Abstract
Aims
To explore differences in B-type natriuretic peptide (BNP) concentration and stability and evaluate BNP accuracy in different collection tubes.
Methods
BNP concentrations in heparin/glass, EDTA/glass, and EDTA/polyethylene terephthalate (PET) tubes were measured on the Mindray CL-6000i at 0.5, 1, 2, and 4 h after collection. Differences were evaluated using Wilcoxon’s paired tests and Bland–Altman plots. BNP stability and measurement accuracies were estimated using Kruskal–Wallis H tests and recovery tests.
Results
BNP concentrations in EDTA/glass tubes were 31.4% higher than those in heparin/glass tubes and 3.04% lower than those in EDTA/PET tubes. BNP stability significantly decreased in the heparin/glass tube. BNP remained stable in EDTA/glass and EDTA/PET tubes at room temperature for 4 h. BNP recovery rates in heparin/glass, EDTA/glass, and EDTA/PET tubes were 77.46, 86.04, and 88.23%, respectively.
Conclusions
Plasma in EDTA/glass and EDTA/PET tubes is suitable for BNP measurement on the Mindray CL-6000i.
Introduction
Heart failure (HF) is a clinical syndrome caused by structural and/or functional cardiac abnormalities that impair ventricular systolic and/or diastolic function. 1 The prevalence of HF in China in 2003 is 0.9%, 2 and a study 3 showed that the in-hospital mortality of patients with HF was 4.1%. In developed countries, the prevalence of HF is approximately 1–2%, which rises to ≥10% in people >70 years old. 4 The incidence and prevalence of HF continue to increase annually, and HF has become the main contributor to cardiovascular disease-related mortality. 5
B-type natriuretic peptide (BNP) was first isolated from porcine brains in 1988 6 and belongs to the natriuretic peptide family that also includes atrial natriuretic peptide (ANP) and C-type natriuretic peptide (CNP). BNP is a polypeptide composed of 32 amino acids which is mainly produced and secreted by myocytes and fibroblasts in the left ventricle in response to increased cardiac wall stress and filling pressure.7,8 Over the recent decades, the role of BNP in heart failure has attracted increasing attention. It is an important biochemical marker for diagnosing HF and can be used to rule out or confirm HF among suspected patients in acute settings. 9 BNP testing is useful in guiding therapeutic regimen design 10 and evaluating prognosis. 11 BNP is also valuable in screening for cardiac dysfunction 12 and the risk stratification of heart failure.10,13 Therefore, accurate testing of BNP concentration is crucial.
In our hospital, plasma was collected into heparin/glass tubes and BNP was detected using the Mindray CL-6000i chemiluminescence immunoanalyzer. The physicians found that BNP results were sometimes low and inconsistent with clinical manifestations. BNP measurements can be easily affected by pre-analytical factors. It was reported that BNP in whole blood and plasma samples was more unstable in EDTA/glass tubes than in EDTA/polyethylene terephthalate (PET) tubes.14,15 Other studies showed that anticoagulants could affect BNP measurements.16,17 IFCC Clinical Committee on Cardiac Biomarkers suggested that EDTA plasma was the only suitable specimen for BNP assays. 18 However, the effects of heparin and EDTA on BNP concentration were conflicting between the Access 2 (Beckman Coulter, CA, USA) and ADVIA Centaur (Siemens Diagnostics, NY, USA) immunoanalyzers.16,17 It was reported that BNP in heparin plasma was more stable than that in EDTA plasma based on the Access 2 system. 17 Although the reagent instructions of the Mindray CL-6000i suggested that BNP should be detected in EDTA/PET tubes, no data demonstrated whether or how much BNP concentrations were affected by the collection tube materials (glass or PET) and/or the anticoagulants used (heparin or EDTA) when using the Mindray CL-6000i.
This study aimed to investigate the differences in plasma BNP concentrations, explore the stability of BNP at room temperature, and evaluate the accuracy of BNP in heparin/glass, EDTA/glass, and EDTA/PET tubes when using the Mindray CL-6000i. We aimed to determine the optimal collection tube type for BNP measurement using the Mindray CL-6000i.
Materials and methods
Participants
Samples were collected from inpatients who required BNP detection at the Department of Cardiology, Jinzhai County People’s Hospital, in January 2021. A total of 55 inpatients were selected and screened according to the exclusion criteria. Exclusion criteria were as follows: (1) specimens with haemolysis, lipaemia, or jaundice; (2) patients whose results were outside of the linear range; or (3) had missing data. Finally, 36 inpatients were included in the analysis.
Four healthy volunteers were recruited. Blood samples from these volunteers were used for recovery tests of the BNP reference material in different collection tubes.
The study was approved by the Ethics Committee of the Jinzhai County People’s Hospital (Approval No. 2021001), and written informed consent was obtained from all participants.
Study design and sample collection
After 8 h of fasting, blood samples from each participant were simultaneously collected into lithium heparin glass (heparin/glass), EDTA/glass, and EDTA/PET tubes. All the tubes were manufactured by Uivers Medical Technology Co., Ltd (Hunan, China). Centrifugation at room temperature at 2000 g for 10 min was performed immediately, and plasma BNP concentrations were measured at 0.5, 1, 2, and 4 h after sample collection.
Recovery test of BNP reference material
Venous blood samples from four healthy volunteers were simultaneously collected into heparin/glass, EDTA/glass, and EDTA/PET tubes. BNP concentrations of these plasma samples were measured, and the results were all <10 ng/L. A total of 400 μL BNP reference material (3734 ng/L) (Mindray Bio-Medical Electronics Co., Ltd.) was added to 346.8 μL plasma from healthy volunteers for a final concentration of 2000 ng/L, and BNP was immediately measured twice again. The recovery rate was calculated as follows: (average BNP concentration/2000) *100%.
BNP measurement
BNP was measured with a double-sided sandwich enzymatic chemiluminescent assay on a CL-6000i automatic chemiluminescence immunoanalyzer (Mindray Bio-Medical Electronics Co., Ltd., Shenzhen, China). The capture antibody is a mouse monoclonal antibody directed to the C-terminal part of the peptide (amino acids 26–32). The ALP-conjugated detection antibody is also a mouse monoclonal antibody against the ring portion of BNP (amino acids 11–17). The original matched BNP reagents (Lot number: 200501), calibration solution (Lot number: 200201), and quality controls (Lot number: 190901) were used. All procedures were performed according to instrument and reagent instructions. The performance of the Mindray CL-6000i immunoanalyzer for BNP detection was verified prior to the study. This included checking for precision, accuracy, linearity, and the reportable clinical range. The intra-assay precisions of BNP in low-value and high-value samples were 1.43% and 0.85%, respectively. The inter-assay precisions in low-value and high-value samples were 2.70% and 3.52%, respectively. The accuracy verification showed that the relative biases of the measured BNP concentrations were −2.5% at 230.11 ng/L and −0.2% at 1793.12 ng/L. The linear range of BNP was 10–5000 ng/L.
Statistical analysis
GraphPad 7.0, SPSS 17.0, and PASS 2022 were used to perform statistical analysis. Data were expressed as mean ± SD or median [Q1, Q3]. Differences between the two groups were analyzed using Wilcoxon matched-pairs signed rank tests and Bland–Altman plots. Differences among multiple groups were estimated using Kruskal–Wallis H tests. The sample size calculation with 90% power at two-sided alpha level of 0.05 revealed that 14 patients were necessary. p < 0.05 was considered statistically significant.
Result
Participant selection
Of the 55 inpatients, 19 were excluded from this study. Of these excluded patients, one had lipaemia, twelve had BNP results <10 ng/L, one had a BNP result >5000 ng/L, and five were sent to the clinical laboratory late and did not have results for the 0.5 h time point. Eventually, 36 subjects (16 females and 20 males) were included in the analysis. The median [Q1, Q3] ages were 71.5 [66, 79] years in female participants and 76 [68, 81] years in males. According to the BNP results in the EDTA/PET tube, there were eight cases with BNP <100 ng/L, sixteen with BNP ranging between 100 and 500 ng/L, and twelve with BNP >500 ng/L.
Comparison of measured BNP concentration in different collection tubes
BNP concentrations in heparin/glass tubes ranged from a minimum value of 11.62 ng/L to a maximum value of 1341.47 ng/L. They ranged from 14.63 to 1684.51 ng/L in EDTA/glass tubes and 16.68 to 1925.46 ng/L in EDTA/PET tubes. As shown in Figure 1, BNP concentrations in EDTA/glass tubes were significantly higher than that in heparin/glass tubes (median [Q1, Q3] = 316.63 [131.51, 724.60] ng/L vs. 219.21 [89.3, 451.18] ng/L, p < .001). Compared with EDTA/glass tubes, BNP concentration is higher in EDTA/PET tubes (289.87 [134.35, 744.95] ng/L, p = .015). Paired comparisons of BNP concentrations in different collection tubes. *p < .05; ***p < .001.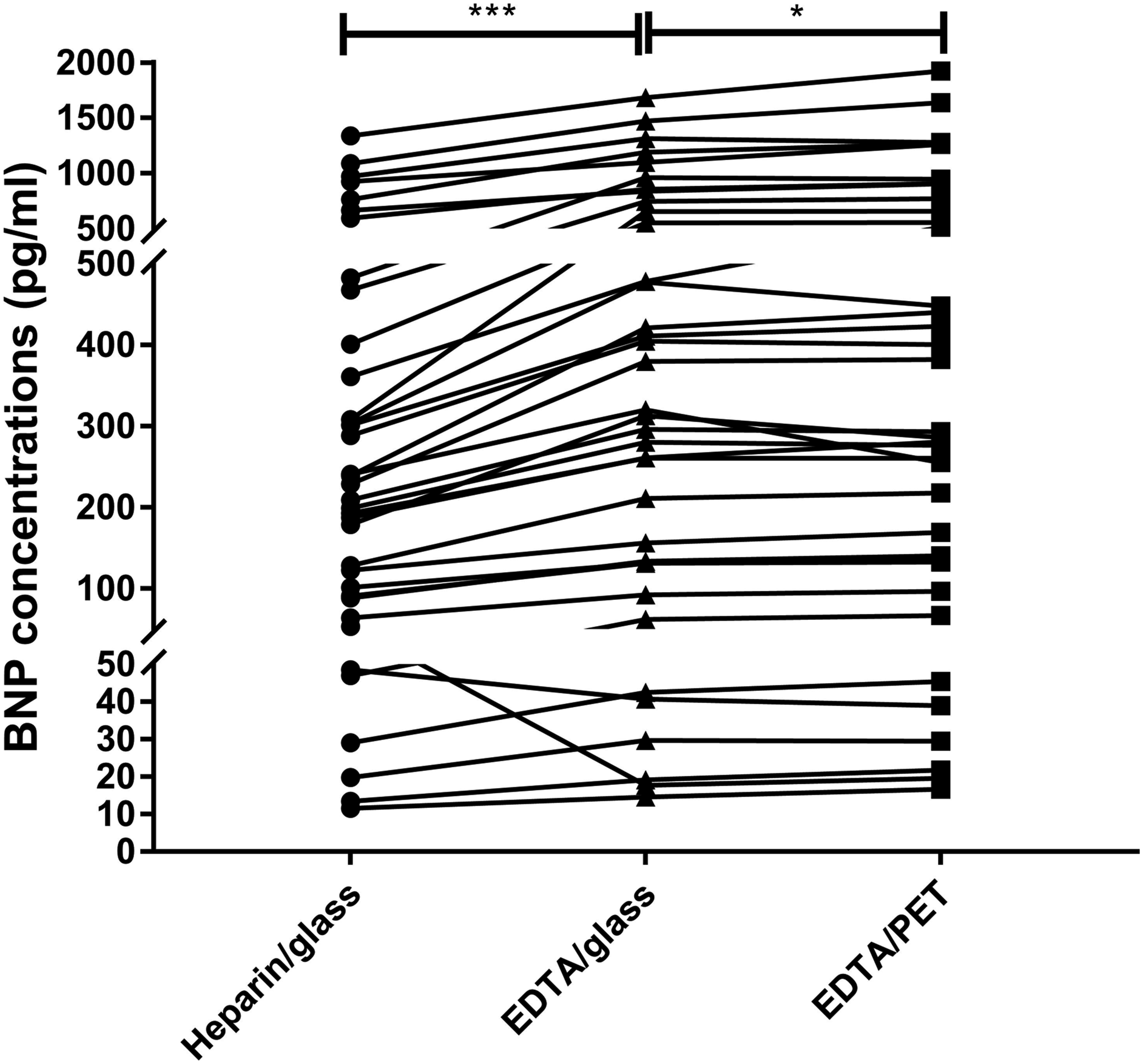
Bland–Altman plots were used to evaluate the difference among different collection tubes. The mean (95% confidence interval) of the difference was 31.4% (−21.6 and 84.5%) between EDTA/glass and heparin/glass and 3.04% (−10.8 and 16.9%) between EDTA/PET and EDTA/glass (Figures 2(a) and (b)). (A) Bland–Altman plot of BNP concentrations measured in EDTA/glass and heparin/glass tubes. (B) Bland–Altman plot of BNP concentrations measured in EDTA/PET and EDTA/glass tubes. Solid blue lines indicate the mean percentage of the difference between different collection tubes; dashed red lines represent the 95% confidence interval for the difference.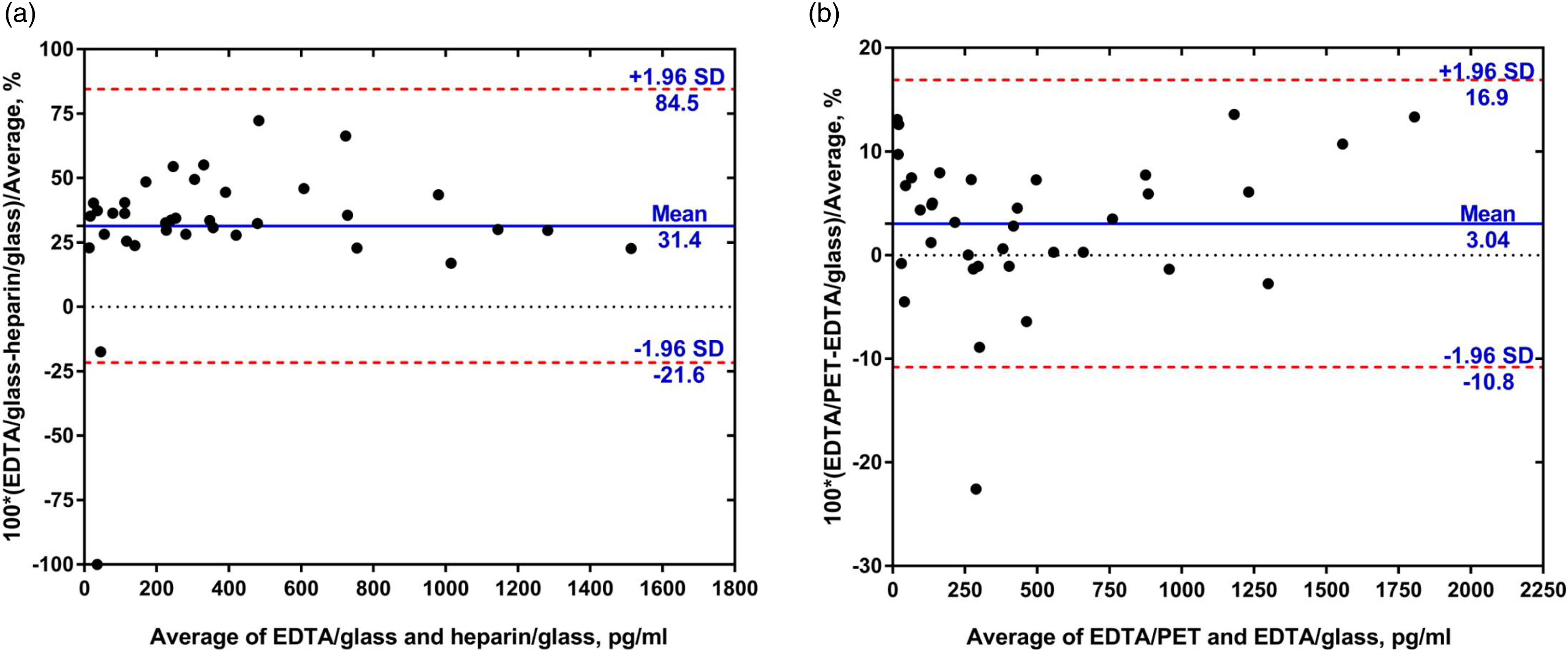
Stability of BNP in different collection tubes
Compared with BNP concentrations at 0.5 h, residual BNP concentrations (%) at 1, 2, and 4 h in the heparin/glass tube were 95.67% ± 7.49%, 86.46% ± 9.27%, and 73.38% ± 8.33%, respectively (p < .001). Although it was statistically significant (p = 0.001), only a 0.73% loss of BNP concentration at 1 h, 0.66% loss at 2 h, and 1.84% loss at 4 h were observed in EDTA/glass tubes. In the EDTA/PET tube, we observed that there was no significant decrease in BNP concentrations at 1, 2, and 4 h (p = 0.910), and residual BNP concentrations were 100.38% ± 1.95%, 100.05% ± 2.45%, and 100.22% ± 2.77%, respectively (Figure 3). Stability of BNP at room temperature in different collection tubes. Following centrifugation at room temperature at 2000 g for 10 min, BNP concentrations of the 36 inpatients were measured at 0.5, 1, 2, and 4 h after sample collection. Residual BNP concentrations (%) at xx h were calculated by (BNP concentration at xx h/BNP concentration at 0.5 h) *100%. The results are presented as mean ± SD of the %residual BNP concentration. ns, no significance; **p < .01; ***p < .001.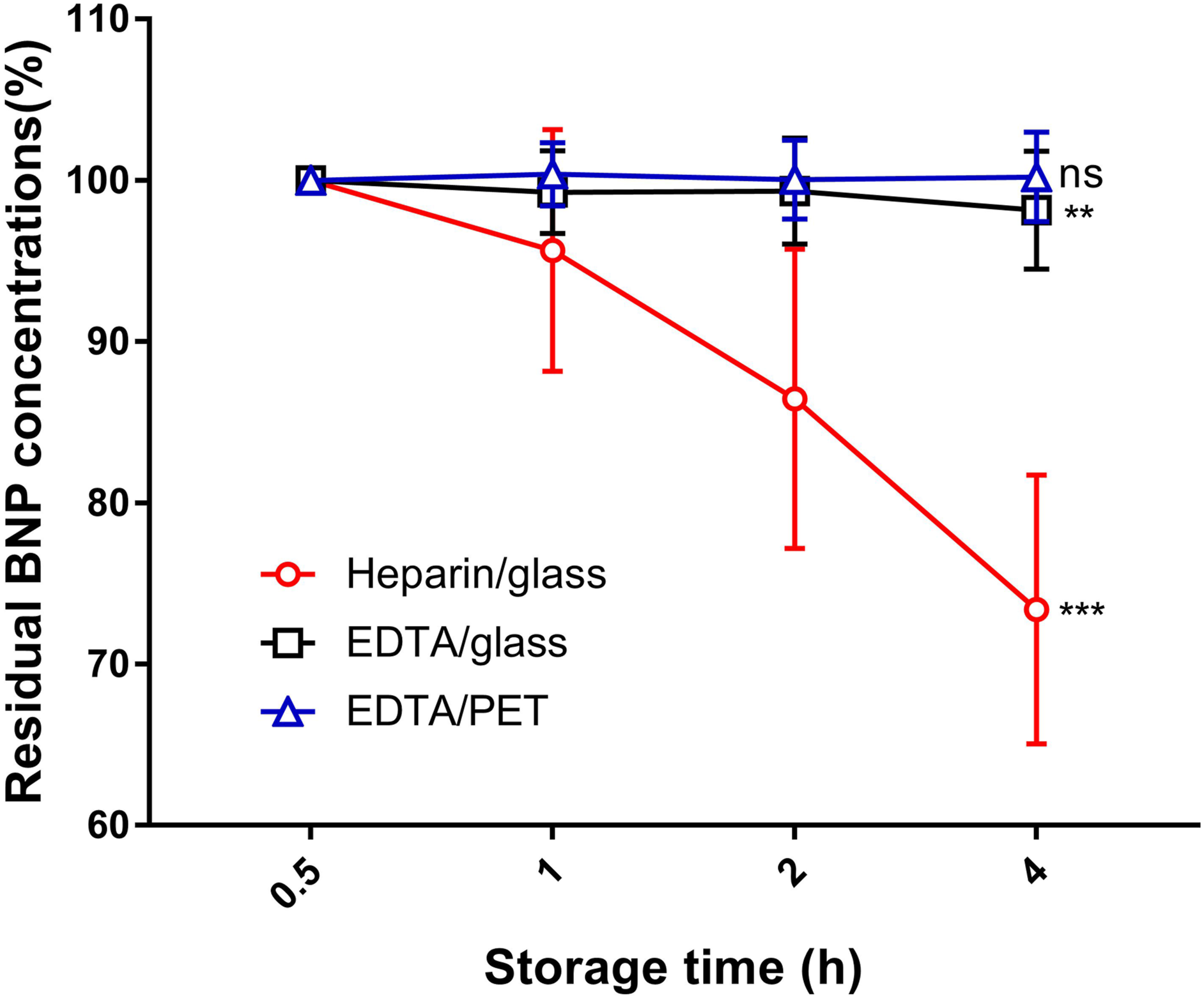
Recovery rate of BNP concentration in different collection tubes
Recovery rate of BNP in different collection tubes on the Mindray CL-6000i.
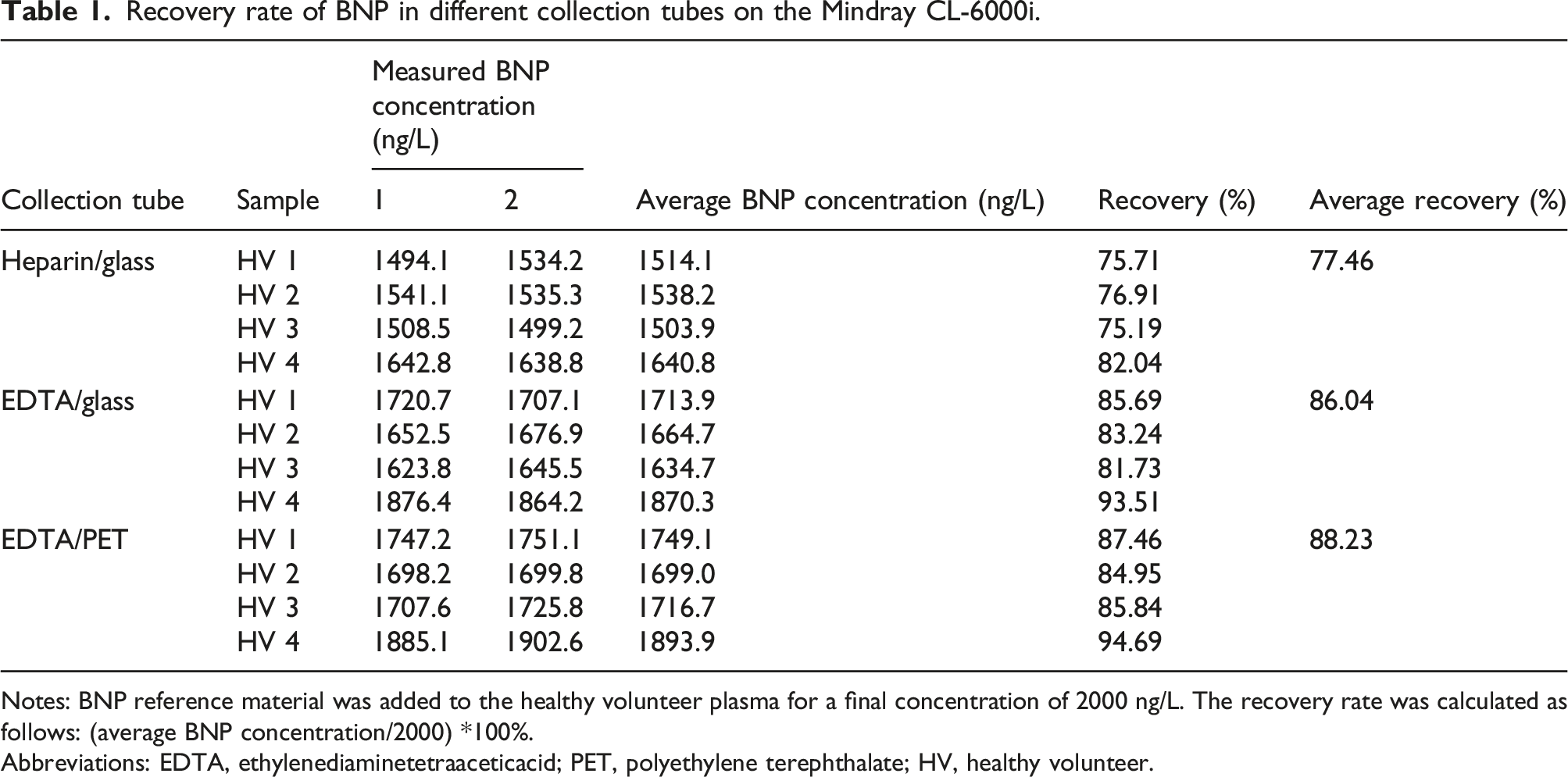
Notes: BNP reference material was added to the healthy volunteer plasma for a final concentration of 2000 ng/L. The recovery rate was calculated as follows: (average BNP concentration/2000) *100%.
Abbreviations: EDTA, ethylenediaminetetraaceticacid; PET, polyethylene terephthalate; HV, healthy volunteer.
Discussion
In recent decades, only a few studies explored the effect of anticoagulants on BNP. Alan H.B. Wu et al. 19 performed a multisite study to evaluate the differences in BNP concentration in collection tubes with different anticoagulants using a Siemens ADVIA Centaur system. The data showed that BNP concentration was 39% lower in heparin tubes than in EDTA tubes. 19 Similar results were observed in other studies using the same analyzer.16,20 On the Beckman Coulter Access Immunoassay System, the effects of anticoagulants on BNP were opposing. Massimo Daves et al. found that samples collected in K2-EDTA showed a mean underestimation of 25.9% when compared to lithium–heparin. 17 Another study showed that BNP concentrations in heparin were higher (mean of 65%) than those in EDTA plasma. 21 On the Architect I2000SR, results were similar to that of the Beckman Coulter Access Immunoassay System, and mean BNP concentrations in heparin and EDTA tubes were 415.64 and 286.11 ng/L, respectively. 22 Our data on the Mindray CL-6000i showed that 34 out of 36 samples had higher BNP concentrations when detected in EDTA/glass tubes (median [Q1, Q3] = 350.1 [139.4, 724.6] ng/L) than in heparin/glass tubes (median [Q1, Q3] = 234.1 [107.0, 451.2] ng/L). In these 34 samples, the mean difference between EDTA/glass and heparin/glass tubes was 45.97%, ranging from 18.57 to 99.37%. The biological matrix can affect BNP detection, and the antigen recognition sites of BNP antibodies from different manufacturers are different,16,21,23 leading to inconsistent BNP results on different analyzers. Therefore, the results obtained in the study cannot be extended to other BNP immunoanalyzers.
The collection tube material was also an important factor affecting BNP measurements. It was reported that there was no significant difference between samples stored in glass and plastic tubes for the Triage BNP or Beckman Access BNP assays. 24 In this study, we found that on the Mindray CL-6000i, 19 samples in the EDTA/PET tubes had higher BNP concentrations than those in their EDTA/glass counterparts at 0.5 h. Seventeen samples had the opposite result. The mean difference ([EDTA/PET − EDTA/glass]/average of EDTA/PET and EDTA/glass *100%) was 3.04%. These results were consistent with a study performed in Fujian Province, which showed that BNP concentrations measured on an ADVIA Centaur XP system in EDTA/glass tubes were 0.9633 times that in EDTA/PET tubes at 0.5 h. 25
The stability of BNP has been investigated in several studies. It was reported that whole blood and plasma BNP concentrations were stable for 4 h from collection when stored at room temperature or at 4°C when measured with the Lumipulse assay. 26 Similar results were found in other studies.24,27 As plasma is generally used for BNP measurements in the clinic, we investigated the plasma stability of BNP in this study. Hiroyuki Shimizu et al. reported that when whole blood samples were stored in EDTA/PET tubes at 25°C for 24 h, about 90% of the BNP immunoreactivity measured by one-step sandwich radioimmunoassay was retained, while 30% was lost in EDTA/glass tubes. This difference increased with storage time. 14 In addition, it was found that BNP in either whole blood or plasma was more stable in EDTA tubes than in heparin tubes.21,28 In this study, we observed the plasma stability of BNP in EDTA/PET, EDTA/glass, and heparin/glass tubes. After 4 h storage at room temperature, residual BNP levels were 100.22% ± 2.77% in EDTA/PET tubes, 98.16% ± 3.65% in EDTA/glass tubes, and 73.38% ± 8.33% in heparin/glass tubes. Compared with our data, BNP degraded faster on the ADVIA Centaur XP system, but residual BNP levels were similar in EDTA/PET (0.8923) and EDTA/glass tubes (0.8546) when stored for 4 h at room temperature. 25 In another study, the stability of BNP immunoreactivity measured by sandwich radioimmunoassay in EDTA/PET tubes at room temperature for 6 h remained around 90%, while 95% was lost in EDTA/glass tubes when chemically synthesized BNP was added into plasma samples. 15 Possible reasons for the discrepancy might include the use of exogenous BNP, different immunoanalyzers, and BNP antibodies that recognize different epitopes. Our study showed that BNP was more unstable in heparin tubes than in EDTA tubes, which is consistent with previous studies.21,28 However, the mechanism for the loss of BNP immunoreactivity in heparin tubes was still unclear.
Although BNP results are known to be affected by anticoagulants and collection tube material, to our knowledge, there is no existing study that evaluates the accuracy of BNP measurements across different collection tubes. Our data indicated that when using the Mindray CL-6000i, BNP results in EDTA/glass and EDTA/PET tubes were more accurate than that in heparin/glass tube, as recovery rates of BNP reference material in heparin/glass, EDTA/glass, and EDTA/PET tubes were 77.46, 86.04, and 88.23%, respectively.
Conclusion
In summary, in heparin/glass tubes, BNP results are often underestimated, and BNP is also unstable. Thus, plasma samples in heparin/glass tubes are not suitable for BNP measurement. BNP concentrations in EDTA/glass and EDTA/PET tubes are closer to true values, and BNP in both types of tubes are stable for 4 h at room temperature. We suggest using EDTA/glass or EDTA/PET tubes when using plasma samples for BNP measurement via the Mindray CL-6000i.
Footnotes
Acknowledgements
We would like to thank all of the participants for their contributions.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was supported by the Key Clinical Specialty Project of Beijing (2020).
Ethical approval
This study was approved by the Ethics Committee of the Jinzhai County People’s Hospital (Approval No. 2,021,001). Written informed consent was obtained from all participants.
Guarantor
YC.
Contributorship
JW found the phenomenon; YC designed the research; XW provided specimens; JF, WZ, YY, RP, and YD detected BNP concentration; WZ and GF analyzed the data and wrote the paper; YC revised the paper.
