Abstract
Objectives:
In accordance with consolidated clinical practice, Diagnostic and Statistical Manual of Mental Disorders, 5th edition suggests a key role of collateral information in the evaluation of retrospective childhood attention-deficit/hyperactivity disorder symptoms in adults despite poor evidence supporting its use. This study aims to assess the incremental value of collateral information on the presence of childhood attention-deficit/hyperactivity disorder symptoms when evaluating adults with attention-deficit/hyperactivity disorder.
Methods:
Adult patients with attention-deficit/hyperactivity disorder (n = 449) and non-attention-deficit/hyperactivity disorder subjects (n = 143) underwent an extensive clinical assessment based on Diagnostic and Statistical Manual of Mental Disorders, 4th edition criteria. For patients, retrospective collateral information regarding childhood attention-deficit/hyperactivity disorder was obtained and used to sort them into two groups: agreement (n = 277) and disagreement (n = 172) between self- and collateral reports. We compared demographic, clinical and response to treatment profiles among groups to test the relevance of collateral information on the specific issue of childhood attention-deficit/hyperactivity disorder symptoms.
Results:
Both attention-deficit/hyperactivity disorder groups had higher rates of several comorbidities (oppositional defiant, conduct, substance use and bipolar disorders; all p < 0.001) and impairments than controls. Disagreement between self- and collateral reports on childhood attention-deficit/hyperactivity disorder symptoms occurred in 38% of patients. Overall, attention-deficit/hyperactivity disorder disagreement and agreement groups had similar profiles in response to treatment and comorbidity, and the few differences detected in impairment measures were of small magnitude (Eta2 < 0.05).
Conclusion:
Although collateral report has an important role for diagnosing attention-deficit/hyperactivity disorder in children, it has no incremental value in the evaluation of childhood attention-deficit/hyperactivity disorder symptoms in adults with a self-reported history of attention-deficit/hyperactivity disorder assessed in clinical settings.
Introduction
Attention-deficit/hyperactivity disorder (ADHD) is characterized by impairing inattentive and/or hyperactive/impulsive symptoms starting in childhood, which persist into adulthood in 15–65% of patients (Faraone et al., 2006a). As a neurodevelopmental disorder, it is mandatory to characterize ADHD symptoms before the age of 12 years (American Psychiatric Association, 2013; Kieling et al., 2010). However, there is evidence that a reduction in the recall of childhood symptoms occurs over time (Todd et al., 2008), diminishing the possibility of diagnosis (Matte et al., 2012). Conversely, the alleged benefits of an ADHD diagnosis given its legal implications (Graf et al., 2013) and the search for cognitive enhancement (Greely et al., 2008) could lead to an unwanted pressure for diagnosis. An approach using self- and collateral reports could minimize those effects, converging in a more precise ADHD diagnosis in adults. In that sense, Diagnostic and Statistical Manual of Mental Disorders, 5th edition (DSM-5) suggests the obtainment of confirmatory reports by informants or collaterals whenever possible (American Psychiatric Association, 2013).
The requirement of collateral information is clearly relevant in the pediatric population, where the diagnostic evaluation is made by assessments including parents, caregivers and teachers (Stanger and Lewis, 1993). It is known that parents are more reliable informants for symptoms and impairment occurring at home and teachers at school (Sayal and Goodman, 2009). In adolescents, findings suggest that the best source of information to evaluate the presence of retrospective ADHD symptoms is the combination of parents’ and teachers’ reports instead of self-report (Sibley et al., 2012a). However, it seems that both adolescents and their parents provide inaccurate reports regarding childhood ADHD symptoms (Miller et al., 2010).
In adults, the relevance of collateral information confirming the presence of symptoms during childhood is less clear (Mannuzza et al., 2002; Miller et al., 2010; Todd et al., 2008). Adults tend to underreport the number and severity of symptoms, and informants could be used to get additional data on the presence of symptoms, its severity and the level of impairment (Barkley et al., 2002; Kooij et al., 2008; Sibley et al., 2012b). The agreement between adult patients and informants in relation to the retrospective diagnosis in childhood can vary, ranging from 21% to 74% (Dias et al., 2008; Kooij et al., 2008; Sibley et al., 2012b; Zucker et al., 2002) in previous studies. However, despite the fact that patients and their parents have poor recall of symptoms, the presence of current symptomatology improves the accuracy of recall (Mannuzza et al., 2002; Miller et al., 2010). Adults with ADHD appear to be the best informants regarding their symptoms (Kooij et al., 2008), and self-report of symptoms is also more correlated with the clinical evaluation than the collateral report (Kooij et al., 2005; Magnússon et al., 2006). More recently, results from a population follow-up study (Moffitt et al., 2015) demonstrated that only 22% of parents of adults diagnosed with ADHD in childhood were able to recall that their children were diagnosed with ADHD or had important symptoms of ADHD during infancy.
Despite conflicting data, some research groups (Adler and Cohen, 2004; Haavik et al., 2010) and the DSM-5 (American Psychiatric Association, 2013) itself encourage clinicians to seek collaterals to corroborate ADHD diagnosis in adults. Since self-report is usually the only source of information available for the majority of adult patients, the effort to obtain collateral information on childhood symptomatology would be justified if a robust evidence of its relevance could be demonstrated (Matte et al., 2012). This study aims to assess the incremental value of collateral information on the presence of childhood ADHD symptoms when evaluating adults with ADHD. To achieve this objective, we compared an extensive clinical profile between adult patients whose informants confirm or not the presence of childhood ADHD symptoms.
Methods
Subjects
The ADHD sample comprised 449 adult patients referred to the ADHD outpatient clinic of the Hospital de Clínicas de Porto Alegre (HCPA), a major teaching hospital in Brazil, evaluated from 2002 to 2012. Subjects were Brazilians of European descent, older than 18 years, fulfilling the diagnostic criteria for Diagnostic and Statistical Manual of Mental Disorders, 4th edition (DSM-IV) ADHD, both currently and in childhood. The exclusion criteria were the presence of significant neurological disease affecting cognition (e.g. epilepsy, history of cerebrovascular accidents or degenerative disorders), lifetime history of psychosis, IQ lower than 70 and, for this analysis, lack of available source of collateral information. All measurements were performed after enrollment.
The control sample consisted of 143 replacement blood donors recruited at the blood bank of HCPA and evaluated by the same diagnostic protocol used for ADHD patients. The inclusion/exclusion criteria were the same as those used for the ADHD sample, except for ADHD diagnosis (an exclusion criterion for controls). Thus, individuals with current and lifetime psychiatric disorders other than ADHD were not excluded.
The study was approved by the institutional review board (IRB) of our university hospital (approved as an IRB by the Office for Human Research Protections, United States of America, IRB 00000921). All participants signed an informed consent.
Diagnostic procedures
The diagnostic evaluations were conducted by trained psychiatrists in a three-step procedure: (a) direct clinical interview with the patient for lifetime ADHD clinical diagnosis, (b) application of the socio-demographic and clinical protocol for formal diagnosis based on DSM-IV (details below) and (c) conflicting cases were discussed in a clinical committee chaired by a senior psychiatrist (E.H.G.; for more detailed information, see Grevet et al. (2006) and Fischer et al. (2007)).
Clinical protocol
All psychiatric diagnoses were based on DSM-IV criteria (American Psychiatric Association, 2000). The ADHD and oppositional defiant disorder (ODD) diagnoses were performed using the Portuguese version of the Schedule for Affective Disorders and Schizophrenia for School-Age Children, Epidemiological version (K-SADS-E) (Mercadante et al., 1995). The K-SADS-E is a semi-structured interview used for children and adolescents aged 6–18 years assessing current and lifetime episodes of psychiatric disorders (Ambrosini, 2000). For its use in adult population, some modifications were necessary. We adapted the examples given by K-SADS-E to describe ADHD symptoms for childhood and adolescence to make them more suitable for its use in adults (Grevet et al., 2005). In addition, the criterion of onset of symptoms was adjusted to age 12 or earlier due to operational advantages and diagnostic reliability (Applegate et al., 1997; Karam et al., 2009; Kieling et al. 2010; Rohde et al., 2000). This modification was based on DSM-IV field trials showing that more hyperactive cases tend to present the onset of symptoms before the age of 7 years, while inattentive cases usually present the onset after this age (Applegate et al., 1997). The age of 12 years was chosen to avoid the inclusion of non-ADHD cases, supported by findings showing that up to 83% of late-onset ADHD cases have their age of onset before 12 years (Faraone et al., 2006b). Finally, we required a self-reported DSM-IV full syndrome during childhood to avoid the inclusion of dubious cases in our sample. The inter-rater agreement for childhood and current ADHD diagnosis using K-SADS-E was evaluated, and the Kappa coefficients were high (Grevet et al., 2005).
The evaluation of conduct disorder and antisocial personality disorder was performed using the Brazilian Portuguese version of the Mini-International Neuropsychiatric Interview (MINI) (Amorin, 2000; Sheehan et al., 1998). All other Axis I psychiatric comorbidities were evaluated using the Structured Clinical Interview for DSM Disorders (SCID-I) (First et al., 1998).
The severity of ADHD symptoms was assessed using the Portuguese version of the Swanson, Nolan and Pelham scale, version IV (SNAP-IV) (Mattos et al., 2006; Swanson, 1992). This is a Likert-type scale including items from DSM-IV criteria for ADHD and ODD based on a 0–3 rating scale (not at all = 0, just a little = 1, quite a bit = 2 and very much = 3). The scores are obtained by summing the result from each item per dimension (inattention, hyperactivity/impulsivity and ODD) and dividing them by the total number of items of certain dimension. Its total score is calculated by summing the means from each dimension divided by 3.
Besides the clinical requirement of significant impairment in at least two settings of social, academic or occupational functioning to establish the ADHD diagnosis, impairment was measured in 10 different areas using Barkley’s current and childhood ADHD symptoms scales. This instrument is based on a 0–3 Likert rating scale which requires patients to report how often symptoms interfere in his or her life (never or rarely = 0, sometimes = 1, frequently = 2 and very frequently = 3) (Barkley and Murphy, 1998).
Treatment protocol
The treatment protocol (Victor et al., 2009, 2014) was designed according to Brazilian guidelines for ADHD (Mattos et al., 2006), similar to worldwide practice (Nutt et al., 2007). A 6-month pharmacologic treatment was initiated for those fulfilling inclusion criteria and that desired to use a stimulant. Immediate-release methylphenidate (IR-MPH) was the drug of choice due to its free availability in the Brazilian public health system. IR-MPH was administered two or three times daily, with weekly increases in the dose until symptoms control was reached or there was the occurrence of intolerable adverse effects. Patients with concomitant psychiatric disorders were first treated for the comorbidities, and IR-MPH was started only after comorbidities stabilization. Thus, the concomitant use of other psychiatric medications was allowed (Gibbins and Weiss, 2007; Newcorn, 2009). The final mean (standard deviation [SD]) IR-MPH dose was 0.52 (0.21) mg/kg per day, ranging from 0.13 to 1.23 mg/kg per day. From the initial sample (n = 449), 232 patients (143 in the agreement group and 89 in the disagreement group) completed the treatment protocol and data on response to treatment were included in the analysis. Treatment response measures were calculated using the difference in the scores of the SNAP-IV obtained before IR-MPH initiation (baseline) and after symptom control (endpoint).
Collateral information assessment
Collateral information was obtained for the ADHD sample based on the collateral report form of Barkley’s scale. This instrument has questions inquiring collateral to inform the frequency of the 18 DSM ADHD symptoms during a specific timeframe from 5 to 12 years of age and the frequency in which these symptoms interfered in 10 different life areas of the patient (Barkley and Murphy, 1998). Researchers asked patients to convey the scale to a parent or a close relative who lived with the patient during his or her childhood and was capable of perceiving his or her functioning. Also, patients were instructed to allow the collateral to fulfill the scale independently of his or her influence. In all, 89% of the ADHD sample obtained information from first-degree relatives (e.g. parents or older siblings), with the remaining collaterals being constituted by second-degree relatives (e.g. grandparents and uncles/aunts).
For patients whose collateral scored 2 or 3 in a particular symptom of the scale, it was considered present during childhood. To reassure that the presence of symptoms during childhood was undoubtedly perceived by collaterals, we used a full syndrome to consider ADHD present during childhood. Thus, once six or more symptoms from the list of nine inattentive or hyperactivity/impulsivity symptoms were fulfilled, childhood ADHD was considered present by collateral report. Therefore, the ADHD sample was divided into two groups: (1) ADHD patients with informants who agree with the diagnosis in childhood and (2) ADHD patients with informants who disagree with the diagnosis in childhood.
Statistical analyses
To assess the importance of collateral report, we compared psychosocial and comorbidity profile, ADHD symptoms severity, impairment scores and response to treatment in adult patients with ADHD whose informants agree (n = 277) or disagree (n = 172) regarding the presence of childhood symptomatology and a control group of non-ADHD adults (n = 143). Categorical variables were analyzed by Pearson’s chi-squared test followed by Bonferroni post hoc. Continuous variables were analyzed by univariate general linear model (GLM) and Bonferroni post hoc comparisons. Since 31 independent tests were carried out, we considered as significant p-values less than 0.0016 (αBonf = 0.05/31). All tests were two-tailed, and the analyses were performed with SPSS 20.0 software.
Effect size estimation
To assess between-groups effect sizes, we used the GLM to measure the proportion of the variance explained (Eta2) for three sets of comparisons: (1) disagreement versus control (Eta2a), (2) agreement versus control (Eta2b) and agreement versus disagreement (Eta2c). We considered Eta2 values as small (⩽0.05), medium (0.06 to 0.13) or large (⩾0.14) according to Cohen (1988).
Results
Demographic and psychosocial profile
ADHD patients and controls did not differ regarding sex and income. Differences observed in age and years of schooling did not survive Bonferroni correction. Patients with ADHD whose collaterals agreed or not in relation to childhood ADHD differed from the control group by presenting more school failure, problems with authority and discipline, problems with the law and police and previous psychopharmacological treatment (all p < 0.001). Patients whose collaterals agreed, as compared with those who did not, presented more school suspensions and problems with authority and discipline (p < 0.001). However, these groups were not different in relation to school failure, problems with the law and police and previous psychopharmacological treatment (Table 1).
Demographic and psychosocial profile.
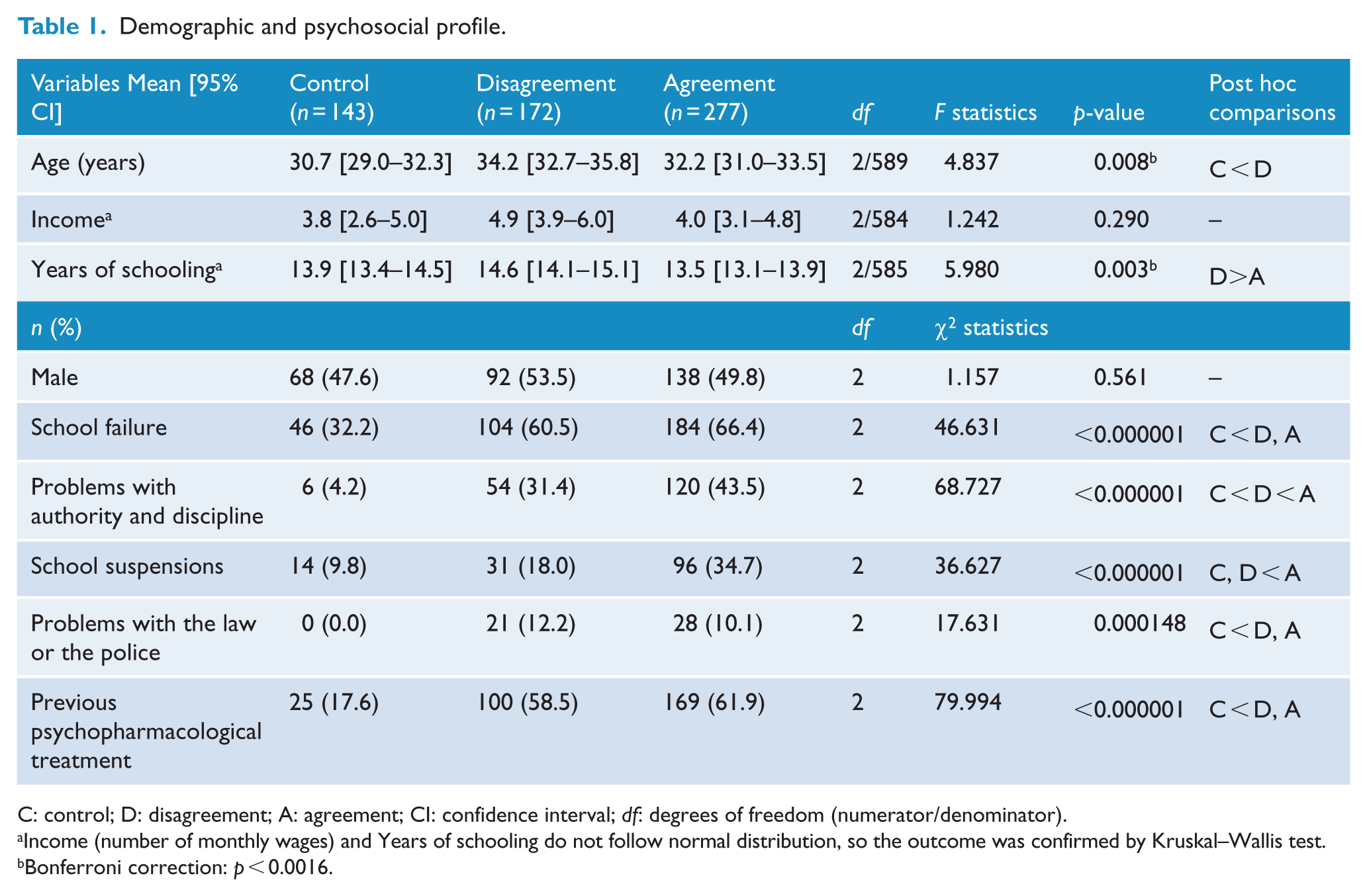
C: control; D: disagreement; A: agreement; CI: confidence interval; df: degrees of freedom (numerator/denominator).
Income (number of monthly wages) and Years of schooling do not follow normal distribution, so the outcome was confirmed by Kruskal–Wallis test.
Bonferroni correction: p < 0.0016.
Comorbidities
The three groups did not differ in relation to the frequencies of major depressive disorder, panic disorder and social and specific phobias. Both ADHD groups differed from the control group (but not between themselves) by presenting higher rates of tobacco use, bipolar disorder, current ODD, conduct disorder and non-alcohol substance dependence (all p < 0.001). The differences observed between ADHD groups and non-ADHD group regarding antisocial personality disorder, alcohol dependence and generalized anxiety disorder did not survive Bonferroni correction (Table 2).
Lifetime comorbidities.
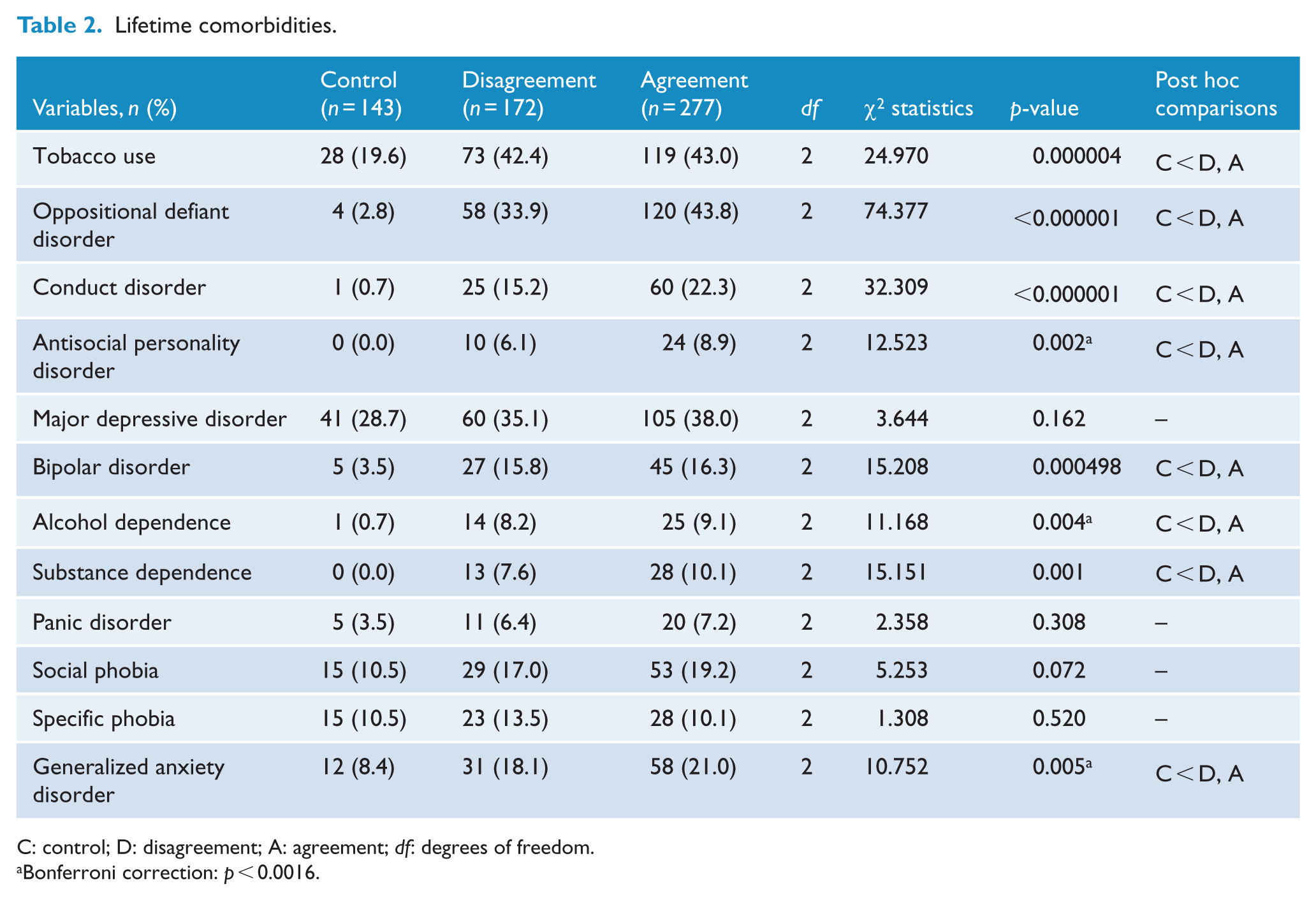
C: control; D: disagreement; A: agreement; df: degrees of freedom.
Bonferroni correction: p < 0.0016.
Severity, impairment and response to treatment
When comparing ADHD symptoms severity and impairment of the three groups, the agreement group presented higher symptom scores than the other groups, while both ADHD groups had higher scores than controls (all p < 0.001). The only exception was the self-report of current problem areas, where both ADHD groups did not differ. The effect sizes observed on symptoms severity and impairment were of large magnitude when comparing both ADHD groups versus non-ADHD group. For the comparison between ADHD groups themselves, the estimations of effect size were of small magnitude, except for childhood impairment that was considered of medium magnitude. There were no differences in response to treatment when comparing the ADHD groups (Table 3).
Severity, impairment and response to treatment.
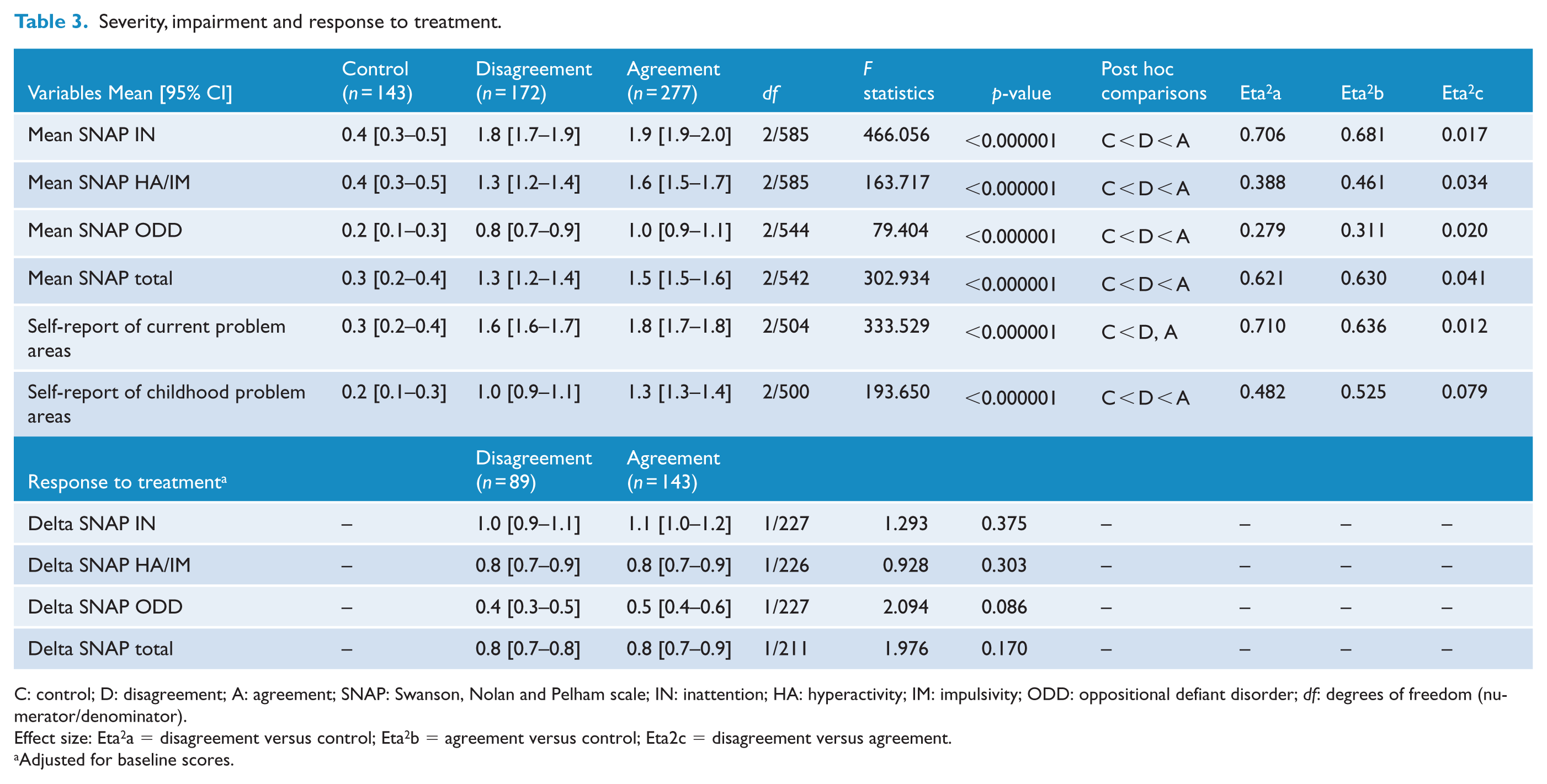
C: control; D: disagreement; A: agreement; SNAP: Swanson, Nolan and Pelham scale; IN: inattention; HA: hyperactivity; IM: impulsivity; ODD: oppositional defiant disorder; df: degrees of freedom (numerator/denominator).
Effect size: Eta2a = disagreement versus control; Eta2b = agreement versus control; Eta2c = disagreement versus agreement.
Adjusted for baseline scores.
Since the retrospective assessment of number of symptoms is always associated with recall bias, we also tested for the entire set of analyses a more rigorous definition of disagreement (cases with three or less inattention and/or hyperactivity/impulsivity symptoms during childhood by informant report). Few differences were observed in relation to the first analysis: age and years of schooling went on to survive the Bonferroni correction; antisocial personality disorder and bipolar disorder were observed more frequently in the agreement group than in controls; and nominal differences in effect sizes (but not in their magnitude) were observed on severity of symptoms and impairment scores (see Supplementary Material).
Discussion
The main finding of this study is that almost 40% of collaterals reported dubious or inexistent ADHD diagnosis during childhood for clinically referred patients with a clear self-reported lifetime history of the disorder. Despite statistically significant differences in discipline problems and ADHD severity, patients whose collaterals do not confirm childhood ADHD have a clear ADHD clinical profile. This implies that the use of such information as a retrospective confirmatory source of childhood ADHD does not have incremental value for diagnostic purposes.
Our findings are in accordance with results from Kooij et al. (2008), showing that collaterals of clinically diagnosed adult patients report childhood symptoms less frequently than patients referred to treatment. Also, these findings partially agree with Zucker et al.’s (2002) results, where correlations between self-report and informants were moderate. However, the study design was different and included a majority of non-ADHD subjects. On the other hand, Sibley et al.’s (2012b) findings pointed to a different direction since they observed that parents report symptoms with more sensitivity. However, it is noteworthy that they investigated young adults that had been diagnosed during childhood. Therefore, it is likely that their parents had a much better recall than in our study since we assessed self-referred adult patients with a mean age of 34 years, without a previous diagnosis of ADHD.
Despite the efforts to make psychiatry a more precise science, a gold standard diagnostic procedure is still missing, and the clinical interview remains the best diagnostic approach (Faraone and Tsuang, 1994). As a neurodevelopmental disorder, ADHD must have a clear beginning in childhood, and the description of symptoms at this developmental stage is essential for the diagnosis (Matte et al., 2012). We did not find robust and clinically significant differences in demography, severity of symptoms, impairment, response to treatment and comorbidity profiles between ADHD groups for whom agreement and disagreement occurred on ADHD symptoms in childhood to rule out ADHD diagnosis in the disagreement group. Reinforcing this idea, both ADHD groups strongly differed from the non-ADHD group on the variables assessed. However, patients from the agreement group presented greater current symptoms severity, more school suspensions, problems with authority and more impairment during childhood, than may be relevant to justify a distinct between-groups clinical understanding and approach. It is important to note that our findings remain true even when using a more strict definition of disagreement (three or less symptoms of inattention/hyperactivity during childhood by collateral report). Therefore, requiring the agreement would preclude the diagnosis in more than one-third of our sample of patients that otherwise clearly presented all other ADHD diagnostic criteria. A noteworthy finding was that patients with school suspensions and problems with authority and discipline had higher probability to belong to the group whose collaterals positively reported ADHD symptoms during childhood. The results reinforce the idea that social misbehavior during childhood is an important issue for collateral recall. These findings are in the same direction of the studies from Young et al. (2009) and Dwyer et al. (2006) that link ADHD with behavior disinhibition and observe that informant rates are more accurate at identifying children at risk of externalizing problems.
Some limitations must be taken into account while interpreting the results of this study. As the collateral information was collected by self-applicable scales and not by face-to-face interviews, it is not possible to rule out measurement biases. However, at least in at-risk populations, rating scales seems to have valid diagnostic properties (Luty et al., 2009; Magnússon et al., 2006) and its use is a routine in different ADHD adult clinics worldwide, where bringing parents to evaluation is difficult. Moreover, our findings are in agreement with a recent longitudinal study where collaterals were assessed in a similar manner as in our study (Moffitt et al., 2015) showing that less than one-fourth of parents could recall that their children had core symptoms or childhood ADHD. Thus, we believe that our results reflect collateral recall bias rather than a measure bias attributable to the fulfilling of scales.
Also, our findings were derived from a clinical population reporting a full syndrome during childhood. Thus, it is not possible to extrapolate our results to those patients with current ADHD symptoms, but with difficulties to recall childhood symptoms. For these patients, collaterals may be a valuable source of information. However, if in an adult sample of well-characterized self-reported childhood ADHD syndrome we observed that almost 40% of the collaterals reported a dubious description, one would not expect a better recall of childhood symptoms from collaterals in populations presenting few ADHD symptoms. This notion is in agreement with the findings from the population-based study by Moffitt et al. (2015).
Our results cannot be generalized to a context of low prevalence or in the presence of subthreshold cases (e.g. non-clinically referred patients or population samples), where collateral information could be relevant. In this regard, the addition of collateral information to those obtained from self-report allowed a more accurate estimative of adult ADHD heritability in a study carried out with a population sample of twins (Chang et al., 2013). Finally, our results concern exclusively to ancillary information on retrospective recall of childhood symptoms and they are not applicable for collateral information on other clinically relevant aspects of ADHD patients. This could include, for example, collateral information concerning the presence of current ADHD symptoms, differential diagnosis of confounding comorbidities and a better estimation of current impairment.
In conclusion, at least in clinical settings of adult patients with a self-report lifetime history of ADHD, the retrospective collateral information does not have incremental value for diagnostic purposes. The use of collateral information in clinical settings could be considered as a source of ancillary information subjected to critical clinical judgment.
Footnotes
Declaration of interest
Eugenio H. Grevet has served as a speaker to Shire and Novartis Pharmaceuticals in the past 3 years. He also received travel awards for taking part in 2014 ADHD World Federation meeting from Shire. Luis A. Rohde has received Honoraria, has been on the speakers’ bureau/advisory board and/or has acted as a consultant for Eli-Lilly, Janssen-Cilag, Novartis and Shire in the last 3 years. He receives authorship royalties from Oxford Press and ArtMed. He also received travel awards for taking part in 2014 APA and 2015 WFADHD meetings from Shire. The ADHD and Juvenile Bipolar Disorder Outpatient Programs chaired by him received unrestricted educational and research support from the following pharmaceutical companies in the last 3 years: Eli-Lilly, Janssen-Cilag, Novartis and Shire. All other authors declare that they have no conflicts of interest.
Funding
The research was supported by grants from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq; Grant 476529/2012-3), Scholarships from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul (FAPERGS-PRONEX; Grant 09/0148-9) and Fundo de Incentivo à Pesquisa e Eventos do Hospital de Clínicas de Porto Alegre (FIPE-HCPA).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
