Abstract
Background
Blood collection tubes with sodium fluoride (NaF) added as a glycolytic inhibitor are widely used for glucose measurement. However, the glycolytic inhibitory effects of NaF are insufficient, and decreases in glucose levels over time after blood collection have become a problem.
Methods
Blood from a volunteer collected using an NaF tube was used to compare the glycolysis inhibitory abilities of ATP and ADP. Blood samples from 10 volunteers were collected in NaF tubes and NaF tubes with added ATP (NaF–ATP tubes). The stability of glucose and haemoglobin (Hb)A1c after whole-blood storage from immediately after blood collection to 24 h later was compared.
Results
ATP and ADP had similar inhibitory effects on glycolysis, but ATP was selected as an additive for blood collection tubes because ADP was more haemolytic than ATP. We verified the ability of NaF blood collection tubes supplemented with ATP to inhibit glycolysis. Mean (± standard deviation) glucose levels (n=10) after storage for 24 h after blood collection decreased to −9.0 ± 2.7 mg/dL (−0.50 ± 0.15 mmol/L) in conventional NaF tubes. NaF–ATP(20) tubes with 20 mg (0.036 mmol) ATP added showed a reduced decrease, with a mean of −5.8 ± 2.9 mg/dL (−0.32 ± 0.16 mmol/L). NaF–ATP tubes also had no effect on HbA1c measurement.
Conclusion
This study reports on a blood collection tube that enables the measurement of glucose and HbA1c. Based on the results of validation, we conclude that NaF–ATP tubes can reduce decreases in glucose over time in stored whole blood compared to conventional NaF tubes.
Introduction
Glucose and haemoglobin (Hb) A1c are frequently measured during diabetes testing. However, sodium fluoride (NaF) tubes have been reported to cause a decrease in glucose in samples stored at room temperature within 4 h after blood collection. 1 As a countermeasure, the American Society of Clinical Biochemistry guidelines recommend immersion in an ice–water slurry immediately after blood collection with heparin–lithium (Hp-Li) for plasmaization within 30 min or the use of NaF and citrate buffer-containing (FC) tubes. 2 However, citric acid in FC tubes is difficult to dissolve, making blood collection operations laborious. Furthermore, it alters red blood cells, making measurement with some HbA1c reagents impossible. Glucose and HbA1c analyzers are available in Japan and other countries, 3 and biochemical analyzers can measure both glucose and HbA1c from a single blood collection tube. 4 Measuring these two items from the same blood collection tube is therefore important, and NaF tubes are currently in wide use for this purpose. In addition, glucose testing can take up to 24 h after blood collection for centrifugation and measurement, as smaller facilities and health screening organizations outsource this testing to outside facilities. In such cases, current glucose tests with NaF tubes may miss diabetes because the measured value is underestimated. In this study, we describe the development of a blood collection tube to maintain stable glucose levels in whole blood for up to 24 h. Since adenosine 3-phosphate (ATP) and adenosine diphosphate (ADP) have been reported to inhibit the function of glucose transporters in erythrocytes (Blodgett DM., 5 Carruthers A., 6 Matsuo S.), 7 we prepared NaF–ATP or ADP tubes by adding ATP or ADP to conventional NaF tubes. The possibility of measuring both glucose and HbA1c was also examined.
Materials & Methods
Subjects
Blood from a volunteer (male; age 54 years, Hct 40.9%) whose glucose decreased substantially in a stability test using an NaF blood collection tube was used to compare the glycolysis inhibitory abilities of ADP and ATP. A 2 mL sample of whole blood was collected in each tube. Immediately after blood collection, each tube was agitated on a mix rotator at 30 rpm for 3 min and then stored under storage conditions until immediately before measurement. Aliquoted blood collection tubes were stored at room temperature (25 ± 0.5°C) for up to 4 h and refrigerated (4°C) for 4–24 h. Furthermore, aliquoted whole blood was plasma-separated by centrifugation immediately after collection, and at 2 h, 4 h, 12 h, and 24 h after collection. For glucose and HbA1c stability studies of blood collection tubes with ATP added to NaF, 10 staff members (five male and 6 female; age range, 25–54 years) were included in the study, with samples taken 5 h after eating. In addition, to study the effect of ATP on blood with high glucose levels, blood samples from an additional five patients (146.4 to 363.2 mg/dL: 8.13 to 20.16 mmol/L) were used. In this study, we used blood collected from patients EDTA blood collection tubes within 1 h after collection.
The study was conducted under a protocol approved by the Ethics Committee of the Graduate School of Medicine, University of Tokyo, after obtaining informed consent from the volunteers (approval no. 2020063NI).
Blood collection tubes
The following eleven types of blood collection tubes were used: 1) EDTA tube – Nipro Neotube A EDTA-2k (granules) NP-EK0205 (Nipro Corporation, Osaka, Japan); 2) NaF tube – Nipro Neotube NaF+EDTA-2Na (granules) OP-FN0205 (Nipro Corporation); 3) NaF-ATP(5) tube – NaF tube containing 5 mg (0.009 mmol) of ATP; 4) NaF-ATP(15) tube – NaF tube containing 15 mg (0.027 mmol) of ATP; 5) NaF-ATP(20) tube – NaF tube containing 20 mg (0.036 mmol) of ATP; 6) NaF-ATP(40) tube – NaF tube containing 40 mg (0.072 mmol) of ATP; 7) NaF-ATP(60) tube – NaF tube containing 60 mg (0.108 mmol) of ATP; 8) NaF-ADP(5) tube – NaF tube containing 5 mg (0.011 mmol) of ADP; 9) NaF-ADP(20) tube – NaF tube containing 20 mg (0.042 mmol) of ADP; 10) NaF-ADP(40) tube – NaF tube containing 40 mg (0.085 mmol) of ADP; and 11) NaF-ADP(60) tube – NaF tube containing 60 mg (0.127 mmol) of ADP. The preparation of ADP or ATP collection tube is described below. First, 25 μL of 200 mg/mL ATP solution was added to filter paper (ADVANTEC Qualitative Filter Paper No. 131; ADVANTEC Co., Ltd., Tokyo, Japan) cut to a diameter of 6 mm, then the ATP–filter paper was dried. One sheet of ATP filter paper (equivalent to 5 mg of ATP) was added to the NaF blood collection tube to make an NaF–ATP (5) tube. NaF–ATP (15, 20, 40, and 60) tubes were made by adding the required number of ATP sheets to NaF tubes. NaF–ADP blood collection tubes with the target concentration were also prepared in the same manner.
Analyzers
A clinical chemistry analyzer (JCA-BM6010 BioMajesty; JEOL, Tokyo, Japan) was used to measure glucose using general-purpose reagents and to measure HbA1c using enzymatic and immunoassay methods. A glycohemoglobin G11 automatic analyzer (Tosoh Co., Tokyo, Japan) was used for HbA1c measurements using the high-performance liquid chromatography (HPLC) method. A multi-item automatic blood cell analyzer (XN-1000; Sysmex Co., Hyogo, Japan) was used to measure haematocrit (Hct). A microplate reader (SYNERGY H1; Agilent Technologies Japan Ltd., Tokyo, Japan) was used to measure haemoglobin (Hb).
Reagents
Glucose was measured using the hexokinase method (Determiner L GLU-HK; Minaris Medical Co., Tokyo, Japan). HbA1c was measured using three methods, comprising the enzymatic method (MetaboLead HbA1c; Minaris Medical Co.), immunoassay method (Determiner L HbA1c; Minaris Medical Co.), and HPLC method with a dedicated column and reagents for a glycohemoglobin G11 autoanalyzer (Tosoh Co.). Haemoglobin was measured using Hemoglobin B test WAKO (FUJIFILM Wako Pure Chemical Co., Osaka, Japan).The reagent for addition of ADP was adenosine 5'-diphosphate disodium salt (45130000; Oriental Yeast Co., Tokyo, Japan). The reagent for addition of ATP was adenosine 5'-triphosphate disodium salt (45142000; Oriental Yeast Co.). The reagent for addition of NaF was sodium fluoride (194-01971; FUJIFILM Wako Pure Chemical Co.).
Glucose and HbA1c measurement methods
Blood was drawn from the 10 staff members. For glucose and HbA1c stability studies, 2 mL of whole blood was collected in each tube. Immediately after blood collection, each tube was agitated on a mix rotator at 30 rpm for 3 min and then stored under storage conditions until immediately before measurement. Aliquoted blood collection tubes were stored at room temperature (25 ± 0.5°C) for up to 4 h and refrigerated (4°C) for 4–24 h. Furthermore, aliquoted whole blood was plasma-separated by centrifugation immediately after collection and at 2 h, 4 h, 12 h, and 24 h after collection. Glucose was measured immediately after separation. For the measurement of HbA1c, a portion of the whole blood before centrifugation for glucose measurement was transferred to another tube and measured according to the recommended protocol for each measurement method.
Changes in glucose concentration in patient whole blood
Patient blood was used within 1 h after collection. NaF collection tubes were prepared by adding NaF to EDTA collection tube patient blood. In addition, NaF–ATP(20) collection tubes were made by adding NaF and ATP filter paper to patient blood (EDTA collection tubes). The prepared blood collection tubes were stored in the same way as previously described, at room temperature (25 ± 0.5°C) for 0–4 h and at 4°C for 4–24 h. Whole blood was plasma-separated by centrifugation immediately after creation and at 24 h after creation. Glucose was measured immediately after separation.
Statistical analysis
All statistical analyses were performed using EZR3 version 1.60 (Saitama Medical Center, Jichi Medical University, Japan), a graphical user interface for R version 2.8-0 (The R Foundation for Statistical Computing, Vienna, Austria). This is a modified version of R Commander in which statistical functions were designed and added for more accurate use of biostatistics. 8 In addition, repeated-measures analysis of variance (ANOVA) and Dunnett’s test were used for statistical processing. Differences were considered significant at the level of P < 0.05.
Results
Comparison of ATP and ADP glycolysis inhibition
Plasma Hb concentrations immediately after adding 20 mg of ATP and ADP to the NaF tube were 12 mg/dL and 17 mg/dL, respectively. And, 24 h after adding 40 mg of ATP and ADP to the NaF blood collection tube, plasma Hb concentrations were 25 mg/dL and 29 mg/dL, respectively. ATP was chosen as the additive for blood collection tubes in this study because ATP causes less haemolysis than ADP. Addition of 20 mg/dL ATP per 2 mL blood collection tube maintained glucose at 97.2% of the initial concentration after 24 h of whole-blood storage at room temperature. Therefore, we set the concentration of ATP added to 20 mg (Figure 1, Supplemental Table 1). Comparison of ADP and ATP glycolysis inhibition. Blood samples were collected from Volunteer (male; age 54 years, Hct 40.9%). Whole-blood collection tubes were stored at room temperature (25 ± 0.5°C) for up to 4 h and refrigerated (4°C) for 4–24 h. Change in blood glucose level with addition of ADP or ATP to NaF blood collection tubes (aliquoted whole blood was plasma-separated by centrifugation immediately after collection and at 2 h, 4 h, 12 h, and 24 h after collection) is shown: a) for addition of ADP to an NaF blood collection tube (glucose concentration), b) for addition of ATP to an NaF blood collection tube (glucose concentration), c) for addition of ADP to an NaF blood collection tube (glucose rate of change), and d) for addition of ATP to an NaF blood collection tube (glucose rate of change) ●:0 h whole blood stored;○:2 h whole blood stored; ▲:4 h whole blood stored; △:12 h whole blood stored;■:24 h whole blood stored.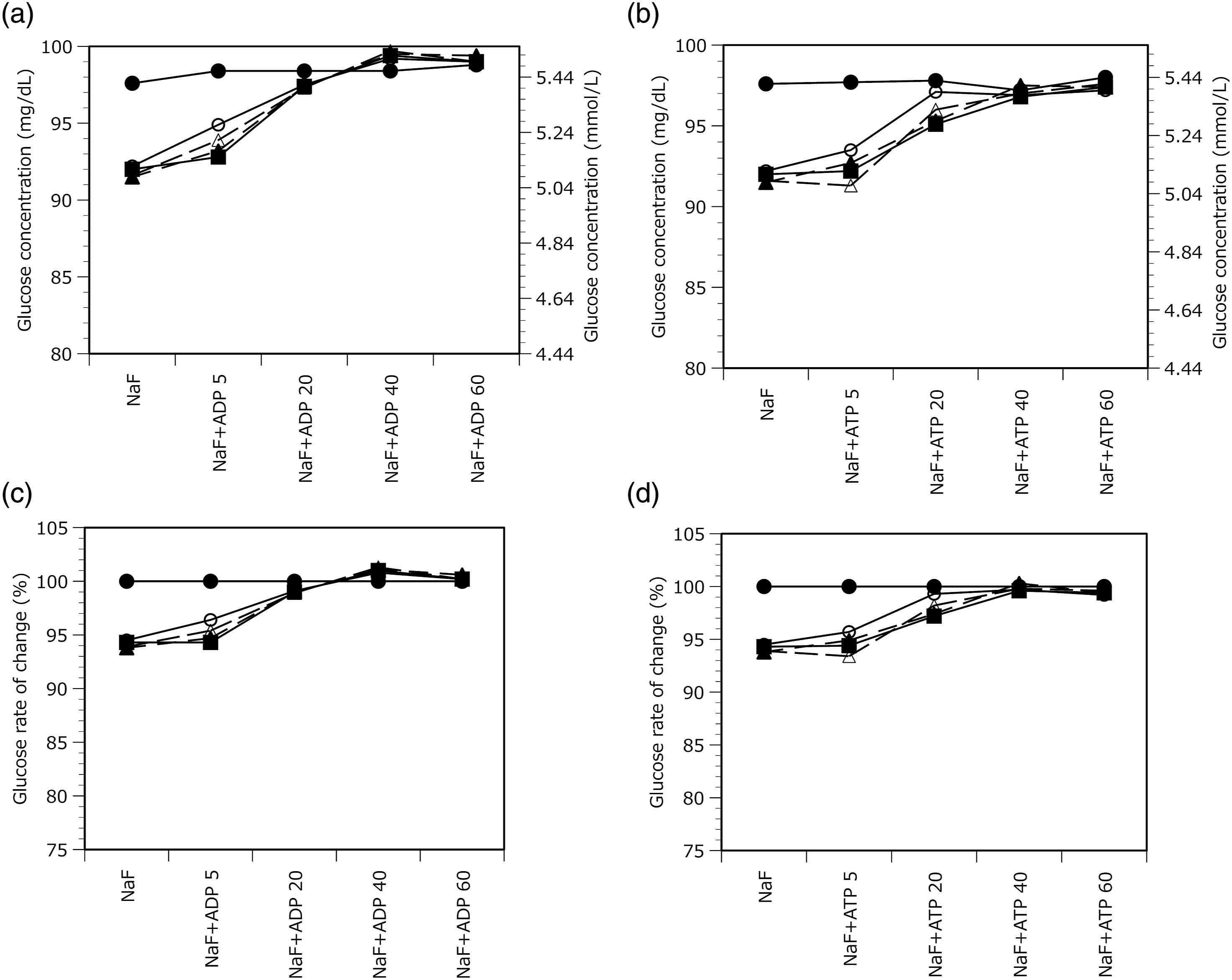
Stability of glucose in the stored blood collection tubes after sampling
Glucose Stability of Whole-Blood Samples.
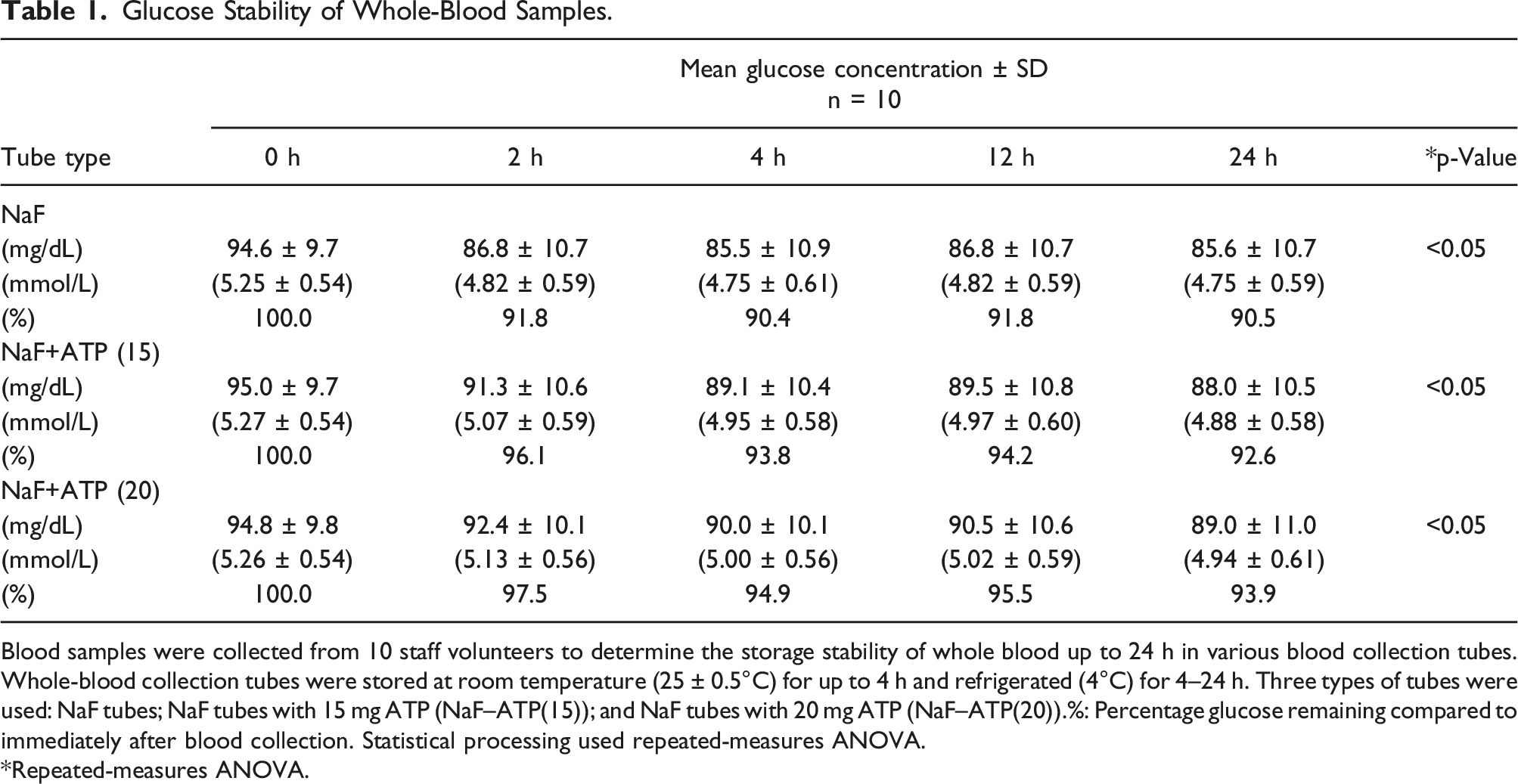
Blood samples were collected from 10 staff volunteers to determine the storage stability of whole blood up to 24 h in various blood collection tubes. Whole-blood collection tubes were stored at room temperature (25 ± 0.5°C) for up to 4 h and refrigerated (4°C) for 4–24 h. Three types of tubes were used: NaF tubes; NaF tubes with 15 mg ATP (NaF–ATP(15)); and NaF tubes with 20 mg ATP (NaF–ATP(20)).%: Percentage glucose remaining compared to immediately after blood collection. Statistical processing used repeated-measures ANOVA.
*Repeated-measures ANOVA.
Amount of Change in Plasma Glucose Reduction From Immediately After Blood Collection for Each Volunteer.
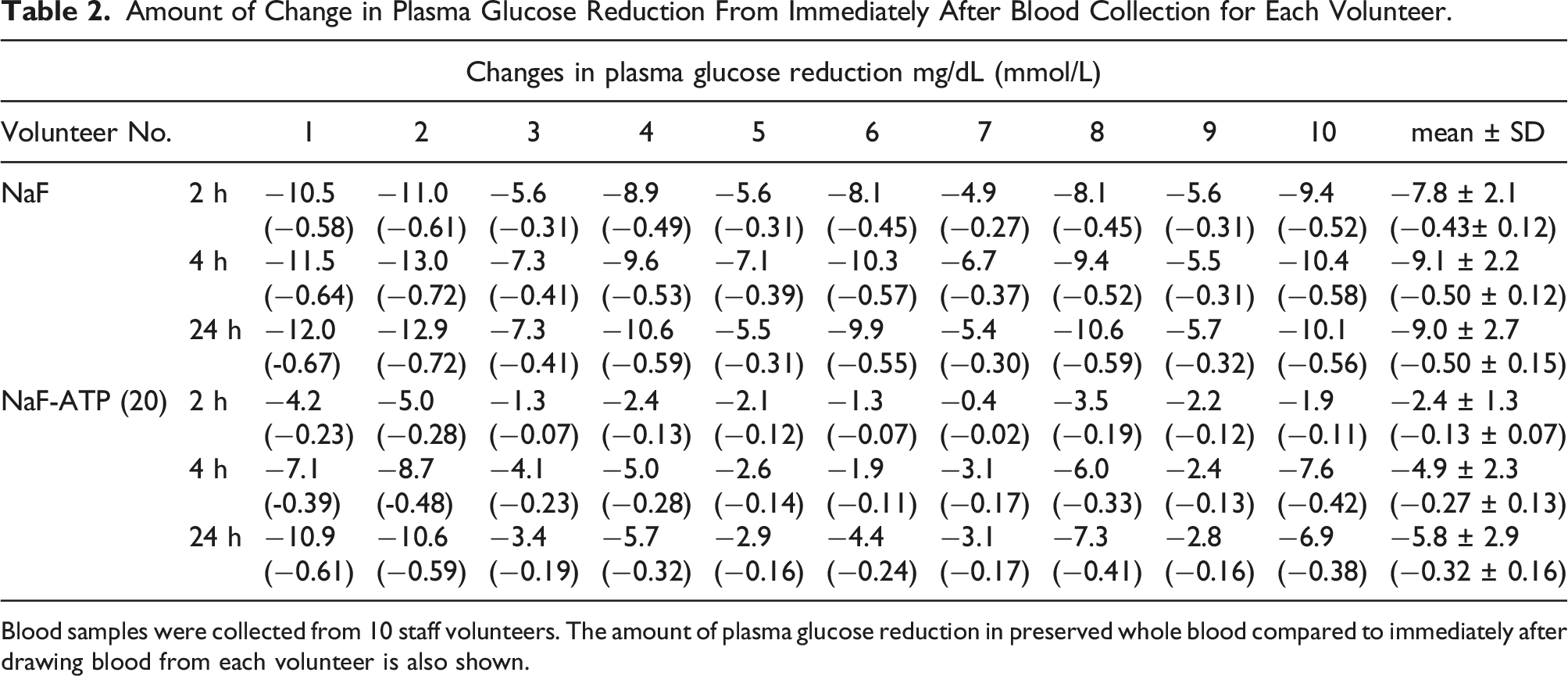
Blood samples were collected from 10 staff volunteers. The amount of plasma glucose reduction in preserved whole blood compared to immediately after drawing blood from each volunteer is also shown.
Initial Plasma Glucose Concentration for Each Volunteer.
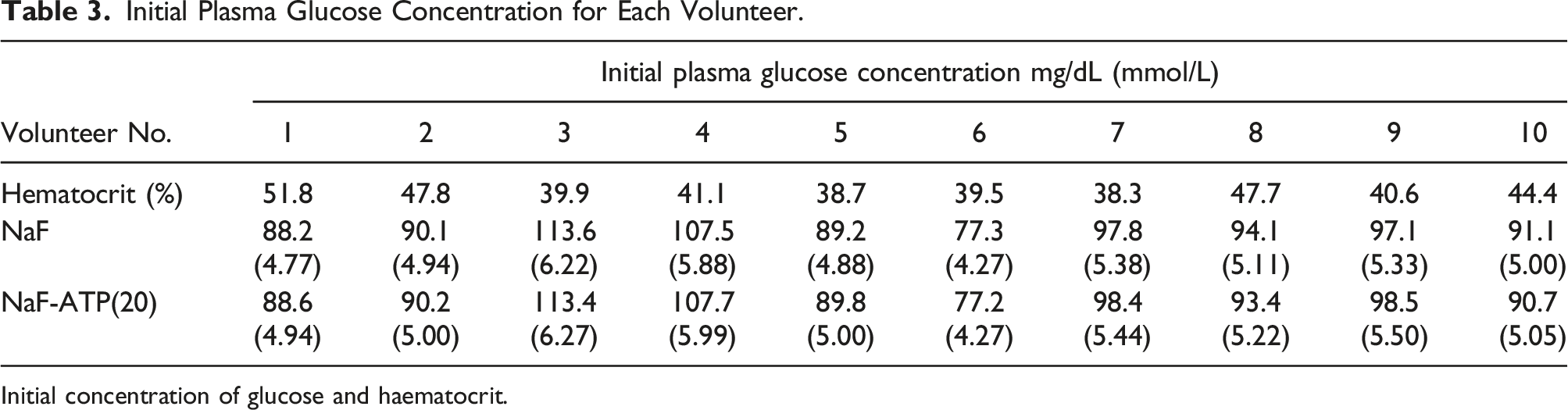
Initial concentration of glucose and haematocrit.
Stability of HbA1c in the stored blood collection tubes after sampling
The Dunnett’s test for HbA1c storage stability in whole blood from 10 volunteers was performed using the 0-h EDTA tube as a control. After 24 h of storing whole blood in the various tubes, no significant differences were seen among the HPLC, enzymatic, and immunoassay methods (Figure 2). Storage stability of HbA1c in various blood collection tubes. Blood samples were collected from 10 staff volunteers. Whole-blood collection tubes were stored at room temperature (25 ± 0.5°C) for up to 4 h and refrigerated (4°C) for 4–24 h. Blood samples were compared for HbA1c values immediately after collection in EDTA tubes (EDTA-0 h) and after 24 h of storage in various blood collection tubes (EDTA, NaF, NaF-–ATP(15), and NaF–ATP(20)). a) HPLC method; b) enzyme method; c) immunoassay method. Dunnett’s test was used for statistical processing.
Changes in glucose concentration in patient whole blood
Changes in Glucose Concentration in Patient Whole Blood.
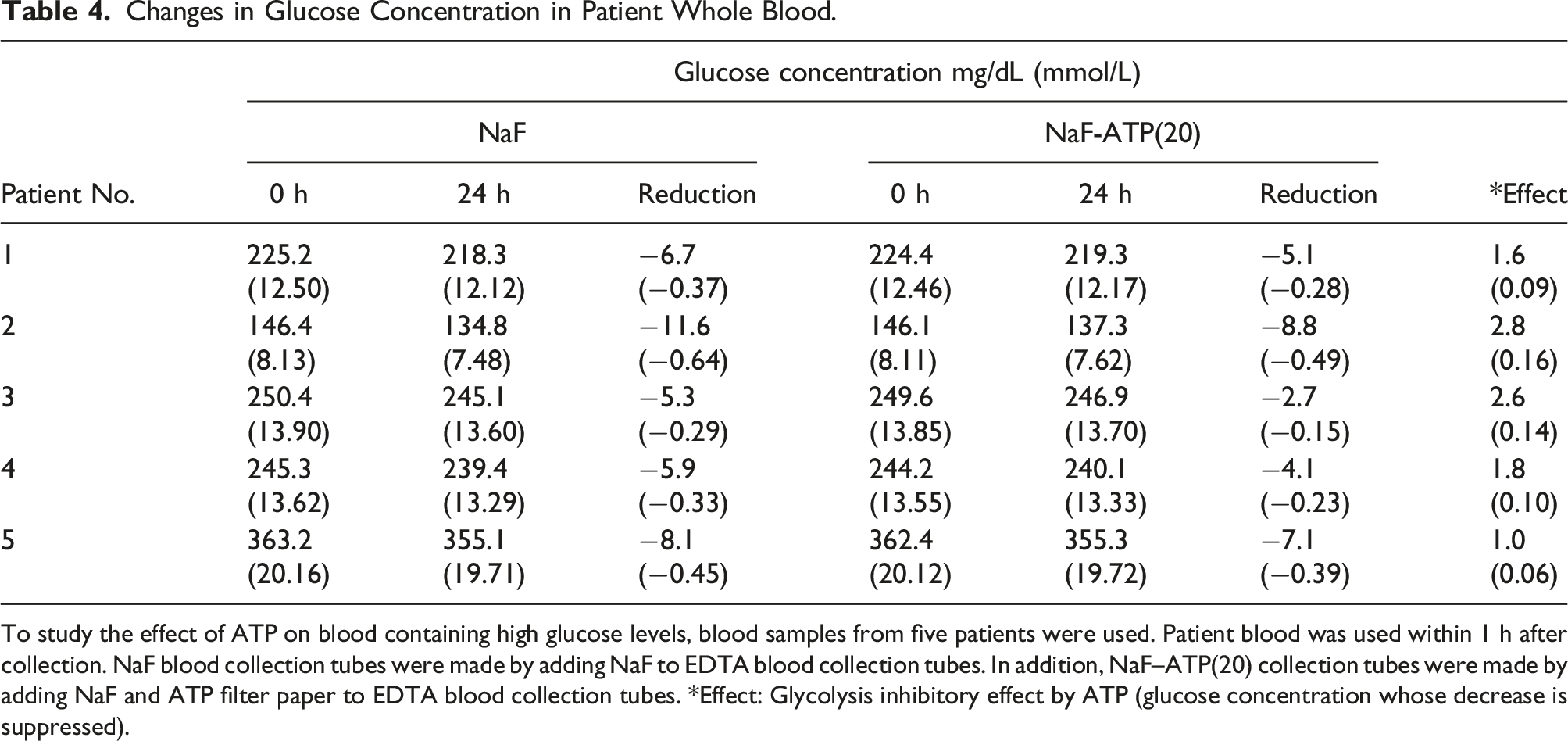
To study the effect of ATP on blood containing high glucose levels, blood samples from five patients were used. Patient blood was used within 1 h after collection. NaF blood collection tubes were made by adding NaF to EDTA blood collection tubes. In addition, NaF–ATP(20) collection tubes were made by adding NaF and ATP filter paper to EDTA blood collection tubes. *Effect: Glycolysis inhibitory effect by ATP (glucose concentration whose decrease is suppressed).
Discussion
ATP and ADP were found to have a concentration-dependent effect on the storage stability of glucose whole blood. Blodgett et al. reported that ATP directly inhibits GLUT1 by promoting pronounced GLUT1 conformational changes (Blodgett DM). 5 This structural change is thought to suppress glucose uptake. Matuo et al. reported that ADP in addition to ATP inhibited GLUT1 (Matuo S). 7 However, ADP was approximately four times more expensive than ATP and was more haemolytic. Therefore, ATP is more suitable than ADP for blood collection tubes. A total of 20 mg or more of ATP should be added, and the amount of ATP added should be adjusted according to the usage conditions (time between blood collection and measurement) at each facility. In addition, the glucose level immediately after blood sampling did not change with the amount of ATP added, suggesting that ATP does not affect glucose measurement. In some cases, blood is stored at the bedside for up to 4 h after collection before being submitted to the laboratory. Conventional NaF tubes have been reported to result in lower measured blood glucose levels up to 4 h after blood collection under room temperature storage, after which the levels stabilized. 1 Therefore, it was considered important to limit the degradation of glucose in NaF blood collection tubes during the first 4 h of storage at room temperature. In addition, guidelines recommend that blood be plasmaized immediately after collection or otherwise stored at low temperature. 2 Furthermore, considering the possibility of simultaneous measurement of HbA1c, it is advisable to store whole blood in a refrigerator if it is to be stored for more than 4 h. NaF had a weak inhibitory effect on glucose depletion in the initial stage, 2 h after blood collection, suggesting that approximately 4 h were required for the inhibitory effect to become apparent. On the other hand, in the ATP(20) tube, the decrease in glucose was slower after 4 h than in the NaF tube and tended to decrease gradually until 24 h. This may be the result of glucose transporter (GLUT1) inhibition on the erythrocyte membrane or allosteric inhibition of phosphofructokinase (PFK), an enzyme that catalyzes the third step of glycolysis, but further study is needed. Individual differences were also observed in the decrease in glucose levels in preserved whole blood. Since these differences are associated with Hct, it was considered that the number and metabolism of red blood cells are related to this. However, despite individual differences, the mean decrease in glucose was higher in the sample containing NaF and ATP than that with NaF alone, suggesting that ATP exerted sufficient sustained antiglycolytic activity from immediately after blood collection to 24 h. The effect of ATP was also confirmed in the whole blood of patients who showed hyperglycaemia. However, since the results of this study were obtained using blood from patients 1 h after collection, the effects of ATP may not be fully reflected in them. In normal use, glycolysis can be inhibited from the early stage, so it is thought that the effect is a little stronger. The stability of HbA1c in whole-blood storage did not differ significantly between EDTA tubes and NaF tubes containing ATP, and stability was confirmed up to 24 h. In addition, because NaF alone causes changes in glucose over time, blood collection tubes containing D-mannose have been reported as a countermeasure,9,10 but problems with D-mannose interfering with some glucose measurement systems have also been reported.11,12 In addition, FC tubes are highly effective in reducing decreases in glucose over time 13 but are unsuitable for HbA1c measurement because citric acid is not easily soluble in blood, resulting in haemolysis. Based on the above results, in this study, we developed a blood collection tube that overcomes the drawbacks of conventional blood collection tubes.
Conclusion
In this study, we developed a blood collection tube capable of facilitating measurement of glucose and HbA1c by adding ATP to NaF. In addition, its ability to stably store whole blood was improved over conventional tubes, and the glucose decrease over time up to 4 h of storage at room temperature, which is a problem with conventional blood collection tubes, was greatly reduced.
Supplemental material
Supplemental Material - Development of blood collection tubes for glucose measurement using adenosine 3-phosphate and sodium fluoride as glycolytic inhibitors
Supplemental Material for Development of blood collection tubes for glucose measurement using adenosine 3-phosphate and sodium fluoride as glycolytic inhibitors by Yukio Kume, Yuji Hirowatari, Makoto Kurano, Yutaka Yatomi, and Makoto Matushita in Annals of Clinical Biochemistry.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by a joint research grant from the University of Tokyo and Nipro Corporation.
Ethical approval
This study was approved by the Ethics Committee of the Graduate School of Medicine at University of Tokyo (approval no. 2020063NI).
Guarantor
MM.
Contributorship
YK and MK designed the study. MM and YY supervised the study. YK carried out the research. YK and MK analysed the data. YK and MK wrote the manuscript. MM and YH reviewed drafts of the manuscript. All authors accept responsibility for the entire content of this manuscript and approved its submission.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
