Abstract
Background
A sample received in the laboratory from a patient receiving total parenteral nutrition (TPN) indicated that the patient may have renal dysfunction, but the results were not considered to be reliable enough to report. Investigations using a reference method for measurement of creatinine confirmed positive interference in the creatinine assay and distribution of samples via an External Quality Assessment (EQA) Scheme showed that this positive interference was method dependent.
Methods
Residual TPN fluid (Nutriflex Lipid Special) left in the bag after the patient had completed the infusion was collected and added to a patient serum pool in increasing amounts and distributed to different laboratories for analysis of creatinine and glucose through an EQA Scheme.
Results
Positive interference in a number of different creatinine assays was identified as a result of a component in the TPN fluid. Positive interference from high concentrations of glucose has been demonstrated to be a cause for falsely high results in Jaffe creatinine assays.
Conclusions
The concern would be that a sample contaminated with TPN fluid would have both abnormal electrolytes and creatinine concentrations and give the impression that the patient was in renal failure due to analytical interference in the creatinine assay and laboratory staff need to be aware of this problem.
Introduction
Laboratory results obtained on the contaminated sample.
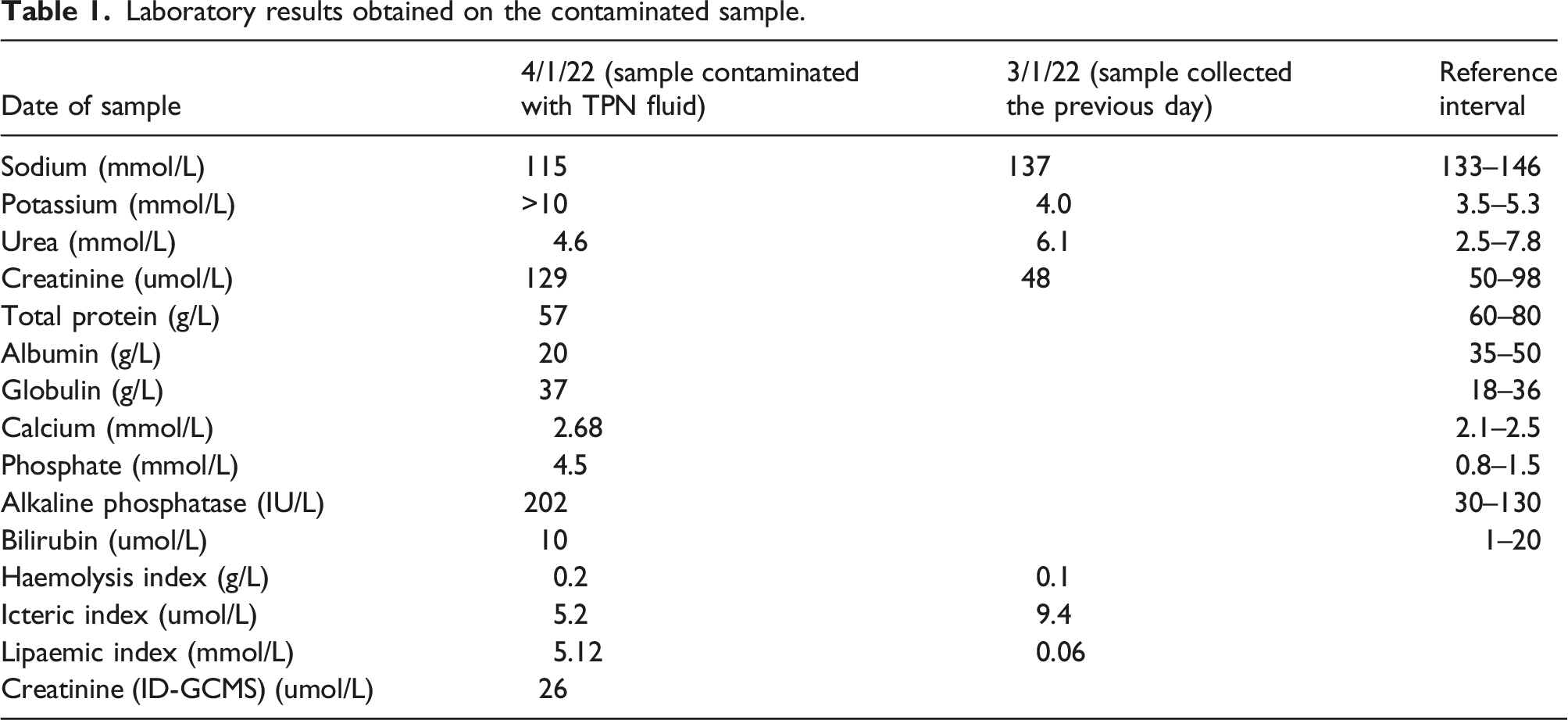
In order to confirm that the increase in creatinine was due to a component of the TPN fluid, the residual TPN fluid was collected after the infusion was completed and the fluid was added to a patient serum pool in increasing amounts using 50 mmol/L aqueous solutions of either ammonium acetate or acetic acid as separate control solutions as these are not known to cause interference in creatinine assays. To investigate whether this interference would affect different manufacturer and different methods employed for creatinine assays, the TPN fluid was added to pooled human serum and distributed via the United Kingdom National External Quality Assessment Services (UK NEQAS) for Clinical Chemistry EQA Scheme. The interference from glucose on creatinine assays has been demonstrated previously by the addition of glucose to samples distributed within Distribution 172 (February 2021) in the UK NEQAS for Acute and Chronic Kidney Disease (known at the time as UK NEQAS for GFR Estimations Scheme).
Materials and methods
TPN Fluid
Total parenteral nutrition fluid (Nutriflex lipid special) was obtained from the clinical team looking after the patient after the infusion had been completed. The TPN fluid contains the following active ingredients per 1000 mL solution: isoleucine 3.28 g, leucine 4.38 g, lysine hydrochloride 3.98 g (= 3.18 g lysine), methionine 2.74 g, phenylalanine 4.92 g, threonine 2.54 g, tryptophan 0.80 g, valine 3.60 g, arginine 3.78 g, histidine hydrochloride monohydrate 2.37 g (=1.75 g histidine), alanine 6.79 g, aspartic acid 2.10 g, glutamic acid 4.91 g, glycine 2.31 g, proline 4.76 g, serine 4.20 g, glucose monohydrate 158.40 g (=144 g anhydrous glucose), soya bean oil 20 g, medium chain triglycerides 20 g, sodium dihydrogen phosphate 2.496 g, sodium hydroxide 1.171 g, sodium chloride 0.378 g, sodium acetate trihydrate 0.25 g, potassium acetate 3.689 g, magnesium acetate tetrahydrate 0.91 g, calcium chloride dihydrate 0.623 g, zinc acetate dihydrate 7.02 mg. Total amino acids 57.6 g, total nitrogen 8 g, total glucose 144 g (800 mmol), total lipids 40 g, osmolality 2090 mmol/kg, Na+ 53.6 mmol, K+ 37.6 mmol, Ca++ 4.24 mmol, Mg++ 4.24 mmol, Zn++ 0.032 mmol, Cl-48 mmol, H2PO4 16 mmol and acetate 48 mmol.
Analytical assays
Creatinine was measured by two assay principles: (1) Compensated Kinetic Jaffe and (2) Enzymatic.
The Abbott Architect Jaffe creatinine assay is a kinetic spectrophotometric assay. At an alkaline pH, creatinine in the sample reacts with picric acid to form a creatinine-picrate complex that absorbs at 500 nm. The rate of increase in absorbance is directly proportional to the concentration of creatinine in the sample.
The Abbott Architect glucose assay is an end-point hexokinase/glucose-6-phosphate dehydrogenase spectrophotometric assay which measures the generation of NADH at 340 nm.
A spiked sample was also analysed using an enzymatic creatinine assay (Radiometer). The Radiometer blood gas enzymatic creatinine assay uses three sensors; a 2-sensor system for detecting creatine and a third sensor for detecting both creatine and creatinine, and the creatinine value is obtained by a difference measurement. The electrodes consist of an internal silver/silver chloride reference electrode, a platinum cathode and a platinum anode. The enzyme layer over the electrodes contains creatinase and sarcosine oxidase for the 2-enzyme sensor with the addition of creatininase enzyme for the 3-enzyme sensor. This enzyme layer is covered by an outer membrane which is permeable to creatinine. Creatinine is converted to creatine by the action of creatininase. Creatine is converted to sarcosine by the action of creatinase and sarcosine is converted to glycine and formaldehyde with the release of H2O2 by sarcosine oxidase. The generation of H2O2 is then measured at the platinum anode and converted into a current which is measured by the analyser.
Preparation and analysis of spiked serum samples in Derriford
All chemicals were purchased from Merck Life Science UK Limited, Gillingham, Dorset, unless otherwise stated. A 50 mmol/L ammonium acetate solution was prepared by the addition of 0.19 g ammonium acetate (H3CC00NH4, Mw 77.08) to 50 mL deionised H2O. A 50 mmol/L acetic acid solution was prepared by the addition of 150 uL 99–100% glacial acetic acid (C2H4O2, Mw 60.05) to 50 mL deionised H2O. Residual patient serum from samples referred to the laboratory for routine analysis was pooled and the same pool of serum was used throughout all of this work in Derriford. Five samples were prepared containing TPN fluid as follows: 500 uL serum pool (0% contamination), 450 uL serum pool + 50 uL TPN fluid or acetate solution (10% contamination), 250 uL serum pool + 250 uL TPN fluid or acetate solution (50% contamination), 50 uL serum pool + 450 uL TPN fluid or acetate solution (90% contamination), 500 uL TPN fluid or acetate solution (100% contamination). A range of samples with final concentration of 0–50% contamination were prepared by the addition of 250 uL of each of these spiked solutions to 250 uL of the serum pool. Samples were analysed in singlicate in one batch on the same Abbott Architect analyser (creatinine reagent lot number 02564UN21^10558, glucose reagent lot number 59672UQ04^24010). The 5% final solution (obtained from a 1:1 dilution of the 10% contamination solution) was referred to WEQAS Reference Measurement Laboratory for measurement of creatinine by ID-GCMS and was also analysed using a Radiometer ABL800 Flex blood gas analyser (Radiometer UK Ltd., Crawley).
Preparation of spiked samples by UK NEQAS
Total parenteral nutrition fluid (Nutriflex Lipid Special), as described previously, was provided by Derriford Combined Laboratory. Three pools were prepared, namely, Pool 706, which did not contain any TPN fluid, Pool 707, which contained 3.4% TPN fluid, and Pool 708, which contained 6.8% TPN fluid. Pools 706 and 707 were corrected for dilution by the addition of Pharmacy grade water. These three Pools were frozen at −40°C, prior to being distributed as Specimens 1122A, 1122B and 1122C, respectively, to all participants in the UK NEQAS for Clinical Chemistry Scheme in March 2022. Over 680 participants, worldwide, but mainly UK, received specimens and had 2 weeks to analyse the specimens and report results for all analytes that the participant was registered for in the UK NEQAS for Clinical Chemistry Scheme. Data was reviewed by Birmingham Quality and method means and standard deviation were calculated for each analyte after Healy Trimming. 1
Results
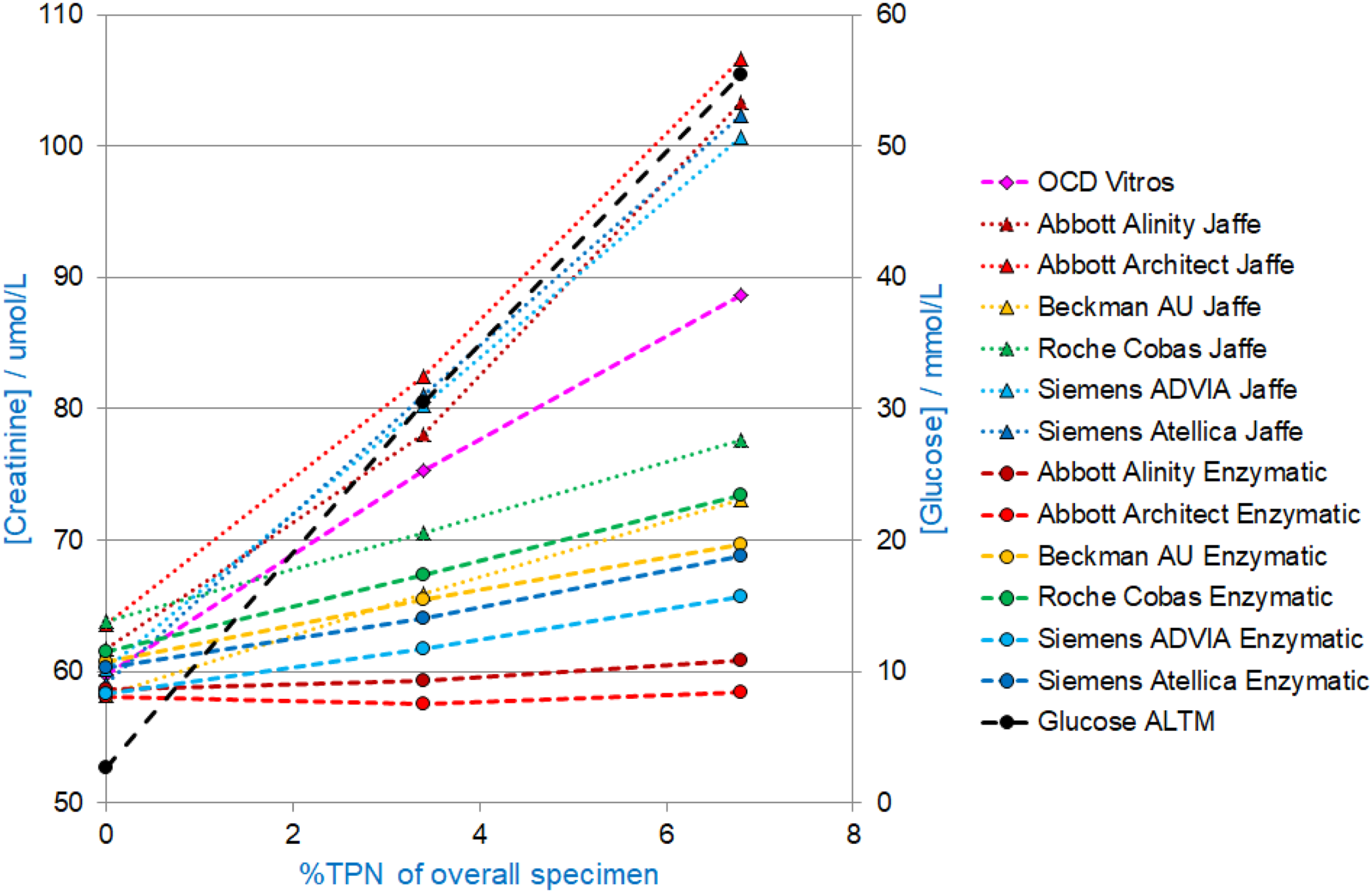
The results obtained from the Abbott Architect in the laboratory at Derriford, ID-GCMS and Radiometer blood gas analyser for the sample with 5% contamination with TPN were as follows: Creatinine (Abbott Architect) 96 umol/L, creatinine (ID-GCMS) 66 umol/L and creatinine (Radiometer ABL800 Flex blood gas) 54 umol/L. Glucose (Abbott Architect) 44.1 mmol/L and glucose (Radiometer ABL800 Flex blood gas) 41 mmol/L. The creatinine results obtained on the other spiked samples are shown in Figure 1. Figure 1 shows the results from work carried out in Derriford. The glucose results were unmeasurable for the samples spiked with 45% or 50% TPN fluid which contains 800 mmol/L glucose. Figure 2 shows the results obtained on samples distributed with UK NEQAS for Clinical Chemistry scheme. Creatinine and glucose concentrations measured on pooled serum with increasing amounts of added TPN fluid or acetate (in the form of 50 mmol/L ammonium acetate or acetic acid – both lines are superimposed on the graph). Creatinine (primary y-axis) and glucose (secondary y-axis) concentrations on specimens with added TPN Fluid distributed to all participants in the UK NEQAS for Clinical Chemistry Scheme, Distribution 1122 March 2022. (Total number of participants n = 680).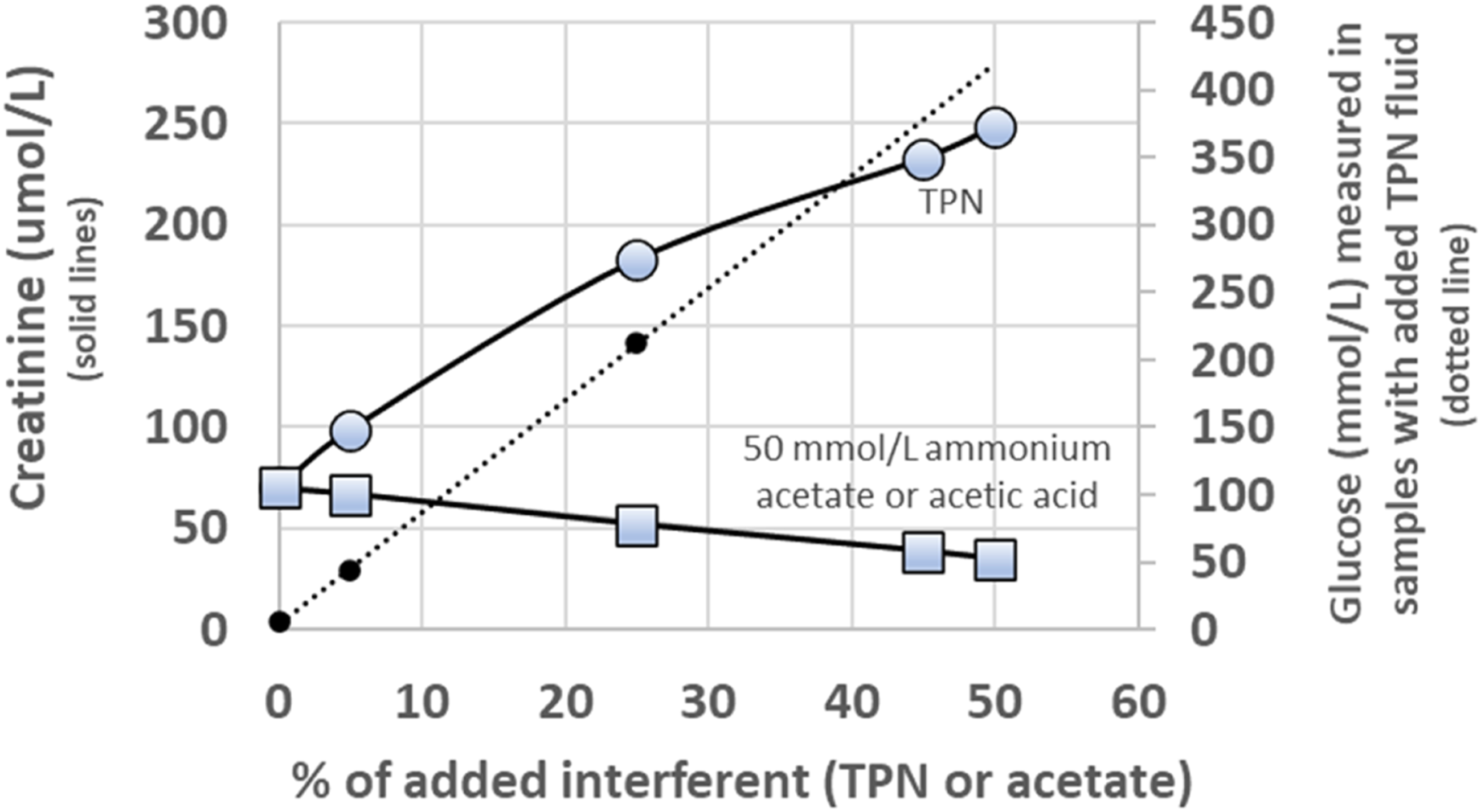
It can be seen from Figure 1 that with increasing amounts of TPN fluid contamination, the creatinine concentration measured by the Abbott Architect Jaffe assay increased. This increase was not seen in the control samples containing increasing amounts of acetate. The control samples showed the expected decrease in creatinine as a consequence of sample dilution.
Figure 2 shows that there is an increase in creatinine with increasing amounts of TPN fluid for both the Compensated Kinetic Jaffe and Enzymatic method principles, for all manufacturers except the Abbott Alinity and Architect Enzymatic assays. Conversely, the Abbott Alinity Jaffe and Abbott Architect Jaffe assays, along with the Siemens ADVIA Jaffe and Siemens Atellica Jaffe are most affected by interference from the TPN fluid.
Discussion
It is of significant concern that a sample contaminated with TPN fluid would show abnormal electrolytes and creatinine and could give the impression that a patient was in renal failure. The proportion of TPN fluid to serum could probably be quite variable, the patient sample shown in Table 1 above was fairly obviously contaminated. The algorithm for detecting AKI based on serum creatinine changes with time, relating to the NHS England patient safety alert (NHS/PSA/D/2014/010), identifies an increase in creatinine of more than 26 umol/L within a 48-h period to be flagged as AKI stage 1 with larger increases in creatinine associated with more severe stages of AKI. 2
The control samples spiked with ammonium acetate or acetic acid did not cause any interference with the creatinine assay, although ammonium acetate did have the expected effect on urea measured using the Abbott Architect photometric urease assay, 3 results not shown. There is published work describing interference in the Vitros creatinine assay from acetic acid, 4 but this is thought likely to be due to pH adjustment of the sample rather than the presence of acetate per-se.
It is clear that glucose in the TPN fluid is contributing, at least in part, to the positive interference in creatinine Jaffe assays. This is supported by data shown in the UK NEQAS for Acute and Chronic Kidney Disease Scheme, Distribution 172 (Figure 3) where glucose was added to serum specimens in different concentrations. The Abbott Alinity/Architect and Siemens ADVIA Jaffe reagents are the most affected. The interference was less significant for the enzymatic creatinine methods (and also the Beckman modified Jaffe). However, some of the creatinine enzymatic assays also show a positive interference in creatinine with increasing concentrations of TPN fluid. This could be due to other components of the TPN fluid or because the specimens are lipaemic. Glucose interference in creatinine assays from the UK NEQAS for Acute and Chronic Kidney Disease Scheme (known at the time as UK NEQAS for eGFR Estimations), Distribution 172 February 2021. (Total number of participants n = 442).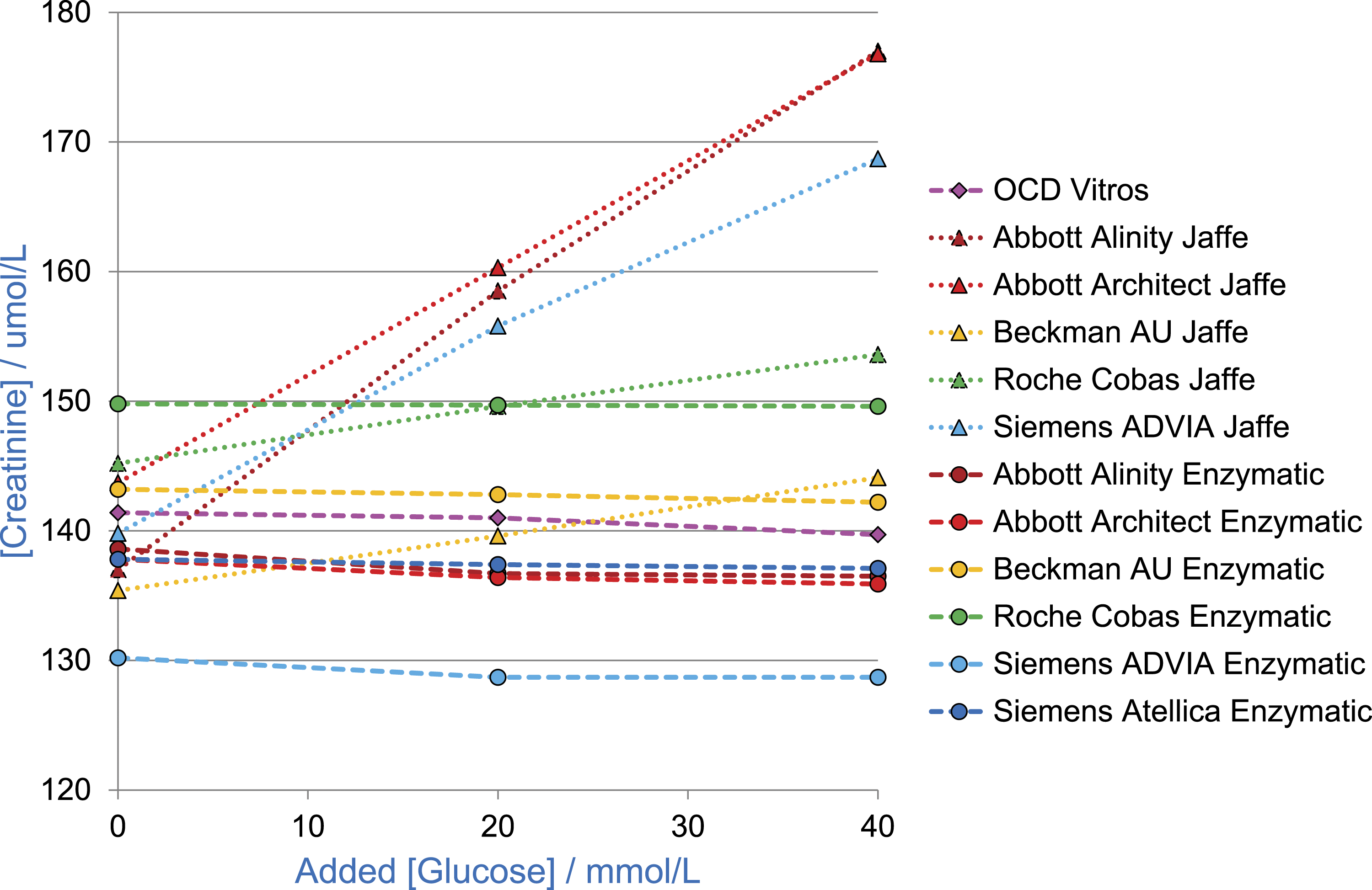
A number of other compounds are known to interfere in Jaffe creatinine assays including ketones, 5 bilirubin, pyruvate,6,7 5-aminolevulinic acid, 8 nitromethane,9,10 eltrombopag, 11 antibiotics 12 and metabolites from methanol and isopropanol ingestion. 13 Not all TPN fluids contain the same ingredients. An overview of intravenous nutrition is provided in the British National Formulary. 14
Interference from turbidity is also a possibility, given that the name of the TPN solution includes the term ‘Lipid Special’. Irrespective of the name, however, most TPN solutions contain approximately 40 g/L lipids to meet energy requirements (25%–30% of the total calories from lipids). This turbidity may affect several assays in either a positive or negative direction. For the data shown in Figure 1, there were lipaemic index flags for creatinine and glucose on the 45% and 50% spiked samples (the lipaemic indices for these two samples were 9.15 and 10.45 and the lipaemic index cut-offs above which lipaemia causes significant interference in the creatinine and glucose assays are 7.50 and 10.00 mmol/L, respectively), but the 5 and 25% samples were not flagged as lipaemic. There was only a relatively modest lipaemic index of ∼2.3 mmol/L (equivalent to ∼200 mg/dL or a category somewhere between 1 and 2 depending on manufacturer) for the UK NEQAS samples (Figure 2).
There are not many published studies relating to analytical interference in laboratory tests as result of sample contamination with TPN fluid. A comprehensive review from 1987 on the subject of TPN in relation to the clinical chemistry laboratory mentions the possibility of physical contamination of the membranes of ion-selective electrodes and interference in Coulter counters, 15 and there has been an article discussing the effect of lipaemia on laboratory tests which describes negative interference in some creatinine assays. 16 One recent article has looked at the effect of fish-oil-based lipid emulsion and concluded that at the concentrations typically found in TPN fluid, there may not be any interference with chemistry analytes, but the higher concentrations of lipids given as rescue therapy for the treatment of drug overdoses will interfere. 17 There is also a recent case report describing interference in a plasma amino acid profile for a child on TPN. 18
One limitation of this study is that many different TPN preparations exist and, in absence of experimental evidence with different TPN solutions, it would be unreasonable to assume that all TPN preparations will cause interference of a similar magnitude.
Conclusion
If an increase in creatinine is noted for a patient receiving TPN infusions, then it would be sensible to check that the patient did not have an active TPN infusion at the time that the sample was collected. Grossly lipaemic samples will be identified in the lab from the lipaemic index, but a lower level of contamination of a sample with TPN fluid could be more difficult to detect. The concern would be that the contaminated sample would have abnormal electrolytes and creatinine and give the impression that the patient was in renal failure. For a quick check in the laboratory, serum glucose could be measured on any samples for which contamination with TPN fluid is suspected.
Footnotes
Acknowledgements
Dr Paula Murphy (Nutrition Support Team Leader Dietician), Lisa Cripps, Steve Edwards (ATO, pharmacy), Jonathan Turton (pharmacist), David Ducroq (Head of the Reference Laboratory WEQAS).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported in part by the provided by Derriford Combined Laboratory for work undertaken by Derriford Combined Laboratory.
Ethical approval
The samples which were used for this study were surplus to requirements and all patient identifiable data was removed before pooling human serum.
Guarantor
JJ.
Contributorship
JJ wrote the article, HM performed the laboratory analysis, FM and RM prepared, distributed samples and reported the results obtained from the UK NEQAS scheme and RM and RG reviewed the final manuscript.
