatic diseases, WBC levels have been used as overall immune system activity markers (Horne et al., 2005; Kannel et al., 1992). We aimed to investigate whether WBC level deviations are associated with mood symptom severity and specific bipolar disorder mood symptoms.
Methods
The study described in this paper is a secondary analysis using data from the Clinical and Health Outcomes Initiatives in Comparative Effectiveness for Bipolar Disorder Study (Bipolar CHOICE) (Nierenberg et al., 2014) and the Lithium Treatment Moderate-Dose Use Study (LiTMUS) (Nierenberg et al., 2009), which were 6-month multi-site, randomized comparative effectiveness trials. Bipolar CHOICE, the discovery sample, compared the classical mood-stabilizer lithium to quetiapine, a commonly used antipsychotic, combined with other medications for bipolar disorder (but not with one another) in a fashion consistent with typical clinical practice (i.e. adjunctive personalized treatment, referred to as APT). LiTMUS, the replication sample, compared lithium treatment combined with optimized personalized treatment (OPT) to OPT alone without lithium. The Institutional Review Boards of the different sites approved the study protocols, and the rationale, design and specific methods of Bipolar CHOICE (Nierenberg et al., 2014) and LiTMUS (Nierenberg et al., 2009) are reported in detail elsewhere. Subjects provided verbal and written informed consent prior to participation. This study examined data from study entry (i.e. at baseline). Importantly, participants could not be treated with lithium or quetiapine at baseline (at least >30 days without treatment) since it is known that, e.g., lithium increases WBC levels (Amitai et al., 2014).
Participants
For Bipolar CHOICE, 692 patients were screened, whereof 482 were randomized. For LiTMUS, 338 patients were screened, whereof 283 were randomized. Both studies used similar inclusion/exclusion criteria. Participants were aged between 18 and 62 years. Limited inclusion and exclusion criteria were defined to maximize heterogeneity of the sample and hence generalizability of the results; however, participants were required to have DSM-IV-TR bipolar I or bipolar II disorder and be at least mildly symptomatic (Clinical Global Impression Scale for Bipolar Disorder [CGI-BP] ⩾3; Spearing et al., 1997) at study entry.
In both studies, psychiatric and substance use disorder diagnoses were determined using the extended Mini-International Neuropsychiatric Interview, an electronic version of a validated structured diagnostic interview (Sheehan et al., 1998). Within Bipolar CHOICE, psychiatric symptom severity was measured with the Bipolar Inventory of Symptoms Scale (BISS) (Bowden et al., 2007; Gonzalez et al., 2008), which includes 44 items covering five specific symptom dimensions of bipolar disorder: depression, mania, irritability, anxiety and psychosis (Thompson et al., 2010). Within LiTMUS, the Montgomery-Aasberg Depression Rating Scale (MADRS) (Montgomery and Asberg, 1979) and the Young Mania Rating Scale (YMRS) (Young et al., 1978) were used. To combine both studies on a single measure, we extracted MADRS score from the BISS items within the Bipolar CHOICE study. Clinical interviews obtained demographic information, psychiatric and medical history (e.g. hypertension, diabetes) and current medications. In both studies, a fasting blood draw (no specific time of day) at study entry assessed the WBC, which was expressed in International units, i.e., ×109/L. Participants were only excluded because of somatic diseases if the disease contraindicated treatment with the study medications.
Statistical analysis
Since the parameter of interest was WBC at baseline visit, we identified the following variables at baseline possibly influencing on WBC levels: BMI; current smoking; and the medical conditions of diabetes, hypertension and hyperlipidemia.
In both studies, mood symptom severity was greater among patients with the highest and lowest WBC counts. Therefore, we used the following statistical approaches to investigate an association between WBC and mood symptom severity. First, we investigated associations between the absolute deviation from the overall median WBC of all patients at baseline (i.e. both higher and lower WBC counts compared to the median of all patients were taken as a positive value) and mood symptom severity at baseline. We grouped participants based on the deviation from the median WBC into the following categories: <1 × 109/L, 1–1.99 × 109/L, 2–2.99 × 109/L and ⩾3 × 109/L. Since the distribution between the deviation from the median WBC and mood symptom severity fitted a linear relationship in both studies, we performed analysis of variance (ANOVA) and linear regression analysis. We set the group with a deviation of <1 × 109/L as the reference group and report p-values. If an overall ANOVA was significant, we performed pairwise comparisons between different groups, applying Bonferroni, Scheffe and Sidak multiple comparison corrections. For the linear regression analyses, we report coefficients including 95% confidence intervals (95% CIs). The coefficient corresponded to the change in mood symptom severity for each 1.0 × 109/L deviation from the median WBC.
Second, we investigated whether higher or lower WBC levels were associated with increased mood symptom severity. Within LiTMUS, we could divide individuals into fewer categories (<6 × 109/L, 6–6.99 × 109/L, 7–7.99 × 109/L, 8–9.99 × 109/L and ⩾10 × 109/L) compared to Bipolar CHOICE (<4.5 × 109/L, 4.5–5.99 × 109/L, 6–6.99 × 109/L, 7–7.99 × 109/L, 8–9.99 × 109/L and ⩾10 × 109/L). We performed multinomial logistic regression analysis and compared all groups to individuals with a WBC of 7–7.99 × 109/L. For the multinomial regression analysis, we report relative risk ratio (RRR) including 95% CI.
We adjusted all analyses for age, gender, BMI, current smoking status, whether the patients were from the Bipolar CHOICE or the LiTMUS study, and a diagnosis of diabetes, hypertension or hyperlipidemia.
As a primary analysis, we performed the above-mentioned analyses on overall MADRS and YMRS in LiTMUS and on overall BISS within Bipolar CHOICE. As secondary analyses within Bipolar CHOICE, we investigated on associations between WBC and specific groups of BISS symptoms (e.g. depressive or manic) (Thompson et al., 2010). In addition, within the Bipolar CHOICE study, we extracted the MADRS score from the BISS and combined the MADRS scores from the Bipolar CHOICE and LiTMUS studies in order to evaluate the association between WBC and MADRS within both studies. Furthermore, we decided to perform all the above-mentioned analyses separated by gender including gender interaction analyses. Finally, since carbamazepine and clozapine can lower WBC levels (Lally and MacCabe, 2015), we performed sensitivity analyses excluding individuals who used these compounds at baseline. All statistical analyses were performed using STATA version 14.
Results
Within the Bipolar CHOICE and LiTMUS studies, 482 (100%) and 283 (100%) participants had a valid WBC count at study entry, respectively. All participants in Bipolar CHOICE were assessed with the BISS, and all participants in LiTMUS were assessed with MADRS and YMRS. The baseline characteristics of both studies are shown in Table 1 with gender separate baseline characteristics shown in Supplementary Table 1. The overall median WBC was 6.8 × 109/L in Bipolar CHOICE and 7.2 × 109/L in LiTMUS, which is similar to the general US population aged 18 years or above (Liu and Taioli, 2015).
Baseline information on 482 patients with bipolar disorder from the Bipolar CHOICE study and 283 patients with bipolar disorder from the LiTMUS study, depending on white blood cell (WBC) counts at study entry.
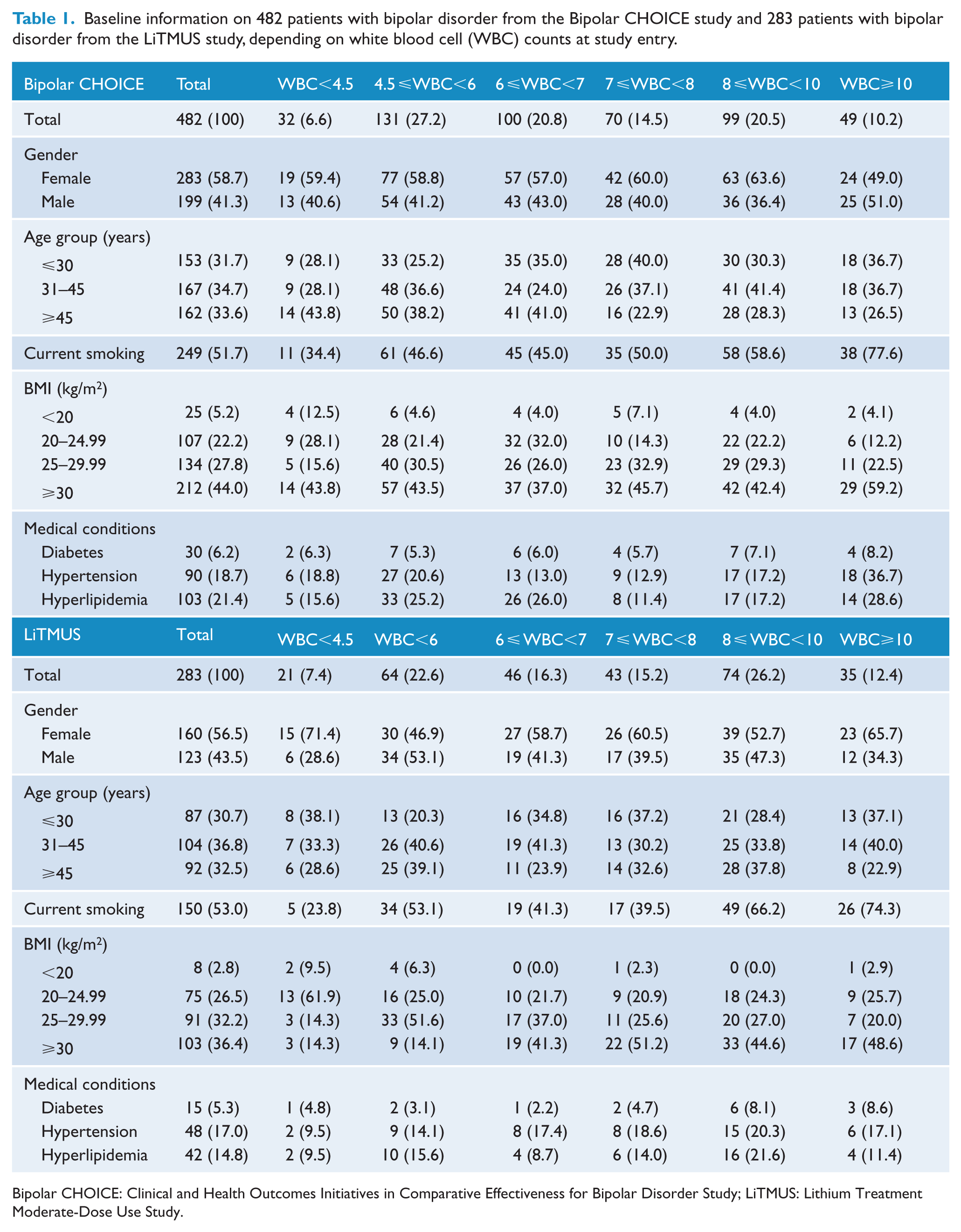
Bipolar CHOICE: Clinical and Health Outcomes Initiatives in Comparative Effectiveness for Bipolar Disorder Study; LiTMUS: Lithium Treatment Moderate-Dose Use Study.
The discovery sample Bipolar CHOICE: WBC levels and BISS symptom severity
Among the 482 Bipolar CHOICE participants, a deviation from the median WBC of 6.8 × 109/L was associated with higher BISS total scores (all p < 0.05) (Tables 2 and 3), which was found among 199 men (p < 0.05), but not among 283 women. The total BISS score increased linearly with a coefficient of 1.74 (95% CI = [0.53, −2.95]) for each 1.0 × 109/L deviation from the median WBC (Table 3). This finding was significant among men (coefficient = 2.13; 95% CI = [0.46, −3.79]; p = 0.013), but not among women (coefficient = 0.87; 95% CI = [−0.87, −1.51]; p = 0.33). Interaction analyses showed a non-significant trend toward higher BISS scores (p = 0.056 and p = 0.20) among men depending on a deviation from the overall median WBC (Tables 2 and 3).
ANOVA a showing the correlation between BISS scores and the absolute deviation from the overall median white blood cell (WBC) count of 6.8 × 109/L among 482 patients (199 men and 283 women) with bipolar disorder from the Bipolar CHOICE study.
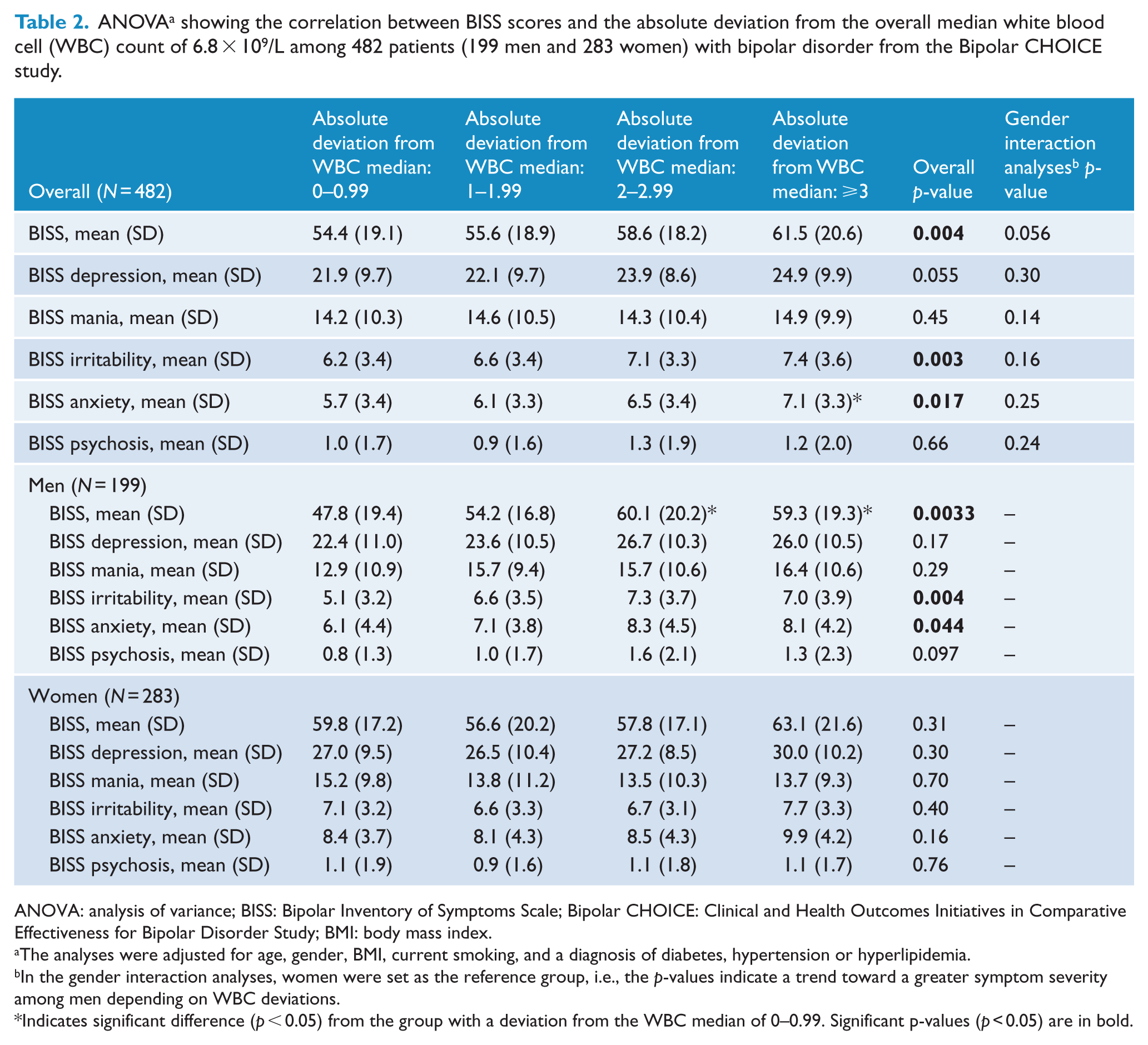
ANOVA: analysis of variance; BISS: Bipolar Inventory of Symptoms Scale; Bipolar CHOICE: Clinical and Health Outcomes Initiatives in Comparative Effectiveness for Bipolar Disorder Study; BMI: body mass index.
The analyses were adjusted for age, gender, BMI, current smoking, and a diagnosis of diabetes, hypertension or hyperlipidemia.
In the gender interaction analyses, women were set as the reference group, i.e., the p-values indicate a trend toward a greater symptom severity among men depending on WBC deviations.
Indicates significant difference (p < 0.05) from the group with a deviation from the WBC median of 0–0.99. Significant p-values (p < 0.05) are in bold.
Linear regression analyses a on the association between the absolute deviation from the overall median white blood cell count (WBC) and BISS among 482 patients (199 men and 283 women) with bipolar disorder from the Bipolar CHOICE study.
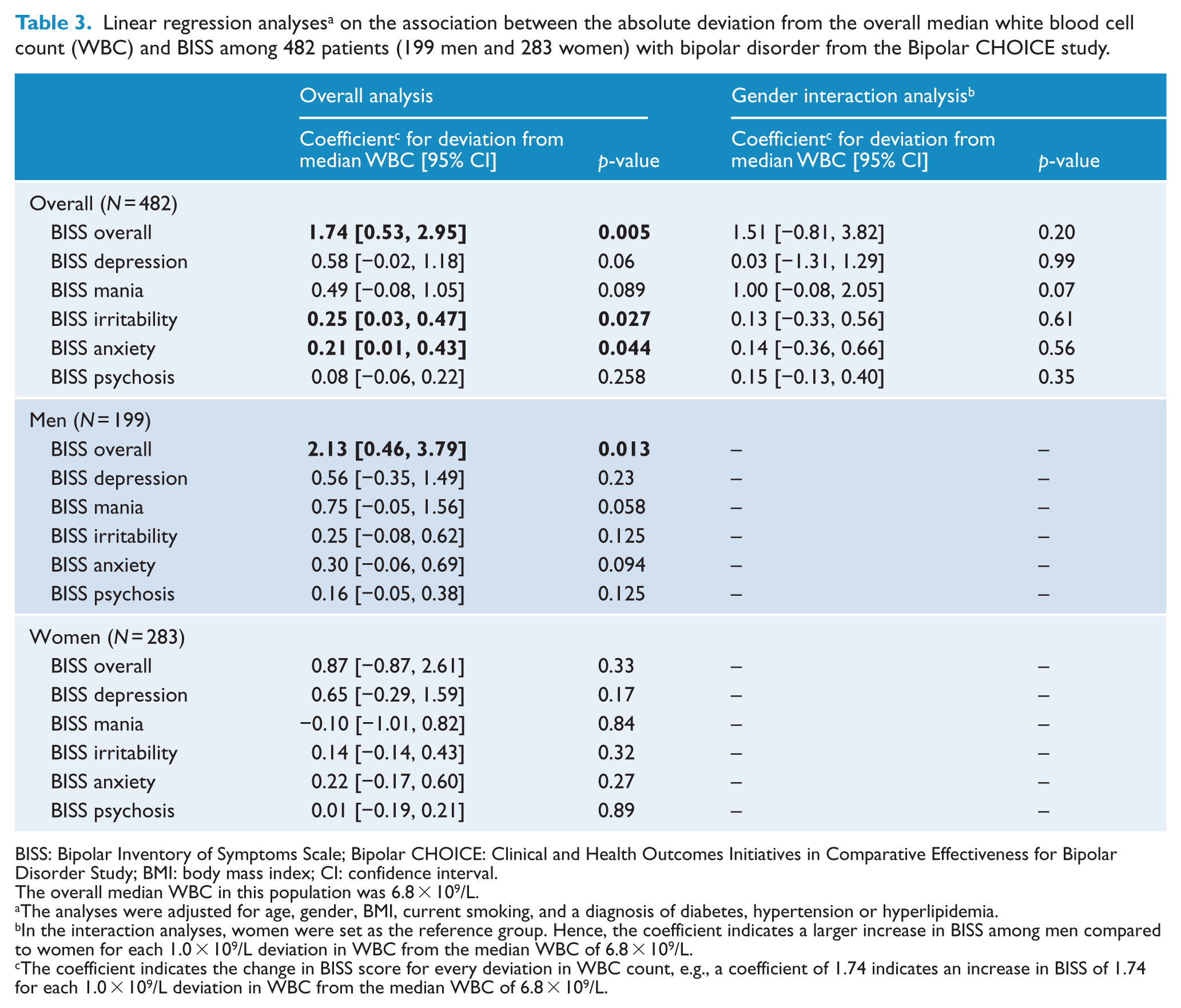
BISS: Bipolar Inventory of Symptoms Scale; Bipolar CHOICE: Clinical and Health Outcomes Initiatives in Comparative Effectiveness for Bipolar Disorder Study; BMI: body mass index; CI: confidence interval.
The overall median WBC in this population was 6.8 × 109/L.
The analyses were adjusted for age, gender, BMI, current smoking, and a diagnosis of diabetes, hypertension or hyperlipidemia.
In the interaction analyses, women were set as the reference group. Hence, the coefficient indicates a larger increase in BISS among men compared to women for each 1.0 × 109/L deviation in WBC from the median WBC of 6.8 × 109/L.
The coefficient indicates the change in BISS score for every deviation in WBC count, e.g., a coefficient of 1.74 indicates an increase in BISS of 1.74 for each 1.0 × 109/L deviation in WBC from the median WBC of 6.8 × 109/L.
Concerning clusters of symptoms, each 1.0 × 109/L deviation from the median WBC was significantly associated with increased anxiety (p = 0.044) and irritability (p = 0.027), whereas depressive and manic symptoms showed borderline significance (Tables 2 and 3). These findings were most pronounced among men. Both lower and higher WBC levels were associated with increased overall BISS total scores, which was most pronounced among men (Supplementary Table 2). Among men, both lower and higher WBC levels showed a trend toward higher manic and anxiety symptoms (Supplementary Table 2). Furthermore, among men, lower WBC levels showed a trend toward increased depressive symptoms, and higher WBC levels showed a trend toward increased irritability. Among women, both lower and higher WBC levels showed a trend toward increased irritability and manic symptoms (Supplementary Table 2).
The replication sample LiTMUS: WBC levels and symptom severity on MADRS and YMRS
Based on our primary findings within Bipolar CHOICE, we were interested if we assessed results in a similar study population, the LiTMUS study. Among 283 LiTMUS participants, a deviation from the median WBC of 7.2 × 109/L showed a tendency toward higher MADRS scores among men (Table 4 and Supplementary Table 3). Men who deviated ⩾3 × 109/L from the median WBC of 7.2 × 109/L had 5 points higher MADRS scores compared to men who deviated <1 × 109/L from the median WBC (Supplementary Table 3). However, the results were not statistically significant. For each 1.0 × 109/L deviation from the median WBC, the overall MADRS showed a trend toward increased scores by a coefficient of 0.56 (95% CI = [−0.36, −1.48]; p = 0.23) (Table 4). Among men, the increase in MADRS was 1.33 (95% CI = [−0.22, −2.89]; p = 0.09), and among women, the increase in MADRS was 0.34 (95% CI = [−0.64, −1.32]; p = 0.50). Both lower and higher WBC levels showed a trend toward increased MADRS scores among men (Supplementary Table 4). We found no associations between WBC deviations and the YMRS score (Table 4 and Supplementary Tables 3 and 4).
Linear regression analyses a showing the association between the absolute deviation from the overall median white blood cell (WBC) count with MADRS and YMRS scores among 283 patients with bipolar disorder (123 males and 160 females) from the LiTMUS study.
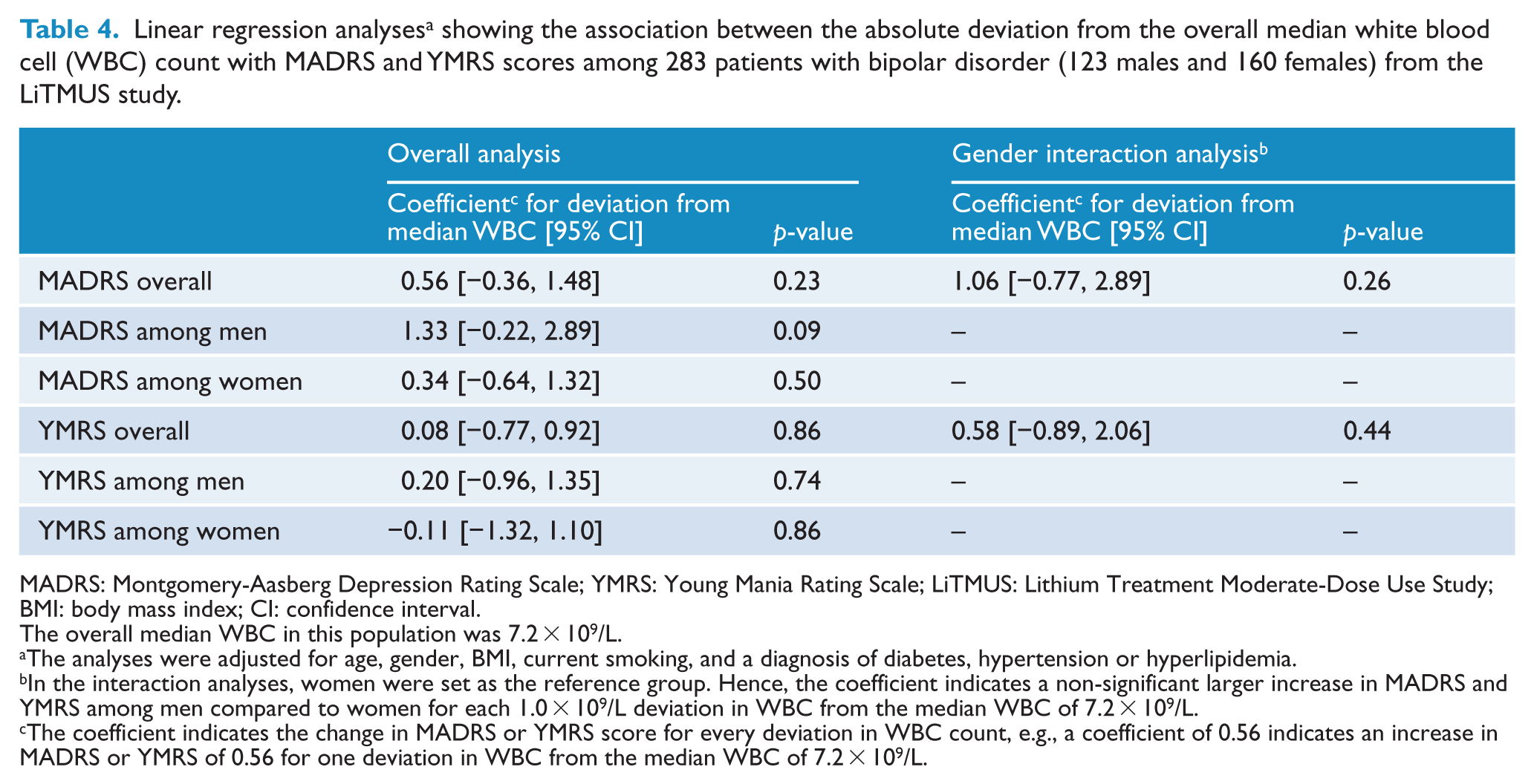
MADRS: Montgomery-Aasberg Depression Rating Scale; YMRS: Young Mania Rating Scale; LiTMUS: Lithium Treatment Moderate-Dose Use Study; BMI: body mass index; CI: confidence interval.
The overall median WBC in this population was 7.2 × 109/L.
The analyses were adjusted for age, gender, BMI, current smoking, and a diagnosis of diabetes, hypertension or hyperlipidemia.
In the interaction analyses, women were set as the reference group. Hence, the coefficient indicates a non-significant larger increase in MADRS and YMRS among men compared to women for each 1.0 × 109/L deviation in WBC from the median WBC of 7.2 × 109/L.
The coefficient indicates the change in MADRS or YMRS score for every deviation in WBC count, e.g., a coefficient of 0.56 indicates an increase in MADRS or YMRS of 0.56 for one deviation in WBC from the median WBC of 7.2 × 109/L.
WBC levels and MADRS scores in Bipolar CHOICE and LiTMUS
Finally, we extracted the MADRS scale from BISS in the Bipolar CHOICE study and combined the MADRS scores from LiTMUS and Bipolar CHOICE. Among 765 patients, MADRS increased with a coefficient of 0.78 (95% CI = [0.24, −1.32], p = 0.005) for each 1.0 × 109/L deviation from the median WBC of 6.9 × 109/L (Table 5). This association was significant among 322 men (coefficient = 1.10; 95% CI = [0.32, −1.89]; p = 0.006) and was weakly associated among 443 women (coefficient = 0.56; 95% CI = [−0.19, −1.31]; p = 0.14). Interaction analyses showed no significant greater symptom severity among men (coefficient = 0.56; 95% CI = [−0.49, −1.60]; p = 0.3). Men who deviated ⩾3 × 109/L from the median WBC of 6.9 × 109/L had 5 points higher MADRS scores compared to men who deviated <1 × 109/L from the median WBC (p = 0.021) (Supplementary Table 5). Both higher and lower WBC levels were associated with increased MADRS scores among men, which was not present among women (Supplementary Table 6).
Linear regression analyses a on the association between the absolute deviation from the overall median white blood cell (WBC) count and MADRS among 765 patients with bipolar disorder from the Bipolar CHOICE and LiTMUS studies.
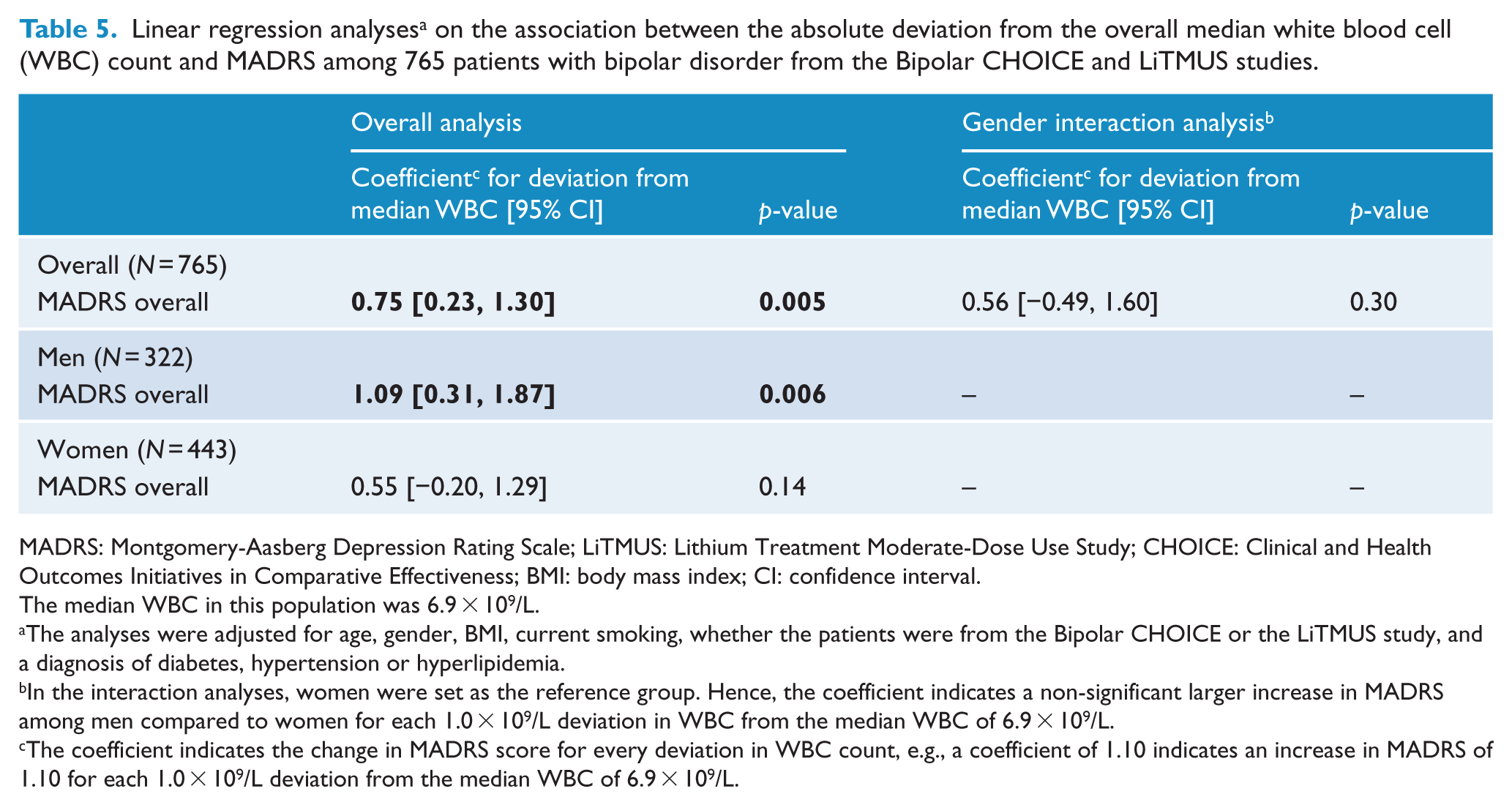
MADRS: Montgomery-Aasberg Depression Rating Scale; LiTMUS: Lithium Treatment Moderate-Dose Use Study; CHOICE: Clinical and Health Outcomes Initiatives in Comparative Effectiveness; BMI: body mass index; CI: confidence interval.
The median WBC in this population was 6.9 × 109/L.
The analyses were adjusted for age, gender, BMI, current smoking, whether the patients were from the Bipolar CHOICE or the LiTMUS study, and a diagnosis of diabetes, hypertension or hyperlipidemia.
In the interaction analyses, women were set as the reference group. Hence, the coefficient indicates a non-significant larger increase in MADRS among men compared to women for each 1.0 × 109/L deviation in WBC from the median WBC of 6.9 × 109/L.
The coefficient indicates the change in MADRS score for every deviation in WBC count, e.g., a coefficient of 1.10 indicates an increase in MADRS of 1.10 for each 1.0 × 109/L deviation from the median WBC of 6.9 × 109/L.
Sensitivity analyses
All significant ANOVA analyses were supported by Bonferroni, Scheffe and Sidak multiple comparison corrections. At study entry, 3 individuals were taking clozapine and 34 were taking carbamazepine. Exclusion of these subjects did not change the results (not shown). Individuals treated with clozapine or carbamazepine tended to have lower symptom severity with higher WBC deviations, although the results were not significant (p = 0.64).
Discussion
This study represents the largest investigation of associations between an overall immune system marker and symptom severity and specific symptoms among 765 patients with bipolar disorder. We present results from two similar clinical trials. Although the results in LiTMUS did not reach significance, the results from Bipolar CHOICE and LiTMUS supported the same trend. The main finding was that in men, for each 1.0 × 109/L deviation from the median WBC of all participants, BISS scale increased by 2.1 points and MADRS scale increased by 1.1 point. No significant association was found among women. Higher and lower WBC levels were associated with greater symptom severity and specific clusters of symptoms, which differed depending on gender but were most pronounced among men. However, interaction analyses only indicated a non-significant trend toward greater symptom severity among men. All results were adjusted for important confounders possibly influencing WBC levels and symptom severity (i.e. age, gender, BMI, smoking, and a diagnosis of diabetes, hypertension or hyperlipidemia).
The immune system and bipolar disorder symptom severity
States of immune system activation represent frequent findings among bipolar disorder patients compared to healthy controls (Bai et al., 2014; Dargel et al., 2015; Drexhage et al., 2010). However, these associations are non-specific, and only a few smaller trials found higher pro-inflammatory markers to be associated with bipolar disorder symptom severity and specific symptom domains (i.e. affective (Hope et al., 2011), psychotic (Hope et al., 2013) and cognitive symptoms (Hope et al., 2015; Lotrich et al., 2014). Another trial found no association between CRP or IL-6 and depressive symptoms (Krogh et al., 2014). Other studies found associations between increased pro-inflammatory markers and cognitive decline in patients with Parkinson’s (Rocha et al., 2014) and Alzheimer’s (Laske et al., 2013) disease, suggesting the observed associations are not specific for bipolar disorder. Nevertheless, these studies were small, did not investigate important baseline characteristics, such as gender, and mostly included pro-inflammatory markers.
On the other hand, the immune system is highly variable, and also depletion of key immune cells, such as T-lymphocytes, may induce a range of neuropsychological problems (Brod et al., 2014). HIV/AIDS represents one of the best examples of T-cell-suppressed emotional imbalance (Rabkin et al., 1994), and treatment with immuno-suppressants can induce depressive and anxiety symptoms (Akaho et al., 2003). Furthermore, CD4 T-cells can enter the central nervous system to yield neuroprotection (Walsh et al., 2014), and animal models have indicated that CD4 T-cell depletion correlates with learning and memory impairments (Brod et al., 2014). In addition, recent studies have indicated that both innate and adaptive immune cells have protective properties in the central nervous system, indicating the importance of a well-balanced immune system (Schwartz et al., 2013). Finally, increased hypothalamic–pituitary–adrenal gland (HPA) axis activity is known to suppress the immune system. However, no clinical trials have investigated the effects of immuno-suppression in patients with bipolar disorder.
Our results are consistent with studies concerning greater symptom severity during inflammatory states (Hope et al., 2013, 2011, 2015; Lotrich et al., 2014), but indicate that increased activity of the immune system may affect several clusters of symptoms. In particular, manic, irritability and psychotic symptoms were increased among individuals with higher WBC levels. Furthermore, our results suggest that also lower WBC levels are associated with increased symptom severity, supporting pre-clinical findings (Brod et al., 2014; Walsh et al., 2014). These pre-clinical studies suggested that low levels of important immune cells, indicating insufficient production, may induce neuropsychological symptoms. This needs to be further investigated in prospective clinical trials which should include several measures over time due to immune system variability. Lower WBC levels tended to be associated with increased symptoms of depression, anxiety, irritability and mania. Hence, this study suggests that both increased activity and suppression of the immune system may affect symptom severity and specific symptoms in bipolar disorder, emphasizing the importance of an optimally balanced immune system. Furthermore, the associations with several clusters of symptoms indicate that the immune system may be of importance in a wide range of neuropsychological disorders. Finally, gender differences appear important since our findings were only significant among men.
WBC in somatic diseases and possible gender differences
WBC deviations have been associated with increased risk of somatic diseases (Horne et al., 2005; Kannel et al., 1992). Within the Framingham study, for each 1.0 × 109/L increment in WBC, the risk for cardiovascular disease (CVD) increased by 32% among men, but not among women (Kannel et al., 1992). Since the comorbidity between bipolar disorder and CVD is well described (Soreca et al., 2008), future studies should investigate whether specific immune system alterations may represent common risk factors and/or etiological pathways.
The apparent gender differences, found in this study and other trials (Kannel et al., 1992), may possibly be explained by the differential effects of sex hormones. Studies in sepsis, representing a complex network between immune and endothelial cell interactions, have shown that female sex hormones exhibit protective effects, whereas male sex hormones can act suppressive on cell-mediated immune responses (Angele et al., 2014). However, we only found a trend toward greater symptom severity among men compared to women.
Implications for personalized treatment
Use of pro-inflammatory markers predicting better treatment response to mood-stabilizing (Li et al., 2015) and anti-inflammatory agents (Arabzadeh et al., 2015; Ayorech et al., 2015) represents promising options for more personalized treatment in bipolar disorder. However, WBC levels yield the advantage of an incorporation of the overall immune system activity. Although this association has not been studied, it may be speculated that lithium treatment may be beneficial in patients with lower WBC levels since lithium increases the amount of circulating WBCs (Amitai et al., 2014). Furthermore, clinical trials need to prospectively investigate whether increased WBC levels, indicating an inflammatory state, may be used as an inexpensive and easy available biomarker predicting use of and response to anti-inflammatory agents. Finally, large clinical trials yield the possibility to identify specific symptoms associated with immune alterations. This knowledge in combination with biomarkers on the immune system may represent a tool for symptom-specific treatment. Hence, large high-quality clinical trials are needed to investigate whether WBC levels may be used as a predictor for more personalized treatment in bipolar disorder.
Strengths and limitations
The LiTMUS and Bipolar CHOICE studies had similar inclusion and exclusion criteria, thus representing populations seen in everyday clinical practice. Both studies had large sample sizes, including in aggregate 765 patients with bipolar disorder. Furthermore, WBC is an easily available and inexpensive measure from peripheral blood, supporting the clinical relevance of our findings. In addition, we were able to adjust for BMI, smoking and specific somatic diseases, important factors concerning WBC levels and symptom severity.
Regarding limitations, we only included measures on overall WBC, a non-specific marker of immune system activity, not the specific subtypes of cells. In CVD, the risk increase associated with WBC deviations seems to depend on specific subtypes of WBCs (Horne et al., 2005). Furthermore, deviations within several immune system cells have been found in depression (Zorrilla et al., 2001), and animal models have indicated potential neuroprotective properties of CD4 T-cells (Brod et al., 2014). Thus, future studies should evaluate specific cell lines. In addition, the use of a wider range of both pro-inflammatory and anti-inflammatory markers may help with a more detailed understanding of the immune system in bipolar disorder. Previous studies have used several pro-inflammatory markers and found that only some, such as IL-1ra and IL-6, were associated with specific clusters of symptoms (Hope et al., 2011, 2013, 2015; Lotrich et al., 2014). This indicates a wide immune system variability, which cannot be measured with one single marker. In addition, WBC may be affected by other factors not accounted for in the analyses. Finally, although we combined two large clinical trials, some analyses on specific symptom-clusters and WBC levels were underpowered, e.g., psychotic symptoms.
Conclusion
Our results indicate that both higher and lower immune system activity, even within the normal range of WBC levels, may impact bipolar disorder symptom severity and specific symptoms. We found that the relationship between WBC deviations from the median WBC and symptoms was statistically significant for men but not women. Future studies should investigate the importance of immune system alterations on mood disorder severity, whether gender differences may be present depending on alterations within the immune system, and the possibilities for using immune system markers as potential predictors of treatment response.
Footnotes
Acknowledgements
The authors would like to thank Dustin Rabideau for important statistical advice.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Köhler has received unrestricted travel grants from the LundbeckFoundation. Dr Nierenberg is a consultant for the Abbott Laboratories, American Psychiatric Association, Appliance Computing Inc. (Mindsite), Basliea, Brain Cells, Inc., Brandeis University, Bristol-Myers Squibb, Clintara, Corcept, Dey Pharmaceuticals, Dainippon Sumitomo (now Sunovion), Eli Lilly and Company, EpiQ, L.P./Mylan Inc., Forest, Genaissance, Genentech, GlaxoSmithKline, Hoffman LaRoche, Infomedic, Lundbeck, Janssen Pharmaceutica, Jazz Pharmaceuticals, Medavante, Merck, Methylation Sciences, Naurex, Novartis, PamLabs, Parexel, Pfizer, PGx Health, Ridge Diagnostics Shire, Schering-Plough, Somerset, Sunovion, Takeda Pharmaceuticals, Targacept and Teva; consulted through the MGH Clinical Trials Network and Institute (CTNI) for AstraZeneca, Brain Cells, Inc, Dainippon Sumitomo/Sepracor, Johnson and Johnson, Labopharm, Merck, Methylation Science, Novartis, PGx Health, Shire, Schering-Plough, Targacept and Takeda/Lundbeck Pharmaceuticals. He receives grant/research support from American Foundation for Suicide Prevention, AHRQ, Brain and Behavior Research Foundation, Bristol-Myers Squibb, Cederroth, Cephalon, Cyberonics, Elan, Eli Lilly, Forest, GlaxoSmithKline, Janssen Pharmaceutica, Lichtwer Pharma, Marriott Foundation, Mylan, NIMH, PamLabs, PCORI, Pfizer Pharmaceuticals, Shire, Stanley Foundation, Takeda and Wyeth-Ayerst. Honoraria include Belvoir Publishing, University of Texas Southwestern Dallas, Brandeis University, Bristol-Myers Squibb, Hillside Hospital, American Drug Utilization Review, American Society for Clinical Psychopharmacology, Baystate Medical Center, Columbia University, CRICO, Dartmouth Medical School, Health New England, Harold Grinspoon Charitable Foundation, IMEDEX, Israel Society for Biological Psychiatry, Johns Hopkins University, MJ Consulting, New York State, Medscape, MBL Publishing, MGH Psychiatry Academy, National Association of Continuing Education, Physicians Postgraduate Press, SUNY Buffalo, University of Wisconsin, University of Pisa, University of Michigan, University of Miami, University of Wisconsin at Madison, APSARD, ISBD, SciMed, Slack Publishing and Wolters Kluwer Publishing ASCP, NCDEU, Rush Medical College, Yale University School of Medicine, NNDC, Nova Southeastern University, NAMI, Institute of Medicine, CME Institute, ISCTM. He was currently or formerly on the advisory boards of Appliance Computing, Inc., Brain Cells, Inc., Eli Lilly and Company, Genentech, Johnson and Johnson, Takeda/Lundbeck, Targacept and InfoMedic. He owns stock options in Appliance Computing, Inc., Brain Cells, Inc and Medavante; has copyrights to the Clinical Positive Affect Scale and the MGH Structured Clinical Interview for the Montgomery Asberg Depression Scale exclusively licensed to the MGH Clinical Trials Network and Institute (CTNI). Dr Tohen was a full-time employee at Lilly (1997 to 2008). He has received honoraria from, or consulted for, Abbott, Actavis, AstraZeneca, Bristol-Myers Squibb, GlaxoSmithKline, Lilly, Johnson & Johnson, Otsuka, Merck, Sunovion, Forest, Gedeon Richter, Roche, Elan, Alkermes, Allergan, Lundbeck, Teva, PamLab, Wyeth and Wiley Publishing. His spouse was a full-time employee at Lilly (1998–2013). During the last 3 years, Dr Terence Ketter has had financial interests/arrangements or affiliations with organizations that could be perceived as real or apparent conflicts of interest. Dr Ketter has received Grant/Research Support (through Stanford University) from the Agency for Healthcare Research and Quality, AstraZeneca Pharmaceuticals LP, Cephalon Inc. (now Teva Pharmaceuticals), Eli Lilly and Company, Pfizer, Inc. and Sunovion Pharmaceuticals; has served as a Consultant/Advisory Board Member for Allergan, Inc., Avanir Pharmaceuticals, Depotmed, Forest Pharmaceuticals, Genentech, Janssen Pharmaceuticals, Merck & Co., Inc., Myriad Genetic Laboratories, Inc., ProPhase, Sunovion Pharmaceuticals and Teva Pharmaceuticals; has received Lecture Honoraria (NOT Speaker’s Bureau payments) from Abbott Laboratories, Inc, GlaxoSmithKline, Otsuka Pharmaceuticals, Pfizer, Inc. and Sunovion Pharmaceuticals; and has received Royalties from American Psychiatric Publishing, Inc. In addition, Dr Ketter’s spouse is an employee of and stockholder of Janssen Pharmaceuticals. The other authors declare no competing interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Institute of Mental Health (Grant/Award Number: ‘NO1MH80001’) and Agency for Healthcare Research and Quality (AHRQ) (Grant/Award Number: ‘1R01HS019371-01’).
