Abstract

To the Editor
Cotard’s syndrome is a rare neuropsychiatric condition which is characterized by nihilistic delusions. There are several neuroimaging studies showing different kinds of brain abnormalities in Cotard’s syndrome. However, 18F-fluorodeoxyglucose-positron emission tomography (18F-FDG PET) scanning has rarely been performed in patients with Cotard’s syndrome (Charland-Verville et al., 2013). Here, we present the first case report of a patient with Cotard’s syndrome in whom hypermetabolism in the basal ganglia has been identified.
A 48-year-old man with schizophrenia was brought to the emergency department by his sister for aggression and thoughts of being dead. Laboratory work-up was normal, and computed tomography (CT) scan showed mild frontal lobe atrophy. He was being prescribed amisulpride 800 mg/day and sertraline 100 mg/day. Neurologic examination revealed cogwheel rigidity in his upper extremities. Because of extrapyramidal system side effects, amisulpride was gradually withdrawn and quetiapine 600 mg/day was started and then increased up to 900 mg/day. Lorazepam 2 mg/day was added to his medication, but no clinical improvement was observed. After 3 weeks of treatment, he was still agitated and his delusions remained. In order to exclude an organic cause, an electroencephalography examination was performed and the findings were normal. Then, 18F-FDG PET imaging was performed, and in three-dimensional stereotactic surface projection analysis, hypometabolism in the frontal–parietal–temporal association cortex, moderate reduction in the cerebellum and severe focal decrease in the left frontal operculum were detected. However notably, hypermetabolic activity was observed in the basal ganglia area (Figure 1).
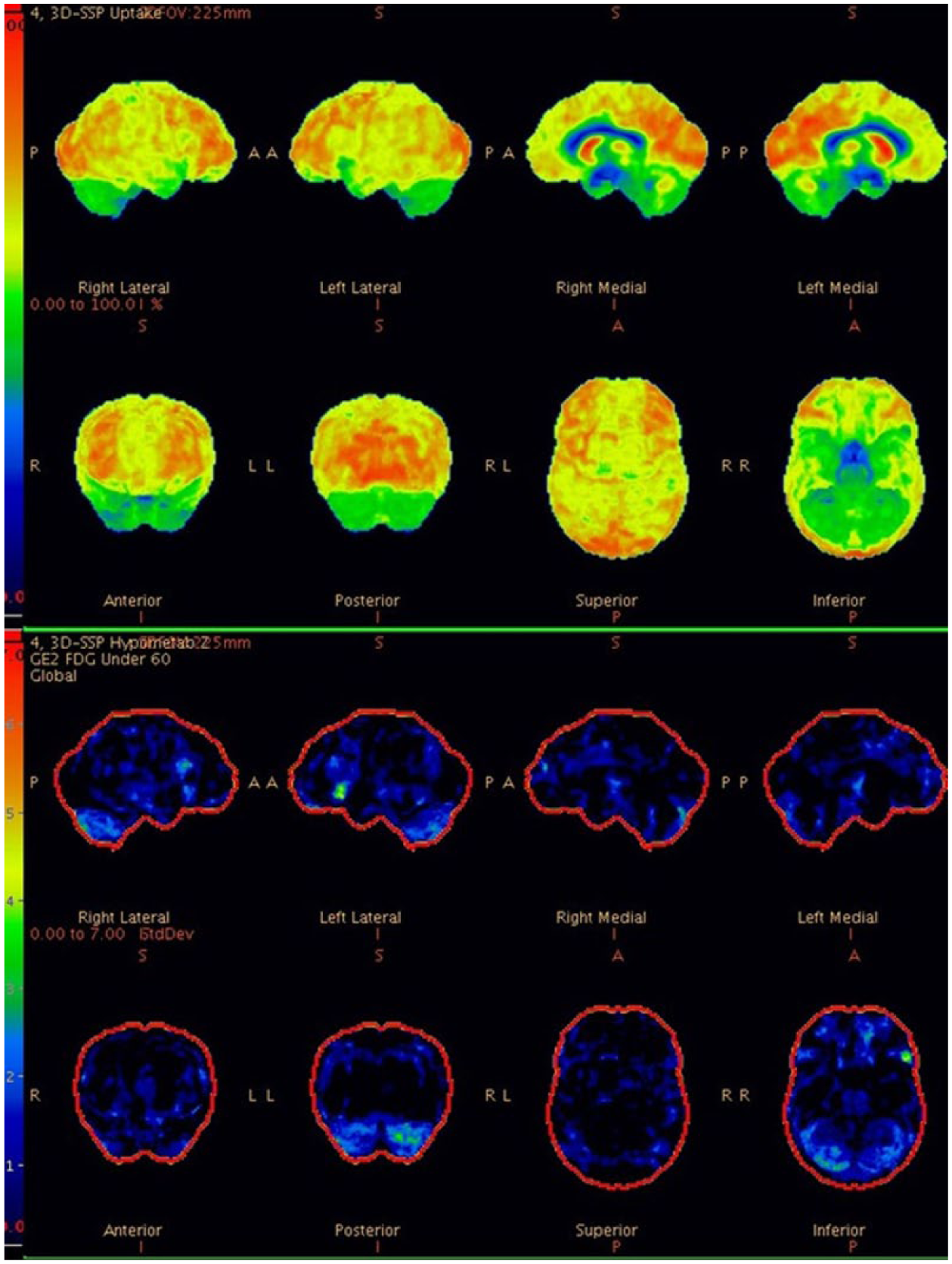
Bilateral metabolic reduction in the frontal–parietal–temporal association cortex with moderate reduction in the cerebellum and severe focal decrease in the left frontal operculum and hypermetabolic activity in the basal ganglia area.
Because of the lack of response to medication, 14 sessions of bitemporal electroconvulsive therapy (ECT) were performed (TM of Somatics, Inc., Lake Bluff). After ECT treatment, his symptoms were improved and he was discharged from hospital. At 6-month follow-up, it was reported that he was still in remission and functioning well.
Here, we report a patient with Cotard’s syndrome in the context of schizophrenia, showing brain metabolism abnormalities in 18F-PDG PET scan and treated with ECT successfully. This report provides new insights regarding possible structural and functional brain abnormalities related to Cotard’s syndrome (Debruyne et al., 2011): hypometabolism in the frontal–parietal–temporal association cortex and hypermetabolism in the basal ganglia might be related to the pathophysiology of the illness and ECT could be first-line treatment in Cotard’s syndrome.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
