Abstract
Objective:
Diagnosing depression in primary care settings is challenging. Patients are more likely to present with somatic symptoms, and typically with mild depression. Use of assessment scales is variable. In this context, it is uncertain how general practitioners (GPs) determine the severity of depressive illness in clinical practice. The aim of the current paper was to identify which symptoms are used by GPs when diagnosing depression and when determining severity.
Method:
A total of 1760 GPs participated in the RADAR Program, an educational program focusing on the diagnosis and management of clinical depression. GPs identified a maximum of four patients whom they diagnosed with depression and answered questions regarding their diagnostic decision-making process for each patient.
Results:
Overall, assessment of depression severity was influenced more by somatic symptoms collectively than emotional symptoms. Suicidal thoughts, risk of self-harm, lack of enjoyment and difficulty with activities were amongst the strongest predictors of a diagnosis of severe depression.
Conclusions:
The conclusions are threefold: (1) collectively, somatic symptoms are the most important predictors of determining depression severity in primary care; (2) GPs may equate risk of self-harm with suicidal intent; (3) educational initiatives need to focus on key depressive subtypes derived from emotional, somatic and associated symptoms.
Introduction
Clinical depression is common in primary care settings. A recent meta-analysis suggested that the point-prevalence of major depression using the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV) criteria is 17% in primary care (Luber et al., 2000), and about the same number have minor depression and/or subsyndromal symptoms of depression (Williams et al., 1995). Therefore, general practitioners (GPs) are ideally placed to detect and treat depression (Armstrong and Earnshaw, 2004) and yet, under-detection and under-diagnosis of depression in primary care settings remains a significant problem (Henriques et al., 2009). GPs typically correctly identify about 50% of cases of major depression (Mitchell et al., 2009), and 33% of minor and subsyndromal depression cases (Mitchell et al., 2011). At the same time, GPs correctly identify 80% of those without symptoms of depression (Mitchell et al., 2009), leading to a small but appreciable false-positive rate.
Reasons for the difficulty in making an accurate diagnosis of depression are complex and multifactorial (Ani et al., 2008), and relate to the very nature of primary care, which necessarily involves time-limited consultations and diverse presentations that encompass a wide range of diffuse symptoms. Additionally, patients with depression usually have medical comorbidity and often focus on their medical condition or somatic symptoms, and avoid disclosure of emotional symptoms, either because they believe their feelings are a component of their medical illness, because of time pressure, or because of the potential stigma of a psychiatric diagnosis (Davidson and Meltzer-Brody, 1999). Understandably, this creates a barrier to prompt diagnosis, as patients presenting with somatic symptoms of depression are less likely to be recognized (Henriques et al., 2009). However, predictors of improved diagnostic accuracy include a better therapeutic relationship, clinician experience and time spent with the patient (Hickie et al., 2001; Mitchell et al., 2009; Nuyen et al., 2005).
Alongside the difficulties in identification, an additional complication is that depression may present differently in the context of physical disease (Meader et al., 2011; Mitchell et al., 2012, 2013). This could perhaps be why, in comparison to psychiatrists, GPs appear to use different symptoms or criteria to diagnose depression (Armstrong and Earnshaw, 2004; Lampe et al., 2012). For example, GPs may be less attuned to detecting psychological and somatic symptoms (Henriques et al., 2009), and are also less likely to elicit symptoms such as worthlessness and guilt in patients with a medical disease. This is partly because GPs can regard these symptoms as ‘normal’ psychological responses to medical illness, and, at the same time, they give priority to thoughts of suicide and sleep dysfunction in their diagnosis of depression. Specifically, suicidal ideation has been shown to increase the likelihood of diagnosing depression fivefold, and individuals with insomnia or hypersomnia are twice as likely to be diagnosed as having depression when compared to individuals without these symptoms (Ani et al., 2008). In one study, stressful life events and occupational, financial, and housing issues were the most influential prompts to a diagnosis (Saltini et al., 2004). Importantly, once a diagnosis of depression is made in primary care, little is known about how GPs determine severity of depression. Many studies highlight that GPs are unlikely to use mood severity questionnaires. For example, Tai-Seale et al. observed that GPs assessed depression in only 14% of visits and almost never used formal depression assessment tools (Tai-Seale et al., 2005). In a second study, most GPs in Germany felt the nine-item Patient Health Questionnaire (PHQ-9) was too time-consuming (Bermejo et al., 2005). Psychiatrists screen for depression severity and depressive subtypes, such as melancholic and psychotic depression, through their association with certain symptoms (Malhi et al., 2005), but GPs are less exposed to severe depression.
Hence, the aim of the current study was to examine the relative importance that GPs attach to specific individual symptoms, as well as groups of symptoms, when assigning a diagnosis of depression and determining its severity.
Method
In conjunction with a national steering committee comprising psychiatrists and GPs with expertise in mood disorders, MediMark International developed an education initiative called RADAR (Recognition, Awareness, Discussion And Review of depression), which was approved by the Northern Sydney Local Health District ethics committee (MediMark is an independent organization that develops educational programs for GPs). One objective embedded within the program was to evaluate the diagnosis and management of depression in primary care, which was conducted prior to the learning component. Each GP was asked to think about the most recent four patients in whom he or she made a diagnosis of depression, and to indicate the following in each case:
The severity of depression on a three-point rating scale (1 = ‘mild’, 2 = ‘moderate’, 3 = ‘severe’).
The extent to which a range of emotional, somatic and associated symptoms featured in making the original diagnosis of depression on a five-point rating scale (1 = ‘not at all’; 5 = ‘to a major extent’). Emotional symptoms included guilt, thoughts of suicide, lack of interest, sadness, and lack of capacity to enjoy pleasure (anhedonia); somatic symptoms included difficulty performing simple everyday activities, change in appetite, decreased concentration, lack of energy, and sleep disturbance or change in sleep patterns; and associated symptoms included tearfulness, excessive worry over physical health, pain, irritability, anxiety or phobias, brooding, and continuous reflection or contemplation.
Whether or not assessment was undertaken for risk of self-harm and risk to others) (yes/no).
The degree of risk of self-harm and risk to others on a five-point rating scale (1 = ‘minimal’; 5 = ‘very likely’).
The presence of any medical or psychiatric comorbidity in the diagnosis of depression (yes/no).
Data analysis
A 3 × 3 mixed design ANOVA was performed to examine differences between the three groups of symptom ratings across the three severity groups. Non-parametric tests (Kruskal–Wallis ANOVA and chi-square analyses) were performed to examine differences according to severity with respect to: (i) GP assessment of risk of self-harm and risk to others; and, (ii) association between depression severity and patients’ degree of risk of self-harm (and harm to others). Two binary logistic regression analyses were conducted to predict depression severity as diagnosed by GPs: severe (coded as 1) or mild. The first regression included all specific individual symptoms as predictors. The second (forward entry) regression controlled for the variables that were significant in the first regression, while adding degree of risk of self-harm and risk to others as predictors.
Results
A total of 1760 GPs participated, and complete data were available for 1026 GPs, each of whom identified a maximum of four patients with depression (n = 4117).
Across all patients, 12.6% were rated as mild, 57.3% as moderate, and 30.1% as severe. The majority (82%) had comorbid medical or psychiatric illnesses, with the most common being an anxiety disorder (59.5%). Substance misuse (33%) and chronic medical illness (23.7%) were the next most commonly diagnosed comorbid illnesses.
Ratings of emotional, somatic, and associated symptoms according to depression severity
Results of the 3 × 3 ANOVA showed that the Mauchly’s test of sphericity was significant, χ2(2) = 60.17, p < 0.05, thus the Greenhouse–Geisser correction was applied. There was a significant main effect for symptoms, F(1.97, 7046.59) = 466.85, p < 0.001, and depression severity, F(2, 3582) = 467.36, p < 0.001. There was also a significant interaction between the symptom and depression severity groups, F(3.93, 7046.59) = 11.43, p < 0.001, such that the discrepancy between somatic and other symptom scores (both emotional and associated) was greatest for those with severe depression (Figure 1).
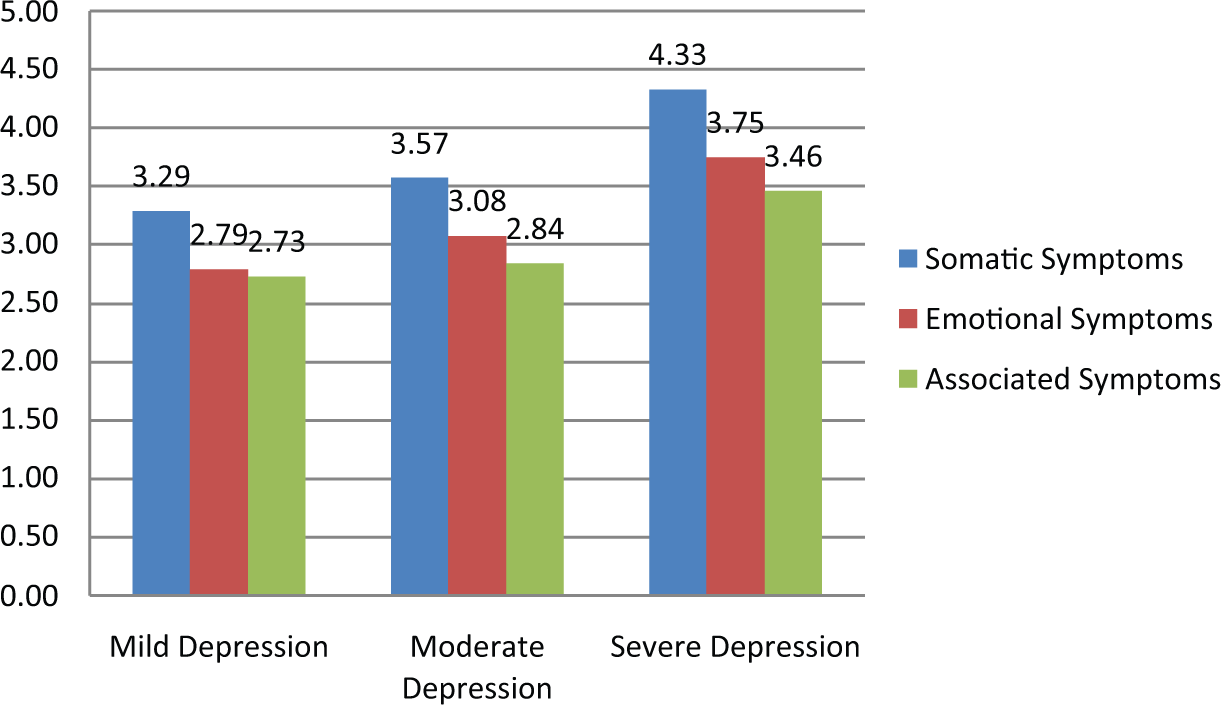
Depressive symptoms (categories averaged) according to depression severity.
Symptomatic predictors of depression severity
Results of the binary logistic regression predicting depression severity showed a significant model, χ2(17) = 767.77, p < 0.001, predicting with 85.8% accuracy. Significant individual predictors of severe depression are indicated in Table 1a. All variables were positively associated with increased odds of predicting severe depression.
(a) Symptomatic predictors of mild versus severe depression in the logistic regression analysis; (b) risk of self-harm and risk to others predicting mild versus severe depression.
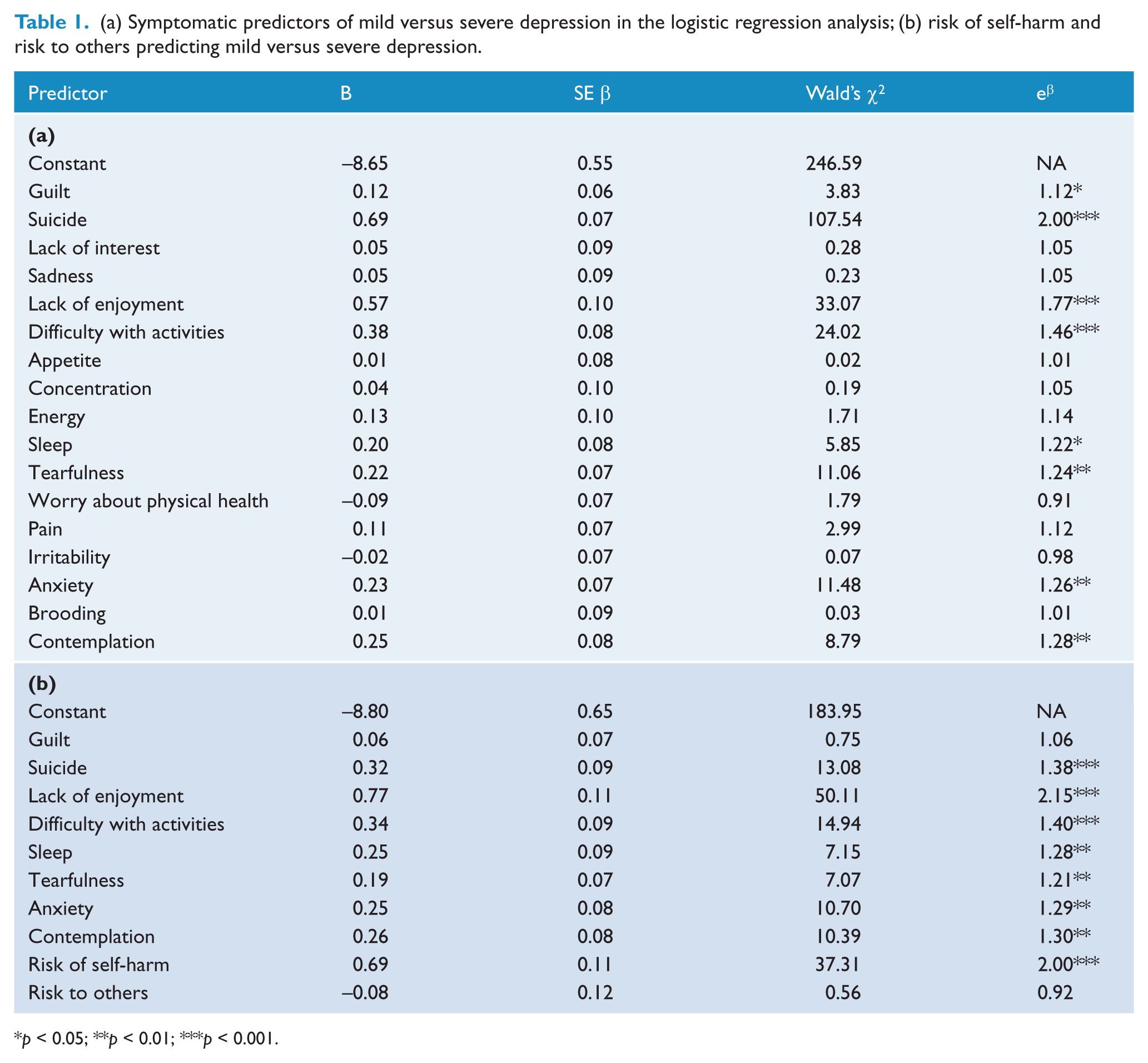
p < 0.05; **p < 0.01; ***p < 0.001.
The strongest symptom predictors of depression severity were suicide (suicidal thoughts), lack of enjoyment, difficulty with activities (functional limitation), contemplation, anxiety, tearfulness, sleep and guilt. These were all statistically significant. No other symptoms were influential in gauging depression severity.
Patients’ risk of harm (to self and others)
Just over half of the patients (59.4%) were assessed for risk of self-harm and 44.7% were assessed for risk to others. GPs were significantly more likely to assess a patient’s risk of self-harm with increasing severity of depression (mild 40.3%, moderate 54.9% and severe 75.5%; χ2(2) = 225.35, p < 0.001), as well as risk of harm to others (mild 34.1%, moderate 43.9% and severe 49.6%; χ2(2) = 35.18, p < 0.001). Similarly, patients’ potential risk of self-harm, χ2(2) = 554.54, p < 0.001, and risk to others, χ2(2) = 57.64, p < 0.001, significantly increased with depression severity.
To examine the predictive power of risk of harm upon depression severity, results of the logistic regression, controlling for the symptoms that were significant in the previous regression, yielded a significant outcome (χ2(2) = 45.00, p < 0.001; overall model, χ2(10) = 609.72, p < 0.001; 87% accuracy). All individual variables were significant predictors, except feelings of guilt and risk to others (Table 1b). According to the model, the log of the odds of a patient being diagnosed with severe depression was positively related to all variables.
Discussion
The RADAR Program provided the opportunity to obtain a real-world picture of GP diagnostic processes in clinical depression, offering new insights into the complexities of detecting and diagnosing major depressive disorder in primary care. The high rates of comorbid illness identified indicate that GPs fully appreciate that depression is predominantly a comorbid illness (Goodwin et al., 2003; Hasin et al., 2005; Katon, 2003).
In keeping with accepted wisdom, our findings show that GPs use a wide range of emotional, somatic, and associated symptomatology to identify depression but, within these domains, emotional symptoms are less likely to contribute to the diagnosis as compared to somatic symptoms. This difference in weighting (favouring somatic symptoms above emotional and associated symptoms) held across all levels of depression severity, but was most significant in severe depression. The importance attached to somatic complaints is understandable because primary care patients with depression typically present with these symptoms as opposed to emotional symptoms (Aragones et al., 2005). Therefore, it is important for GPs to be able to identify which somatic symptoms are indicative of depression or other mental health problems as opposed to those associated with a medical illness. In the context of physical diseases such as cancer and epilepsy, somatic complaints may have special significance. For example, they may be present frequently in non-depressed patients causing possible false-positive diagnoses. Recent work has actually suggested only subtle differences in depression with physical disease, with possible contamination from the symptoms of agitation, retardation or weight change. In these conditions, almost all somatic symptoms retained their importance in terms of diagnostic significance (Mitchell et al., 2012, 2013).
It remains unclear to what extent somatic complaints actually interfere with the diagnosis of depression in primary care. Although a presentation with somatic symptoms of depression reduces prompt detection of depression (Henriques et al., 2009), the presence of physical comorbidity may or may not have the same effect. For example, Ani and colleagues (2008) found comorbidity had no effect of recognition accuracy. Borowsky et al. (2000) found superior detection of depression if comorbid diabetes or hypertension were present, and O’Conner et al. (2001) found that comorbid pain positively influenced detection of late-life depression.
Although somatic symptoms, as a group, appear to be important for diagnosing depression and assigning severity, individual emotional symptoms of suicidal thoughts, anhedonia and risk of self-harm served as strong predictors of a rating of severe depression, which is in keeping with a more severe melancholic subtype of depression (Taylor and Fink, 2006). However, only just over half of patients were assessed for risk of self-harm, with the number of risk assessments increasing as depression severity increased. Therefore, it is unknown whether risk assessment informs decisions around severity, or whether making an assessment of severe depression prompts a risk assessment. Another important issue concerns the possibility that GPs equate risk of self-harm with suicidal intent, given that both variables were very strong indicators of depression severity and were highly inter-correlated. Previous studies have used Rasch analysis to determine which symptoms relate to overall severity. For example, Mitchell et al. (2011) found that suicidal ideation and guilt (‘I thought my life had been a failure’) strongly related to severe depression.
In sum, these findings suggest assessment of depression severity in primary care was influenced more by somatic symptoms than emotional symptoms. At the symptom level, suicidal thoughts, risk of self-harm, lack of enjoyment and difficulty with activities were amongst the strongest predictors of a diagnosis of severe depression. It is likely that a rating of severe depression in primary care is not equivalent to a specialist’s diagnosis of severe depression. GPs appear to be most influenced by the number, and perhaps severity, of somatic symptoms. Hence, it is possible that GPs are diagnosing severe depression using a narrower symptom base, and as such, depression that is rated ‘severe’ in primary care is likely to be somewhat different to severe depression in specialist psychiatric practice.
Limitations
It is presumed that melancholic and psychotic depression in primary care is rare. Since the current study did not employ a structured diagnostic assessment for all patients attending a GP’s practice in a given time frame, what cannot be answered by the present study was whether some patients with melancholic depression may have been missed. Whilst this is possible, research suggests that GPs follow their patients over time and will pick up on those whose symptoms, in contrast to the usual pattern, do not appear to improve (Lampe et al., 2012).
GPs within the RADAR Program were self-selecting, and thus potentially more likely to have an interest in mental health and/or more likely to diagnose a patient compared to those who did not participate. Therefore, the data may not be representative of GPs who chose not to participate in the program.
Conclusion
This large and innovative study has identified key gaps in knowledge concerning the detection and diagnosis of depression in primary care. These findings are important and should be targeted by future educational initiatives. In particular, GPs need to be better informed about subtyping depression and the associated treatment implications, as well as differentiating self-harm and suicidal intent so as to guide interventions more effectively. Given that GPs will remain the gate-keepers to the effective management of depression, they need to be provided with the appropriate keys so as to effectively detect the illness and direct its treatment.
Footnotes
Funding
The RADAR Program was supported by MediMark International through an unconditional education grant from Eli Lilly Australia.
Declaration of interest
None of the expert steering committee had any contact at any time with the sponsor of the Program. Further, at no time in the development or delivery of the Program was the sponsor advertised or promoted to the committee or general practitioners.
In the past 3 years, Professor Gin Malhi has served on a number of international and national pharmaceutical advisory boards, received funding for research and has been in receipt of honoraria for talks at sponsored meetings worldwide involving the following companies: AstraZeneca, Lundbeck, Sanofi-Aventis, Eli Lilly, Janssen-Cilag, Organon, Pfizer and Wyeth. Dr Lisa Lampe has received honoraria for lectures, consultancy, workshops, and educational material in the past 3 years from the following companies: Lundbeck, Pfizer, Sanofi, Servier, Janssen-Cilag and AstraZeneca. She has received travel assistance from Wyeth. Dr Bill Lyndon has been a member of the advisory boards of Lundbeck Australia, AstraZeneca Australia and Eli Lilly, and has participated in developing educational material for Eli Lilly and AstraZeneca Australia. Professor Malcolm Hopwood has received support from the following sources over the last 3 years: speaker’s fees/honoraria – AstraZeneca, Boehringer, Bristol-Myers-Squibb, Eli Lilly, Janssen-Cilag, Lundbeck, Pfizer, Schering Plough, Servier, Wyeth; advisory board – Boehringer, Eli Lilly, Lundbeck, MediMark International, Lifeblood, MD Briefcase, Schering Plough, Summer Foundation, Wintringham; travel support – AstraZeneca, Boehringer, Eli Lilly, Janssen-Cilag, Lundbeck, Pfizer, Sanofi-Aventis, Servier; and clinical trials/research support – AstraZeneca, AHMRF, Bristol-Myers-Squibb, Cephalon, Eli Lilly, Forrest, Lundbeck, GSK, Sanofi-Aventis, Servier, Sunovion, VNI, Weary Dunlop Foundation.
Dr Carissa Coulston, Dr Kristina Fritz, Danielle Bargh, Dr Michael Ablett, Dr Rick Sapsford, Dr Michael Theodoros, Derek Woolfall, Dr Andrea van der Zypp, and Dr Alex Mitchell have no competing interests to report.
