Abstract

Recent evidence suggests a compelling link between autoimmunity and bipolar disorder (Rege and Hodgkinson, 2013). We report a case of a 21-year-old Caucasian female who presented with a first episode of mania and was initiated on risperidone (2 mg) and sodium valproate (20 mg/kg/day). The diagnosis was bipolar affective disorder with a current episode of mania. After 1.5 weeks, she showed no response to treatment and was transferred to the high-dependency unit due to increasing psychomotor agitation. She exhibited disinhibition, inappropriate giggling, psychomotor agitation, a decreased need for sleep and elevated mood. On-going observation showed fluctuating levels of cognition and poor memory. Her history revealed a recent bereavement 3 months previously and an episode of viral illness with fever 2 weeks before the onset of the current episode. There was no family history or previous history of mental illness. There was no drug or alcohol misuse. An organic screen revealed an anti-nuclear antibody (ANA) level of 1:640 (nucleolar type, normal range < 1:40) with a double-stranded DNA (dsDNA) level of 17 (normal range < 4). The patient’s white-cell count was normal and her mid-stream urine showed no evidence of infection. There were no electrolyte abnormalities. An electroencephalogram (EEG) showed generalised slowing that was especially prominent in the frontal region. Magnetic resonance imaging (MRI), positron emission tomography (PET) and single-photon emission computed tomography (SPECT) scans were normal. Cerebrospinal fluid (CSF) showed no infectious or inflammatory profiles. A provisional diagnosis of autoimmune encephalopathy was made, and an empirical trial of methylprednisone (1 g/day for 5 days) was instituted. The patient responded dramatically within 3 days and showed a marked improvement in her mental state; she was transitioned to 1 mg/kg/day of prednisone in two divided doses. She remained stable during this period, and no changes were made to the psychiatric medication. During the reduction of prednisone from 80 mg to 50 mg, she relapsed, which prompted maintenance on the previous dose. She was started on metformin due to raised blood sugars. The prednisone was ceased after 2 weeks, but 2 weeks after this cessation she relapsed. On this occasion, she was initially treated with intravenous gamma globulin with no response; she was then treated with plasmapheresis and showed an excellent response within 2 days. She was discharged on mycophenolate mofetil (1 g BD), which was changed after 2 months to azathioprine (100 mg daily) because of concerns about alopecia. The psychiatric medication was gradually reduced and ceased. The patient’s serum tested positive for anti-neuronal antibodies on indirect immunofluorescence (Figure 1). She was negative for all of the specific antibodies tested for, including: amphiphysin, ANNA-1/anti-Hu, ANNA-2/anti-Ri, PCA, Yo antibodies, phMA2 and Ma2/Ta. The anti-N-methyl-
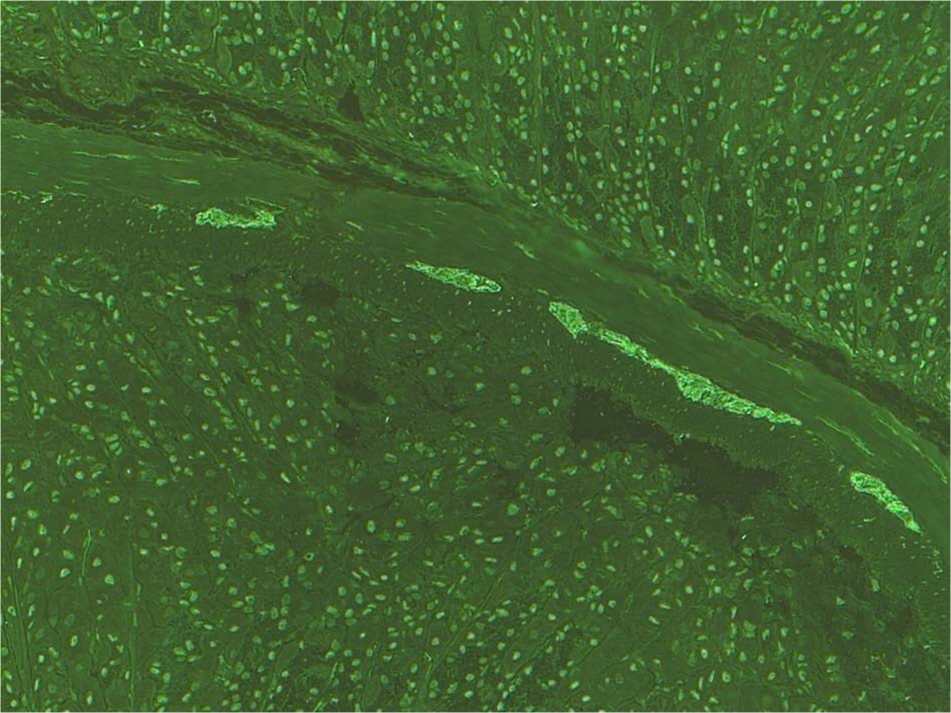
The patient’s serum was tested on monkey brain tissue. The immunofluorescence illustrates staining of neuronal structures, indicating the presence of anti-neuronal antibodies binding to the neuronal structures. This staining, however, is not consistent with any of the previously known antigens described in synaptic encephalitis. The clinical significance of this pattern is currently unknown and it is not possible to comment on the pathogenic potential. Further research is required to clarify its significance.
Neuroinflammation and peripheral immune dysregulation is postulated in the pathogenesis of bipolar disorder. This involves a complex relationship between the immune cells of the periphery (T and B lymphocytes) and the central nervous system (microglia, astroglia and oligodendrocytes) resulting in cellular damage through excitotoxicity, oxidative stress and mitochondrial dysfunction. Neuropsychiatric systemic lupus erythematosus (NPSLE) and synaptic encephalitides, particularly anti-NMDA encephalitis, are important differentials for the psychiatrist to consider in presentations of psychosis. In NPSLE, evidence suggests the brain may be affected early in the course of the disease (Joseph et al., 2007) with a negative correlation with systemic manifestations (Karassa et al., 2000). In neurology, Dalmau’s group have identified autoantibodies acting on specific synaptic sites in the brain (Rosenfeld and Dalmau, 2011) and the question has been raised about whether some psychiatric presentations are manifestations of autoimmunity. Seventy-five per cent of anti-NMDA encephalitis cases initially present to psychiatrists and milder cases (formes frustes) may be missed due to the absence of overt neurological symptomatology. Anti-NMDA encephalitis and NPSLE involve different subunits of the NMDA receptor: anti-NMDA encephalitis is associated with NR1 (Kayser and Dalmau, 2011) whilst NPSLE is associated with antibodies to NR2A/B, which may explain their different phenotypic presentations (DeGiorgio et al., 2001). Antibodies to dsDNA, which is highly specific in SLE, cross-reacts with the NR2 receptor (DeGiorgio et al., 2001), linking the peripheral antibodies with the brain. The exact nomenclature of the disorder in our case report is subject to debate and may be classified broadly as autoimmune encephalopathy. Our case had antibodies found in SLE (ANA and dsDNA) but no other American College of Rheumatology (ACR) criteria for SLE. This finding raises the question of whether this disease is a sub-clinical form of NPSLE, as previous studies have shown that neuropsychiatric presentations can be the initial manifestation of NPSLE. The patient’s mother also described possible photosensitivity 2 years prior to her presentation and a fever with swollen glands (glandular fever?) in the patient and her brother 2 weeks prior to the initial presentation. Anti-neuronal antibodies and the acute behavioural change following viral illnesses are characteristic of anti-NMDA encephalitis. It is unclear if the viral illness was glandular fever, but Epstein–Barr virus (EBV), which is associated with glandular fever, is well known to be associated with SLE and has been included in the St Thomas alternative criteria for the diagnosis of lupus (Hughes, 1998). The mechanism of EBV-induced antibody production is postulated to be through molecular mimicry. Indirect immunofluorescence testing showed staining for neuronal structures in serum, indicating anti-neuronal antibodies but not in a pattern known for previously defined antibodies (Figure 1). Antibodies to the NR2 receptor have been shown to be elevated in acute episodes of mania (Dickerson et al., 2012). The poor response to psychotropics, followed by a good response to immunosuppressants, indicates immune dysfunction and is consistent with synaptic encephalitis. The good outcome in this case may have been due to early intervention, as in the course of some autoimmune limbic encephalopathies (Kayser and Dalmau, 2011). The case raises questions about the role of autoimmunity in psychiatry and underscores the importance of vigilance for autoimmunity in psychiatry and of a collaborative multispecialty approach due to its therapeutic implications.
The immune/inflammatory hypothesis in bipolar disorder creates a paradigm shift in psychiatry that has significant research and therapeutic implications. From a treatment perspective, consideration of the immune hypothesis opens up a new avenue for research into the role of immunomodulators in psychiatry. The relationship between psychiatric and autoimmune disorders has a high biological plausibility, and the final effector mechanism may be the glutamatergic pathway through the NMDA receptors situated in the hippocampus, frontal regions and limbic system (Mak et al., 2009). Owing to continued under recognition, with subsequent inappropriate treatment and poor outcomes (Thachil et al., 2013), it is a matter of urgency that a robust discussion around these disorders takes place. Owing to the variability in diagnostic and treatment pathways, a consensus working group may be a practical first step to create clinical pathways to guide clinicians in these complex cases.
See Review by Rege and Hodgkinson, 2013, 47(12): 1136–1151.
Footnotes
Acknowledgements
Written informed consent was obtained from the patient and the family for the publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Declaration of interest
Dr Sanil Rege is a founding trainer at Psych Scene, a company providing educational courses for psychiatry exams. Dr Suzanne Hodgkinson has no conflict of interest.
