Abstract
Objective:
Agomelatine is a new antidepressant with unique melatonin receptor type 1A (MTNR1A) and 1B ( MTNR1B) agonism and serotonergic receptor 5-hydroxytryptamine receptor 2C (5-HT-2C) antagonism. Several studies of patients with major depressive disorder (MDD) have confirmed the superior efficacy and safety of agomelatine in comparison with established treatments, such as selective serotonin reuptake inhibitors (SSRIs) or serotonin–norepinephrine reuptake inhibitors (SNRIs). This meta-analysis comprehensively shows the efficacy, acceptability, and safety of agomelatine in comparison with SSRIs and SNRIs used as antidepressants in MDD.
Method:
Comprehensive electronic database searches were performed to identify reports of head-to-head randomized controlled trials that have compared agomelatine with SSRIs or SNRIs in terms of efficacy/effectiveness in treating MDD. Response and remission rates at both acute (6–12 weeks) and follow-up (24 weeks) phases, Clinical Global Impression-Improvement Scale response and remission rates, changes in depression scale scores, improvements in subjective sleep, dropout rates, and side effect rates were extracted and analysed.
Results:
The meta-analysis included six head-to-head trials involving 1871 patients. In the acute phase, agomelatine had higher response rates (relative risk (RR) 1.08, 95% confidence interval (CI) 1.02–1.15) compared to SSRIs and SNRIs. In the remission analysis, only acute remission rates (RR 1.12, 95% CI 1.01–1.24) significantly differed. The action of agomelatine was superior on the Leeds Sleep Evaluation Questionnaire-Quality of Sleep score (mean difference 4.05, 95% CI 0.61–7.49). Discontinuation due to inefficacy did not differ between agomelatine and SSRIs/SNRIs (RR 0.74, 95% CI 0.42–1.28). Compared to SSRIs and SNRIs, however, agomelatine revealed a lower rate of discontinuation due to side effects (RR 0.38, 95% CI 0.25–0.57).
Conclusions:
Agomelatine has significantly higher efficacy and potential acceptability compared to SSRIs and SNRIs when treating MDD. However, the difference in efficacy is not considered clinically relevant. Because of its unique chronobiotic effects, agomelatine may be useful for the management of some MDD patients with circadian disturbance.
Introduction
The global lifetime prevalence of major depressive disorder (MDD) is an estimated 6.7% (Waraich et al., 2004). According to a World Health Organization (WHO) report, MDD is also the third leading contributor to the global disease burden (Mathers et al., 2008). Therefore, MDD presents a major challenge for health-care systems worldwide. Selective serotonin reuptake inhibitors (SSRIs) and serotonin–norepinephrine reuptake inhibitors (SNRIs) are the first-line therapy for treating MDD because of their superior tolerability and safety profiles relative to monoamine oxidase inhibitors and tricyclic antidepressants (Nutt et al., 2010). Therapeutic application of these inhibitors is based on the monoamine hypothesis of depression. Unfortunately, over half of MDD patients do not achieve remission after a 12-week treatment with SSRIs. Studies have also shown that one-third of patients treated with successive antidepressant therapies do not achieve remission even after a 1-year intervention (Rush et al., 2006). Therefore, novel antidepressants that target different pathophysiological aspects of depression have attracted strong interest.
A common feature of MDD is the disturbance of circadian rhythm and sleep homeostasis, such as insomnia/hypersomnia, non-restful sleep, diurnal variation in mood, decreased slow-wave sleep, and reduced latency to the first rapid eye-movement sleep cycle. Proposed approaches to resynchronizing the circadian system include light therapy, restructuring of sleep/wake timing, and melatonin treatment. Patients with a history of depression reportedly show altered expression of circadian genes (Gouin et al., 2010). In antidepressant therapy, restoration of normal chronobiology is an important marker of effectiveness, whereas failure to restore a normal rhythm is highly predictive of advancing symptoms or early relapses (Hickie and Rogers, 2011). Since monoamine-based antidepressants do not directly affect depression-associated circadian disorders, treatments targeted at resynchronizing the circadian system may be effective for treating depression.
Agomelatine is a new antidepressant with a unique mechanism of action that has been approved for clinical use in Europe and Australia, but not in the United States. Several double-blind, randomized, placebo-controlled trials have suggested its efficacy and safety for treating MDD (Kennedy and Emsley, 2006; Lôo et al., 2002; Montgomery and Kasper, 2007). Agomelatine has potent melatonin receptor type 1A (MTNR1A) and 1B (MTNR1B) agonism and serotonergic receptor 5-hydroxytryptamine receptor 2C (5-HT-2C) antagonism. The agonistic effects of agomelatine on the MTNR1A and MTNR1B receptors located in the hypothalamic nucleus may contribute to resynchronization of the circadian system. The antagonistic properties of the 5-HT-2C receptor produce concomitant dopamine and norepinephrine/noradrenaline overflows in the frontal cortex. Melatonin and monoamine reportedly exhibit a particularly robust synergistic efficacy in MDD (Kasper and Hamon, 2009).
Although earlier studies have evaluated the efficacy of agomelatine for treating MDD, these reviews have not provided meta-analytic summaries (Demyttenaere, 2011; Hickie and Rogers, 2011; Kennedy and Rizvi, 2010), or have focused on comparing agomelatine and placebo (Montgomery and Kasper, 2007). Singh et al. (2011) performed a meta-analysis of five placebo-controlled trials and five active comparator trials, but did not examine the response rate (the most common measure of antidepressant efficacy) or remission rate (the clinical state characterized by minimal residual symptoms, which is the main treatment goal). Kasper et al. (2013) also performed a pooled analysis of six active comparator trials but excluded studies that did not use the Hamilton Rating Scale for Depression (HRSD). Additionally, both of these meta-analyses were limited to short-term (6–12 weeks) trials. Therefore, the purpose of our study was to conduct a meta-analysis for a comprehensive depiction of the efficacy, acceptability, and safety of agomelatine for treating MDD compared to other antidepressants, such as SSRIs and SNRIs.
Methods
Database searches
The database search for this study included PubMed (from 1966 to 23 July 2013), CINAHL (from 2007 to 23 July 2013), PsycINFO (from 1998 to 23 July 2013), EMBASE (from 1974 to 23 July 2013), and the Cochrane Central Register of Controlled Trials (CENTRAL; 2013, Issue 5). The trial databases of the following drug approval agencies were also searched: the U. S. Food and Drug Administration, the Medicines and Healthcare Products Regulatory Agency (UK), the European Medicines Agency (EU), and the Therapeutic Goods Administration (Australia). Reports of trials were also sourced from the WHO International Clinical Trials Registry Platform, the ClinicalTrials.gov website, and reports of conference proceedings. The references provided in the selected studies and systematic reviews were further checked for additional citations of published or unpublished reports. The Trip database and Google Scholar were also searched. The keywords used in the search were agomelatine, major depression, and MDD. The search was limited to clinical trials involving human subjects. No language restrictions were applied.
Double-blind, randomized controlled trials (RCTs) were included if they had performed a head-to-head comparison of agomelatine with SSRIs (fluoxetine, fluvoxamine, citalopram, escitalopram, paroxetine, sertraline) or SNRIs (venlafaxine, duloxetine, milnacipran) in terms of efficacy/effectiveness for treating MDD. The presence of placebo was suspected to affect the outcome of antidepressant clinical trials. The influence of placebo on antidepressant response had been observed between placebo-controlled trials and active comparator trials (Rutherford et al., 2009); thus, placebo-controlled trials in which a comparator antidepressant was used to check assay sensitivity were excluded (Khan et al., 2003). The inclusion criteria were age 18 years or older and a primary diagnosis of MDD according to the Diagnostic and Statistical Manual of Mental Disorders, fourth edition or according to the International Statistical Classification of Diseases and Related Health Problems, 10th revision.
Two of the authors (KLH and WCL) independently selected the trials based on the previously described criteria, and disagreements were resolved by a third author (YYW). The included studies were then assessed for quality using the Cochrane risk of bias tool. The assessed risk of bias factors included sequence generation, allocation concealment, blinding of the participants and personnel, blinding of the outcome assessors, incomplete outcome data, selective outcome reporting, and other potential sources of bias (e.g. inconsistent distribution of baseline characteristics and industry initiation or funding of the study).
Data extraction
Two of the authors (KLH and WCL) extracted the data independently, with any disagreements resolved through discussion and consensus with other team members (YYW and GCH). For each trial, data related to the characteristics of the trial and the reported results were extracted. The primary efficacy outcome measures were the number of patients with a treatment response that included a reduction of at least 50% in their HRSD score or Montgomery–Åsberg Depression Rating Scale (MADRS) score and the number who had a much improved or a very much improved score (1 or 2, respectively) on the Clinical Global Impression-Improvement Scale (CGI-I). The secondary efficacy outcome measure was the number of patients who had achieved remission according to their HRSD, MADRS, or CGI-I scores (scores ≤ 7, ≤12, and 1, respectively), Leeds Sleep Evaluation Questionnaire (LSEQ) subjective sleep score changes, the difference between post- and pre-trial symptom mean scores (mean differences (MDs) for the scores from the same scale, standardized mean differences (SMDs) for different scales), and their pooled standard deviation for agomelatine and SSRIs/SNRIs. Outcome data were extracted for both acute (6–12 weeks) and follow-up (24 weeks) phases. Acceptability outcomes included total dropout rate, dropout rate due to inefficacy, and dropout rate due to side effects. The tolerability outcome measures were the percentage of patients who had experienced at least one side effect and the percentage of patients who had experienced at least one treatment-related side effect.
Statistical analysis
RevMan version 5.2 (Cochrane Collaboration, Oxford, United Kingdom) was used for the data analysis. For continuous data (mean score changes from baseline), MDs and SMDs were calculated at 95% confidence intervals (CIs). The SMDs were used for pooled scores for the HRSD and MADRS scales; MDs were used for pooled scores for the same scale. For dichotomous data (response, remission, and dropout rates; percentages of side effects), the relative risk (RR) was calculated at the 95% CIs. Pooling was performed in both fixed-effect and random-effect models. Heterogeneity was assessed with the I2 index and Cochran’s Q (X2) statistics. We considered an I2 value greater than 50% and p < 0.05 as indicative of heterogeneity. If the I2 index showed significant heterogeneity between the study results, a random-effect model was used. The variables included in the sensitivity analyses were dosing (flexible dosing only) and primary depression efficacy scale score (HRSD only).
Results
Of the 213 potentially relevant articles identified in the literature search, 185 were excluded by the consensus of two authors. In cases of discordant exclusions by the two authors, a third author made the final selection (CCH). Thus, 29 full-text manuscripts were retrieved for detailed evaluation. Of these, 23 were excluded for the following reasons: five papers were excluded because they were review articles (Carney and Shelton, 2011; Di Giannantonio and Martinotti, 2012; Hickie and Rogers, 2011; Howland, 2011; McAllister-Williams et al., 2010); eight papers were excluded because they were not head-to-head trials (European Medicines Agency, 2008 (CL3-022, CL3-023, and CL3-024 trials); Kennedy and Emsley, 2006; Lôo et al., 2002; Novartis, 2009; Stahl et al., 2010; Zajecka et al., 2010); three papers were excluded because they were discontinuation studies (Goodwin et al., 2009; Montgomery et al., 2004; Novartis, 2010); four papers were excluded because they were open-label studies (Di Giannantonio et al., 2011; Langan et al., 2011; Laux, 2012; Quera Salva et al., 2007); two papers were excluded because they did not provide data for meta-analysis: Martinotti et al., 2012 (open-label study) and Stein et al., 2013 (anxiety symptoms in major depression); one paper was excluded because there were no relevant outcomes (Karaiskos et al., 2013; open-label study, comorbid diabetes mellitus). Finally, six studies (1871 patients) were retained for full data extraction for the meta-analysis. Figure 1 summarizes the process used to select the relevant full-text articles and Table 1 summarizes the characteristics of the trials included in the meta-analysis.
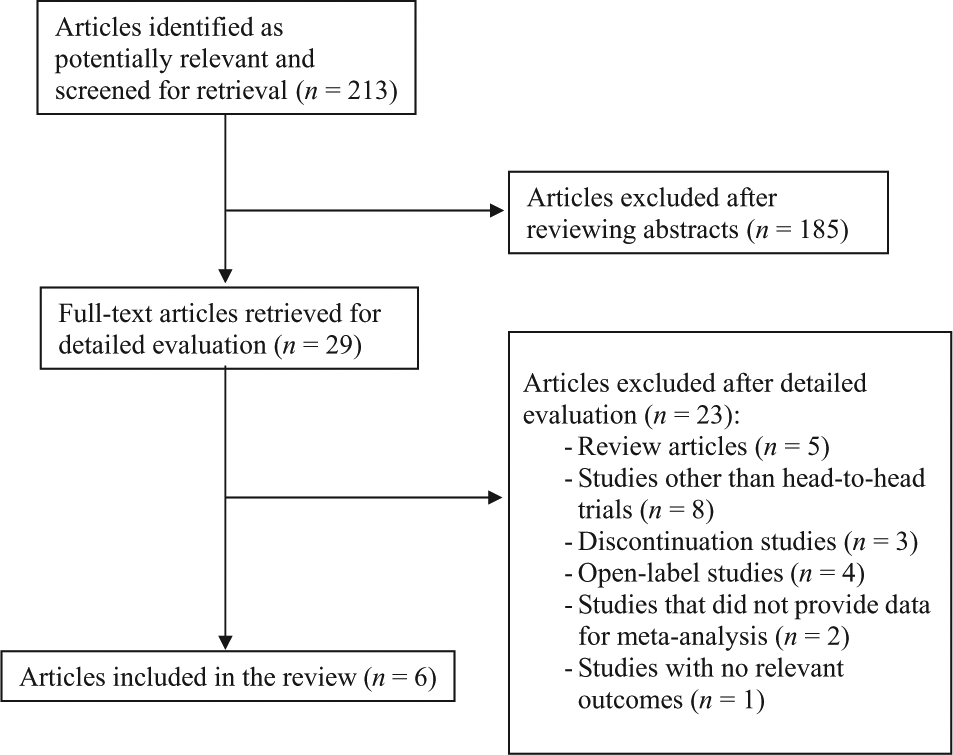
Process used to select the relevant full-text articles, which were then included in the meta-analysis.
Characteristics of the studies included in the meta-analysis (n = 6).
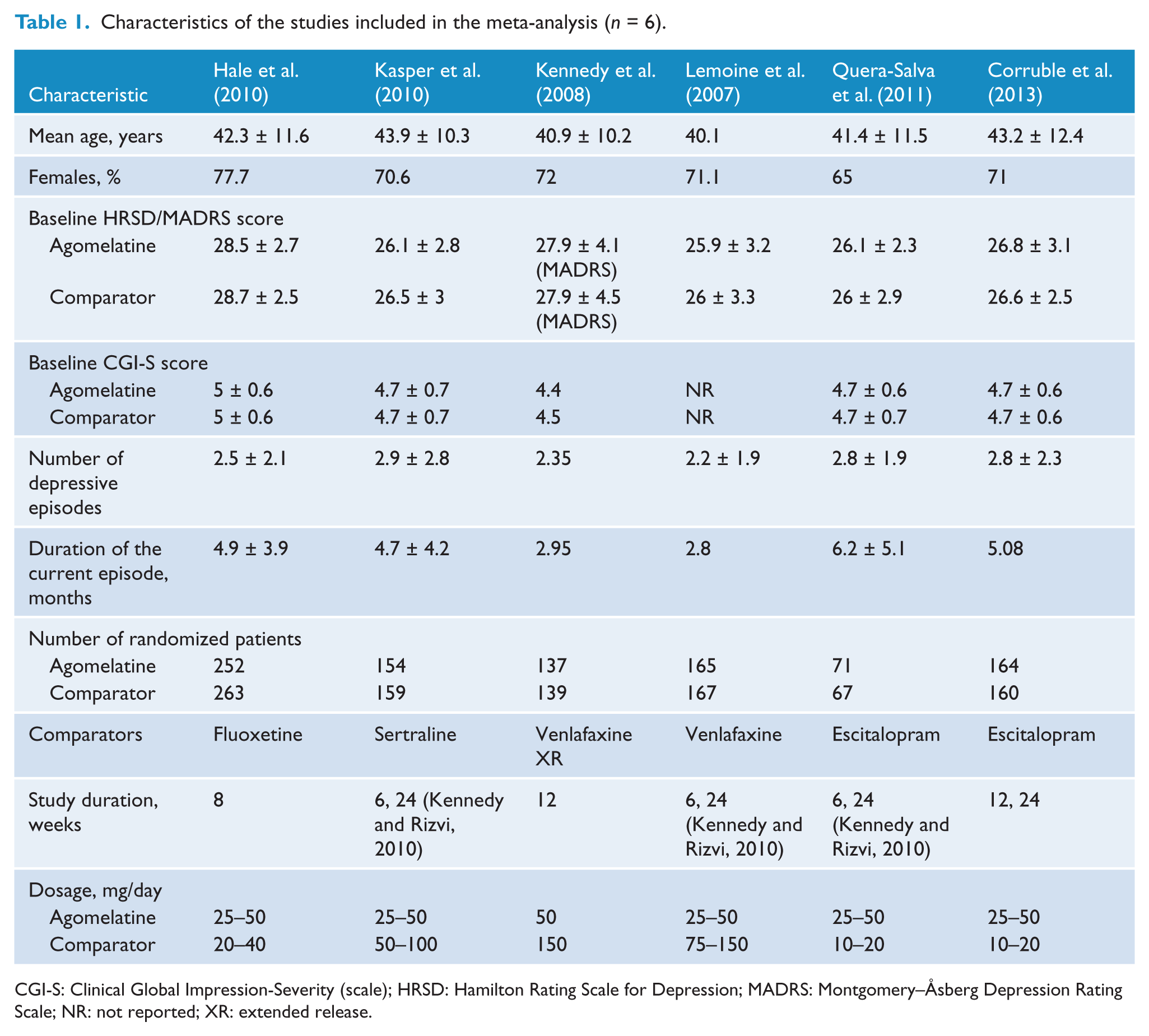
CGI-S: Clinical Global Impression-Severity (scale); HRSD: Hamilton Rating Scale for Depression; MADRS: Montgomery–Åsberg Depression Rating Scale; NR: not reported; XR: extended release.
Quality of the studies included
Figure 2 compares the overall risk of bias in each study. All included studies were RCTs. One trial (Kennedy et al., 2008) did not report adequate information about sequence generation and blinding of outcome assessors. Three trials (Hale et al., 2010; Kasper et al., 2010; Kennedy et al., 2008) did not report the allocation concealment in sufficient detail. All RCTs used a double-blind design. All included trials had dropout rates lower than 20% and all reported the study protocol. All included studies were funded by the drug industry. Overall, the included studies had good quality methodology.
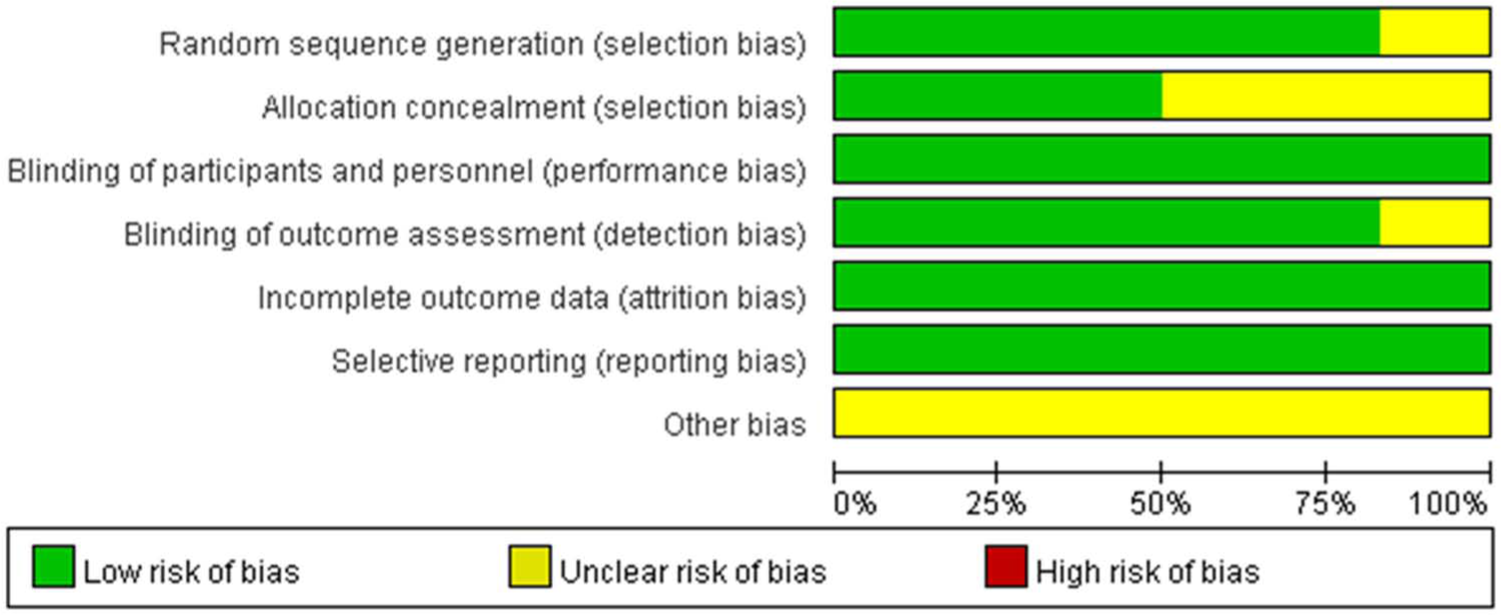
Overall risk of bias in the individual studies (n = 6).
Antidepressant efficacy
The analysis revealed that, in the acute phase, agomelatine had a higher response rate compared to SSRIs and SNRIs (RR 1.08, 95% CI 1.02–1.15) with no heterogeneity observed (X2 = 2.06, p = 0.84, I2= 0%; see Figure 3). However, in the follow-up phase, the response rate for agomelatine was not significantly higher than that recorded for SSRIs (RR 1.08, 95% CI 0.96–1.21). The CGI-I response analyses showed that agomelatine had a better response rate in the acute phase response (RR 1.12, 95% CI 1.05–1.19), but the response rate did not significantly differ during the follow-up (RR 1.06, 95% CI 1.00–1.12). The remission analysis showed that agomelatine had a significantly better remission rate in the acute phase (RR 1.12, 95% CI 1.01–1.24; see Figure 4). The RR for the remission rate in the follow-up phase was 1.10 (95% CI 0.96–1.25). Additionally, the CGI-I remission rates (in the acute and follow-up phases) did not significantly differ between agomelatine and SSRIs/SNRIs (RR 1.08, 95% CI 0.94–1.23 for acute phase; RR 1.08, 95% CI 0.99–1.17 for the follow-up phase). The total HRSD scores in the acute phase differed significantly: agomelatine obtained a significantly larger reduction compared to SSRIs and SNRIs (MD -1.05, 95% CI from -1.87 to -0.23). The action of agomelatine was superior to that of SSRIs and SNRIs on the LSEQ (quality of sleep) measure at week 6 (MD 4.05, 95% CI 0.61–7.49).
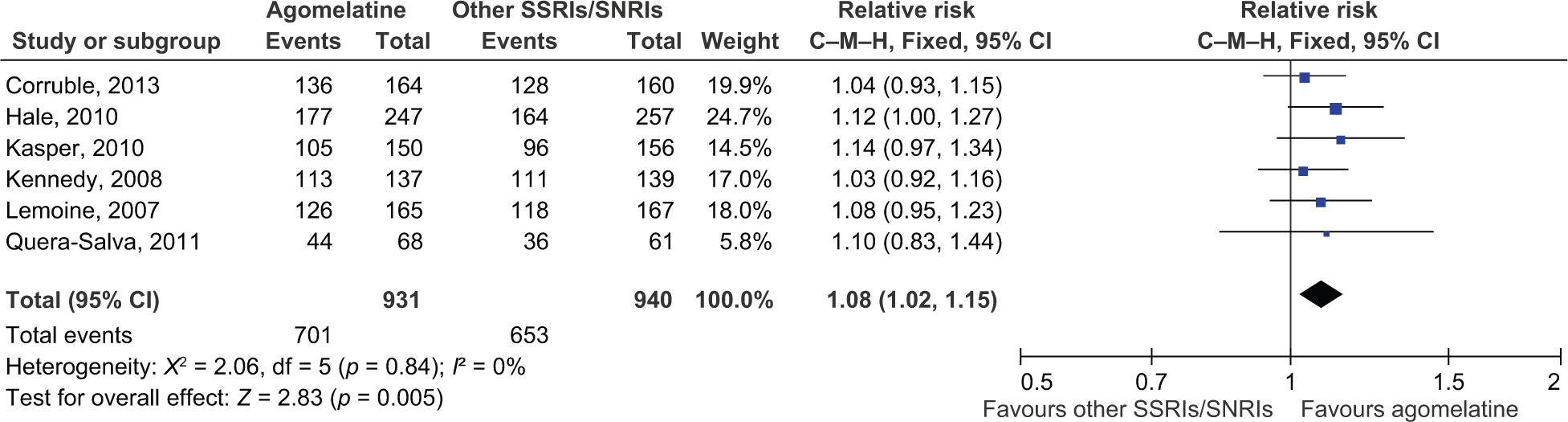
Acute phase response. Agomelatine was shown to have a higher response rate compared to SSRIs and SNRIs. CI: confidence interval; C–M–H: Cochran–Mantel–Haenszel; SNRI: serotonin–norepinephrine reuptake inhibitor; SSRI: selective serotonin reuptake inhibitor.
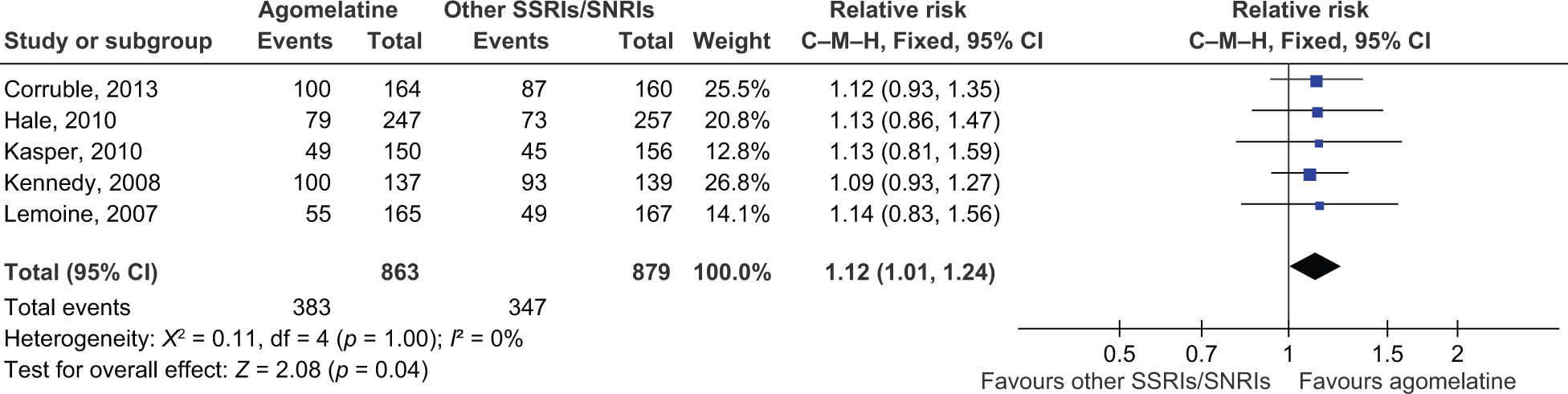
Acute phase remission. The remission analysis showed that agomelatine had a significantly better remission rate in the acute phase. CI: confidence interval; C–M–H: Cochran–Mantel–Haenszel; SNRI: serotonin–norepinephrine reuptake inhibitor; SSRI: selective serotonin reuptake inhibitor.
Acceptability
Agomelatine was associated with a lower total dropout rate compared with SSRIs and SNRIs (RR 0.64, 95% CI 0.50–0.82). Discontinuation due to inefficacy did not differ significantly between agomelatine and SSRIs/SNRIs (RR 0.74, 95% CI 0.42–1.28). However, there was evidence showing that the discontinuation rate due to side effects was lower in agomelatine than in SSRIs and SNRIs (RR 0.38, 95% CI 0.25–0.57; see Figure 5). None of the comparisons showed statistically significant heterogeneities.
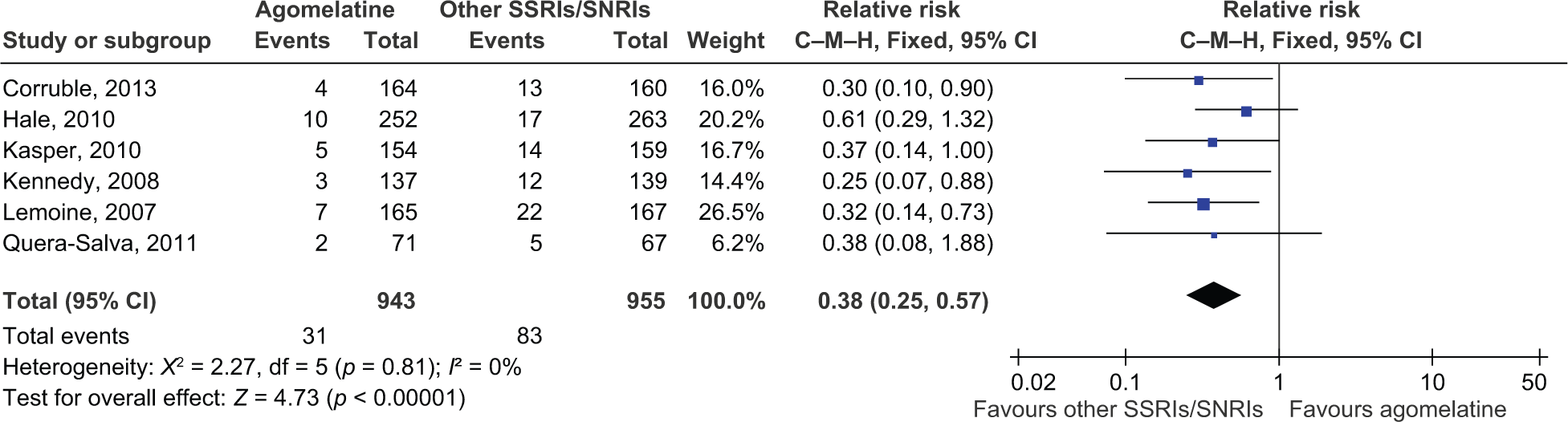
Dropout rate due to side effects. Evidence showed that the discontinuation rate due to side effects was lower when using agomelatine than when using SSRIs and SNRIs. CI: confidence interval; C–M–H: Cochran–Mantel–Haenszel; SNRI: serotonin–norepinephrine reuptake inhibitor; SSRI: selective serotonin reuptake inhibitor.
Tolerability
Agomelatine and SSRIs/SNRIs did not significantly differ in terms of the percentage of patients who had experienced at least one side effect (RR 0.93, 95% CI 0.86–1.01). However, the percentage of patients who had experienced at least one treatment-related side effect differed significantly between agomelatine and SSRIs/SNRIs (RR 0.68, 95% CI 0.50–0.92). This comparison revealed high heterogeneity (X2 = 11.86, p = 0.008, I2 = 75%).
Sensitivity analyses
After excluding trials that applied a fixed dosing schedule and those that did not use the HRSD, the sensitivity analysis included five trials. After excluding the trial by Kennedy et al. (2008), the RR was 1.09 (95% CI 1.03–1.16) for the response rate in the acute phase. The analysis revealed a significantly higher response rate for agomelatine. Further repetition of the meta-analysis obtained similar conclusions.
Discussion
The aim of this meta-analysis was to assess the clinical efficacy, acceptability, and safety of agomelatine compared with other SSRIs and SNRIs in patients with MDD. This review included six RCTs comprising 1871 participants. All six studies had a similar design. The antidepressants included in the analysis were three SSRIs (fluoxetine, sertraline, escitalopram) and one SNRI (venlafaxine). The pooled data indicated that, compared to the other antidepressants, agomelatine achieved a marginally superior response rate and reduction in HRSD score when used to treat MDD in the acute phase. Remission rates in the acute phase were also significantly higher in patients treated with agomelatine compared to those treated with SSRIs and SNRIs. The CGI-I analyses showed that agomelatine obtained a significantly larger CGI-I response in the acute phase but not in the follow-up phase. However, the CGI-I remission rates did not differ between agomelatine and SSRIs/SNRIs in either the acute or follow-up phases. The pooled analysis also revealed a significantly lower dropout rate due to side effects in the agomelatine group compared to the SSRI and SNRI groups. Finally, the rate of treatment-related adverse events was significantly lower in the agomelatine group than in the SSRIs and SNRIs groups.
Despite the use of different effect measures in this study (RRs vs. between-group difference), the findings of this study were consistent with the literature (Demyttenaere et al., 2013; Kasper et al., 2013; Singh et al., 2011). The pooled response rates in the acute phase were 75.3% for agomelatine and 69.5% for other SSRIs and SNRIs (between-group difference (standard error (SE)) = 5.83 (2.06), 95% CI 1.78–9.87, p = 0.005). In an earlier study by Kasper et al. (2013), the pooled response rate in the acute phase was 71% for agomelatine and 66% for the other SSRIs and SNRIs (between-group difference (SE) = 5.08 (2.02), 95% CI 1.11–9.05, p = 0.012). However, their review excluded studies in which primary efficacy scales were not evaluated by the HRSD, whereas we included studies that used the HRSD and MADRS scales to measure the change in the severity of depression. Overall, the estimated difference between agomelatine and other SSRIs in terms of response rate in the follow-up phase was 6.78 (3.28, p = 0.039). In another review by Demyttenaere et al. (2013), the overall estimated difference was 5.09 (2.59, p = 0.031). Their review identified one additional unpublished trial, which enhanced the precision of the results. We note that the relatively small sample sizes at the follow-up phase may not provide sufficient statistical power to distinguish between the two groups. However, the SMD of our pooled estimate was -0.14 (95% CI from -0.25 to -0.03), which approximates that reported in an earlier study by Singh et al. (2011), who obtained an SMD of -0.11(95% CI from -0.21 to -0.02) in a comparison between agomelatine and other antidepressants.
In studies submitted for licensing approval, a difference of 10% or more between the treatments is generally considered sufficient to establish a clinical advantage (Montgomery and Möller, 2009). Although the data discussed previously showed that the effect of agomelatine statistically differed from that of SSRIs and SNRIs, the difference was not clinically significant.
Moreover, sleep-related symptoms were weighed more heavily in the HRSD (3-items) scale compared to the MADRS (1-item) scale, which may have caused a small improvement (by up to six points) in the depression score for the HRSD scale. Patients with sleep disturbance may then have a better response to agomelatine, which is a melatonergic compound.
Agomelatine has been shown to have better acceptability than SSRIs and SNRIs in MDD patients. There were significantly fewer dropouts owing to adverse events from agomelatine (RR 0.38, 95% CI 0.25–0.57). In a pooled analysis of head-to-head studies (Kasper et al., 2013), significantly fewer patients withdrew because of side effects with agomelatine (6.9%) than with SSRIs and SNRIs (10.7%, p < 0.001). In a pooled analysis of head-to-head trials lasting 24 weeks (Demyttenaere et al., 2013), the discontinuation rate due to adverse events was 6.6% for agomelatine and 9.4% for SSRIs (p = 0.065). The results of these studies indicated that agomelatine had an advantage in terms of acceptability. Agomelatine also demonstrated greater tolerability. The proportion of patients who had experienced at least one adverse event related to treatment was significantly lower with agomelatine compared with SSRIs and SNRIs (RR 0.68, 95% CI 0.50–0.92).
One limitation of this analysis is the generalization of individual drugs into drug classes. Effects and adverse reactions may differ among a single class of drugs. However, meta-analyses that have compared one SSRI against another for treating MDD have generally reported comparable efficacy (American Psychiatric Association (APA), 2010; Gartlehner et al., 2011), i.e. no compelling evidence shows that one SSRI is more efficacious than another. Moreover, the only SNRI included in these studies was venlafaxine. Previous meta-analyses (Demyttenaere et al., 2013; Kasper et al., 2013; Singh et al., 2011) have encountered the same problem because of the absence of comparative studies between agomelatine and SNRIs other than venlafaxine (e.g. duloxetine, milnacipran).
In addition, a recent meta-analysis of published and unpublished trials (Koesters et al., 2013) showed that treatment with agomelatine in the acute phase was associated with a statistically significant superiority over placebo of -1.51 HRSD points (99% CI from -2.29 to -0.73). All unpublished, short-term efficacy trials (European Medicines Agency, 2008; Novartis, 2009) did not find agomelatine to be better than placebo. Since it has been suggested that a difference of two HRSD points is clinically important, Koesters et al. (2013) concluded that the clinical relevance of agomelatine in the treatment of MDD in the acute phase may be questionable. However, it is also noteworthy that active comparators were used to evaluate assay sensitivity in all unpublished, short-term, placebo-controlled trials (European Medicines Agency, 2008 (CL3-022, CL3-023, and CL3-024 trials); Novartis, 2009). Assay sensitivity is a property of a clinical trial defined as the ability of a trial to distinguish an effective treatment from a less effective or ineffective intervention. Without assay sensitivity, a trial is not internally valid and is thus not capable of comparing the efficacy of two interventions (European Medicines Agency, 2001). However, active comparators were not more effective than placebo in the two unpublished trials (European Medicines Agency, 2008 (CL3-023 and CL3-024 trials)). They should be identified as failed trials due to the lack of assay sensitivity (European Medicines Agency, 2001; Kennedy and Rizvi, 2010). Study failure may be due to a large placebo effect (European Medicines Agency, 2008). In fact, only two unpublished trials (European Medicines Agency, 2008 (CL3-022 trial); Novartis, 2009) in the study by Koesters et al. (2013) showed a negative result of agomelatine in the treatment of MDD in the acute phase. Furthermore, Kennedy and Rizvi (2010) found an agomelatine-placebo difference of 2.43 HRSD points from the meta-analysis of three published trials and one unpublished negative trial (European Medicines Agency, 2008 (CL3-022 trial)). One should cautiously interpret the result of failed trials because only positive or negative trials are useful in evaluating whether a new drug works.
Despite the thorough literature search in this study, the lack of data from unpublished studies likely resulted in publication bias. Although efforts were made to maximize data retrieval, data from some trials were not included. Additionally, all the studies included had been sponsored by drug companies manufacturing agomelatine, which raises concerns of sponsorship bias. Further, the doses of active comparators were relatively lower in our study compared to the doses reported in the literature. The consensus guideline is a fluoxetine dose of 20–60 mg/day (APA, 2010). In Hale et al. (2010), patients were administered fluoxetine doses in the range of 20–40 mg/day. The same patterns were observed in sertraline and venlafaxine (typical dose: sertraline 50–200 mg/day, venlafaxine 75–375 mg/day; typical dose in trial: sertraline 50–100 mg/day, venlafaxine 75–150 mg/day). Sinyor et al. (2012) suggested that dosing differences may affect the efficacy results of antidepressants obtained by RTCs. Therefore, the potential inflation of the efficacy of agomelatine cannot be ruled out.
Conclusion
The results of this meta-analysis indicate that the clinical efficacy of agomelatine is significantly higher than that of SSRIs and SNRIs in terms of response and remission rates when used in the acute phase of MDD. Compared to other antidepressants, agomelatine also obtained a significantly better CGI-I response in the acute phase. However, the difference in efficacy was not considered clinically relevant. Agomelatine significantly improves the subjective quality of sleep after 6 weeks of treatment. In terms of dropout rate due to side effects, agomelatine performed better than SSRIs and SNRIs. Agomelatine did not differ from SSRIs and SNRIs in terms of percentages of patients with at least one treatment-emergent adverse event. Because of its unique chronobiotic effects and good tolerability, agomelatine may have important roles in the management of some MDD patients with circadian disturbance.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
