Abstract
Objective:
Oestradiol has been implicated in the pathogenesis of schizophrenia. Women with schizophrenia often suffer with menstrual dysfunction, usually associated with low oestradiol levels, but whether menstrual dysfunction has an effect on their psychiatric symptoms is not well researched. The aim of this study is to document the menstrual characteristics of women with chronic schizophrenia with focus upon menstrual regularity, menstrual cycle length and menstrual symptoms. To determine which patient characteristics are associated with irregular menses and whether irregular menses are associated with the severity of psychotic symptoms, menstrual symptoms or depressive symptoms.
Method:
Cross-sectional analyses using baseline data of women enrolled in a clinical trial. Inclusion criteria include
Results:
Of the 139 women, 77 (55.4%) had regular menses, 57 (41%) had irregular menses and 5 (3.6%) women had missing data on their menstrual cycle. Use of atypical antipsychotics associated with hyperprolactinaemia was positively associated with irregular menses (odds ratio = 4.4, 95% confidence interval = [1.8, 10.9],
Conclusion:
Regular menses are associated with higher oestradiol levels and higher rates of cyclical mood symptoms but are not associated with Positive and Negative Syndrome Scale scores. Understanding the effect the menstrual cycle can have on psychiatric illness, such as premenstrual exacerbations, is important for the holistic care of women with schizophrenia.
Introduction
Schizophrenia is a severe mental illness that affects approximately 1% of the population worldwide and causes considerable morbidity (Prince et al., 2007). Gender differences in the presentation of schizophrenia have been recognised for over a century (Kraepelin, 1909-15). Women with schizophrenia are at higher risk of acute psychotic symptoms at times of physiologically low or decreasing serum oestradiol levels, such as postpartum (Matevosyan, 2011) and the menopause transition (Seeman, 2012b). The fluctuations in serum levels of oestradiol in the menstrual cycle also have a significant effect on psychopathology in some women (Seeman, 2012a). Admissions to psychiatric units are reported to be more common in the late luteal and early follicular menstrual phase, and lowest in the high oestradiol ovulatory phase (Lande and Karamchandani, 2002). We recently reported that adjunctive treatment with high-dose oestradiol patches has an antipsychotic effect in women with treatment-resistant schizophrenia (Kulkarni et al., 2014), further adding to the weight of evidence that hormones related to the menstrual cycle play an important role in modulating the symptoms of schizophrenia.
Menstrual dysfunction is common in women with schizophrenia and is at least partially accounted for by antipsychotic induced hyperprolactinaemia (Chiang et al., 2011; Magharious et al., 1998; Prentice and Deakin, 1992; Smith et al., 2002; Wieck and Haddad, 2003). Irregular menses are usually associated with anovulation (Munro et al., 2011) and therefore lack of rise in oestradiol levels in the luteal phase of the menstrual cycle.
There has also been a suggestion that poorer cognition may be associated with menstrual dysfunction in women with schizophrenia (Prentice and Deakin, 1992). Although several studies have documented the changes in symptoms of psychosis across the menstrual cycle (Bergemann et al., 2007; Hallonquist et al., 1993; Seeman, 2012a), there is a dearth of information on menstrual symptoms in women with schizophrenia.
Therefore, in this study, we have documented the menstrual cycle characteristics of a group of women with treatment-resistant schizophrenia, with a particular focus upon menstrual regularity, menstrual cycle length and symptoms associated with menstruation. Our aims were to determine (1) which patient characteristics are associated with irregular menses and (2) whether irregular menses are associated with psychotic symptoms, menstrual symptoms or depressive symptoms.
Methods
Participants
Cross-sectional analyses were performed using data taken from baseline characteristics of women enrolled in a clinical trial of adjunctive oestradiol for treatment-resistant schizophrenia. The inclusion criteria for this study were
The study had approval from the Human Ethics Committee of the Alfred Hospital (202/04) and was registered at ClinicalTrials.gov (NCT00357006).
Characterising the menstrual cycle
Women were asked questions on menstrual cycle regularity, the date of the last menstrual cycle and usual menstrual cycle length. Menses were classified as regular if they occurred once every 3–5 weeks. Cycle lengths outside this time frame were considered irregular and included women with amenorrhoea. Menstrual cycle length was assessed in women who had menstruated within the last 6 months. Women were also asked whether they experienced a series of physical symptoms in relation to their menses, or whether their psychotic symptoms or mood worsened before, during or after their period.
Hormone assays
Hormone assays were performed by the Department of Biochemistry, Alfred Hospital. A single 10 mL blood sample was collected at baseline for serum oestradiol, prolactin and testosterone.
Psychopathology
Symptoms of schizophrenia were measured using the Positive and Negative Syndrome Scale (PANSS) (Kay et al., 1987). The PANSS is a semi-structured interview that consists of 30 items across three subscales – positive, negative and general psychopathology. The subscales are added to create a total PANSS score, with a range of 30–210. Cognition was measured using the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) (Randolph, 1999). Depression was assessed using the Montgomery–Asberg Depression Rating Scale (MADRS), a 10-item researcher rated scale of depression (Montgomery and Asberg, 1979).
Antipsychotic medications
Antipsychotic medications act as dopamine antagonists and can be classified as either ‘typical’ or ‘atypical’. Atypical antipsychotics differ from the older generation typical antipsychotics in their greater specificity for the dopamine D2 receptor. Dopamine inhibits the secretion of prolactin from the pituitary gland, with greater affinity for the D2 receptor associated with hyperprolactinaemia.16 In particular, risperidone, amisulpride and paliperidone are well known to cause hyperprolactinaemia.(Haddad and Wieck, 2004; Peuskens et al., 2014). For the purposes of these analyses, antipsychotic medications were clustered into three categories: typical antipsychotics (TA), atypical antipsychotics that are not likely to cause hyperprolactinaemia (AA) and atypical antipsychotics that are likely to cause hyperprolactinaemia (PAA) (Table 1).
Categories of antipsychotics.

Statistical analysis
For the purposes of these analyses, women using a hormonal contraceptive (the combined oral contraceptive pill, depot progestin or progestin implant) were excluded. Descriptive statistics are presented as number (percent) for categorical data and mean (standard deviation) for descriptive data.
Bivariate regression was used to determine factors associated with irregular cycles. A multiple logistic regression model was constructed with the outcome defined as ‘irregular cycles’ versus ‘regular cycles’. Factors significantly associated with irregular cycles in bivariate analysis were used to construct a multiple logistic regression model with the outcome defined as ‘irregular cycles’ versus ‘regular cycles’. All analyses were undertaken using STATA version 12.0 (StataCorp). An alpha of 0.05 was used to determine a significant effect.
Results
Overall, between 2006 and 2011, 180 women were recruited to the study. Of these, 41 (22.7%) were excluded as they were using hormonal contraception. The remaining 139 women comprise the study sample.
The mean age of the group was 35.0 ± 8.2 years, with a mean duration of illness of 9.6 ± 7.5 years (Table 2). TA were used by 23 (16.6%) women, AA by 81 (58.3%) and PAA by 35 (25.2%) women, with a mean total antipsychotic dose in risperidone equivalents of 7.7 ± 4.9 mg.
Characteristics of entire sample.
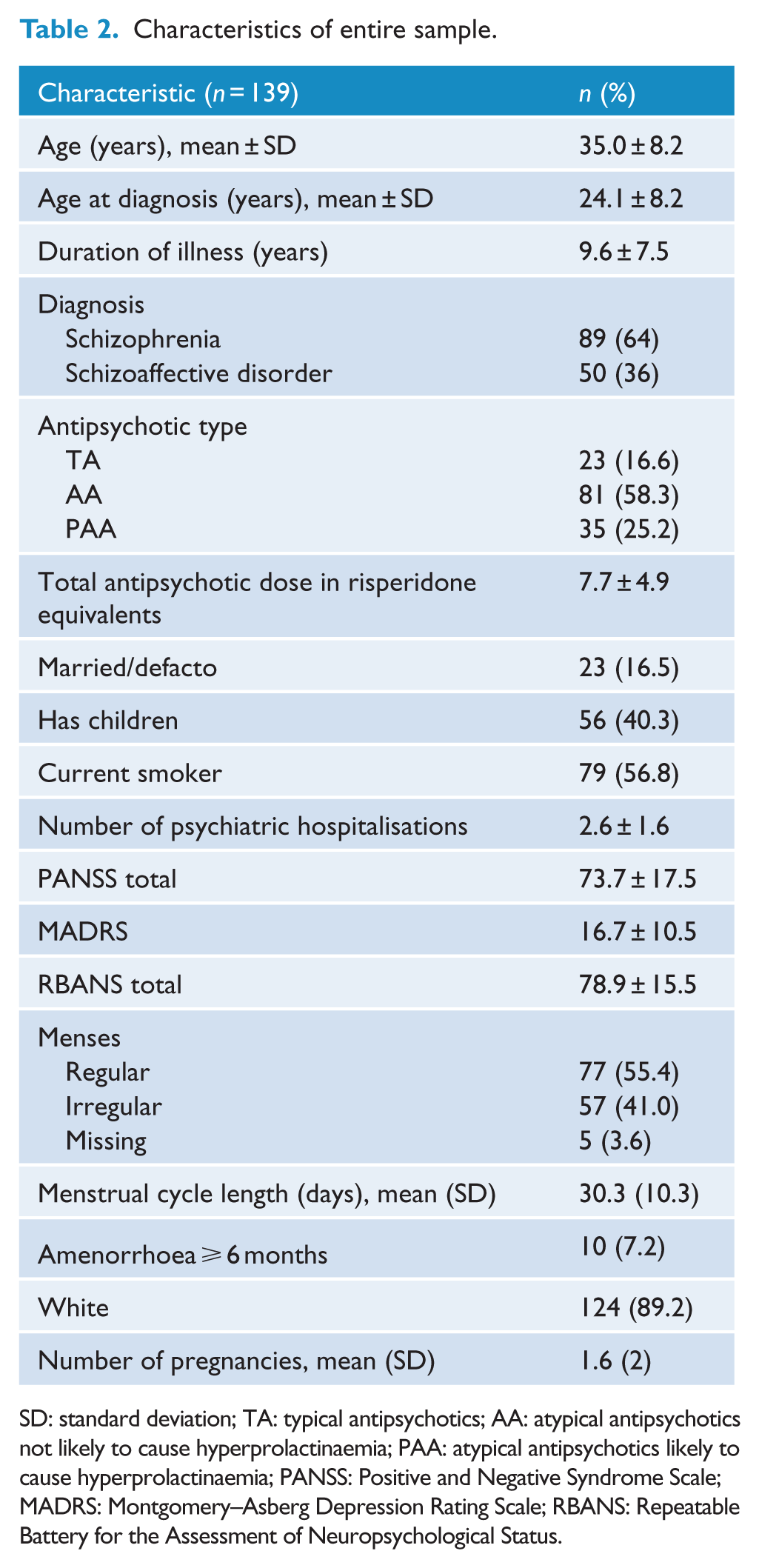
SD: standard deviation; TA: typical antipsychotics; AA: atypical antipsychotics not likely to cause hyperprolactinaemia; PAA: atypical antipsychotics likely to cause hyperprolactinaemia; PANSS: Positive and Negative Syndrome Scale; MADRS: Montgomery–Asberg Depression Rating Scale; RBANS: Repeatable Battery for the Assessment of Neuropsychological Status.
Menstrual associated symptoms
Worsening of psychosis symptoms with the menstrual cycle was reported by 45 (32.4%) women. The most common menstrual associated symptoms were decrease in mood with the menstrual cycle (64.8%) and bloating (64.8%), followed by cramps (59.7%) and back pain (36%) (Table 3).
Menstrual cycle associated symptoms.
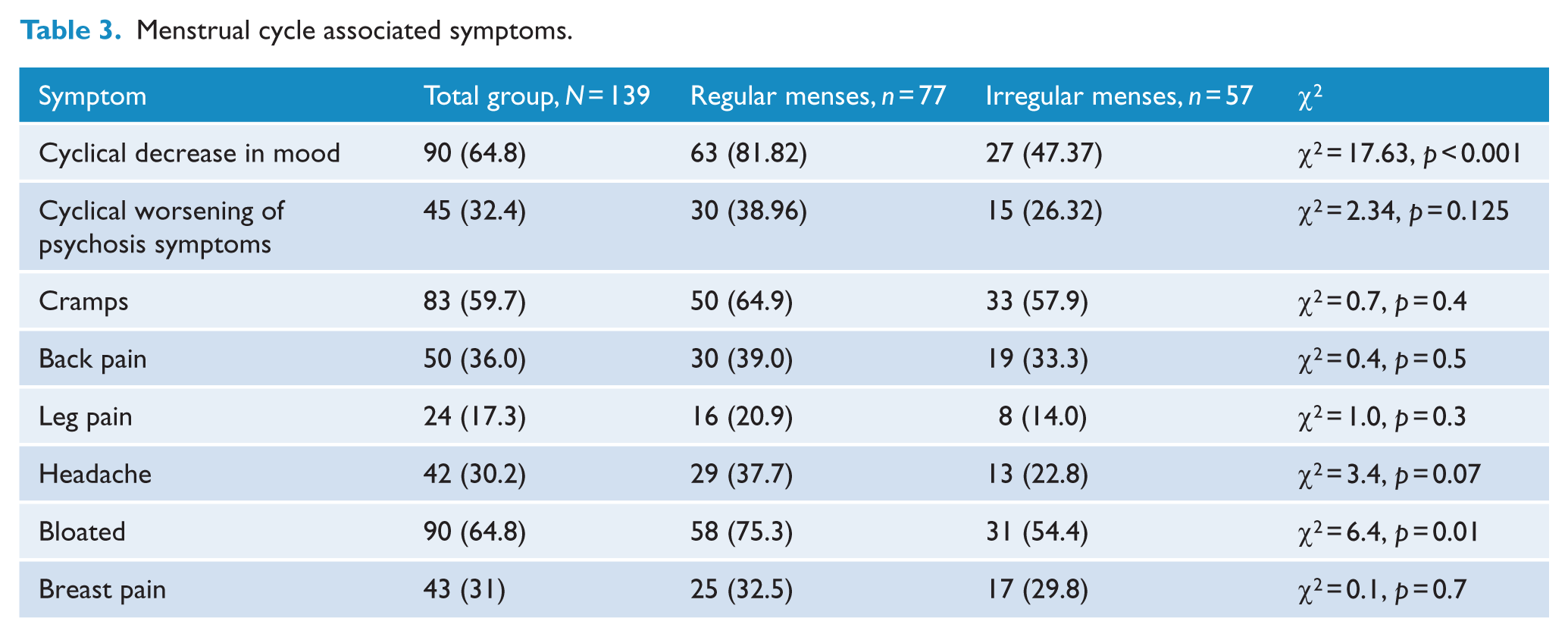
Menstrual cycle regularity
Of the 139 women in the sample, 77 (55.4%) had regular menses, 57 (41%) had irregular menses and 5 (3.6%) women had missing data on their menstruation status.
Women with regular menses as compared to women with irregular cycles had higher serum oestradiol levels (299.0 ± 27.3 vs 213.2 ± 25.0,
Menstrual regularity was not associated with the PANSS or MADRS but was associated with a higher mean total RBANS (78.77 ± 16.47 vs 73.14 ± 14.27,
Factors associated with irregular menses.
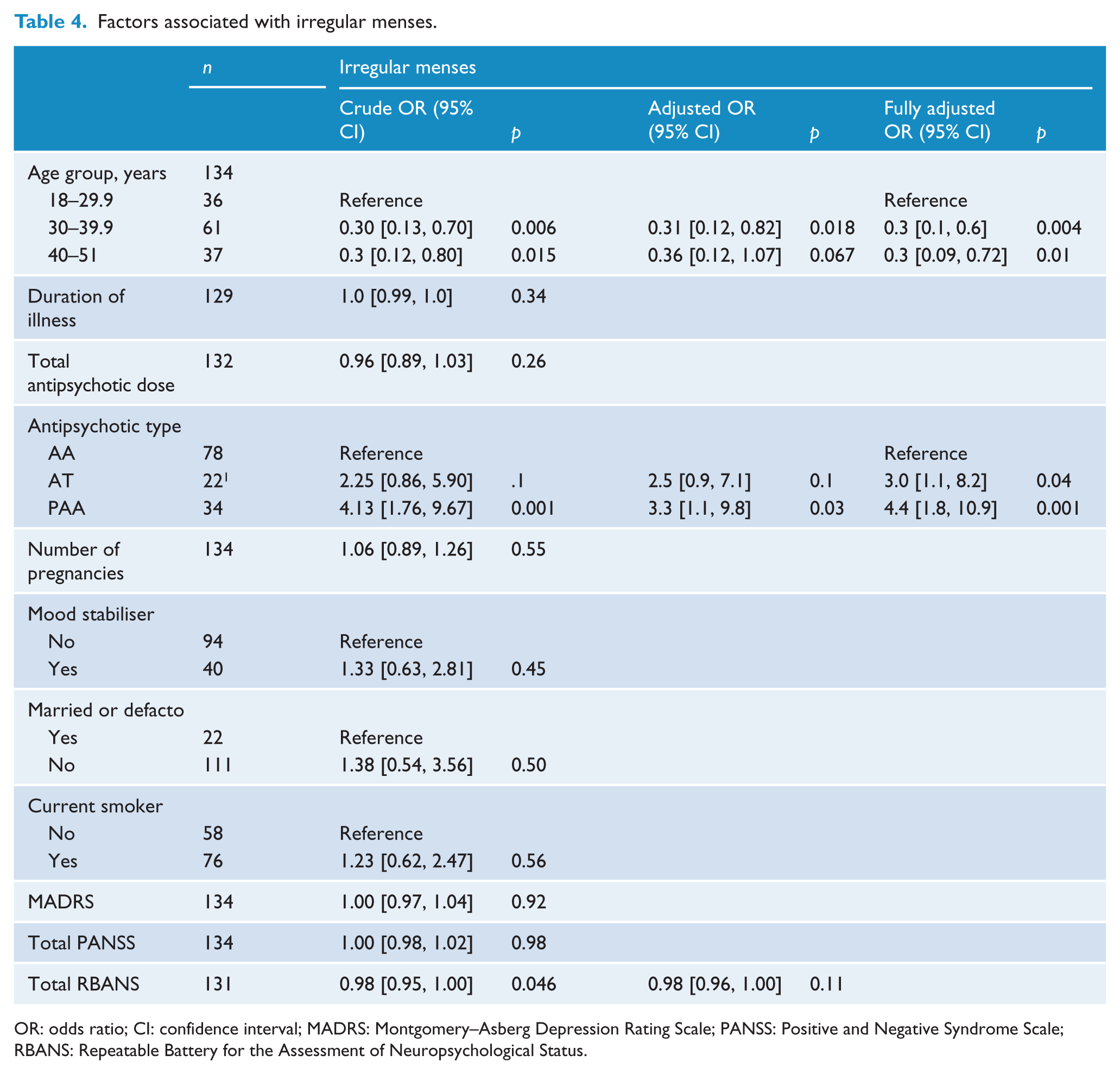
OR: odds ratio; CI: confidence interval; MADRS: Montgomery–Asberg Depression Rating Scale; PANSS: Positive and Negative Syndrome Scale; RBANS: Repeatable Battery for the Assessment of Neuropsychological Status.
In the fully adjusted logistic regression model, women aged more than 30, as compared to women aged less than 30, were significantly less likely to have irregular cycles (OR = 0.3, 95% CI = [0.1, 0.6],
Hormone levels differed significantly with women with regular cycles having higher oestradiol (299.0 ± 27.3 vs 213.2 ± 25.0,
Menstrual cycle length
There was a significant difference in menstrual length between those with regular and irregular cycles (28.5 ± 3.2 vs 34.8 ± 17.9
Menstrual cycle length and hormone levels by antipsychotic group.
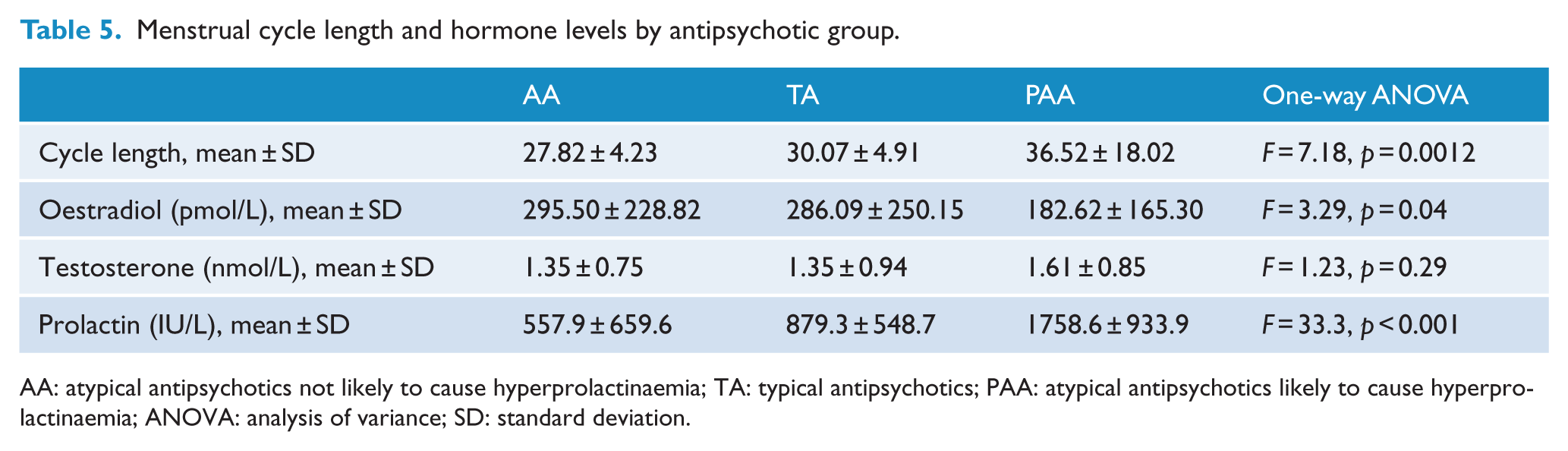
AA: atypical antipsychotics not likely to cause hyperprolactinaemia; TA: typical antipsychotics; PAA: atypical antipsychotics likely to cause hyperprolactinaemia; ANOVA: analysis of variance; SD: standard deviation.
Discussion
In this sample of women with chronic, treatment-resistant schizophrenia, there was a very high rate of menstrual dysfunction, with 41% of all women and more than two-thirds of women taking PAA. Although women on risperidone, amisulpride and paliperidone had the highest prolactin levels, elevated prolactin levels were observed in all antipsychotic groups. Atypical antipsychotics have various effects on prolactin levels, with clozapine, quetiapine and olanzapine showing little or no elevation (Tollefson and Kuntz, 1999) compared to risperidone and amisulpride, which have shown marked and sustained elevation (Wieck and Haddad, 2003). The increase in prolactin levels associated with antipsychotics is believed to be due to the antipsychotic drugs themselves, as patients with acute or chronic psychiatric disorders who have not yet been placed on antipsychotic medication having normal prolactin levels (Rao et al., 1994).
Young women were also more likely to have menstrual dysfunction than older women. It would be expected that older women would have more irregular cycles, as varying cycle length is a feature of the menopause transition, which usually begins in the fifth decade. However, the majority of women in the study were in their 30s and early 40s, and it has been found in community-based studies of healthy women that menses tend to be more regular in this age group compared to women in their early 20s (Davison et al., 2005; Hahn et al., 2013). Irregular and long menstrual cycles are associated with chronic anovulation and subsequently reduced exposure to oestradiol. Women with irregular cycles in this study did have significantly lower oestradiol levels. Menstrual irregularity can also be a feature of polycystic ovary syndrome, which may explain the higher testosterone levels seen in women with irregular cycles. The median testosterone level of 1.7 nmol/L in women with irregular menses is noteworthy, as only 10% of healthy women in the community will have a testosterone level of 1.7 nmol/L or more. Many of these women therefore have significant hyperandrogenism.
There was no difference in PANSS or MADRS scores between those with regular and irregular cycles. There was no association between cognitive function and menstrual irregularity, contrary to an earlier, but smaller study (Prentice and Deakin, 1992). On the other hand, women with regular menses were significantly more likely to experience change in mood with the menstrual cycle. Premenstrual dysphoria has been observed in a substantial number of women with schizophrenia, although it is unclear whether such symptoms are due to schizophrenia per se or superimposed premenstrual syndrome or premenstrual dysphoric disorder (Choi et al., 2001; Seeman, 2012a).
Smokers had shorter menstrual cycles than non-smokers, although this was not a significant difference, presumably due to the sample size. Shorter cycles in smokers have been seen in large studies in women without schizophrenia (Hahn et al., 2013). This is important to note as the prevalence of smoking among patients with schizophrenia is much higher than the general population (Lohr and Flynn, 1992). It has been suggested that smoking can lead to a hypoestrogenic state in women which in turn can lead to abnormal menstrual cycle characteristics including menstrual length variability (Grossman and Nakajima, 2006).
The main strengths of this study are the large sample size and breadth of data collected in this very hard to recruit patient group. This is not a random sample of women and so cannot be used to provide an estimate of the prevalence of menstrual dysfunction in women with schizophrenia. Interpretation of hormone assays in this study is also limited as blood collection was not collected at specific times in the menstrual cycle. Ideally, blood samples would be drawn on day 3–5 of the menstrual cycle; however, because of the nature of the study, this was not practical. However, this study has identified factors associated with menstrual dysfunction in women with treatment-resistant schizophrenia, of which there is little research to date. Understanding the relationship between menstrual cycle characteristics and schizophrenia is important in helping to maximise successful treatment and supportive care.
Given the beneficial effects of oestradiol on psychotic symptoms (Kulkarni et al., 2014) and the negative medical consequences of oligo-amenohorrea, attention to menstrual dysfunction in women with schizophrenia is an important clinical priority. These results suggest that the severity of cyclical mood symptoms should be taken into consideration whenever attempts are made to normalise menstrual cycles in women with schizophrenia.
In conclusion, in women with chronic, treatment-resistant schizophrenia, menstrual dysfunction is very common and associated with antipsychotics known to induce hyperprolactinaemia and younger age. Irregular menses are associated with lower oestradiol levels and a high prevalence of hyperandrogenism. Many women experience cyclical mood symptoms and worsening of psychotic symptoms premenstrually, particularly if they have regular cycles. There was no association between menstrual cycle regularity and PANSS scores.
Footnotes
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
Funding
The funding for this study was provided by the Stanley Medical Research Institute, Washington, USA. Grant ID: 05T-742
