Abstract
Background
It is important that blood glucose concentrations be accurately and conveniently measured in infants. However, especially in the early neonatal period, point-of-care testing devices used for adults may not accurately measure blood glucose concentrations in neonates.
Methods
In Study 1, the accuracy of neonatal whole-blood glucose measurements was evaluated for the existing glucose analyser Glutest Mint® (hereinafter MINT1; Sanwa Kagaku Kenkyusho, Nagoya, Japan) by comparing the data with reference blood glucose concentrations. In Study 2, we used MINT2, which was modified based on the findings from Study 1, to measure whole-blood glucose concentrations in newborns, and the accuracy of the measurements was compared with that of MINT1.
Results
Blood glucose concentrations were measured in 100 infants each in Study 1 and 2. In Study 1, the whole-blood glucose concentrations measured using MINT1 were found to be significantly lower than the reference blood glucose concentrations in early neonates. The results of Study 1 suggested that characteristics of erythrocyte membranes in early neonates affected the measurements. Therefore, we conducted Study 2 using MINT2, which was modified to be less susceptible. MINT2 was found to accurately measure whole-blood glucose concentrations in the early neonatal period.
Conclusion
The study showed that the point-of-care testing device could be improved to allow for accurate whole-blood glucose measurements in the early neonatal period.
Introduction
Fetuses are supplied with glucose in utero by their mothers. However, at birth, the supply of glucose from the mother via the placenta is stopped, and the blood glucose concentration of the newborn rapidly decreases. At this time, the blood glucose concentration of a healthy neonate is maintained within the range of the infant’s physiological requirement by the blood glucose level maintenance mechanism.
However, neonates with backgrounds that put them at risk for hypoglycaemia, such as a low birth weight, preterm birth and maternal diabetes, may not maintain adequate blood glucose concentrations and may have disorders such as hypoglycaemic encephalopathy. To protect neonates from hypoglycaemic injury, blood glucose concentrations should be repeatedly assessed, and interventions should be considered at appropriate times. 1
Portable blood glucose metres, which are frequently used for blood glucose control in patients with diabetes, are useful for neonatal blood glucose monitoring because they can easily measure blood glucose with a very small volume of blood, as has been reported in many studies.2–4 However, it has been suggested that the accuracy of measurements needs to be assessed when blood glucose metres developed for use in adults are used in neonates. 5
The glucose analyser Glutest Mint® (Sanwa Kagaku Kenkyusho, Nagoya, Japan) is a point-of-care testing (POCT) device. In adults, Glutest Mint can accurately measure glucose concentrations in plasma sample and wide range of haematocrit values (20%–70%) whole-blood sample. 6 Therefore, this device is expected to be useful for measuring blood glucose concentrations in neonates with physiological polycythaemia.
As the neonatal period is a phase of adaptation from the in utero to the out-of-utero environment, the blood of neonates is not only polycythaemic but also has a variety of specific characteristics that are different from those in the blood of adults. The major environmental change for erythrocytes during the adaptation to the out-of-utero environment is a marked increase in the partial pressure of oxygen in tissues. During the fetal period, the fetus depends on the gas exchange in the placental circulation, and the partial oxygen pressure of 29–50 mmHg in the placenta is the maximum to which fetal erythrocytes are exposed and adapted. 7 These erythrocytes are then exposed to a large change in the oxygen environment at birth. Once the pulmonary circulation is established and is used for gas exchange, erythrocytes of the newborn are exposed to a partial oxygen pressure of approximately 100 mmHg in the pulmonary interstitium, which exposes the cell membranes to oxidative stress. 8 In addition, we have previously reported that neonatal erythrocytes produce approximately twice as much superoxide as adult erythrocytes under similar oxygen conditions. 9
We have speculated that these early neonatal erythrocyte characteristics might affect the readings of a portable whole-blood glucose metre. Therefore, this study aimed to improve the accuracy of the measurement of blood glucose concentrations in the early neonatal period using a portable blood glucose metre.
Methods
Study design
In Study 1, we evaluated the accuracy of neonatal blood glucose measurements using the existing glucose analyser Glutest Mint (hereinafter MINT1), and in Study 2, MINT2 was used as a glucose analyser, which was modified based on the results of Study 1. The subjects for both studies were term infants born at the Kagawa University Hospital, for whom blood sampling was deemed necessary. The number of subjects for each study was 100. In both studies, blood glucose concentrations measured by ABL800 and ABL90 FLEX (Radiometer, Copenhagen, Denmark) were used as reference values. To verify the accuracy of the ABL800 in neonatal blood, we compared blood glucose levels measured with the ABL800 to those obtained with a standard blood glucose meter, the GA1152 (Arkray, Kyoto, Japan). Our analysis of 104 neonatal samples confirmed a strong correlation, evidenced by a slope of 1.0 and an R^2 value of 0.98.
This study received approval from the institutional ethics committee of Kagawa University (approval numbers Heisei 27-094/Heisei 30-111). Prior to the measurements, the parents of all neonates enrolled in the study were fully informed about its nature and provided written informed consent. Additionally, this study was funded by PHC Holding Corporation as a contract research project. The authors disclose that the funding for this research was provided by the manufacturer of the equipment used during the study. It is important to note that aside from the financial support for the study, the authors have not received any other form of benefit, financial or otherwise, from the equipment manufacturer. This disclosure is made to ensure transparency and maintain the integrity of the research findings.
Study 1
When venous blood was drawn for clinical purposes, glucose concentrations in both whole blood and plasma were measured using MINT1, while whole-blood glucose levels were assessed with the ABL800 (Radiometer). The date of the blood collection was recorded, and haematocrit, bilirubin, pH, Na+ and pO2 values were also measured.
Study 2
Assuming that the oxidation of the electron mediator in the glucose analyser was the cause for the discrepancy in whole-blood glucose readings, the electron mediator used in MINT1 (9, 10-phenanthrenequinone-2-sulphonic acid sodium salt) was changed to 3-(2′,5′-disulphophenylimino)-3H-phenothiazine-bis-sodium salt in MINT2. When blood samples (including venous and heel blood) were collected for clinical needs, blood glucose concentrations were measured using ABL800 and ABL90 FLEX analysers simultaneously with MINT2.
Statistical analysis
Bland–Altman analysis was performed on whole-blood and plasma data to determine deviations in blood glucose concentrations measured by MINT1 from reference values determined using ABL800. To analyse the deviations in blood glucose concentrations between ABL800 and MINT1, as well as haematocrit, bilirubin, pH, Na+ and pO2 value subjects were divided into the following two groups: before and after 7 days of age. P < .05 was considered significant, and comparisons between the two groups were made using Student's t test. For Bland–Altman analysis of deviations in whole-blood glucose concentrations measured by MINT2, ABL800 and ABL90 FLEX blood glucose readings were used as reference values. Among the daily blood glucose deviations between MINT1 obtained in Study 1 and MINT2 obtained in Study 2, P < .05 was considered significant for the early neonatal group, and MINT1 and MINT2 data were compared by Student's t test. Analyses were performed using GraphPad Prism version 6.0 for Windows (GraphPad Software, San Diego, CA, USA).
Results
Study 1
Data for 99 of the 100 neonates were analysed, excluding one case for whom the measurement could not be performed owing to a mechanical failure of ABL800. No deviations were found between the reference glucose values and MINT1-measured plasma glucose concentrations (Figure 1(a)); however, low whole-blood glucose concentrations were obtained using MINT1 (Figure 1(b)). Deviation of blood glucose measurements from plasma and whole blood in MINT 1 (Bland–Altman analysis).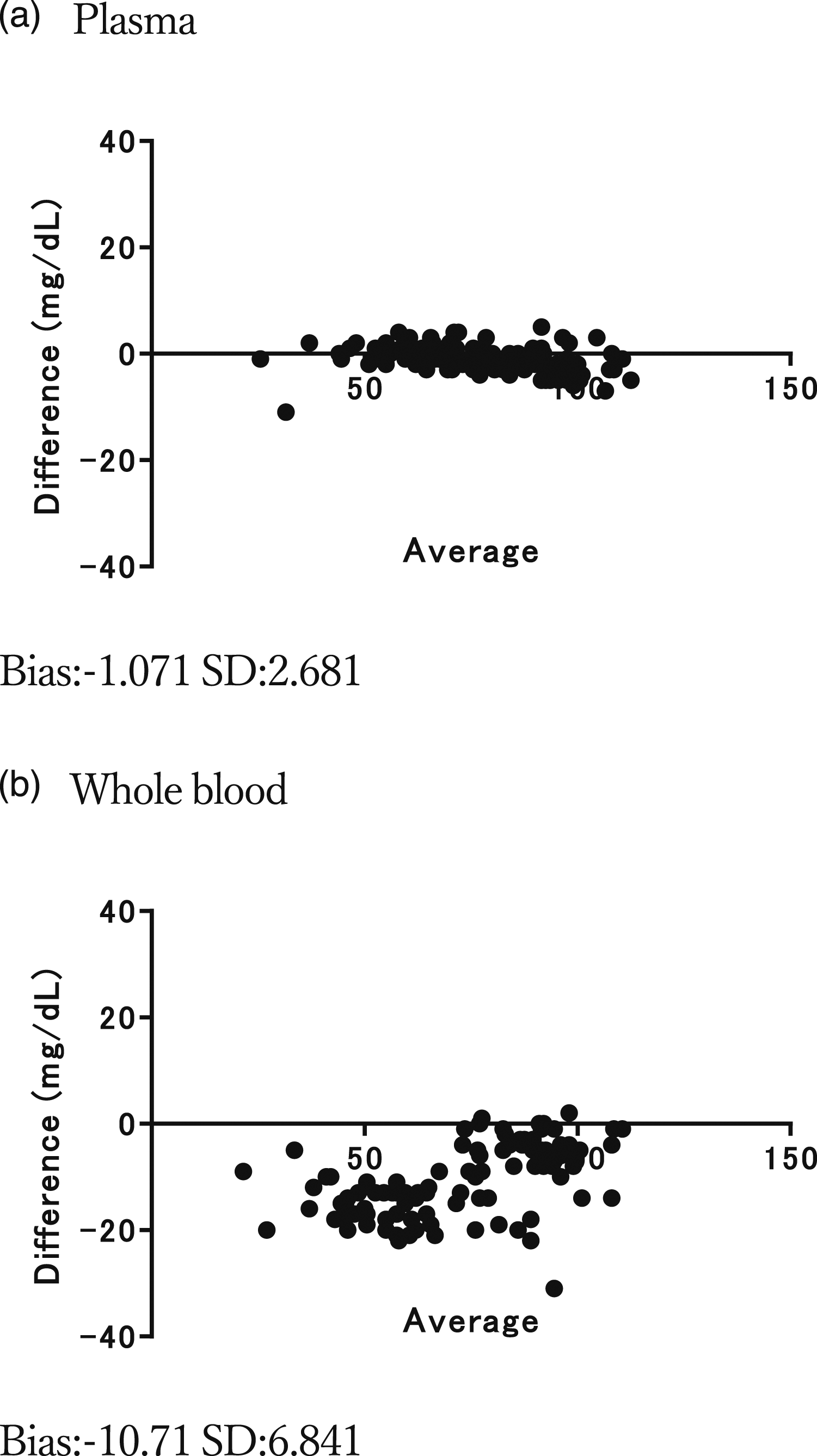
Comparison of Measured Parameters Between the Early Neonatal and Post-7-day Groups in Study 1.
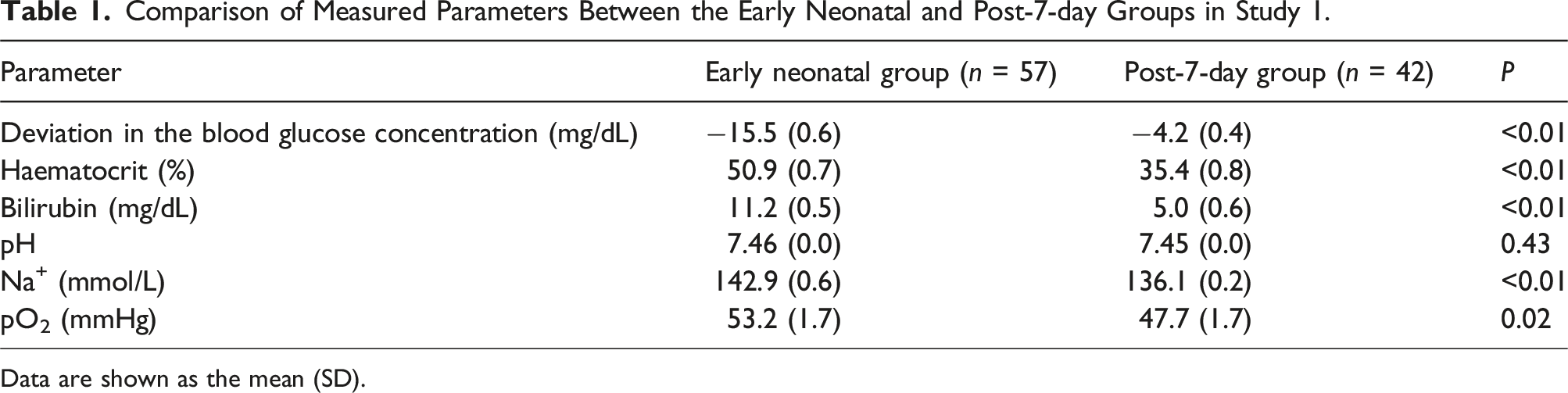
Data are shown as the mean (SD).
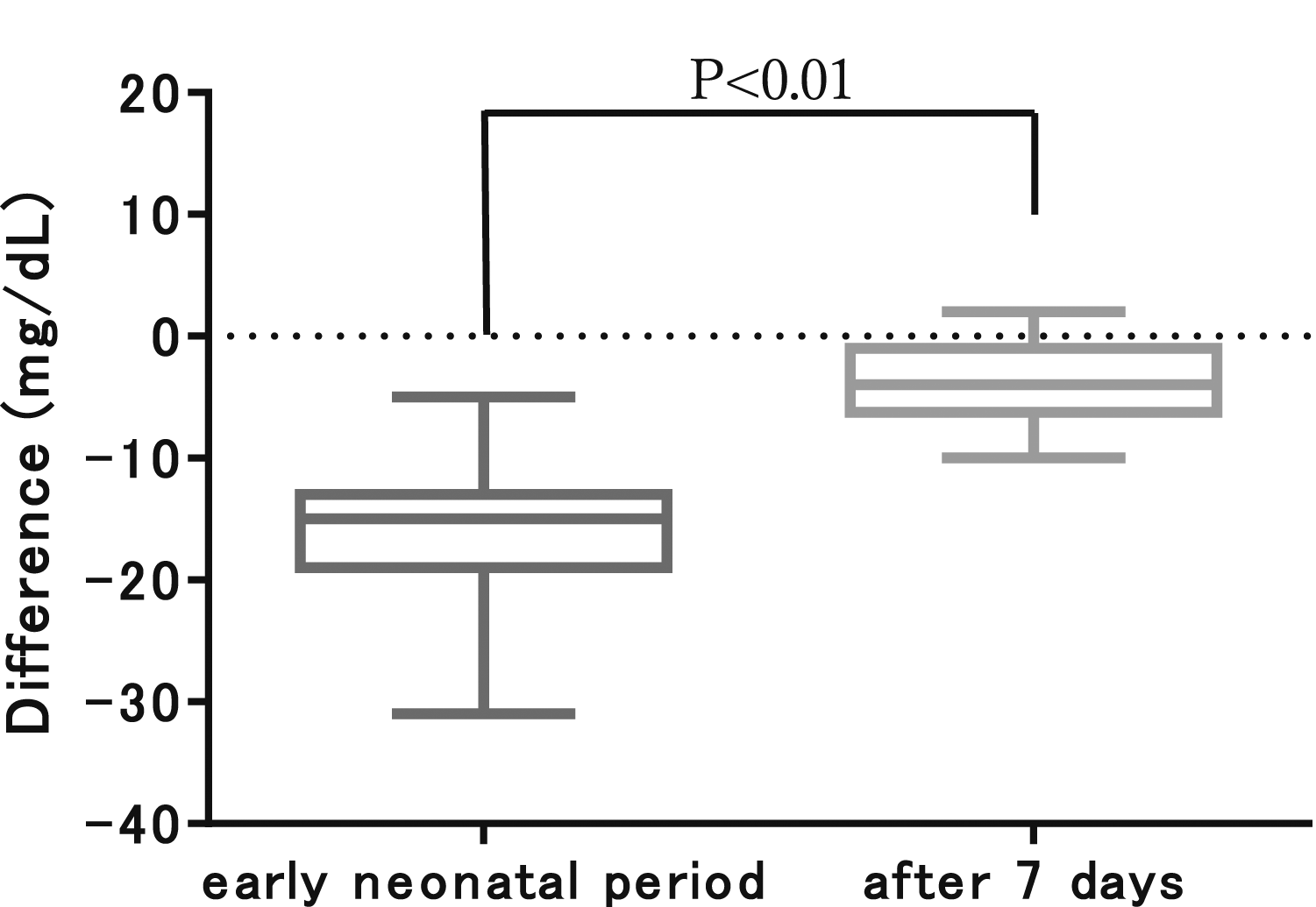
Comparison of blood glucose deviations in MINT1 between the two groups in the early neonatal period and after day 7.
In addition to the differences in blood glucose concentrations, there were significant differences in haematocrit, bilirubin, Na+ and pO2 values between the two groups, while pH values did not differ between the groups (Table 1).
Study 2
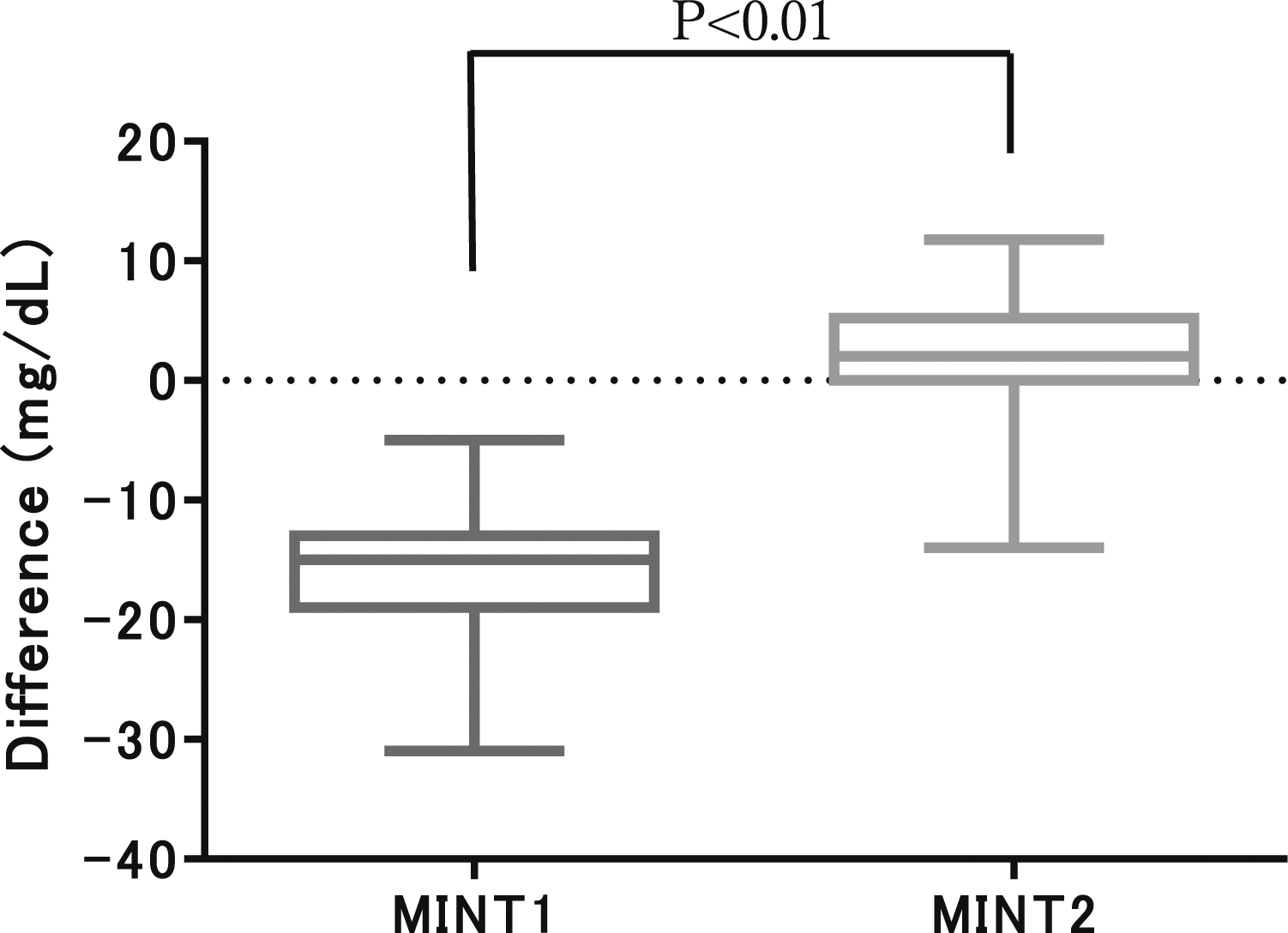
No deviations were found between the reference blood glucose concentrations and those measured with MINT2 using whole blood (Figure 3). Among the daily blood glucose deviations between MINT1 in Study 1 and MINT2 in Study 2, MINT1 (n = 57) showed significantly greater deviations than MINT2 (n = 89) in the early neonates (Figure 4). Deviation of blood glucose measurements from whole blood in MINT 2 (Bland–Altman analysis). Comparison of blood glucose deviation in early neonatal group at MINT1 and MINT2.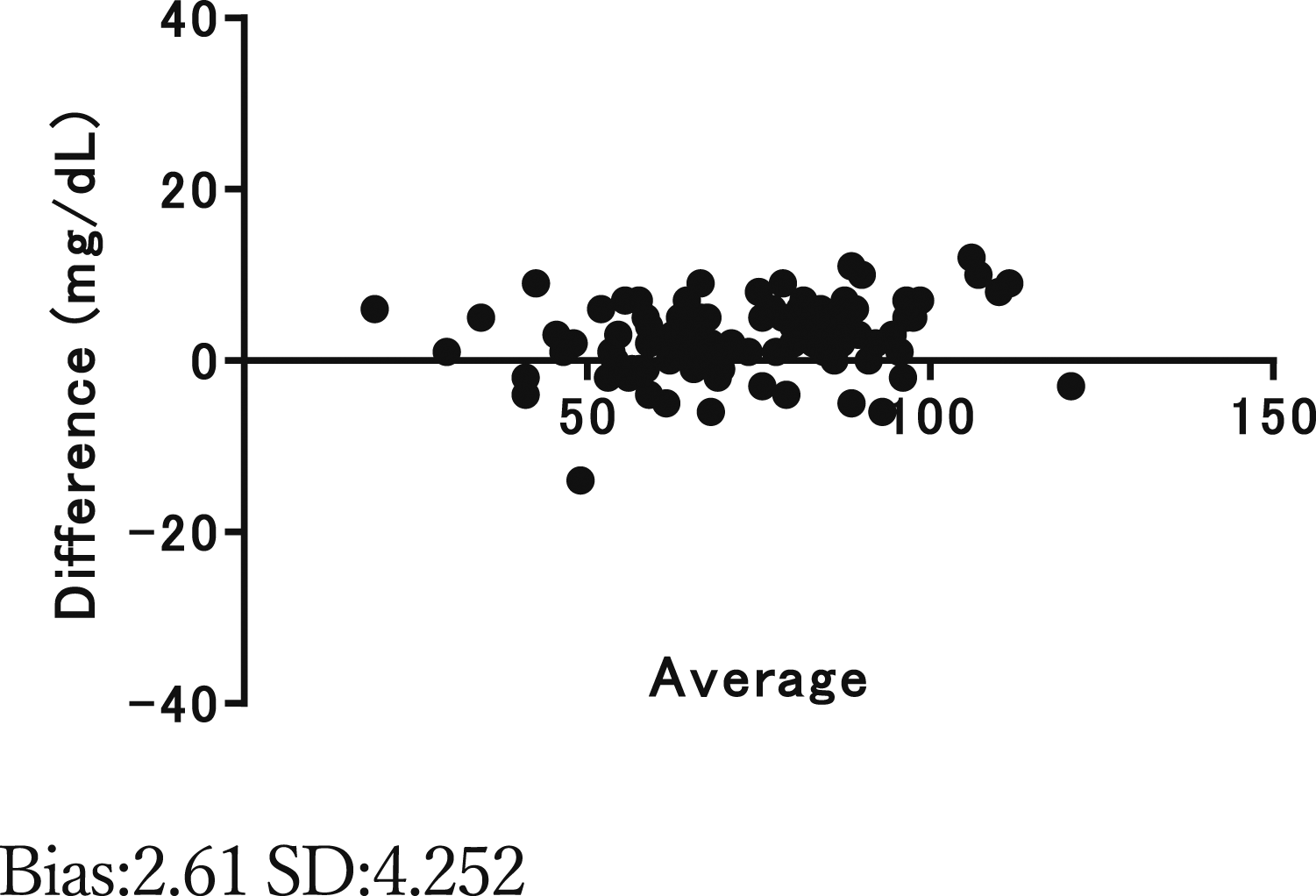
Discussion
When whole blood was used in the MINT1 study, the blood glucose concentrations were lower than the reference values in the early neonatal period. Since no deviations from the reference values were observed when using plasma, it was thought that the deviations were caused by blood cell components in neonates.
In addition, whole-blood readings were also taken with other POCT devices from other manufacturers at the time of the Study1, and these devices showed significantly higher blood glucose readings compared to ABL readings.
The lower blood glucose readings with the MINT1 were presumed to be due to MINT1-specific causes.
The reason for the greater deviations in the blood glucose concentrations from reference values during earlier days of age was thought to be the presence of fetal-type erythrocytes in the blood of neonates in the early postnatal period. It has been reported that more reactive oxygen species (superoxide, hydrogen peroxide and hydroxyl radicals) are present or produced on the surface of fetal-type erythrocytes than in adults.9,10
In MINT1 and MINT2, glucose in the specimen reduces the oxidized mediator through an enzymatic reaction. When the reduced mediator is oxidized again, an electric current is generated to the electrode, and the blood glucose concentration is measured by measuring the current.
The reason for the low blood glucose readings by MINT1, especially in the early neonatal group, was assumed to be the oxidation of the mediator, phenanthrene hydroquinone, by reactive oxygen species, such as superoxide, on the surface of fetal-type erythrocytes, resulting in a low response at the electrode. 11
To avoid the reactions with superoxide, we considered a change to a mediator that had a higher oxidation–reduction potential than quinone compounds and was less affected by reactive oxygen species. Therefore, we utilized a phenothiazine compound with a high oxidation–reduction potential and high water solubility to increase solubility in blood, as a mediator. No studies have been conducted on any of the supplies other than 3-(2′,5′-disulphophenylimino)-3H-phenothiazine-bis-sodium salt. As a result, MINT2 was able to accurately measure blood glucose concentrations even in early neonates, without generating low values.
This study revealed that even instruments that can stably measure blood glucose concentrations in adult samples do not necessarily maintain the same level of accuracy when used for early neonates, who are in the process of adjusting from the in-utero environment to the out-of-utero environment. When using biochemical analysers for children, especially neonates, it is important to verify the accuracy of measurements before clinical applications.
In Study 1, many blood samples were collected on day 2 for medical reasons, which could explain significant differences in haematocrit, bilirubin and Na+ values. In particular, high haematocrit and Na+ values could be due to physiological dehydration on day 2, and high bilirubin concentrations could be due to a physiological increase on day 2. We thought that the main reason for the deviations in the blood glucose concentrations was related to the characteristics of erythrocytes in the early neonatal period. Accordingly, we were able to eliminate the difference in blood glucose readings by changing the electron mediator. However, we did not directly examine early neonatal erythrocyte characteristics in this study, and this is only a hypothesis. Although the difference was small, pO2 levels were significantly higher in the early neonatal group. Consequently, the possibility that this elevated dissolved oxygen content in MINT1 contributed to the notable measurement error in the early neonatal group cannot be dismissed. In investigating the impact of pO2 on MINT2, we analyzed the correlation between measurement errors and pO2 values compared between MINT2 and ABL. However, we found no correlation (Figure 5). Thus, we concluded that pO2 does not influence the accuracy of blood glucose level measurements by MINT2. However, the limitation of this study is that it did not allow ruling out that differences in haematocrit, bilirubin and Na+ values were responsible for differences in blood glucose concentrations. Correlation Between pO2 levels and blood glucose measurement deviations in MINT2.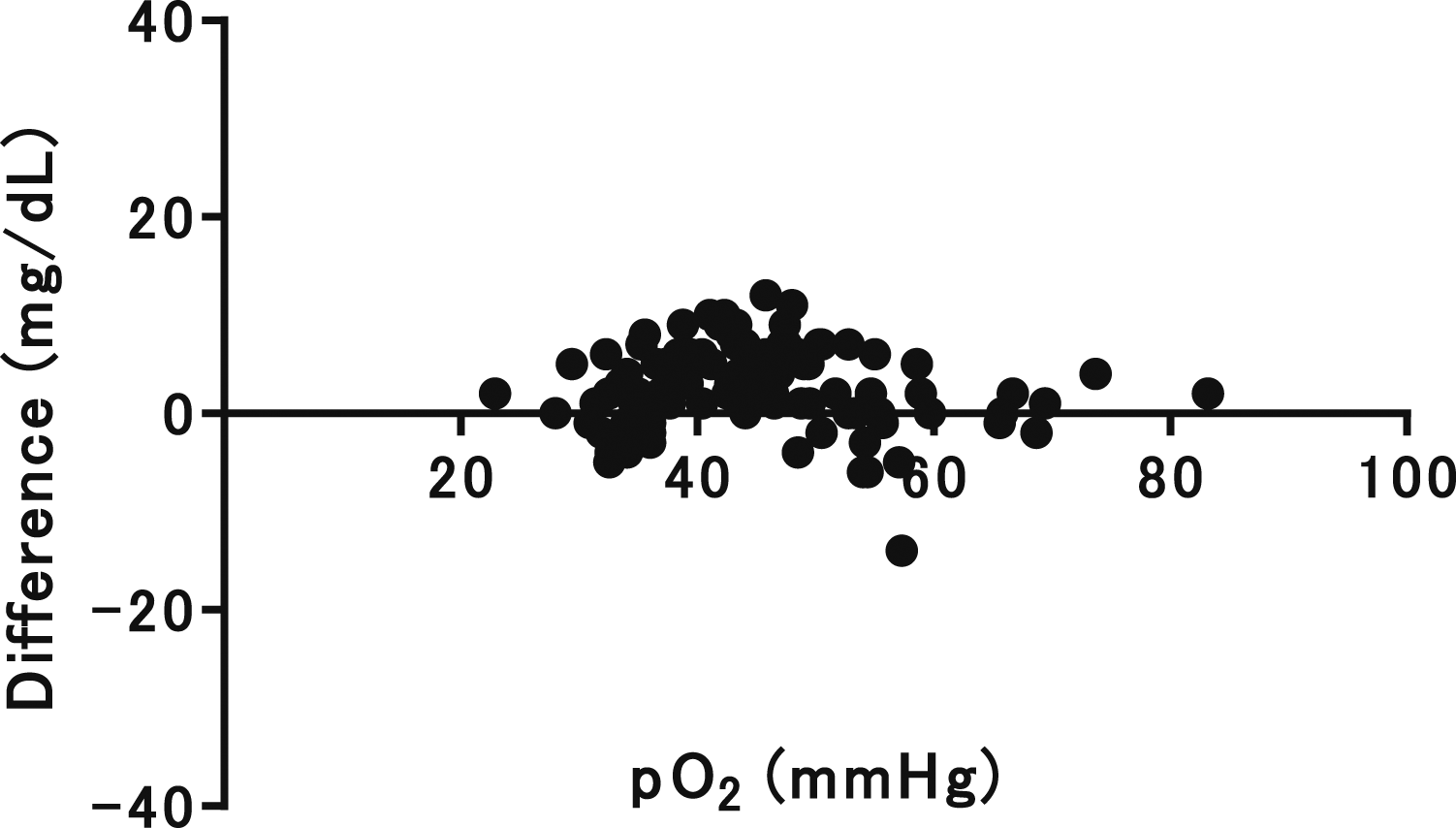
Conclusion
Considering the characteristics of fetal-type erythrocytes, we were able to improve the performance of a POCT device to allow accurate measurement of whole-blood glucose concentrations in the early neonatal period.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by PHC Holding Corporation as a contract research project. The authors disclose that the funding for this research was provided by the manufacturer of the equipment used during the study.
Correction (July 2024):
Ethical approval
The study was approved by the institutional ethics committee (Kagawa University Heisei 27-094 / Heisei 30-111).
Guarantor
KK and MS.
Contributorship
KK, SN, SY, TK researched literature and conceived the study, were involved in protocol development, KK, HM, SK, KY, TK were responsible for gaining ethical approval, and AM, MA, YN, MS collected samples. KK wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final manuscript.
