Abstract
Background
Acute respiratory distress syndrome (ARDS) is a severe disease with high mortality, and its primary cause is sepsis. The aim of this study was to detect and evaluate the role of Human epididymis protein 4 (HE4) in sepsis-related ARDS.
Methods
One hundred and twenty-three critically ill sepsis patients with/without ARDS and 102 healthy controls were enrolled in this study. Blood samples were collected upon admission for quantitative testing of HE4 by chemiluminescent microparticle immunoassay (CMIA). ROC curve analysis and Spearman’s correlation analysis were conducted to determine the diagnostic and prognostic value of HE4.
Results
Compared with controls, the serum HE4 concentrations of sepsis patients were elevated, and levels in sepsis patients with ARDS were significantly higher (all p < 0.0001). Moreover, HE4 concentrations were strongly correlated with the clinical severity characteristics of sepsis patients, and ROC curve suggested that the AUC of HE4 applied to discriminate sepsis-ARDS patients from sepsis patients was 0.903. HE4 was also found to be a prognostic biomarker of clinical severity and 28-day mortality among critically ill sepsis patients. Logistic regression analysis showed that HE4 was an independent factor for diagnosis of ARDS. Meanwhile, ROC curve analysis showed that the cut-off value of serum HE4 to discriminate 28-day mortality from sepsis patients (AUC: 0.782) was 646.5 pmol/L.
Conclusions
The concentration of serum HE4 in patients with sepsis-related ARDS was markedly increased and was significantly correlated with mortality, which suggests that serum HE4 could be a promising diagnostic and prognostic biomarker for ARDS in sepsis patients.
Introduction
Acute respiratory distress syndrome (ARDS) is one of the most frequent causes of severe hypoxemic respiratory failure in critically ill patients, and is characterized by acute dysregulated inflammatory lung injury, damaged alveolar capillary permeability, increased lung fluid accumulation, profound hypoxemia and need for mechanical ventilation.1,2 ARDS is diagnosed most frequently in cohorts of sepsis patients, accounting for 10% of all patients enrolled in intensive care units (ICUs) worldwide. 3 Although numerous preclinical and clinical studies of ARDS have made strides in elucidating its pathogenesis in the last two decades, specific drug therapies other than supportive measures relying on lung protection ventilation are still insufficient, and mortality still reaches 30–46%.2–4 Although a series of biomarkers have been proposed as possible diagnostic and prognostic indicators for ARDS, their potential clinical value remains undetermined.5,6 Therefore, investigators are searching for potential biomarkers that could discriminate patients with a high risk of ARDS during admissions of critically ill individuals, and predict clinical outcomes.
The pathogenesis of ARDS is characterized by two phases, the exudative and fibroproliferative phases, accompanied by a series of inflammatory and secondary injury responses, pathological changes in the lung matrix and fibroproliferative process. 7 A proliferative response mainly appears during the first to third week of ARDS; however, it can also start within the first week, which usually eventually results in irreversible pulmonary fibrosis. 8 Marshall et al. 9 found that high levels of N-terminal procollagen peptide III (N-PCP-III, a marker of collagen synthesis) could be detected in the bronchoalveolar lavage fluid of ARDS patients as early as the first 24 h after onset of the illness. This indicates that fibroproliferation may occur in the very early stages of lung injury. Consequently, pulmonary fibrosis-related biomarkers may have potential value in the diagnosis and prognosis of ARDS.
Human epididymis protein 4 (HE4) is a secretory protein that was first detected in the epithelium of the distal epididymis, and serves as a protease inhibitor associated with sperm maturation.10,11 Subsequent studies have shown that elevated serum HE4 levels in patients with ovarian cancer can be a valuable biomarker for the diagnosis and prognosis of epithelial ovarian cancer.12,13 HE4 is encoded by the HE4 gene (also known as WAP 4-disulfide core domain-2 or Wfdc2) and is the most upregulated gene in fibrosis-associated myofibroblasts. 14 Recent studies have demonstrated that serum HE4 levels are elevated in patients with kidney disease, heart failure and pulmonary cystic fibrosis, and could be used as a candidate diagnostic and prognostic biomarker for those diseases.15–17 Although the function of HE4 is not fully understood, the whey acidic protein (WAP) domain has been indicated in the activity of serine proteinase inhibitors, which inhibit the proteolytic enzyme elastase, indicating that HE4 may participate in fibrosis.14,18 Indeed, elevated serum HE4 concentrations were reported in a series of studies to be associated with pulmonary fibrosis, and presumably a potential biomarker for the evaluation of the diagnosis, severity and progression of pulmonary fibrosis.17,19–21 However, the concentrations of serum HE4 in ARDS patients have not yet been explored. In this study, we tested the serum HE4 levels of patients with ARDS and investigated its clinical value for the diagnosis and prognosis of ARDS.
Materials and methods
Subjects
This is prospective study. One hundred and twenty-three adult participants meeting the sepsis criteria as defined by the Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3) 22 were enrolled from 2017 to 2019 at the Department of Medical Intensive Care Unit and Division of Respiratory Diseases of the Third Affiliated Hospital of Sun Yat-sen University (Guangzhou, China). The exclusion criteria included patients younger than 18 years, those with noninfectious conditions, chronic renal disease, malignant cancer, acquired immune deficiency syndrome, organ transplantation, underlying pulmonary interstitial fibrosis and pulmonary tuberculosis. Patients with sepsis were subdivided into a sepsis without ARDS (non-ARDS sepsis) group and a sepsis-induced ARDS (sepsis-ARDS) group according to whether they developed sepsis complicated by ARDS during their hospitalization. Patients diagnosed with acute lung injury (ALI) or ARDS met the criteria defined by the American-European Consensus Conference (AECC). 23 Venous blood specimens were obtained after informed consent and within 24 h of sepsis onset. Clinical data were documented. They included sex, age, Acute Physiology and Chronic Health Evaluation (APACHE) II scores, Sequential Organ Failure Assessment (SOFA) scores, arterial partial pressure of carbon dioxide (PaCO2), arterial partial pressure of oxygen (PaO2), the ratio of PaO2 to the fraction of inspired oxygen (FiO2) (also termed the oxygenation index, OI), white blood cell (WBC) counts, plasma lactate, C-reactive protein (CRP) levels, length of ICU stay and 28-day mortality. One hundred and two age-matched healthy controls without underlying medical conditions and who had normal physical examination, routine laboratory tests and chest X-ray or chest CT scans were recruited from the Health Management Center of this hospital.
The study was reviewed and approved by the medical ethics committee of Third Affiliated Hospital of Sun Yat-sen University ([2020]02-123-01), Guangzhou, China, under the guidelines of the Helsinki Conventions. Written informed consent for participating in this study and use of their blood samples was acquired from all subjects or their guardians.
Measurement of serum HE4 concentrations
Venous samples were collected in BD Vacutainer SST tubes from the patients diagnosed with sepsis or sepsis-induced ARDS within 24 h after admission. Venous samples were also obtained from healthy controls. The samples were quickly centrifuged at 3500 r/min at ambient temperature for 5 min, and the sera were separated and stored at −80°C for later testing. The serum HE4 concentration was measured by chemiluminescent microparticle immunoassay (CMIA) with an ARCHITECT I2000sr System (Abbott Diagnostics, Abbott Park, IL, USA) following the manufacturer’s instructions. Measurement was performed with the same number of calibrators, reagents and controls with a single analyzer.
Statistical analysis
All statistical analyses and graphics were performed with SPSS version 18.0 (SPSS Inc., Chicago, IL), Deepwise & Beckman Coulter DxAI platform (https://dxonline.deepwise.com), and GraphPad Prism version 5.0 (GraphPad Inc., San Diego, CA), respectively. Data with a normal distribution are presented as the mean ± standard deviation (SD) and were analyzed with Student’s independent t-test or ANOVA for two or more than two groups, respectively. Clinical variables and laboratory data with a nonnormal distribution are presented as medians and interquartile ranges (IQRs) and were analyzed with the Mann–Whitney U test. Categorical variables are presented as numbers and percentages, and the differences were analyzed using the chi-squared test. The correlation analysis was presented and calculated with a scatter plot and Spearman’s correlation analysis, respectively. To determine the predictive power of serum HE4 concentrations for the development of ARDS or 28-day mortality, areas under the curve (AUCs) of the receiver operating characteristic (ROC) curve were calculated, and the optimal cut-off values were calculated with the Youden Index through each ROC curve. For all tests, two-tailed values p < 0.05 were considered to be statistically significant.
Results
Characteristics of the study subjects
A total of 225 adult subjects composed of 123 critically ill patients with sepsis and 102 healthy controls were enrolled in this study. Sepsis patients without ARDS were assigned to the Non-ARDS sepsis group (n = 58), and sepsis-induced ARDS patients were assigned to the sepsis-ARDS group (n = 65). The 102 healthy controls included 52 (51.0%) males and 50 (49.0%) females, with a median age of 50.0 years [interquartile range (IQR): 43.0–56.0]. The characteristics of the subjects are presented in Supplementary Table 1.
Serum HE4 concentrations increased in ARDS patients
Baseline clinical characteristics of patients with sepsis, and sepsis-ARDS subjects.
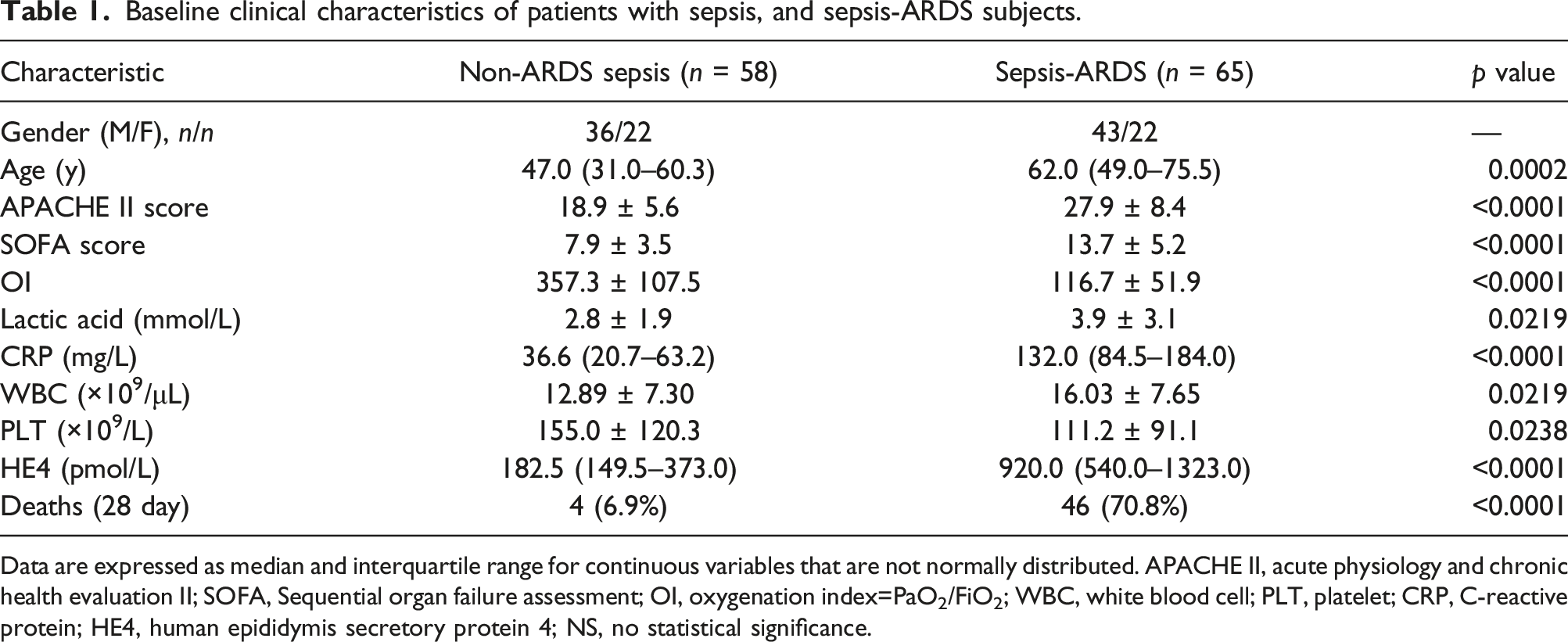
Data are expressed as median and interquartile range for continuous variables that are not normally distributed. APACHE II, acute physiology and chronic health evaluation II; SOFA, Sequential organ failure assessment; OI, oxygenation index=PaO2/FiO2; WBC, white blood cell; PLT, platelet; CRP, C-reactive protein; HE4, human epididymis secretory protein 4; NS, no statistical significance.
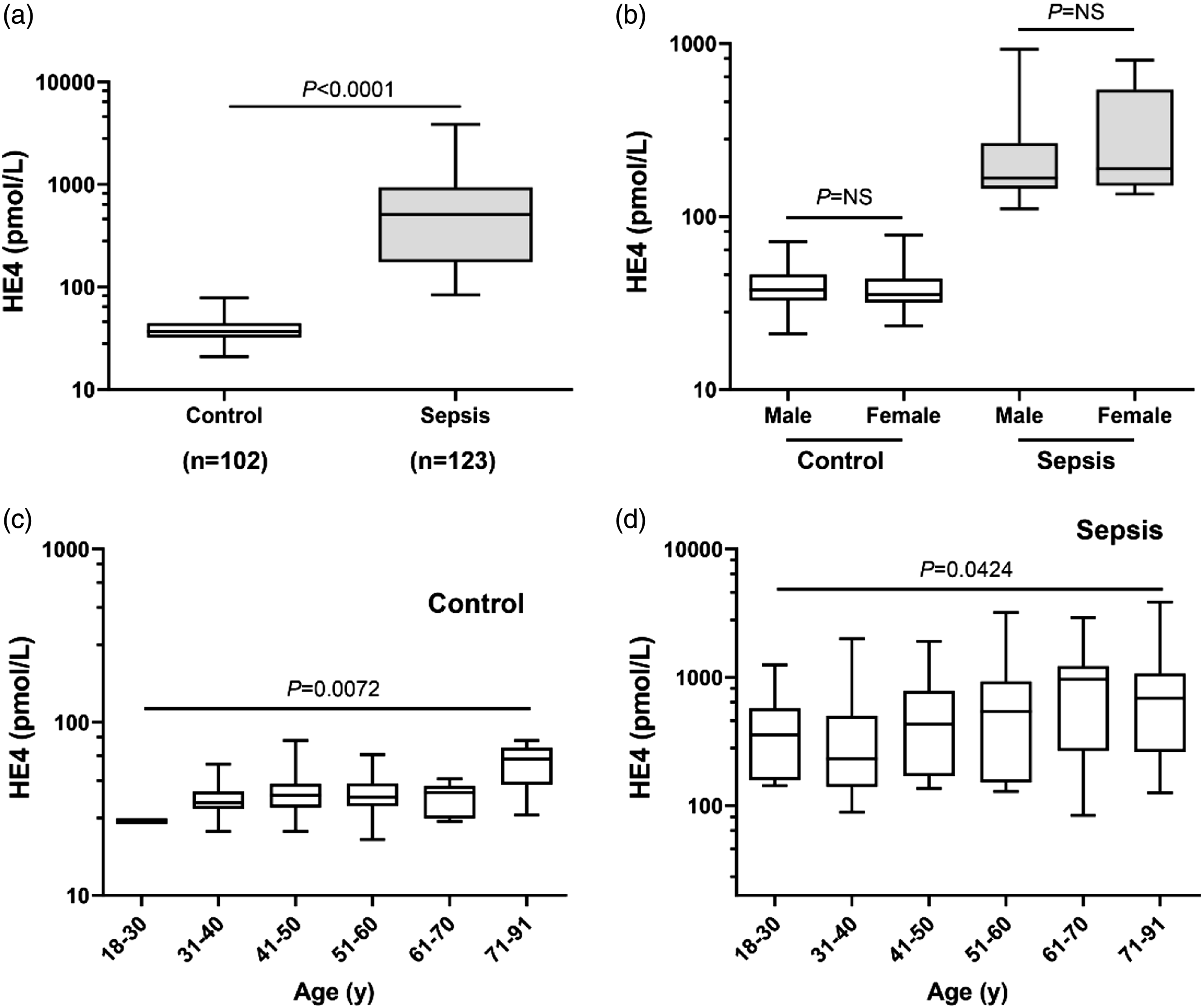
Serum HE4 concentrations in the healthy control, sepsis and sepsis-ARDS groups. (A) Serum HE4 concentrations in the healthy controls (n = 102), and sepsis group (n = 123). (B) The distribution of serum HE4 levels in the male and female subgroups in the healthy control, and sepsis groups. (C) The distribution of serum HE4 levels in control group. (D) The distribution of serum HE4 levels in sepsis patients.
Correlation between serum HE4 concentrations and clinical characteristics
We also assessed the correlations of serum HE4 concentrations with clinical risk characteristics of critically ill patients with sepsis (Figure 2). We observed a notable positive correlation of serum HE4 concentrations with SOFA and APACHE II scores (r = 0.3400 with p = 0.0001 and r = 0.2964 with p = 0.0009, respectively) (Figures 2(a) and (b)). Meanwhile, we also found that serum HE4 concentrations were strongly correlated with the levels of WBC and CRP (r = 0.1793 with p = 0.0472 and r = 0.3140 with p = 0.0004, respectively) (Figures 2(c) and (d)). The correlation of serum HE4 concentrations with SOFA scores, APACHE II scores, levels of lactic acid and CRP in critically ill patients with sepsis. (A) Serum HE4 concentrations were positively correlated with SOFA scores in critically ill patients with sepsis (r = 0.3400, p = 0.0001). (B) Serum HE4 concentrations were positively correlated with APACHE II scores in critically ill patients with sepsis (r = 0.2964, p = 0.0009). (C) Serum HE4 concentrations were positively correlated with levels of WBC in critically ill patients with sepsis (r = 0.1793, p = 0.0472). (D) Serum HE4 concentrations were positively correlated with CRP levels in critically ill patients with sepsis (r = 0.3140, p = 0.0004).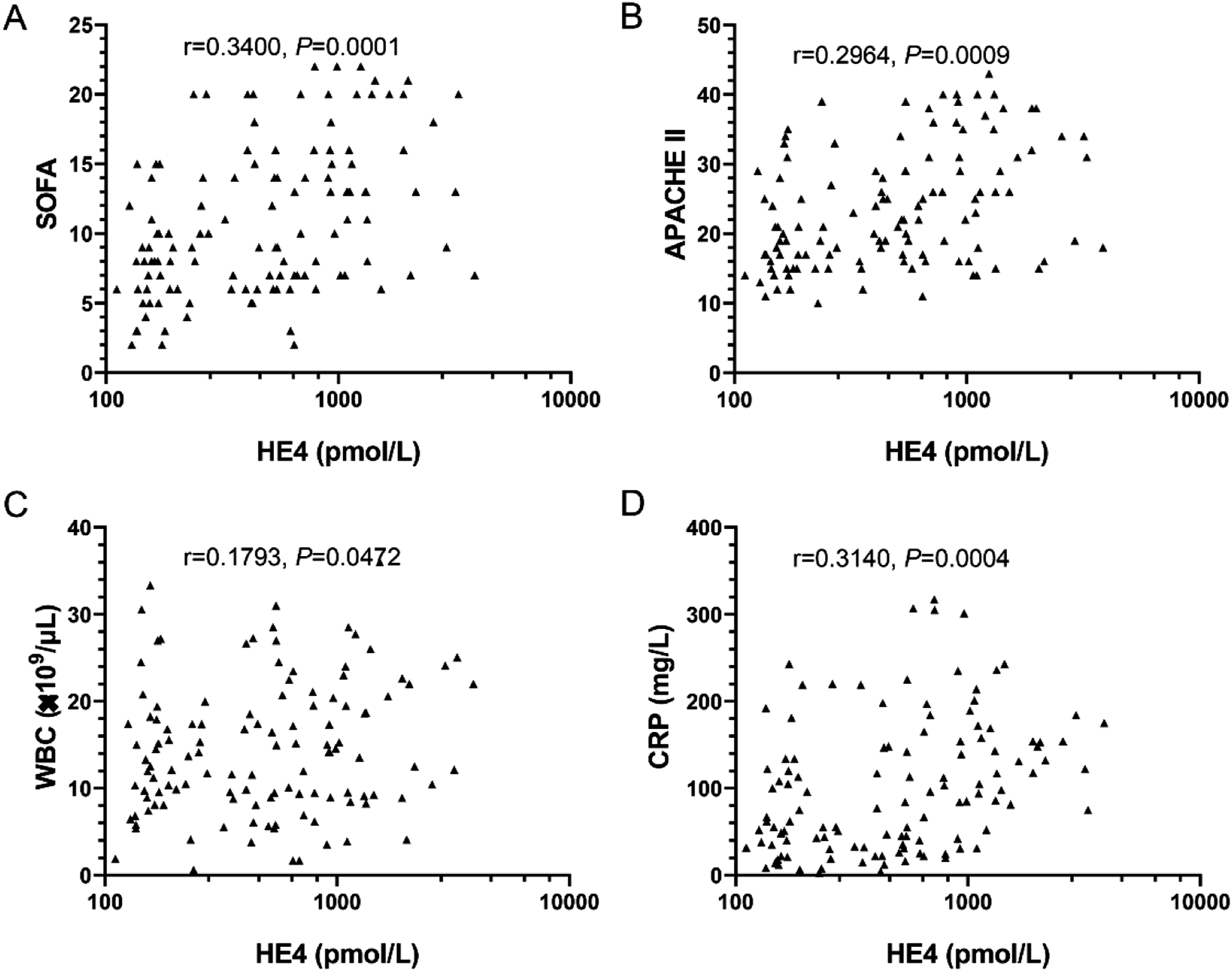
Serum HE4 could be a potential biomarker of sepsis-associated ARDS
We further evaluated the discriminatory value of HE4 for differentiating between sepsis-induced ARDS and sepsis-non ARDS with ROC curves (Figure 3). The ROC for HE4 (0.903 [95% CI: 0.851–0.956]; p < 0.001) was significantly higher than that for APACHE II (0.802 [95% CI: 0.724–0.879]; p < 0.001), SOFA (0.806 [95% CI: 0.731–0.880]; p < 0.001), WBC (0.622 [95% CI: 0.523–0.722]; p = 0.020), and CRP (0.848 [95% CI: 0.779–0.918]; p < 0.001) and lower than OI (0.984 [95% CI: 0.966–0.999]; p < 0.001). The cut-off points (sensitivity, specificity) for HE4, APACHE II, SOFA, OI, WBC and CRP were 533.0 pmol/L (78.5%, 87.9%), 25.5 (79.5%, 89.7%), 12.5 (63.1%, 87.9%), 211.7 (95.4%, 91.6%), 18.56 × 109/μL (60.0%, 84.5%) and 76.0 mg/L (81.5%, 79.3%), respectively. Univariate analysis showed that HE4, APACHE II, SOFA, OI and CRP were statistically significant (p < 0.05). Multivariate logistic regression analysis showed HE4 and OI were independent risk factors for ARDS (Table 2). ROC curves of HE4, APACHE II, SOFA, OI, WBC and CRP in discriminating sepsis-ARDS from sepsis-non ARDS patients. The AUC of HE4 was 0.903, APACHE II score was 0.802, SOFA score was 0.806, OI was 0.984, WBC was 0.622 and CRP was 0.848. The optimized cut-off value according to ROC-analysis for HE4 was 533.0 pmol/L. Univariate and multivariate logistic regression analysis of related indexes to predict ARDS in sepsis patients. HE4, human epididymis secretory protein 4; APACHE II, acute physiology and chronic health evaluation II; SOFA, Sequential organ failure assessment; OI, oxygenation index=PaO2/FiO2; CRP, C-reactive protein; OR, odds ratio; 95% CI, 95% confidence interval.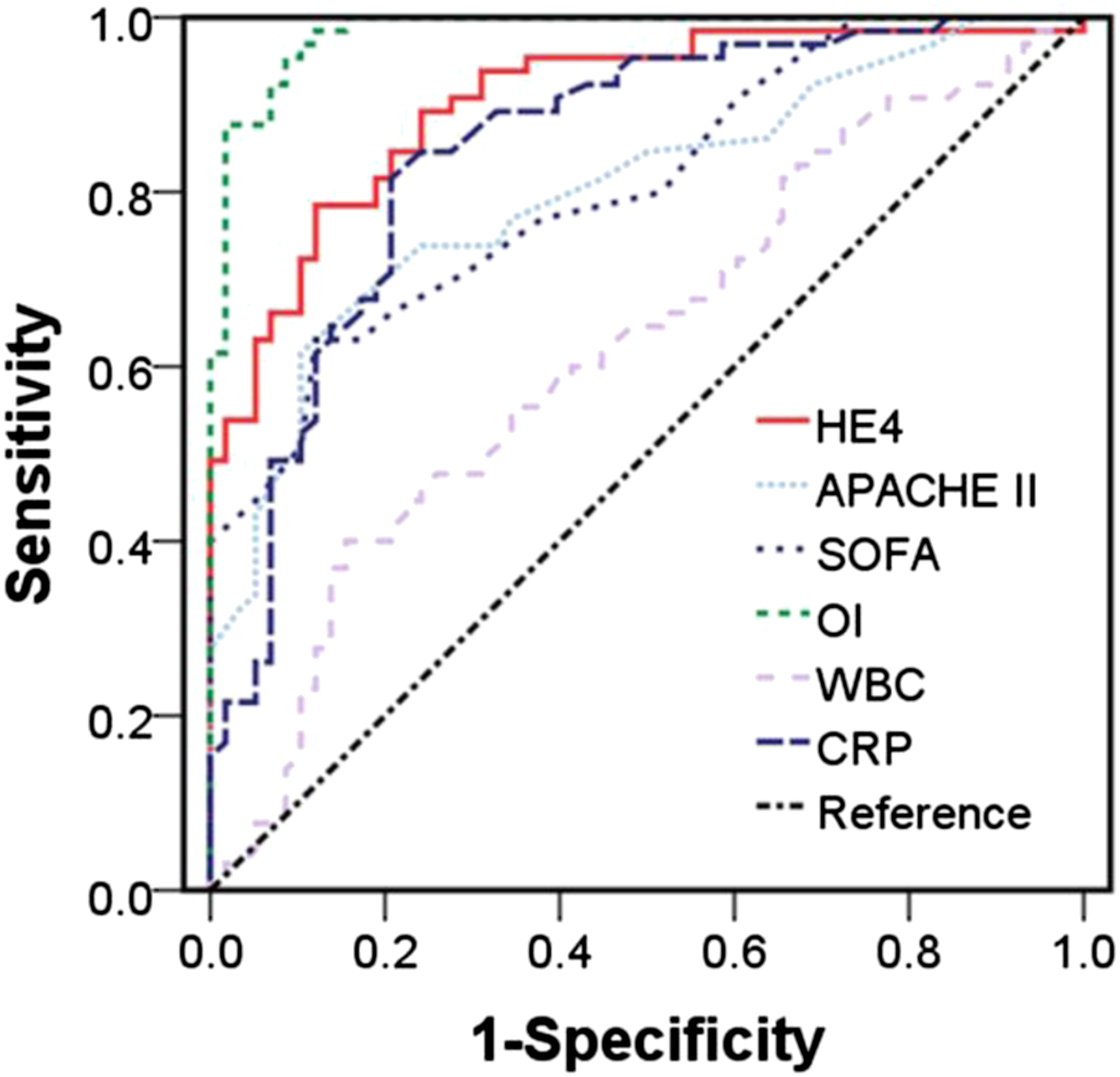
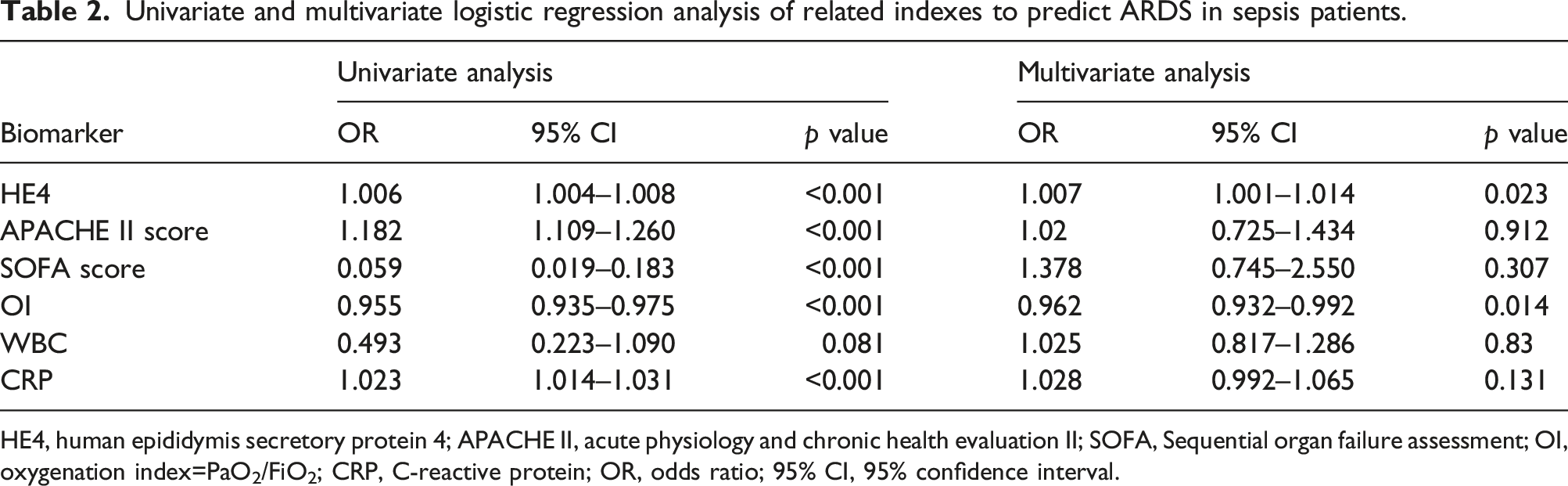
Prognostic value of serum HE4 concentrations in sepsis patients
To determine the prognostic value of serum HE4 concentrations in patients with sepsis, 123 patients with sepsis were divided into surviving and deceased subgroups based on 28-day mortality during hospitalization. As presented in Figure 4(a), serum HE4 concentrations in the deceased group (851.5 pmol/L, IQR: 433.8–1347.0) were higher than those in the surviving group (241.0 pmol/L, IQR: 157.0–600.5) (p < 0.0001). ROC/AUC analyses were also performed to evaluate the value of serum HE4 in predicting the 28-day mortality of sepsis patients (Figure 4(b)). The results showed that the AUCs of HE4, APACHE II, SOFA, OI, WBC and CRP were 0.782 (95% CI: 0.700–0.865, p < 0.001), 0.845 (95% CI: 0.776–0.914, p < 0.001), 0.814 (95% CI: 0.733–0.895, p < 0.001), 0.921 (95% CI: 0.863–0.978, p < 0.001), 0.575 (95% CI: 0.470–0.679, p = 0.161) and 0.718 (95% CI: 0.627–0.809, p < 0.001), respectively. The corresponding cut-off points (sensitivity, specificity) for HE4, APACHE II, SOFA, OI, lactic acid and CRP to predict were 646.5 pmol/L (64.0%, 80.8%), 24.5 (76.0%, 80.8%), 12.5 (72.0%, 83.6%), 171 (92.0%, 86.3%), 16.02 × 109/μL (48.0%, 68.5%) and 71.0 mg/L (80.0%, 63.0%), respectively. Univariate analysis also showed that HE4, APACHE II, SOFA, OI, WBC and CRP were statistically significant (p < 0.05). Multivariate logistic regression analysis showed OI were independent prognostic factors for 28-day mortality (Supplementary Table 2). Serum HE4 served as a biomarker of mortality in critically ill patients with sepsis. (A) Serum HE4 concentrations in surviving (n = 73) and deceased (n = 50) subgroups (p < 0.0001). (B) ROC curves of HE4, APACHE II, SOFA, OI, WBC and CRP in discriminating 28-day mortality among survivors in critically ill patients with sepsis. The AUC of HE4 was 0.782, APACHE II score was 0.845, SOFA score was 0.814, OI was 0.921, WBC was 0.575 and CRP was 0.718. The optimized cut-off value according to ROC-analysis for HE4 was 646.5 pmol/L.
Discussion
Although significant progress has been made in the diagnosis and management of sepsis in the past few decades, the mortality of severe sepsis and sepsis-induced ARDS remain unacceptably high.22,24 ARDS with sepsis as the primary predisposing factor is the most serious form of acute lung injury, and is a devastating clinical syndrome with a mortality rate of 30–60%.25,26 At present, there are no effective drugs for improving the clinical outcome of ARDS. Therefore, early diagnosis and stratification and timely intervention could be helpful to reduce mortality; this is largely dependent on searching for a novel efficient biomarker.
In this study, we showed that the concentration of serum HE4 was significantly increased in ICU patients with sepsis, especially in sepsis-induced ARDS patients. Moreover, serum HE4 concentration was associated with SOFA scores, APACHE II scores and levels of WBC and CRP, which suggested that HE4 is associated with disease severity in sepsis patients. We also demonstrated that HE4 could be a powerful biomarker for differentiating sepsis-associated ARDS from patients with sepsis. Meanwhile, serum HE4 concentrations were strongly associated with 28-day mortality in ICU patients with sepsis, which indicated that HE4 could be a valuable prognostic biomarker for sepsis patients. To the best of our knowledge, this is the first study of serum HE4 concentrations in ICU patients with sepsis.
For ARDS patients, the mortality rate is unacceptably high. 5 Moreover, the primary treatment for ARDS depends on reasonable use of mechanical ventilation and avoidance of lung damage by cytokine storms. ARDS biomarkers have shown promise in the diagnosis, stratification and evaluation of prognoses. 27 Epithelial biomarkers, such as surfactant protein D (SP-D), vascular endothelial growth factor (VEGF), Krebs von den Lungen-6 (KL-6), soluble receptor for advanced glycation end-products (sRAGE) and thymic stromal lymphopoietin (TSLP) have been shown to be associated with disease severity and mortality among patients with ARDS.28–30 The endothelial biomarkers angiopoietin-2 (Ang-2) and von Willebrand factor (VWF) and the inflammatory cytokines IL-8 and IL-18 were also correlated with the mortality of ARDS patients.31,32 However, to date, most of these serum biomarkers are related to the exudative phase of ARDS, and few biomarkers have been reported to be associated with the fibroproliferative phase of ARDS. HE4 was found to be strongly expressed by myofibroblasts, and its serum concentration was associated with disease severity and tissue fibrosis.14,17,33 Disordered healing and fibrous tissue proliferation occurred primarily in the fibroproliferative phases of some ARDS patients and eventually resulted in irreversible lung fibrosis. Therefore, increased HE4 in ARDS patients could be caused by the process of tissue fibrosis, which also indicates that serum HE4 could be a biomarker for ARDS.
In this study, the serum HE4 concentrations of patients with sepsis were significantly increased, and there were no differences by sex (Figure 1(b)), which was consistent with the results of our previous study. 34 We also demonstrated that serum HE4 concentrations were strongly correlated with the clinical severity (SOFA score, APACHE II score, CRP, WBC) of patients with sepsis-ARDS. Therefore, these results indicated that HE4 could be a potential biomarker for identifying the risk of ARDS in patients with sepsis and assessing the severity of sepsis.
We further found that serum HE4 concentrations were significantly increased in and associated with mortality among sepsis patients. Compared with routine clinical characteristics (SOFA score, APACHE II score, OI, CRP, WBC), the ROC curve showed that HE4 could be a valuable biomarker for predicting 28-day mortality in sepsis patients. Previous studies have shown that serum HE4 could serve as a biomarker for predicting death and prognosis in ovarian cancer, chronic heart failure patients and progressive fibrosing interstitial lung diseases.13,16,35 Taken together, we believe that serum HE4 is a biomarker of ARDS induced by sepsis and is associated with the prognosis of sepsis.
There were some limitations in our study. First, this was a single-center study with a small sample size, and more large multicenter studies need to be performed to validate our findings. Second, because lung biopsy might aggravate sepsis patients, we were not able to obtain pathological lung specimens from patients to confirm the direct relationship between HE4 and ARDS-related pulmonary fibrosis. Third, the serum HE4 concentrations were merely tested at the time of ICU admission, and the dynamic changes in serum HE4 concentrations accompanied by the pathogenesis of sepsis and sepsis-ARDS are still unknown. Last, the source and pathogenesis of serum HE4 in sepsis-ARDS patients require further study, and additional experimental studies need to be done.
Conclusions
This study demonstrated that serum HE4 concentrations in sepsis patients, especially in septic ARDS patients, are significantly elevated and significantly related to the mortality of sepsis-ARDS patients. Serum HE4 may be a promising prognostic biomarker for ARDS.
Supplemental Material
Supplemental Material - Serum human epididymis secretory protein 4 correlates with sepsis-associated acute respiratory distress syndrome and 28-day mortality in critically ill patients
Supplemental Material for Serum human epididymis secretory protein 4 correlates with sepsis-associated acute respiratory distress syndrome and 28-day mortality in critically ill patients by Jinmei Luo, Jingjing Liang, Shaofang Wang, Shaoqiong Huang, Laizhi Zhou, Yunfeng Shi, Jingcong Zhang, Yanhong Wang, Ben-Quan Wu and Laisheng Li in Annals of Clinical Biochemistry
Supplemental Material
Supplemental Material - Serum human epididymis secretory protein 4 correlates with sepsis-associated acute respiratory distress syndrome and 28-day mortality in critically ill patients
Supplemental Material for Serum human epididymis secretory protein 4 correlates with sepsis associated acute respiratory distress syndrome and 28-day mortality in critically ill patients by Jinmei Luo, Jingjing Liang, Shaofang Wang, Shaoqiong Huang, Laizhi Zhou, Yunfeng Shi, Jingcong Zhang, Yanhong Wang, Ben-Quan Wu and Laisheng Li in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
We thank all subjects and their family members for providing assistance for this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Young Teacher Foundation of Sun Yat-sen University (20ykpy61), the Third Affiliated Hospital of Sun Yat-sen University for the Cultivate Special Funding Projects of 2020 National Natural Science Foundation of China (2020GZRPYQN26). The funder had no role in the design of the study, data collection and process, preparation of the manuscript, or the decision to publish.
Ethical approval
This study was approved by the medical ethics committee of Third Affiliated Hospital of Sun Yat-sen University (Ethical approval No.: [2020]02-123-01).
Guarantor
B-QW.
Contributorship
B-QW and LSL conceived the study. JML, JJL, SFW, SQH, LZZ, YFS, JCZ, YHW, B-QW, and LSL conducted the experiment, collected the data, and helped to prepare the manuscript. All authors read and approved the final manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
