Abstract
Introduction
Thalassaemias are inherited disorders caused by reduced production of structurally normal haemoglobin chains. The most common forms are α- and β-thalassaemia, which result from pathogenic variants in the α- and β-globin genes, respectively. In healthy adults, haemoglobin A (HbA) is the predominant form, comprising 96–98% of total haemoglobin and consisting of two alpha and two beta chains (α2β2). Minor fractions include haemoglobin A2 (HbA2), which accounts for 2–3% of total haemoglobin and consists of two alpha and two delta chains (α2δ2), and fetal haemoglobin (HbF), which makes up less than 1% of total haemoglobin and is composed of two alpha and two gamma chains (α2γ2).
The diagnostic evaluation of thalassaemias involves integration of erythrocyte indices, phenotypic characterization of haemoglobin fractions and genetic testing. High-performance liquid chromatography (HPLC) and capillary electrophoresis are the principal methods used to quantify haemoglobin fractions. In clinical practice, HbA2 quantification is especially important for identifying β-thalassaemia trait. Elevated HbA2 levels above 3.5% are considered a hallmark of β-thalassaemia trait, reflecting decreased β-globin chain production and compensatory incorporation of δ-globin chains. However, HbA2 values are not interpreted in isolation. Red cell indices, particularly mean corpuscular haemoglobin (MCH) and mean corpuscular volume (MCV), are essential for diagnostic interpretation. In β-thalassaemia trait, elevated HbA2 is typically accompanied by low MCH (<1.70 fl) and microcytosis (MCV <80 fl), despite a normal or mildly decreased haemoglobin level. In contrast, although α-thalassaemia trait usually presents with similar hypochromic and microcytic red cell indices, HbA2 levels are typically normal or mildly decreased. Thus, interpreting HbA2 levels in conjunction with red cell indices is essential for the accurate diagnosis of thalassaemias.
Iron deficiency anaemia is an important confounding factor in the laboratory assessment of thalassaemias. Iron deficiency reduces the expression of HbA2, which may cause diagnostic confusion by mimicking an α-thalassaemia phenotype. 1 In addition, iron deficiency can compromise the diagnosis of β-thalassaemia trait by masking its characteristic HbA2 increase. 2 Thus, insight into the factors that modulate HbA2 levels is critical for correct interpretation of laboratory results during the diagnostic workup in cases where thalassaemia is suspected.
Haemoglobin A1c (HbA1c) is a glycated form of haemoglobin A that reflects the average plasma glucose concentration over the life span of erythrocytes. HbA1c levels are greatly enhanced in patients with chronic hyperglycaemia and represent the gold standard for the evaluation of glycaemic control in patients with diabetes.
Despite the widespread use of HbA2 in thalassaemia diagnostics, limited attention has been paid to the potential impact of hyperglycaemia on HbA2 measurements. The rationale for this study arose from clinical observations that patients with markedly elevated HbA1c values often showed unexpectedly low HbA2 fractions, complicating thalassaemia interpretation. This prompted the hypothesis that high HbA1c levels may interfere with the accurate quantification of HbA2, thereby acting as a confounding factor in thalassaemia diagnosis. The aim of this study was to investigate whether chronic hyperglycaemia can affect the diagnostic reliability of HbA2 in the evaluation of thalassaemia.
Materials and methods
Data collection
The patient cohort was selected retrospectively from individuals over 1 years of age that underwent haemoglobin analysis by HPLC at the Amsterdam UMC. Data were collected between 2021 and 2023, unless state otherwise. In our laboratory, haemoglobin phenotyping with HPLC is performed in patients who present with markedly elevated HbA1c levels (>110 mmol/mol), as initially detected on routine HbA1c analysis using the Tosoh G11 analyser. In these cases, HPLC is used to rule out the presence of haemoglobin variants that co-elute with HbA1c and thereby lead to falsely elevated results. 3 This approach enabled us to investigate the potential impact of elevated HbA1c levels on the measurement of the HbA2 fractions by HPLC.
The following patient groups were selected. First, a control group (n = 80) was selected by including all patients over a period of 3 years that met the following criteria: HbAA-phenotype, fetal haemoglobin < 1%, haemoglobin concentration ≥ 7.5 mmol/l, iron concentration ≥ 8 µmol/l and ferritin concentration ≥ 25 µg/l. Second, the groups with elevated HbA1c levels were selected by including all patients over a 3-year period with moderately (70–80 mmol/mol; n = 15) or strongly (110–180 mmol/mol; n = 49) elevated HbA1c levels, HbAA-phenotype, haemoglobin concentration ≥ 7.5 mmol/l, iron concentration ≥ 8 µmol/l and ferritin concentration ≥ 25 µg/l. Third, an iron deficiency group (n = 32) was selected by including all patients over a 3-year period with a HbAA-phenotype and ferritin concentration < 15 µg/l. Finally, four α-thalassaemia groups were selected by including all -α/αα patients (n = 65) over a 3-year period, all -α/-α patients (n = 45) over a 6-year period (from 2018 through 2023) and all --/αα (n = 34) and --/-α (n = 7) patients over a 10-year period (from 2014 through 2023; Table S1). Because -α/-α, --/αα and --/-α-thalassaemias are relatively rare, the collection period was extended beyond 3 years to include sufficient patients in these groups. Individuals included in the control and iron deficiency groups were negative for the seven most common deletions within the α-globin gene cluster (-α3.7, - α4.2, -α20.5, --SEA, --MED, --THAI and --FIL). Individuals included in the groups with elevated HbA1c levels did not undergo genetic α-thalassaemia testing.
HbA1c analysis
EDTA anticoagulated peripheral blood samples were measured using cation-exchange HPLC on the Tosoh G11 (Tosoh Bioscience, Japan) within 48 h after venipuncture.
Hemocytometry
EDTA anticoagulated peripheral blood samples were analysed with the XN-10 or XN-20 haemocytometer (Sysmex, Japan) within 24 h after venipuncture.
High-performance liquid chromatography
HPLC was conducted either at the physician’s request, due to suspected thalassaemia, or when routine HbA1c analysis using the Tosoh G11 analyser showed an HbA1c level exceeding 110 mmol/mol. Erythrocytes were lysed in 150 µl demineralized water followed by the addition of 500 µl potassium cyanide (2 mmol/l KCN in 0.5% BIS-TRIS, pH 6.98). Next, samples were centrifuged (10 min at 1500 RCF) and the resulting supernatant was filtered through a 0.22 μm membrane filter. Haemoglobin fractions were separated using an Ultimate 3000 pump (ThermoFisher Scientific) equipped with a 100 × 4.6 mm PolyCAT-A column (particle size 3 μm, pore size 1500 Å; PolyLC INC, #104ct0315). Haemoglobin fractions were detected at 410 nm using an Ultimate 3000 fluorescence detector (ThermoFisher Scientific). The HPLC system was controlled using Chromeleon 7.2.10ES software (ThermoFisher Scientific). This software package was also used for data analysis.
Statistical analysis
All groups were compared to the control group using the Kruskal-Wallis test with Dunn’s post-hoc correction. All data were analysed using GraphPad Prism 10.
Results
Patients with strongly elevated HbA1c levels (range: 110–180 mmol/mol) showed an average HbA1c of 130 mmol/mol and this was associated with a significant reduction in HbA2 levels compared with controls (2.0% vs 2.4%, P < .0001; Figure 1a–(b)). Patients with moderately elevated HbA1c levels (range: 70–80 mmol/mol) showed an average HbA1c of 76 mmol/mol, which was associated with a trend towards decreased HbA2 levels compared with controls, though without reaching statistical significance (2.2% vs 2.4%, P = .32). The reduced HbA2 fraction in patients with strongly elevated HbA1c levels was not observed when HbA2 was expressed relative to non-glycated HbA (i.e. HbA0) instead of all haemoglobin fractions (Figure 1(c)). Neither the MCH, the MCV nor the haemoglobin concentrations were affected in patients with elevated HbA1c levels (Figure 1(a), (d)). HbA2 levels, red cell indices, haemoglobin concentrations and erythrocyte count in patients with elevated HbA1c, iron deficiency or α-thalassaemia versus controls. (a) HbA2 level relative to all haemoglobin fractions, mean corpuscular haemoglobin (MCH) and mean corpuscular volume (MCV) in patients with elevated HbA1c levels, iron deficiency anaemia or α-thalassaemia versus controls. (b) HbA1c levels in patients with moderate elevated HbA1c levels (70–80 mmol/mol) and strongly elevated HbA1c levels (110–180 mmol/mol). (c) HbA2 levels relative to non-glycated HbA (i.e. HbA0) in patients with strongly elevated HbA1c levels (110–180 mmol/mol) versus controls. (d) Haemoglobin concentration and erythrocyte count in patients with elevated HbA1c levels, iron deficiency anaemia or α-thalassaemia versus controls. Data points in (a)–(d) represent the median (horizontal lines) from N = 9-80 individual patients, with each data point representing an individual patient. The green colour in (a)–(d) corresponds to the reference range established by the host laboratory and the red colour corresponds to the out-of-reference range. Reference ranges: HbA2, 2–3%; MCH, 1.7–2.1 fmol; MCV, 80–100 fl; HbA1c, 20–42 mmol/mol; haemoglobin concentration, 7.5–10.5 mmol/l; erythrocyte concentration, 4.5–5.5*109/l. The statistical differences in (a) and (d) are based on comparisons with the control group using the Kruskal-Wallis test with Dunn’s post-hoc correction. ****, P < .0001.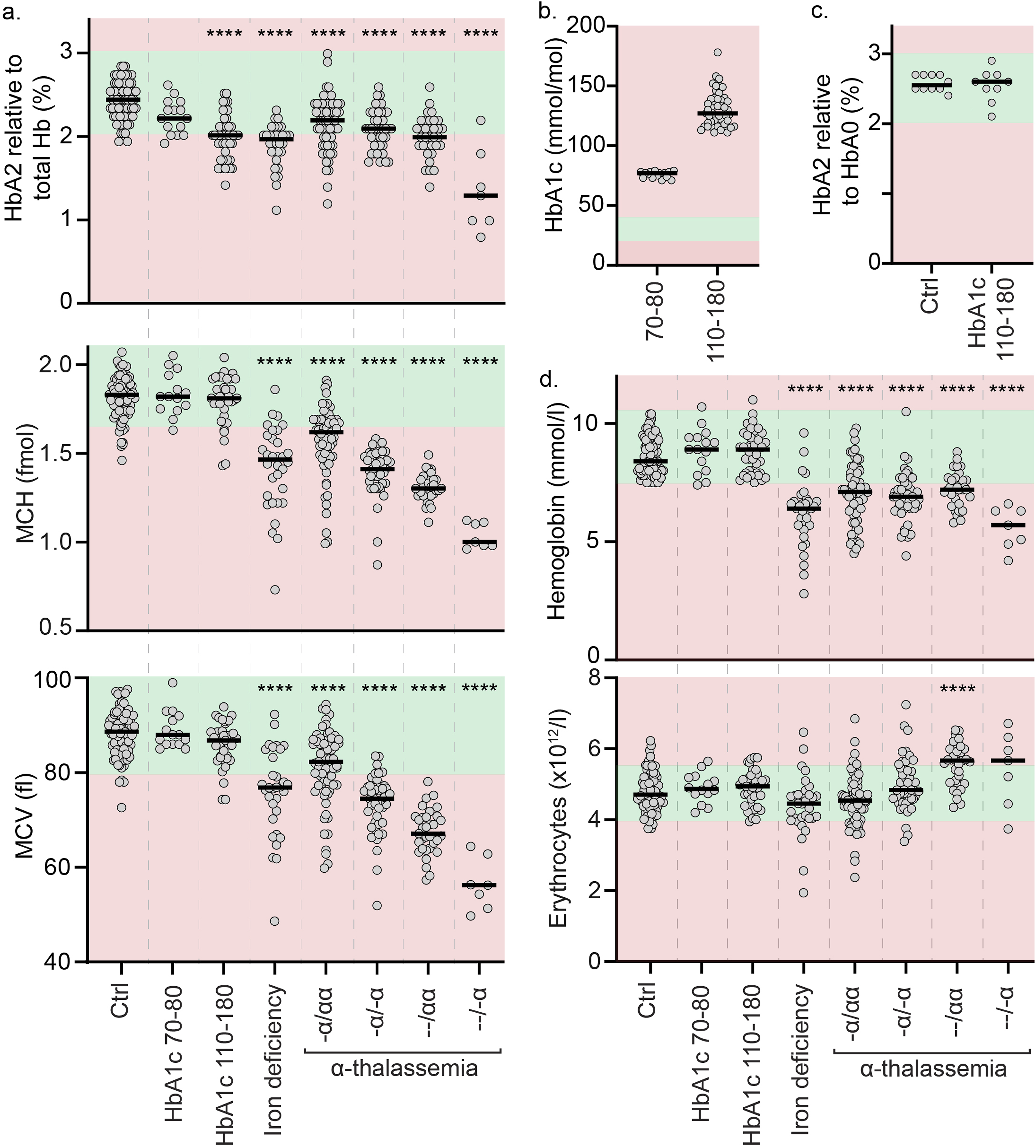
Patients with α-thalassaemia showed median HbA2 levels between 1.3 and 2.2% with a gradual decrease in MCH and MCV depending on the number of alpha gene deletions (Figure 1(a)). Iron deficiency resulted in a median HbA2 level of 2.0% with reduced MCH and MCV. In patients with strongly elevated HbA1c levels, the HbA2 fractions were reduced to a degree comparable to that seen in the α-thalassaemia cohort.
Discussion
Insight into the factors that modulate HbA2 levels is critical for correct interpretation of the laboratory results during diagnostic workup of patients suspected of thalassaemia. This study revealed that chronic hyperglycaemia, as reflected by elevated HbA1c levels, appears to be an important confounding factor in the diagnostic evaluation of thalassaemias as it results in significantly lower HbA2 levels.
Chronic hyperglycaemia increases the non-enzymatic addition of sugar residues to haemoglobin, including HbA2. We therefore hypothesize that glycated forms of HbA2 gain extra negative charge, causing it to be accelerated in the cation-exchange resin resulting in an earlier elution and thereby decreased levels of non-glycated HbA2. This hypothesis is supported by the observation that non-glycated HbA2 levels decrease in a dose-dependent manner with rising HbA1c concentrations. Furthermore, the reduced HbA2 fraction in patients with strongly elevated HbA1c levels was not observed when HbA2 was expressed relative to non-glycated HbA rather than total haemoglobin, suggesting that HbA2 glycation parallels the glycation of HbA. Thus, reduced HbA2 levels in uncontrolled diabetes reflect a shift in relative proportions due to glycation rather than an absolute reduction in HbA2 production.
Increased glycation of HbA2 in patients with uncontrolled diabetes has important clinical implications. Specifically, chronic hyperglycaemia may mask the elevated HbA2 levels typically used to diagnose β-thalassaemia trait, potentially leading to missed or delayed diagnoses. In addition, increased glycation of HbA2 may account for otherwise unexplained low HbA2 levels. To improve diagnostic accuracy, we recommend that laboratories consider HbA1c levels when interpreting HbA2 results in patients with poorly controlled diabetes. In such cases, expressing HbA2 levels relative to non-glycated HbA may provide a more accurate assessment and help clarify whether the HbA2 reduction reflects true pathology or is a consequence of glycation.
We compared the reduced HbA2 percentages during hyperglycaemia to other known causes of reduced HbA2 levels, such as iron deficiency and thalassaemia. Strongly elevated HbA1c levels reduced HbA2 fractions similarly to iron deficiency and α-thalassaemia. On the other hand, an altered lifespan of erythrocytes in patients with thalassaemia or iron deficiency anaemia will affect HbA1c levels in comparison to healthy individuals, thereby frustrating glycaemic control.
In conclusion, chronic hyperglycaemia, as reflected by elevated HbA1c levels, contributes to the diagnostic complexity of thalassaemias by lowering the HbA2 fraction and should be regarded as a potential explanation for reduced levels of non-glycated HbA2 during the evaluation of a suspected thalassaemia.
Supplemental Material
Supplemental Material - Elevated haemoglobin A1c confounds thalassaemia diagnosis by lowering HbA2 levels
Supplemental Material for Elevated haemoglobin A1c confounds thalassaemia diagnosis by lowering HbA2 levels by Jeroen Slaats, Erfan Nur, Harriët Heijboer, Eva Rettenbacher, Rob van Zwieten, Esther Miltenburg, Joyce Reijerse, Nienke Verhaart, Wouter van den Eshof,Bart J. Biemond and Henrike M. Hamer in Annals of Clinical Biochemistry.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Due to the retrospective design, the study was not subject to the Medical Research Involving Human Subjects Act since it does not impose any act or mode of behaviour on the subjects. All data were analysed anonymously.
Guarantor
HMH.
Contributorship
JS, EN, HH, ER, RZ, WE, BB and HMH conceived the study. EM, JR and NV performed the measurements. JS, EM, JR and NV analysed the data. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
