Abstract
Objective
Unique clinical courses were observed in two asymptomatic patients receiving warfarin who referred to our hospital because of suspected central hyperthyroidism. We eventually diagnosed these patients with falsely elevated thyroid hormone levels caused by macroscopically invisible fibrin. Although false results caused by fibrin interference in vitro have been identified in various immunoassays, especially in blood samples from patients receiving anticoagulant therapy, no studies on thyroid function testing have been reported. The experience in evaluating these cases prompted us to investigate the independent influence of oral anticoagulants via putative fibrin interference on thyroid function testing.
Methods
We retrospectively reviewed known contributing factors that affect thyroid function testing including age, gender, medication history, body mass index, estimated glomerular filtration rate, smoking status, alcohol consumption, and the seasons of hospital visits from participants who presented the Department of Health Checkup between April 2010 and December 2020.
Results
A propensity-matched analysis revealed that the median serum free thyroxine levels under oral anticoagulant were significantly higher (17.9 pmol/L, n = 60) than those without anticoagulants (16.0 pmol/L, n = 60; p < 0.001). It was noted that this difference was the largest among contributing factors we analyzed. No significant differences were noted in serum thyroid-stimulating hormone levels.
Conclusions
We report two patients receiving warfarin with falsely elevated thyroid hormone levels caused by fibrin interference resembling central hyperthyroidism for the first time. Our retrospective study suggests that the medication status of oral anticoagulants should be considered when evaluating thyroid function tests.
Keywords
Introduction
In the laboratory, the time interval from a patient’s blood draw to the reporting of their results is classified into three phases: pre-analytical, analytical, and post-analytical. Fibrin interference has been recognized as a possible source of pre-analytical errors in immunodiagnostic testing. 1 Residual fibrin in serum samples has been reported to interfere with antigen–antibody binding, especially in blood samples from patients receiving anticoagulant treatment or those with blood coagulation diseases.1,2 False-positive results associated with fibrin were identified in various immunoassays, including hepatitis B and C markers, cardiac troponin I (cTnI), and human immunodeficiency virus (HIV) testing.2–5 The methods to confirm this type of interference include comparing the results using the initial serum samples, serum samples after recentrifugation, and matched plasma samples.6,7
Central hyperthyroidism or inappropriate secretion of TSH (IST) is defined as detectable serum thyroid-stimulating hormone (TSH) in the presence of elevated free thyroid hormone concentrations.8,9 Similarly, the term syndrome of inappropriate secretion of thyrotropin (SITSH) is widely used in Japan to refer to a closely related condition; 10 however, unlike central hyperthyroidism, an elevated serum free triiodothyronine (FT3) concentration is not a requisite criterion for diagnosis. 11 Central hyperthyroidism, IST, and SITSH are important indicators of TSH-secreting pituitary adenomas and resistance to thyroid hormone β (RTHβ).9,12 Other conditions, such as methodological interference or hysteresis in the hypothalamic–pituitary–thyroid axis, have been reported to cause laboratory findings resembling central hyperthyroidism.11,13 Examples of the former include heterophilic antibodies, thyroid hormone autoantibodies, anti-streptavidin antibodies, and substances interfering with ruthenium. 13 However, to our knowledge, laboratory findings resembling central hyperthyroidism associated with fibrin interference have not been reported.
Here, we report the first case series of laboratory findings resembling central hyperthyroidism associated with interference against serum thyroid hormone levels induced by macroscopically invisible fibrin observed in patients receiving the oral anticoagulant warfarin. The experience in evaluating these cases prompted us to investigate the effect of oral anticoagulant (OAC) therapy on thyroid function testing in a large sample of medical checkup participants. Our findings suggest that falsely elevated thyroid hormone levels associated with fibrin interference may occur more frequently than generally considered.
Case 1
An asymptomatic 65-year-old Japanese woman visited the Division of Endocrinology, Department of Internal Medicine, Seirei Hamamatsu General Hospital, because of suspected central hyperthyroidism. The hospital is the largest tertiary hospital in the western part of Shizuoka Prefecture, Japan. She was diagnosed with Graves' hyperthyroidism, atrial fibrillation, and chronic heart failure at the age of 58 years and was started on therapy with thiamazole, warfarin, bisoprolol, and diuretics. Thiamazole treatment was discontinued four years later due to the remission of Graves’ disease. She had no family history of thyroid disease and did not smoke or consume alcohol. The patient was 144 cm tall and weighed 57 kg (body mass index = 27.4 kg/m2). Her vital signs were as follows: blood pressure, 141/92 mmHg; pulse rate, 95/min; and body temperature, 36.3°C. Palpation revealed diffuse goiter. Routine blood tests, including complete blood cell count, electrolytes, and renal and liver function, were all within the reference intervals.
To evaluate thyroid function, blood samples were collected into vacutainer transparent blood collection tubes (SMD108CG, Insepack™ II-D, Sekisui Medical Co., Ltd., Tokyo, Japan) containing silica as a coagulation enhancer and a separating gel. First, blood samples were mixed by inverting at least five times according to the standard phlebotomy guidelines published by the Japanese Committee of Clinical Laboratory Standards 14 and left at room temperature for at least 10 min. Then, the supernatants were obtained by centrifugation at 2000×g for 10 min. After no visible fibrin formation was macroscopically confirmed, serum samples were immediately measured using the ARCHITECT i2000SR immunoassay analyzer with the ARCHITECT TSH Reagent Kit, the ARCHITECT Free T4 Reagent Kit, and the ARCHITECT Free T3 Reagent Kit (Abbott Japan, Tokyo, Japan).
Timeline of TSH (mIU/L), free T4 (pmol/L), and free T3 (pmol/L) levels determined by the different assay methods in Case 1.
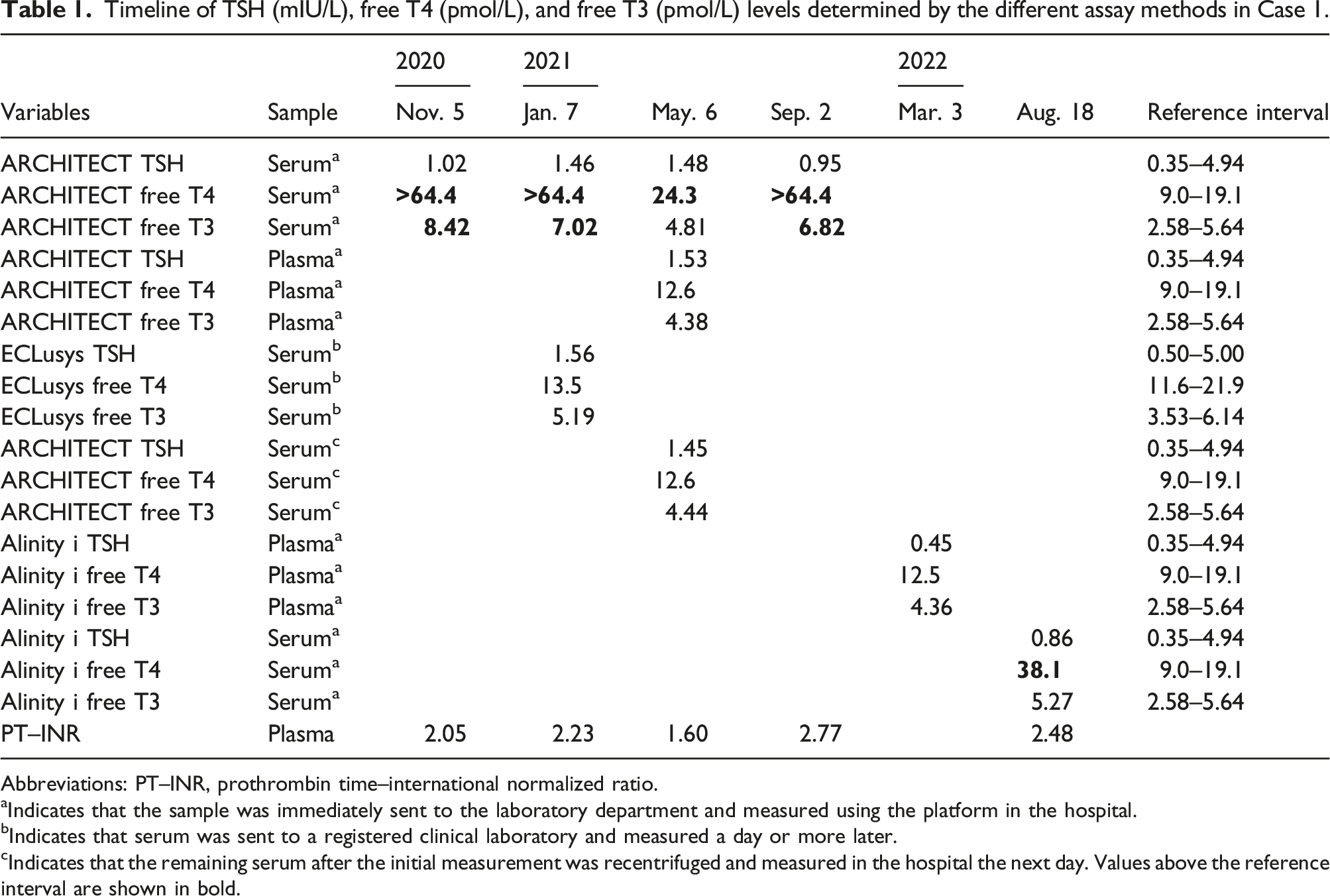
Abbreviations: PT–INR, prothrombin time–international normalized ratio.
aIndicates that the sample was immediately sent to the laboratory department and measured using the platform in the hospital.
bIndicates that serum was sent to a registered clinical laboratory and measured a day or more later.
cIndicates that the remaining serum after the initial measurement was recentrifuged and measured in the hospital the next day. Values above the reference interval are shown in bold.
As the patient was treated with warfarin anticoagulation, fibrin interference was suspected. Therefore, thyroid function tests were performed using the following methods. Blood samples were drawn simultaneously to obtain serum and plasma samples. After the initial centrifugation, the serum sample showed an elevated FT4 level once again. On the other hand, blood for plasma specimens was collected into Vacutainer transparent blood collection tubes (VP-H100K, Venoject™ II, Terumo Co., Ltd. Tokyo, Japan), containing heparin sodium as an anticoagulant. Plasma supernatants were prepared using the same procedure as for the serum samples. The remaining serum aliquots were stored at 2–8°C and recentrifuged the next day. Both the plasma sample after the initial centrifugation and the serum sample after the second centrifugation demonstrated thyroid hormone levels within the reference intervals. Based on these findings, the patient was diagnosed with falsely elevated thyroid hormone levels caused by fibrin interference.
During the eight-month follow-up, the results of periodic thyroid hormone profiles measured using the plasma sample remained stable with hormone levels within the reference intervals. The hospital replaced the Architect platform with the newer Alinity platform (Abbott Japan, Tokyo, Japan) in March 2022. Thyroid hormone tests measured using the Alinity i system with the Alinity i TSH Reagent Kit, the Alinity i Free T4 Reagent Kit, and the Alinity i Free T3 Reagent Kit again revealed an elevated FT4 level when serum but not plasma was analyzed.
To evaluate the effect of different coagulation statuses on assay interference, we retrospectively reviewed the concurrent measurements of prothrombin time–international normalized ratio (PT–INR) and thyroid function tests measured with the Architect platform from the medical records of the patient (Table 1; n = 4). The relationship between PT–INR and FT4 (or FT3) was analyzed by Pearson’s correlation test after FT4 levels more than the maximum detectable level of 64.4 pmol/L were defined as 64.4 pmol/L. Although FT4 levels appeared to correlate positively with PT–INR, no significant correlations were observed (FT4, r = 0.775, p = 0.500; FT3, r = 0.200, p = 0.917).
Case 2
Timeline of TSH (mIU/L), free T4 (pmol/L), and free T3 (pmol/L) levels determined by the different assay methods in Case 2.
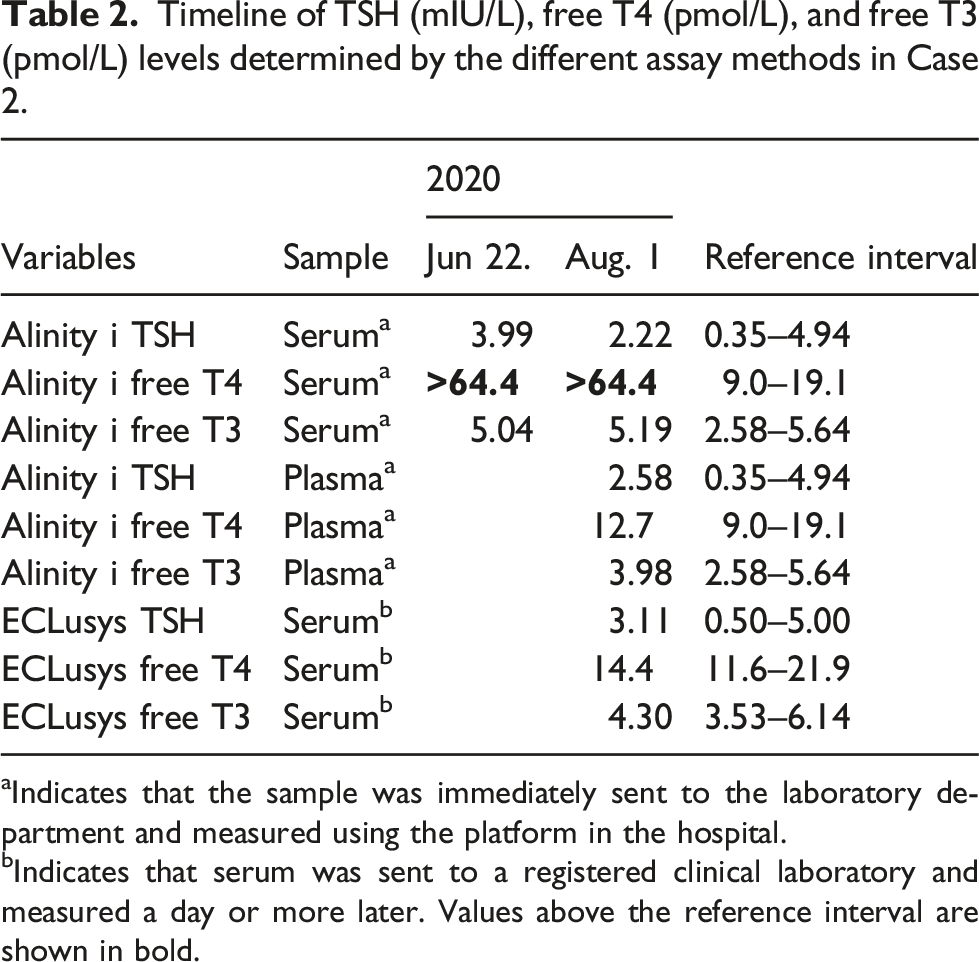
aIndicates that the sample was immediately sent to the laboratory department and measured using the platform in the hospital.
bIndicates that serum was sent to a registered clinical laboratory and measured a day or more later. Values above the reference interval are shown in bold.
Written informed consent was obtained from both patients for the publication of this report.
The experience in evaluating these two patients prompted us to explore the independent influence of possible fibrin interference on serum thyroid hormone profiles in participants receiving OACs.
Methods
Study design
It has been reported that serum FT4 levels are affected by various factors, including age, sex, obesity, kidney function, smoking, and season of hospital visit.15–18 Therefore, we retrospectively reviewed concurrent measurements of FT4, TSH, estimated glomerular filtration rate (eGFR), and body mass index in participants who visited the Department of Health Checkup, Enshu Hospital between April 2010 and December 2020. Participants with thyroid diseases were included in this study. The effect of fibrin interference on serum thyroid hormone profiles was also evaluated in participants receiving antiplatelet therapy or omega-3 fatty acid administration, in addition to OAC treatment. All patients provided written informed consent to participate before the start of the study and at each study visit.
Procedures
The checkup program included an interview on health status. Information, such as medication history or smoking status, was manually retrieved from their medical records. The OACs defined in this study were warfarin and direct oral anticoagulants (DOACs; i.e., dabigatran, rivaroxaban, apixaban, and edoxaban). The antiplatelet drugs included in this study were aspirin, clopidogrel, ticlopidine, cilostazol, dipyridamole, sarpogrelat, and limaprost. Current smoking was defined as smoking at least once daily. Frequent alcohol consumption was defined as the consumption of alcohol six or seven times per week. We categorized the seasons of hospital visits into spring–summer (from March to August) and autumn–winter (from September to February) because the FT4 concentrations were reported to be significantly higher in summer and significantly lower in autumn. 17
Blood was drawn in the early morning between 8:30 AM and 9:30 AM after fasting for at least 12 h and collected into plastic tubes containing a silica-coated film (Venoject II VP-AS109 K, Terumo, Tokyo, Japan). Serum samples were mixed by inverting at least five times and were left at room temperature for at least 15 min. The supernatants were obtained by centrifugation at 2000×g for 7 min. After no visible fibrin formation was macroscopically confirmed, serum samples were immediately measured using the Cobas e411 modular analyzer with dedicated reagents (Roche Diagnostics, Tokyo, Japan). Reference intervals used were as follows: TSH 0.5–5.0 mIU/L and FT4 11.6–21.9 pmol/L (0.9–1.7 ng/dL). There were no analyzer upgrades between 2010 and 2020. Serum TSH concentrations were measured using the ECLusys reagent TSH. Serum FT4 concentrations were measured using the ECLusys reagent FT4, ECLusys reagent FT4II, and ECLusys reagent FT4III between 2010 and 2013, 2013 and 2019, and 2019 and 2020, respectively. The monthly interassay coefficient of variation (CV) of within-laboratory precision for low concentration (approximately 1.0 ng/dL) was around 4.1%; that for high concentration (approximately 3.0 ng/dL) was around 3.2% between 2010 and 2020. To minimize any concern on the accuracy of the measurements from 2010 to 2020, year of visit was also considered as a confounding factor when conducting the following analyses.
Statistics
Data are expressed as the mean ± standard deviation or median with an interquartile range. FT4 levels more than the maximum detectable level of 64.4 pmol/L were defined as 64.4 pmol/L. TSH levels less than the minimum detectable level of 0.01 mIU/L were defined as 0.01 mIU/L. Serum FT4 and TSH levels were Box-Cox transformed before statistical analyses because of their skewed distributions. The unpaired t-test, Fisher’s exact test, or Mann–Whitney test were used for comparisons. To minimize the effects of potential confounding factors, a propensity-matched analysis was conducted. In all cases, 1:1 matching without replacement was used with a caliper of width equal to 0.2 of the standard deviation of the propensity score and with the nearest neighbor method. The relationship between FT4 or TSH (Box-Cox transformed) and other variables were examined using simple and multiple linear regression analyses. All variables with p < 0.20 after simple regression analysis and without collinearity between two variables were investigated using multiple regression analysis.
All statistical analyses were conducted using IBM SPSS Statistics (version 28.0.1.1; IBM, Chicago, IL, USA). Statistical significance was defined as p < 0.05, with two-sided p-values.
Results
We identified 5892 concurrent measurements from 2814 individuals (Table S1). Among the 66 measurements under OAC treatment, no data fulfilled the diagnostic criteria for SITSH.
Predictors of free T4 levels in the study population.
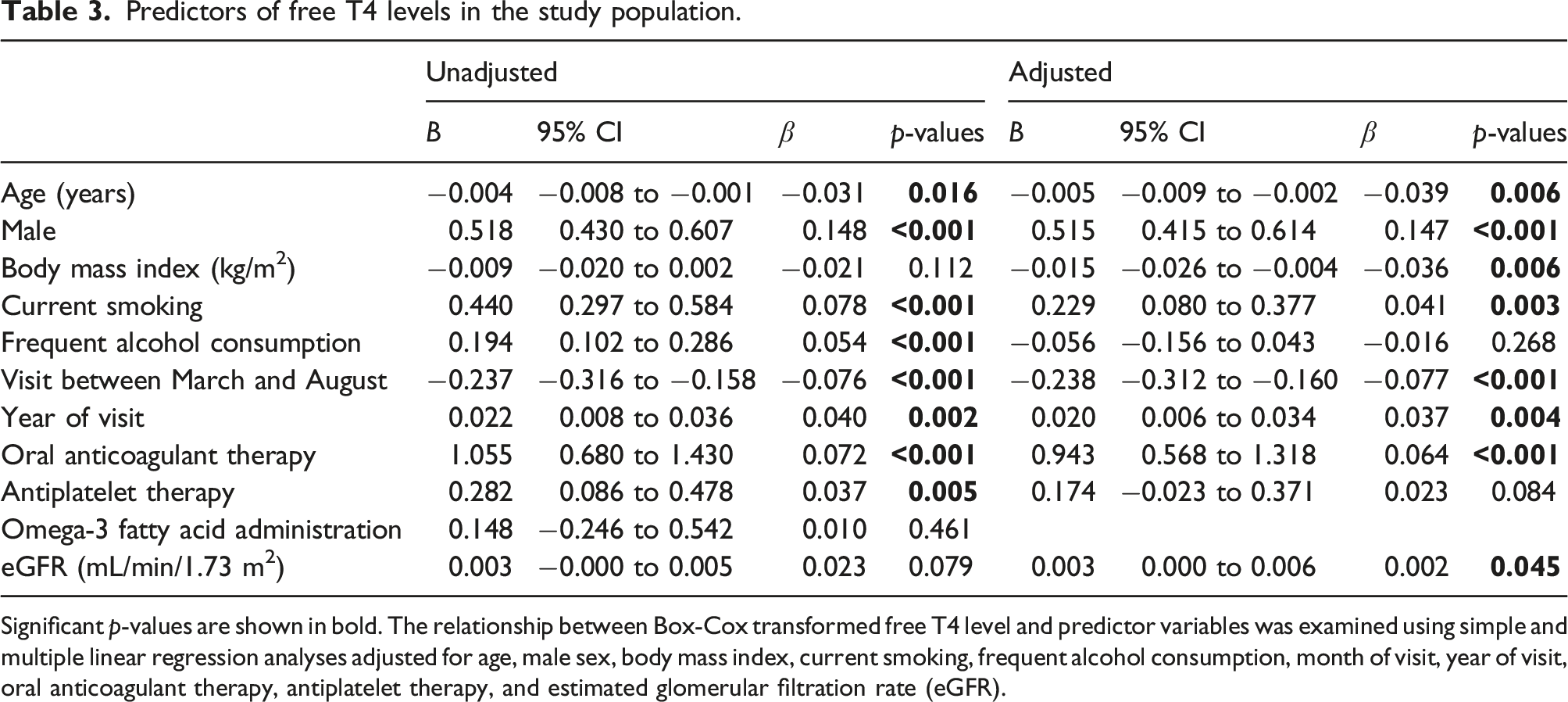
Significant p-values are shown in bold. The relationship between Box-Cox transformed free T4 level and predictor variables was examined using simple and multiple linear regression analyses adjusted for age, male sex, body mass index, current smoking, frequent alcohol consumption, month of visit, year of visit, oral anticoagulant therapy, antiplatelet therapy, and estimated glomerular filtration rate (eGFR).
Participants’ characteristics in the propensity-matched model.
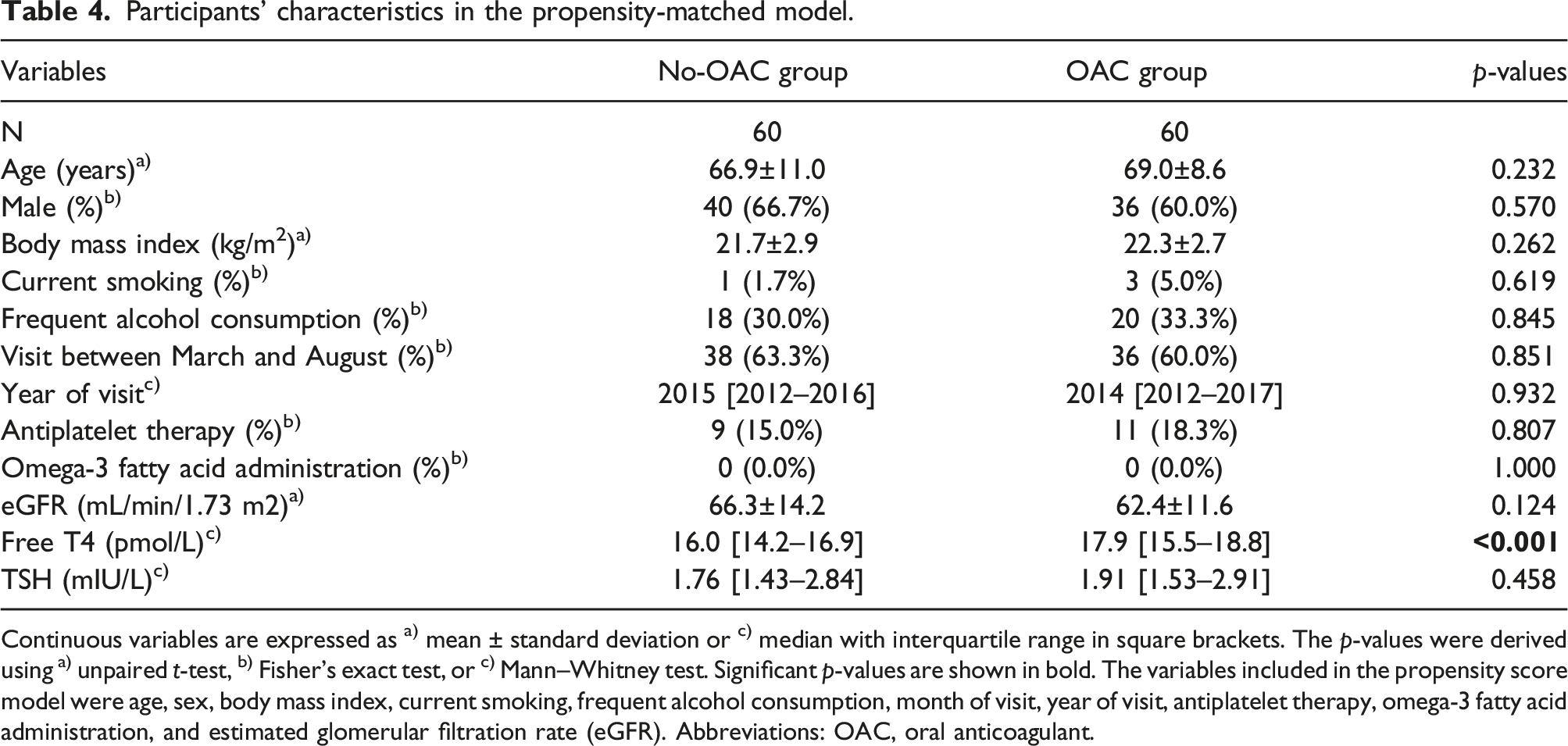
Continuous variables are expressed as a) mean ± standard deviation or c) median with interquartile range in square brackets. The p-values were derived using a) unpaired t-test, b) Fisher’s exact test, or c) Mann–Whitney test. Significant p-values are shown in bold. The variables included in the propensity score model were age, sex, body mass index, current smoking, frequent alcohol consumption, month of visit, year of visit, antiplatelet therapy, omega-3 fatty acid administration, and estimated glomerular filtration rate (eGFR). Abbreviations: OAC, oral anticoagulant.
The difference in free T4 levels after propensity-matched adjustment.
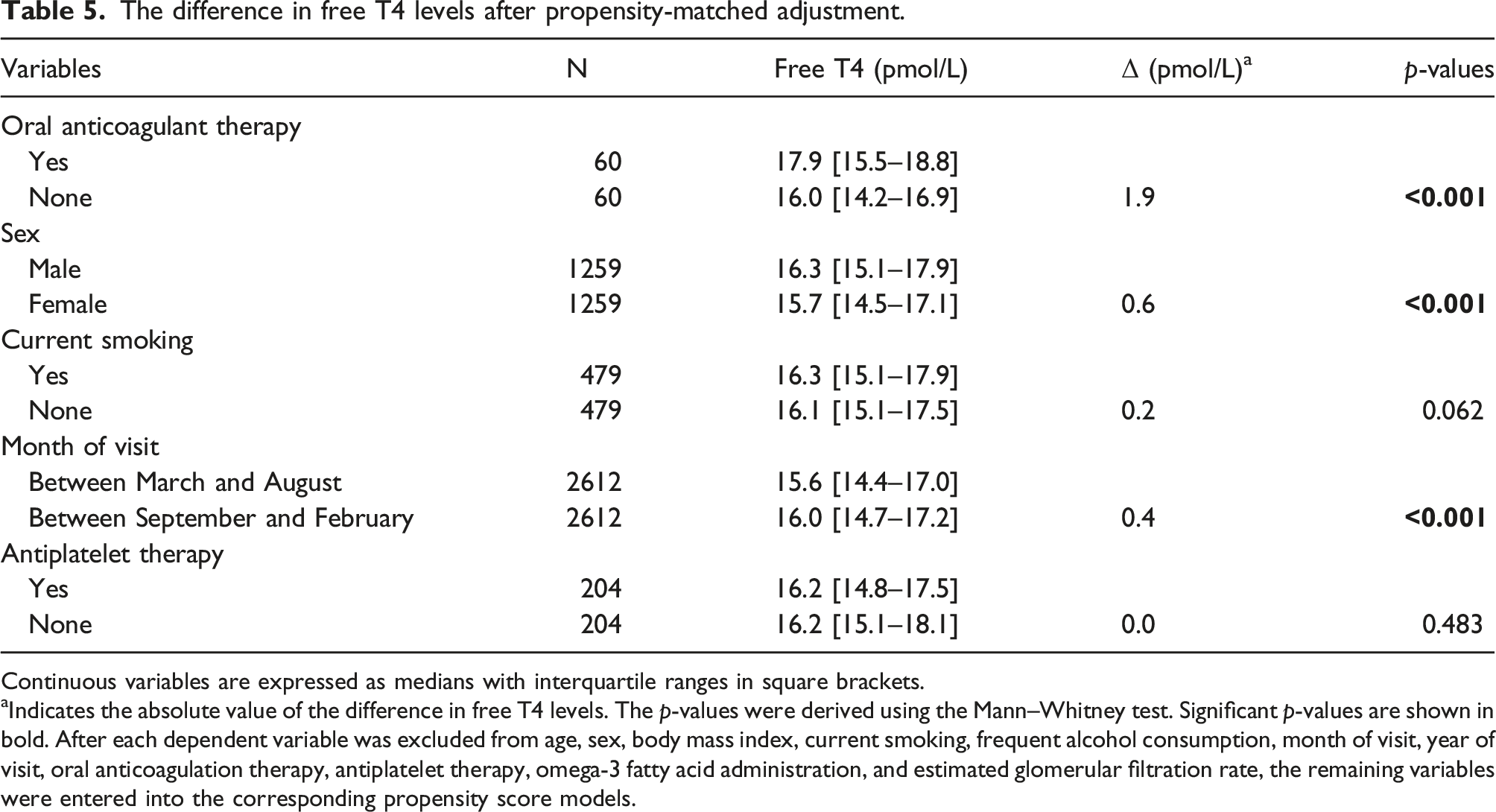
Continuous variables are expressed as medians with interquartile ranges in square brackets.
aIndicates the absolute value of the difference in free T4 levels. The p-values were derived using the Mann–Whitney test. Significant p-values are shown in bold. After each dependent variable was excluded from age, sex, body mass index, current smoking, frequent alcohol consumption, month of visit, year of visit, oral anticoagulation therapy, antiplatelet therapy, omega-3 fatty acid administration, and estimated glomerular filtration rate, the remaining variables were entered into the corresponding propensity score models.
We also evaluated the factors affecting TSH levels using regression analysis (Table S2). Multiple linear regression analysis demonstrated that significant predictors included age, sex, smoking status, year of visit, and eGFR, but not OAC therapy status. In addition, the differences in median TSH levels between the OAC and no-OAC groups were not significant after propensity-matched adjustment (Table 4).
Discussion
Here, we report two patients receiving warfarin with falsely elevated thyroid hormone levels associated with fibrin interference resembling central hyperthyroidism for the first time. We also found that FT4 levels in participants receiving OAC treatment were significantly higher than those in participants who did not receive OACs. Our findings suggest that interference with thyroid hormone levels induced by fibrin in patients receiving OAC may be more frequent than generally considered. The strength of our retrospective study lies in the premise that various known confounders of FT4 levels were considered.
Fibrin has long been recognized as a substance that induces possible interference in clinical laboratories. 1 The simultaneous occurrence of hepatitis B surface antigen (HBsAg) and antibodies against HBsAg was reported in the early 1970s. 19 Grangeot-Keros et al. found a slower clotting time in patients with blood coagulation disorders as a possible cause of false-positive reactions in hepatitis B virus (HBV) testing. 2 The same group further suggested that the assay interference was probably due to residual fibrin strands. Subsequent studies have extensively investigated the clinical characteristics of fibrin interference, particularly in cardiac troponin I (cTnI) assays.3,6 Roberts et al. first reported that incomplete serum separation contributed to false-positive increases in cTnI values. 3 The same group further proposed that the following methods helped eliminate fibrin interference: the use of plasma rather than serum, repeated centrifugation of serum, and the addition of a clot activator to serum. In addition to HBV markers and cTnI values, fibrin interference has been identified in several immunodiagnostic tests, including cardiac troponin T, protein induced by vitamin K absence or antagonist-II (PIVKA-II), HCV markers, and HIV testing.4,5,20,21 However, to our knowledge, there have been no reported cases of fibrin interference in thyroid function tests. Since fibrin interference could theoretically affect other hormone assays, this type of interference may have been overlooked.
The precise mechanism underlying fibrin interference remains poorly understood. Previous studies have suggested that residual fibrin binds nonspecifically to antibodies or traps indicator enzymes.4,6 We previously reported a possible mechanism by which interfering substances against ruthenium cause laboratory findings resembling central hyperthyroidism in the ECLusys platform. 22 First, immunoassays are classified as competitive or noncompetitive sandwich assays. For the competitive assay, including FT4 and FT3, the amount of emission produced by the excited ruthenium inversely correlates with hormone concentrations. In contrast, in sandwich assays, such as TSH, the amount of emission is positively proportional to the concentration of the hormones present. Second, an interfering substance against ruthenium reduces the amount of emission produced by the excited ruthenium, which results in falsely elevated or decreased hormone levels in the competitive or sandwich assays, respectively. Third, sandwich assays involve more ruthenium than competitive assays. Less significant interference in the sandwich assay than in the competitive assay appears to result in smaller changes in TSH levels than in FT4 levels. Given that fibrin could eventually reduce the amount of emission produced by the chemiluminescent substrate (the Alinity or Architect platform) or ruthenium (the ECLusys platform), a similar mechanism reported for interfering substances against ruthenium could account for the thyroid hormone profile resembling central hyperthyroidism observed in this case series.
Fibrin interference has been reported to cause positive errors in measurements obtained using sandwich assays, including HBV markers, cTnI values, and HIV testing.2,4 Because interference appears to have opposite effects on the values measured by the sandwich assay and those measured by the competitive assay, one may expect that fibrin interference has a negative effect on values obtained by competitive assays. However, our two cases revealed falsely elevated thyroid hormone concentrations in a competitive assay. In this regard, heterophilic antibodies, including human antimurine antibody (HAMA), have been reported to cause false-positive errors as well as false-negative results.
23
False-positive results are caused by the cross-linking of assay antibodies by heterophilic antibodies, whereas false-negative results occur when heterophilic antibodies block assay antibodies to bind antigens. Although the precise mechanism has not been clarified, invisible fibrin may result in falsely elevated values by blocking antigen binding (Figure 1). Putative models of false elevation caused by invisible fibrin in competitive immunoassays. Abbreviations: Ab, antibody; Ag, antigen.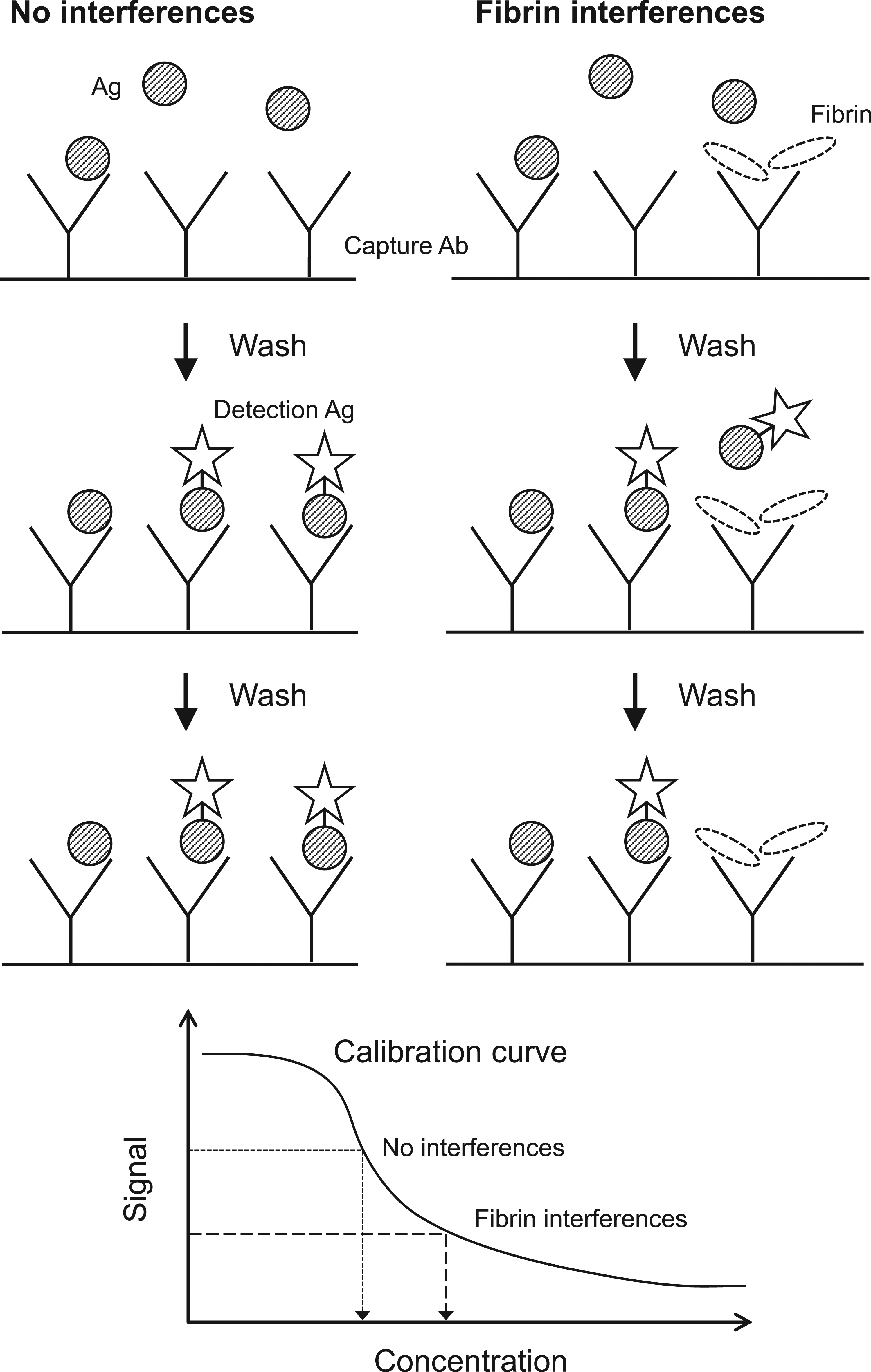
In 1969, heparin, a commonly used injectable anticoagulant, was reported to induce falsely elevated FT4 levels. 24 Numerous studies have revealed that this phenomenon is initiated in vivo but not in vitro. 25 Briefly, heparin induces lipoprotein lipase activity in vivo, which increases non-esterified fatty acid (NEFA) concentrations in vitro during sample storage or incubation. Increased NEFA generation interferes with thyroid hormone binding to serum transport proteins, resulting in an artifactual increase in free thyroid hormone concentration. Various drugs commonly prescribed effect on thyroid hormone testing. 25 However, studies of OACs have not yet been reported. Our findings suggest, for the first time, that the medication status of OACs should be considered when evaluating thyroid function tests.
In our two cases, the serum FT4 values measured using the ECLusys platform at a registered clinical laboratory a day or more after sample collection were within the reference intervals. One possible reason for the absence of obvious interference is the longer time between sample collection and analysis compared to that measured using the Architect or Alinity platform in the hospital. Another possibility is that the degree of fibrin interference depends on the platform. Stiegler et al. reported that the effect of fibrin interference on the cTnI assay was different across assay platforms. 20 The possible platform-specific fibrin interference against thyroid hormone testing is an important issue that needs to be addressed further.
We identified no participants whose thyroid hormone profiles fulfilled the diagnostic criteria for SITSH among the participants receiving OAC treatment. This finding suggests that the prevalence of thyroid hormone profiles resembling SITSH or central hyperthyroidism, caused by fibrin interference, is probably low. One possible explanation is that the degree of assay interference varies from serum to serum and that significant interference appears to be rare. In this connection, Kazmierczak et al. reported that falsely elevated cTnI values in 16 patients, whose repeat analysis revealed values less than the minimum detectable level of 0.3 μg/L, ranged from 2.4 μg/L to 24.0 μg/L. 26 Unfortunately, only serum, not plasma, samples were measured in our retrospective study. The possible difference in the degree of fibrin interference against the FT4 assay from serum to serum is also an interesting issue that needs further investigation.
Previous studies have recommended that serum samples be completely clotted and sufficiently centrifuged to avoid fibrin interference.1,6 In addition, the manufacturers recommend that samples from patients on anticoagulation therapy or with coagulation abnormalities should be subjected to longer coagulation and centrifugation times. However, the optimal procedure remains elusive. It has also been reported that laboratories often shorten the time allotted for a specimen to clot under pressure to provide a rapid result. 1 Accordingly, the optical conditions required for the preparation of fibrin-free serum must be further clarified. Kazmierczak et al. reported that falsely elevated cTnI values in 10 samples, in which repeat analysis revealed values less than the minimum detectable level of 0.3 μg/L, were macroscopically judged to be free of fibrin after the initial centrifugation. 26 Further studies to develop a simple and sensitive method for detecting macroscopically invisible fibrin are important for appropriate diagnosis and treatment.
This study had several limitations. First, the putative association between fibrin interference and higher FT4 levels in subjects taking OAC in our retrospective study is not elucidated. Further studies to compare the values determined by the initial serum samples with those determined by serum samples after the second centrifugation are required using the ECLusys platform. Second, the exact clotting time before centrifugation was not recorded. Third, our retrospective study included participants with thyroid disorders and those undergoing medical treatments other than OACs and antiplatelet drugs. Accordingly, the influence of possible confounders on thyroid function tests, including clotting time, medication status, and presence of thyroid autoantibodies, cannot be excluded. Last, the interference caused by the components of the blood collection tube cannot be excluded. For example, falsely elevated FT3 and total T3 levels, possibly mediated by separating gels, have been reported in serum separator tubes.27,28 Further detailed studies are required to evaluate the effects of fibrin interference on hormone assays, including thyroid function tests.
In conclusion, we identified, for the first time, falsely elevated thyroid hormone levels associated with fibrin interference. Serum samples should be completely clotted and sufficiently centrifuged to evaluate thyroid function, especially in patients receiving OAC treatment or with coagulation disorders.
Supplemental Material
Supplemental Material - Falsely elevated thyroid hormone levels associated with fibrin interference in patients receiving oral anticoagulant therapy
Supplemental Material for Falsely elevated thyroid hormone levels associated with fibrin interference in patients receiving oral anticoagulant therapy by Mitsuaki Tokumaru, Kenji Ohba, Yumiko Kashiwabara, Hiroyuki Takase, Chiga Hayashi, Takayuki Iwaki, Yasuhide Suzuki, Akio Matsushita, Shigekazu Sasaki, Takafumi Suda and Masato Maekawa in Annals of Clinical Biochemistry
Footnotes
Acknowledgement
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by the Japan Society for the Promotion of Science KAKENHI Grant (19K17981 for KO).
Ethical approval
The study protocol was approved by the ethics committee of Enshu Hospital (EH20190823) and conducted in accordance with the Declaration of Helsinki.
Guarantor
KO.
Contributorship
MT drafted the original manuscript. KO is the corresponding author and organized the study. TI, AM, SS, TS, and MM were involved in reviewing and editing the manuscript. YK, HT, CH, and YS contributed to the acquisition of data.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
