Abstract
Background
It is recommended that samples for plasma ammonia analysis are kept chilled and processed promptly as in vitro metabolism causes falsely elevated results. Rejection of unsuitable samples can cause delayed diagnosis and treatment of hyperammonaemia with potentially serious clinical consequences. The Metabolic Biochemistry Network (MetBioNet) hyperammonaemia guideline recommends analysis of samples not collected under ideal conditions and reporting with appropriate comments. An audit found that some laboratories did not follow this guidance. An investigation was performed into whether storage at controlled room temperature and delayed sample processing affected interpretation of plasma ammonia results.
Methods
Eleven healthy volunteers provided informed consent. Blood was taken from each into 14 paediatric EDTA blood sample tubes, one placed immediately on ice, the others in a rack at room temperature. The chilled and baseline room temperature samples were centrifuged and plasma analysed by the Roche Ammonia (NH3L2) method. Samples stored at room temperature were analysed at 10-min intervals up to 2 h.
Results
Baseline room temperature ammonia was higher than in the chilled sample (19 ± 6.6 µmol/L [mean ± standard deviation] and 18 ± 6.6 µmol/L, respectively). Ammonia increased further by 0.09 ± 0.02 µmol/L per minute to 30 ± 8.4 µmol/L at 2 h. No result was above the reference range (50 µmol/L). No healthy subject with normal baseline ammonia would have been erroneously identified as having hyperammonaemia.
Conclusions
Results support MetBioNet guidance that laboratories accept blood samples for ammonia analysis which are not processed under ideal conditions.
Background
Ammonia is highly neurotoxic, and hyperammonaemia is a medical emergency. Clinical features are subtle and non-specific, so prompt laboratory analysis of plasma ammonia is critical to identify high levels and ensure appropriate treatment to avoid devastating long-term neurological consequences. 1 Blood ammonia increases in vitro due to release from erythrocytes and plasma amino acid deamination. It is therefore recommended that blood is collected carefully into a cooled tube which is transported to the laboratory on ice and analysed promptly to avoid falsely elevated results. Some guidelines recommend no more than 15 min between venepuncture and sample analysis.2,3 This is difficult to follow in practice since clinicians may not be aware of the requirements, ice may not be readily available and prompt transport to the laboratory not possible. These pre-analytical challenges may prevent or delay ammonia requesting and measurement. 4
The UK Metabolic Biochemistry Network (MetBioNet) guideline for investigation of hyperammonaemia was updated in 2018 to recommend laboratories accept blood samples for ammonia analysis which are not collected and transported under ideal conditions and to advise urgent repeat if plasma ammonia is increased. 5 A national audit of adherence to the revised guideline was performed in 2019 by the Association for Clinical Biochemistry and Laboratory Medicine Audit Committee. 4 Responses from 76 laboratories showed that 64% rejected samples not received in the required time following venepuncture and 25% rejected samples not transported on ice. Rejection of samples not sent in ideal conditions necessitates repeat venepuncture and has the potential to delay detection of hyperammonaemia, development of ammonia neurotoxicity and associated increased health-care costs.
The studies on which the ideal sample requirements are based are over 20 years old 3 and involve equipment and methods no longer in routine use. An investigation into plasma ammonia concentration in blood taken from healthy volunteers and stored at controlled room temperature before analysis was performed using current, widely used methods to determine whether following the MetBioNet guideline would affect interpretation of results.
Methods
Healthy volunteers over 18 years of age, non-smokers not taking diuretics or valproate, were invited to participate in the investigation. Blood was taken from each subject into a syringe, the needle was removed and the blood transferred carefully to 14 Microvette 0.5 mL K2EDTA blood tubes (Sarstedt, Nümbrecht, Germany). The tube caps were replaced as soon as the blood had been added. One tube was put into a container full of ice, and the others were placed in a rack and stored on the laboratory bench. The sample on ice and baseline room temperature sample were centrifuged within 15 min of venepuncture at 4°C for 5 min at 2000g. Plasma was removed, transferred to an analytical tube and placed on the Roche c702 analyser (Roche Diagnostics, Burgess Hill, UK) for analysis by the Roche Ammonia (NH3L2) method. Every following 10 min a tube was removed from the rack, centrifuged and plasma ammonia analysed. The assay was calibrated before each set of samples and internal quality control (IQC) analysed at baseline, 60 min and 120 min for each set. The study was performed over 5 days in an ISO 15189-accredited laboratory. Temperature was recorded using a Hanwell temperature monitoring device (Hanwell, Hertfordshire, UK) calibrated and assured to ISO 15189 standards. External quality assurance (EQA) samples were analysed during the study period. The analytical coefficient of variation (CV) was 5.1% at 52.9 µmol/L, less than half the reported intra-individual biological variation of ammonia (13.8%), thus accepted as fit for purpose. 6
Results of ammonia analysis at different time points were compared using a paired t-test and the relationship between ammonia concentration and time of storage compared by regression analysis with differences considered significant when P < .05. Data analysis was performed using Microsoft 356 Excel (Microsoft UK, Reading, UK).
The study was approved by the Heath Research Authority and Health and Care Research Wales, REC reference 21/NI/0058, IRAS project ID 292835.
Results
Eleven healthy volunteers provided informed consent for a blood sample to be taken for the study. Plasma ammonia in samples placed on ice was 18 ± 6.6 µmol/L (mean ± standard deviation). Ammonia was higher in the baseline room temperature sample analysed at the same time, 19 ± 6.6 µmol/L (P = .02).
After storage for 120 min at room temperature, ammonia increased to 30 ± 8.4 µmol/L (P < .001). The mean increase in ammonia was linear (R2 = 0.9896, P < .001) (10 ± 2.3 µmol/L over 120 min, or 0.09 ± 0.02 µmol/L per minute) (Figure 1). No blood sample analysed had a plasma ammonia concentration above the reference limit of 50 µmol/L. Plasma ammonia concentration in blood samples stored at ambient temperature analysed every 10 min for 2 hours. Results show the mean (black squares) ± standard deviation (vertical bars). Linear regression is shown by the dotted line and R2 value.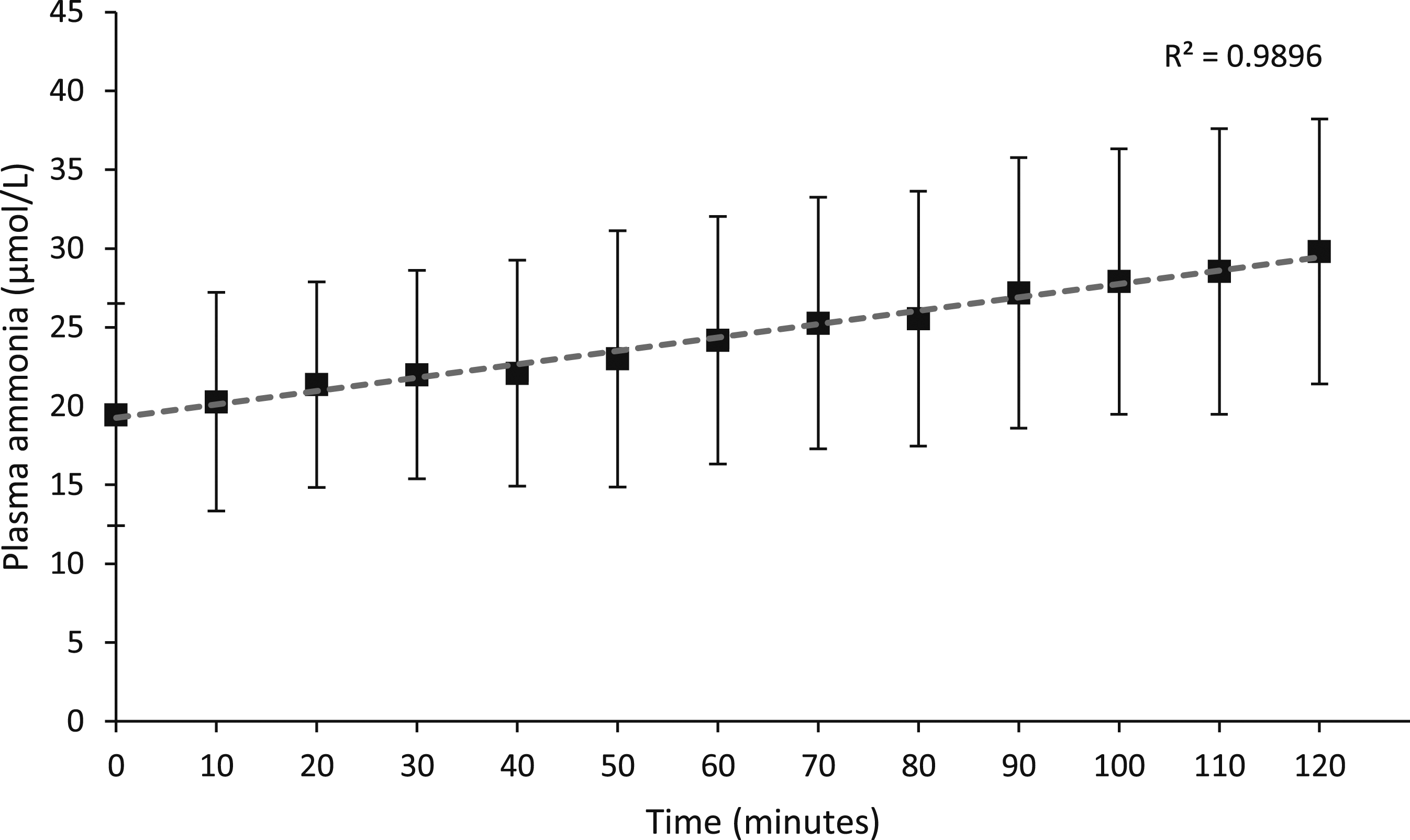
During the study, IQC and EQA results were within acceptable limits and the laboratory temperature was 20°C–21°C.
Discussion
Blood stored in paediatric sample tubes at controlled room temperature showed increased ammonia up to 15 min after venepuncture compared with samples placed on ice, as reported previously. 3 Over the following 120 min there was a further small, linear increase in plasma ammonia but no subject was misclassified as having hyperammonaemia.
These results indicate that when investigating patients to determine whether they have hyperammonaemia, transport of samples on ice is not necessary if samples are processed within 2 hours, supporting the MetBioNet guidance. 5
The data may not be applicable to monitoring patients with hyperammonaemia such as those with urea cycle disorders or hepatic encephalopathy since a clinically significant change in ammonia has not been established and patients with high circulating ammonia were not included in the study. Further work is required to determine a clinically significant change in ammonia and whether the results of this study can be reproduced in samples from patients with hyperammonaemia.
Conclusion
Blood samples that were stored at room temperature up to 120 min before analysis demonstrated a small, linear increase in plasma ammonia that did alter interpretation of the result. This supports the MetBioNet guidance that laboratories accept blood samples for investigation of hyperammonaemia which are not collected and transported under ideal conditions.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
