Abstract
Background
Post-operative hyponatraemia is common following arthroplasty. Clinical hyponatraemia guidelines lack detail on when treatment is necessary, and there is a paucity of literature to guide best practice.
Methods
Data were collected within retrospective service evaluations over two time periods in a single high throughput joint unit. The hospital’s electronic database identified 1000 patients who were admitted electively between February 2012 and June 2013 and again between November 2018 and April 2019 for primary total hip, total knee or uni-compartmental knee arthroplasty. Hyponatraemia and non-hyponatraemia groups were compared. Logistic regression analysis was used to identify independent predictors of post-operative hyponatraemia, length of stay (LOS), re-attendance or re-admission to hospital.
Results
Between 2012–2013 and 2018–2019, 32.1% and 25.7% of patients, respectively, developed post-operative hyponatraemia (serum sodium (s[Na]) ≤135 mmol/L). Those with post-operative hyponatraemia were significantly older, weighed less, were more comorbid and had lower pre-operative sodium. Multivariate analysis showed that increased age, knee surgery and lower pre-operative s[Na] independently predicted post-operative hyponatraemia. Post-operative hyponatraemia did not independently predict LOS, re-attendance or re-admission to hospital, within 90 days, in either cohort.
Conclusion
Post-operative hyponatraemia is common and may be a marker of pre-operative vulnerability. In these cohorts, it was not independently associated with LOS, re-attendance or re-admission to hospital. We suggest that otherwise well patients with mild hyponatraemia can be safely discharged earlier than is often the case and may not require extensive investigation. Further examination and research is required to develop a pre-operative approach to predict which patients will develop significant post-operative hyponatraemia.
Background
It is not yet clear to what extent hyponatraemia after surgery reflects an appropriate transient physiological response, and to what extent it reflects pre-existing physiological vulnerability. It is also unclear what risks (if any) accompany a minor, short-lived, self-limiting drop in serum sodium concentration (s[Na]). An accumulating body of evidence suggests that routine post-operative laboratory blood testing may not always be necessary and can perhaps be safely omitted in some settings.1–3 Hyponatraemia, defined here as a s[Na] ≤ 135 mmol/L, is a common electrolyte disorder that, when encountered following surgery, often triggers a series of investigations and a prolonged hospital stay.4,5 As enhanced recovery after surgery (ERAS) programmes become more popular, and the focus moves towards proactive and pragmatic perioperative care with shorter inpatient stays, there is a need to better identify those individuals who are likely to come to harm as a result of hyponatraemia, whilst minimising unnecessary tests for others who are likely to tolerate a small and short-lived drop in s[Na] without complication. 6 Whilst thorough guidelines regarding identification and management of hyponatraemia exist and reference post-operative hyponatraemia, specific evidence and guidance related to the management of post-operative hyponatraemia in ERAS for arthroplasty is lacking.7–9
We conducted a large service evaluation of post-operative inpatient medical complications following arthroplasty in 2012–2013. Adjustments have since been made to perioperative practices, and in order to reassess the needs of our patients prior to the rolling out of an ERAS programme, we re-assessed the incidence of hyponatraemia and associated factors. Here, we present large-scale data captured in an effort to establish: (i) the incidence of, and risk factors associated with, hyponatraemia following elective primary Total Hip Arthroplasty (THA), Total Knee Arthroplasty (TKA) or Uni-Compartmental Knee Arthroplasty (UKA); and (ii) associations between post-operative hyponatraemia and other post-operative complications.
Methods
Data were collected within retrospective service evaluations of inpatient complications and unplanned re-attendances at hospital in consecutive elective primary THA, TKA and UKA patients in a high throughput elective primary joint unit. The evaluations were registered with the Healthcare Trust’s Standards, Quality and Audit Department: 2012–2013 reference number 4232 and 2018–2019 reference number 5996. The hospital’s electronic database was used to identify 1000 patients who were admitted electively from 29 February 2012 until 25 June 2013 (2012–2013) and from 26 November 2018 until 30 April 2019 (2018–2019). Patients from 2012 to 2013 were admitted under the care of a single surgeon. In comparison, for the second collection period, we broadened the remit to include all surgeons in the unit, only THA and TKA patients were included, and less data were collected, for example, medications and inpatient events were not recorded.
Perioperative practices
All THAs were performed in the lateral decubitus position using the posterior approach using both cemented and cementless fixation although the majority of acetabulae were cementless. All TKAs were performed using a medial parapatellar approach without patellar resurfacing and again with both cemented and cementless fixation with the majority, unlike the rest of the UK being cementless. 10 All UKAs were performed using the medial Oxford uni-compartmental implant (Biomet, Warsaw, IN, USA) with the majority being cementless. Antimicrobial prophylaxis was administered according to the unit’s policy. Between 2012 and 2018, antibiotic prophylaxis changed from 2 g of flucloxacillin and 1.5 mg/kg of gentamicin given intravenously prior to knife to skin, followed by three further intravenous doses of 2 g of flucloxacillin at 6, 12 and 18 h post-operatively, to cefuroxime 1.5 g with 1.5 mg/kg of gentamicin given intravenously prior to knife to skin, followed by two further intravenous doses of 1.5 g of cefuroxime at 8 and 16 h post-operatively. Patients with a history of penicillin allergy or methicillin-resistant Staphylococcus aureus received 10 mg/kg teicoplanin and 1.5 mg/kg gentamicin intravenously once only prior to knife to skin. For cemented components, intravenous gentamicin was not administered as the bone cement utilised contains gentamicin. Tranexamic acid was only administered to patients deemed at high risk of bleeding, including those with rapidly progressive osteoarthritis during the first audit period but was more widely used in the second. Venous thromboembolism prophylaxis consisted of foot pumps for all combined with either enoxaparin for 28 days or aspirin for six weeks as per hospital guidelines and then surgeon preference. All high-risk patients received enoxaparin.
Routine practice during these time periods included checking of electrolytes on post-operative Day 1 and Day 2. Post-operative hyponatraemia was investigated and managed by the attending medical team in line with regional published guidelines. 11
Between 2012–2013 and 2018–2019 normal practice changed to include routine default holding of antihypertensive medications in the immediate post-operative period.
Data collection
Written clinical notes and local/regional electronic databases (including regional radiology systems and regional Electronic Care Records) were interrogated during data collection capturing data including Emergency Department (ED) attendances and episodes of hospital re-admission within 90 days. Additional data were manually extracted from medical and nursing notes by trained research staff or assistants (nursing and medical staff). Hyponatraemia was defined as a s[Na] ≤ 135 mmol/L. This was further categorised as mild: 130–135 mmol/L; moderate: 125–129 mmol/L; or severe: <125 mmol/L.4
When available, pre-operative results, measured on the day of surgery, were utilised. If not available, s[Na] taken at pre-operative assessment were used. One patient in the 2012–2013 group and three patients in the 2018–2019 group did not have pre-operative results available.
Statistical analysis
Patients undergoing more than one surgery during the collection period were considered as different entries. All statistical analyses were carried out using SPSS for Windows (IBM, Version 26.0, Armonk, NY, USA) and all data were assessed for normality using the Shapiro–Wilk test. Chi-square analysis (or Fisher’s Exact test as appropriate) and the Mann–Whitney U test were used to compare categorical and continuous variables, respectively, between those with and without post-operative hyponatraemia. The Kruskal–Wallis test was used to compare s[Na] between THA, TKA and UKA patients since previous studies have found varying susceptibility to hyponatraemia by type of arthroplasty.12,13 The Wilcoxin Signed-Rank test was used to compare pre- and post-operative s[Na]. Logistic regression analysis was used to determine factors associated with hyponatraemia, length of stay (LOS), re-attendance and re-admission. Variables which displayed P < 0.1 in univariate analysis for either cohort were added to the multivariate logistic regression model. Otherwise, statistical significance was set at the P < 0.05 level. More data were available for the 2012–2013 group however, only variables which were available for both groups were used in these analyses.
Results
2012–2013 cohort: A total of 1015 patients underwent elective THA, TKA or UKA. Of these, one underwent non-arthroplasty surgery, one was a duplicate record and notes were not available for 13 patients, leaving 1000 arthroplasties on 969 patients; any bilateral procedures were staged as opposed to simultaneous.
2018–2019 cohort: A total of 994 patients underwent elective THA or TKA (1000 arthroplasties); again any bilateral procedures were staged. Three patients did not have pre-operative s[Na] results and as such were removed from the analysis.
Incidence and predictors of post-operative hyponatraemia
Pre-operative patient characteristics and comparison of those with and without post-operative hyponatraemia.
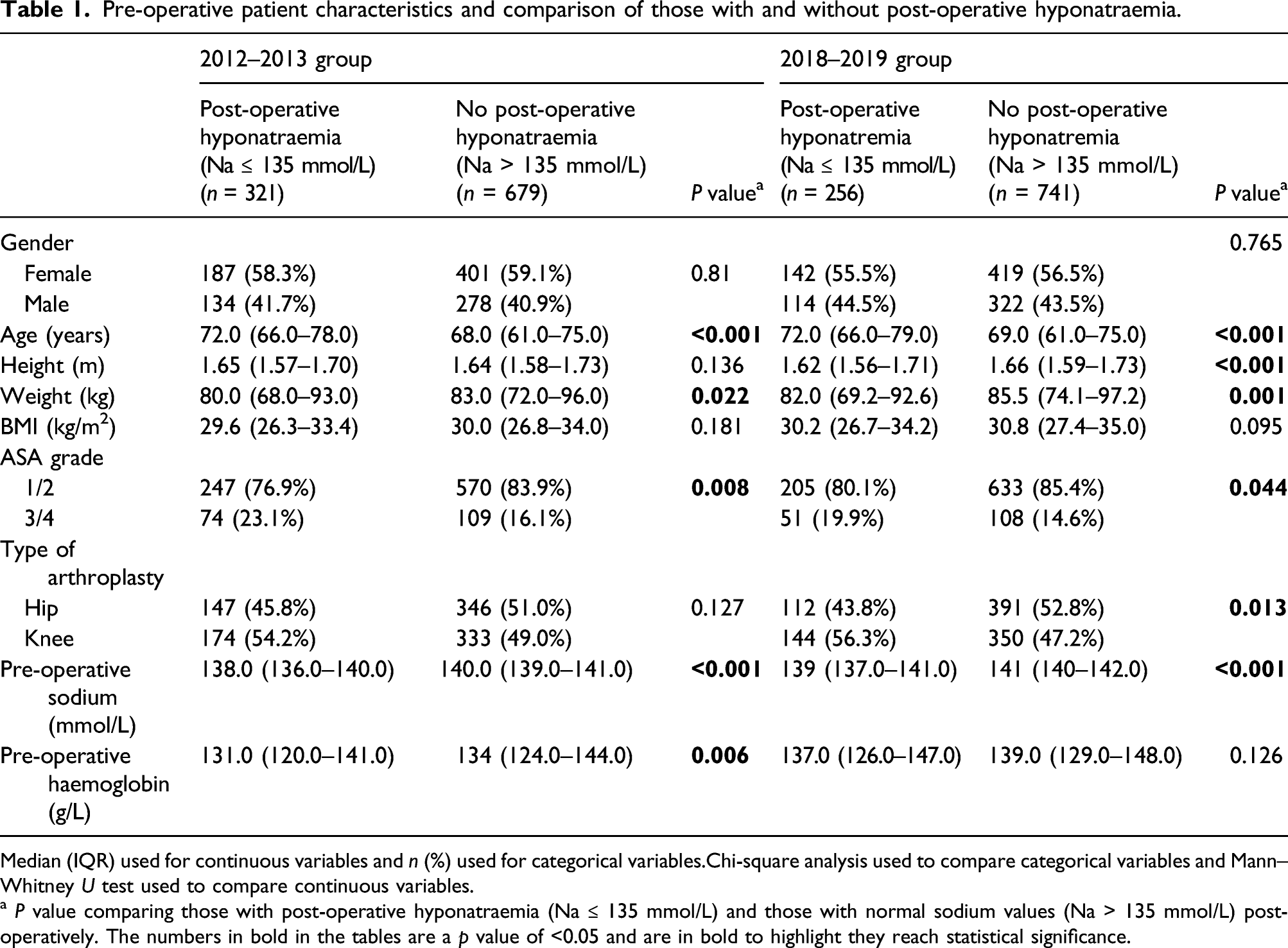
Median (IQR) used for continuous variables and n (%) used for categorical variables.Chi-square analysis used to compare categorical variables and Mann–Whitney U test used to compare continuous variables.
a P value comparing those with post-operative hyponatraemia (Na ≤ 135 mmol/L) and those with normal sodium values (Na > 135 mmol/L) post-operatively. The numbers in bold in the tables are a p value of <0.05 and are in bold to highlight they reach statistical significance.
Logistic regression analysis of factors associated with post-operative hyponatraemia (Na ≤ 135 mmol/L) following elective primary hip and knee arthroplasty.
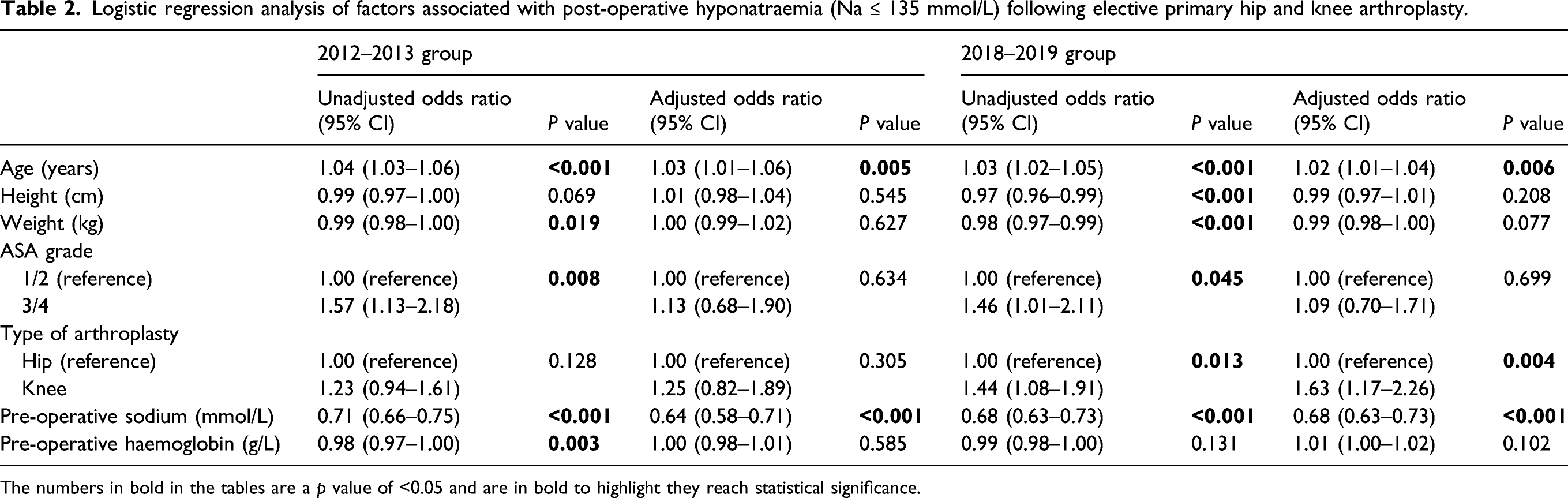
The numbers in bold in the tables are a p value of <0.05 and are in bold to highlight they reach statistical significance.
The perioperative change in s[Na] by pre-operative hyponatraemia classification can be seen in Figure 1.
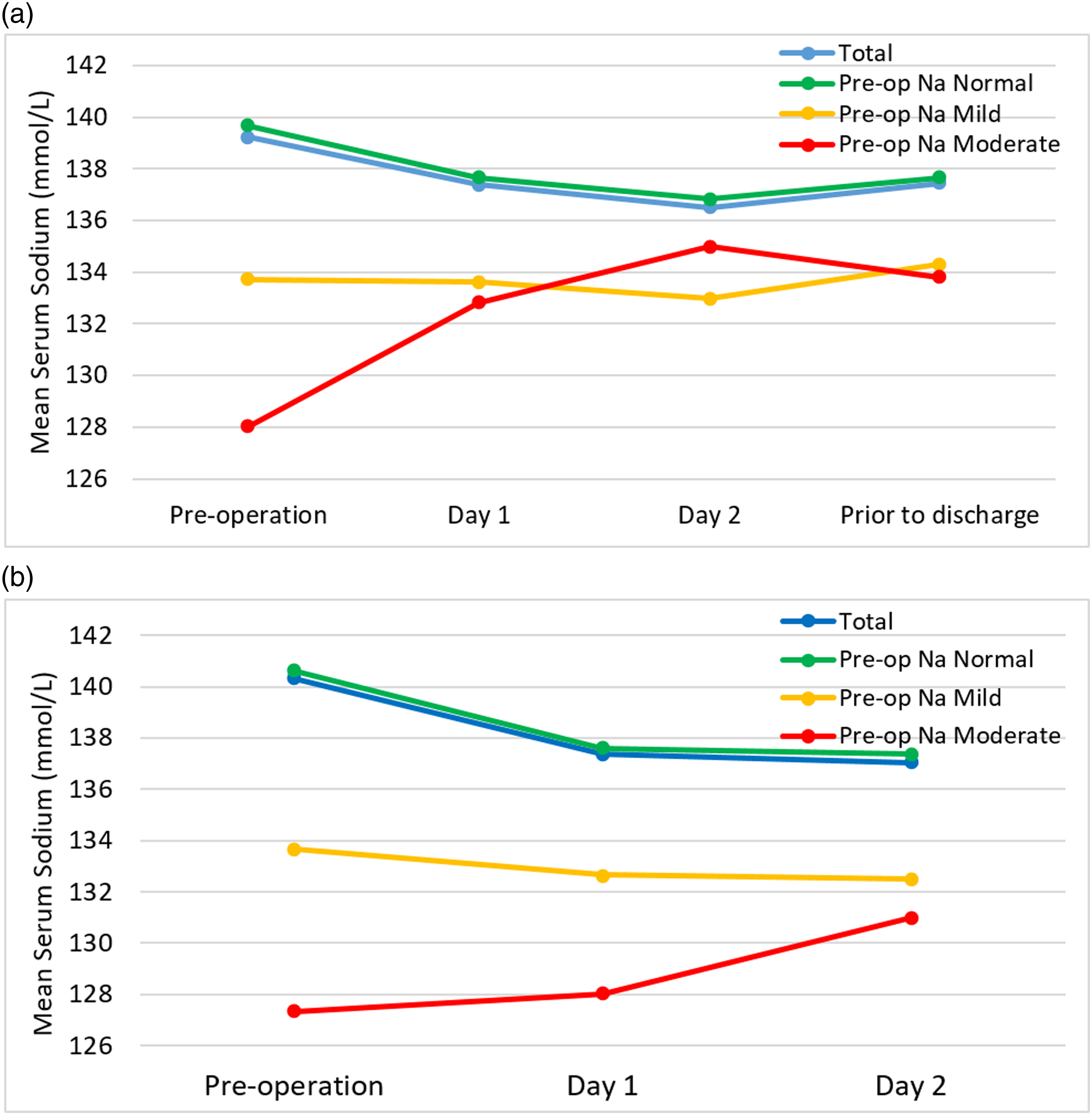
(a) 2012–2013 - Mean change in serum sodium categorised by pre-operative sodium levels. Number of patients within each group: Total = 999, Pre-op normal s[Na] = 930, Pre-op Mild s[Na] = 64, Pre-op Moderate s[Na] = 5. (b) 2018–2019 – Mean change in serum sodium categorised by pre-operative sodium levels. Number of patients within each group: Total = 994, Pre-op normal s[Na] = 957, Pre-op Mild s[Na] = 34, Pre-op Moderate s[Na] = 3.
Associations between post-operative hyponatraemia and other post-operative complications
Post-operative outcomes with comparison of those with and without post-operative hyponatraemia.
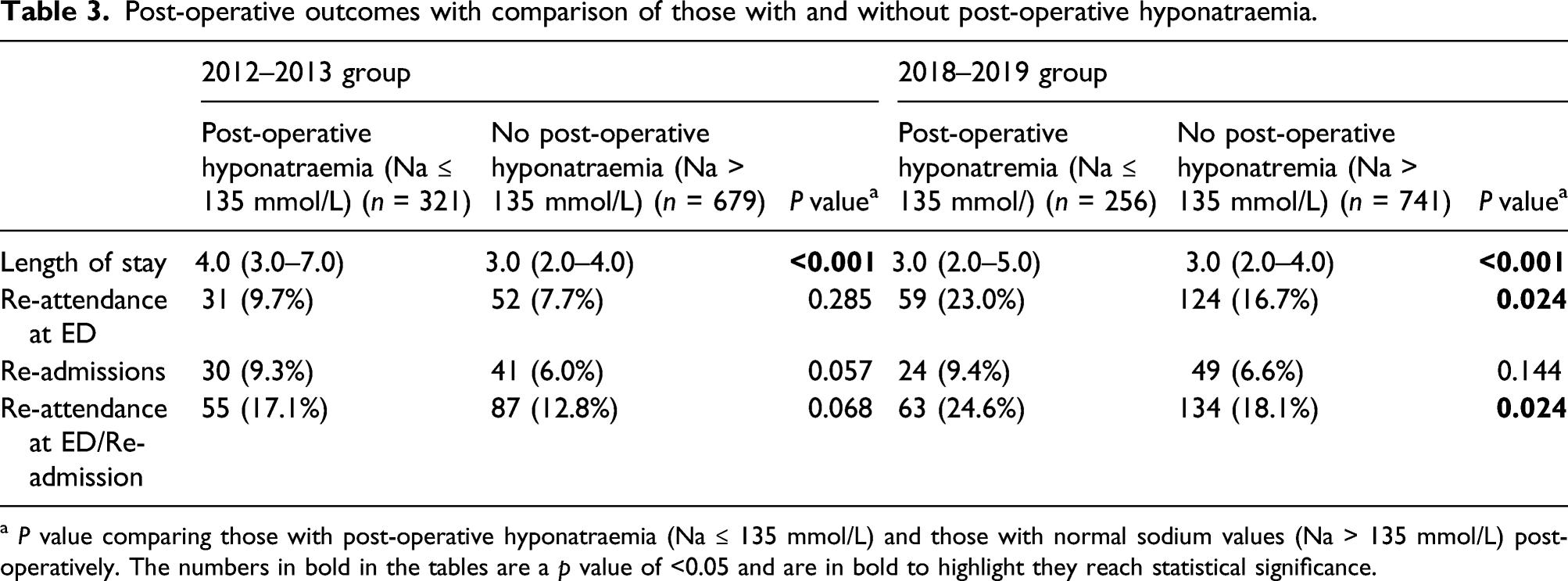
a P value comparing those with post-operative hyponatraemia (Na ≤ 135 mmol/L) and those with normal sodium values (Na > 135 mmol/L) post-operatively. The numbers in bold in the tables are a p value of <0.05 and are in bold to highlight they reach statistical significance.
Logistic regression analysis of factors associated with re-attendance or re-admission following elective primary hip and knee arthroplasty.
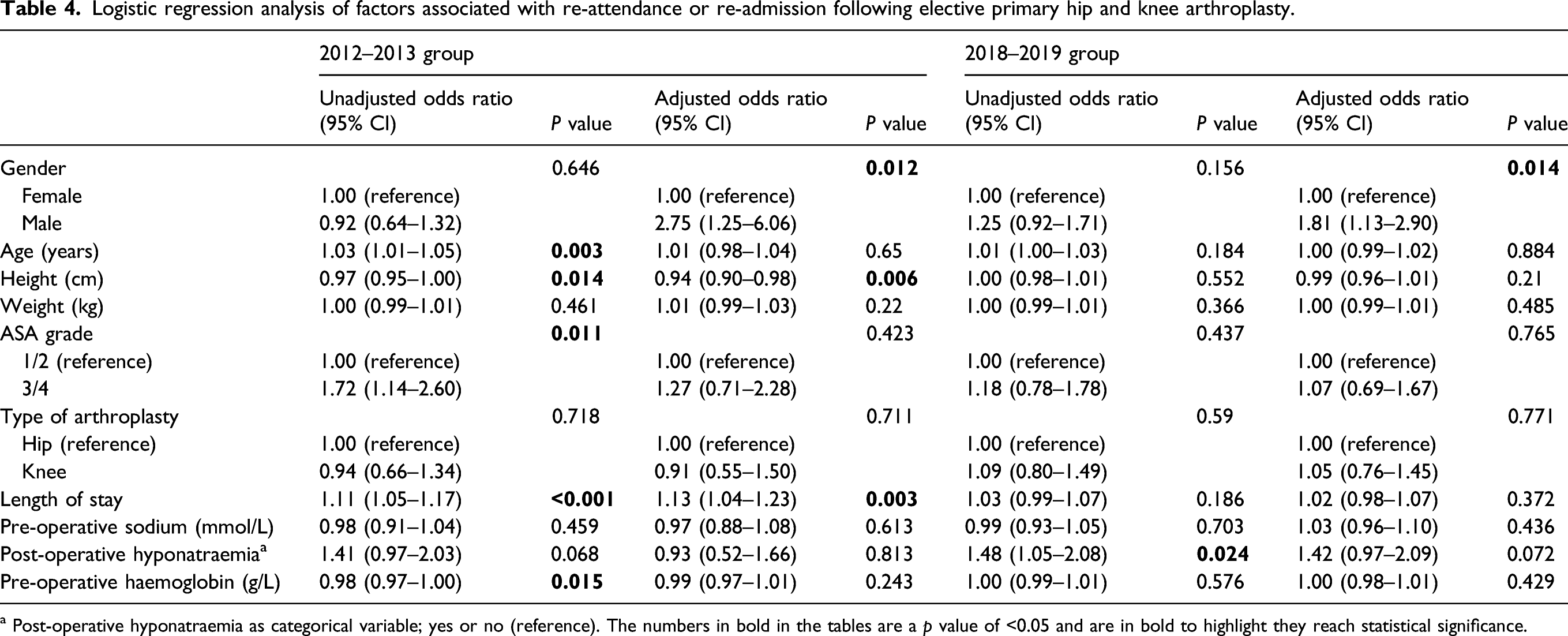
a Post-operative hyponatraemia as categorical variable; yes or no (reference). The numbers in bold in the tables are a p value of <0.05 and are in bold to highlight they reach statistical significance.
Discussion
Main findings
Within our arthroplasty population, post-operative hyponatraemia is common with around one-quarter (2018–2019 data) to one-third (2012–2013 data) of patients affected. It appears to be a marker of pre-operative vulnerability, as patients developing post-operative hyponatraemia following elective primary arthroplasty were significantly older, weighed less, were more comorbid and had lower pre-operative sodium concentrations. We have found that post-operative hyponatraemia does not independently predict LOS or re-attendance at ED, and/or re-admission to hospital within 90 days. This suggests that increased rates of re-attendance at ED, and/or re-admission to hospital within 90 days seen in people with post-operative hyponatraemia could be related to other reasons as opposed to being directly related to the post-operative hyponatraemia.
Regulation of sodium and water are multifaceted, and surgery may lead to a reduced s[Na] via several pathways. These likely include blood loss, inflammation, pain, vomiting, fever, positive pressure ventilation, anaesthesia and analgesia leading to the production of antidiuretic hormone (ADH), appropriately or inappropriately.12,14–16 Increasingly popular ERAS programmes concentrate assessments and interventions pre-operatively, aiming to minimise post-operative inpatient stays and may reduce post-operative complications.6,9 In addition, evidence is accumulating that suggests routine post-operative electrolyte testing is not necessary for all patients.1–3 Our finding that post-operative hyponatraemia is common but does not independently predict re-attendance at ED, and/or re-admission to hospital within 90 days suggests that omitting routine post-operative electrolyte testing would leave common, mild, post-operative hyponatraemia unrecognised but that this may be safe. We acknowledge that patients captured here had their hyponatraemia recognised and, if appropriate, managed according to local guidelines, 11 and this may have affected outcome. Recently published further analysis of the 2012–2013 data showed that there were no significant differences in re-attendance at EDs and/or re-admission within 90 days between those who had post-operative hyponatraemia whilst in hospital (39/217 = 18.0%) and those who did not (103/783 = 13.2%), or between those who were discharged with hyponatraemia (18/108 = 16.7%) and those discharged with normal s[Na] (124/880 = 14.1%). 17
We have shown that whilst patients developing post-operative hyponatraemia tended to be older, more comorbid and more anaemic, only age and lower pre-operative s[Na] independently predicted post-operative hyponatraemia. As we move to better pre-operative characterisation of perioperative risk, more accurate information will be required to predict those who need post-operative laboratory testing. Electrolytes are tested to assess problems other than hyponatraemia, acute kidney injury being the other main example, so again more information is needed before we can accurately identify patients likely to need post-operative laboratory testing.
Our findings are in keeping with the current literature. Post-operative hyponatraemia usually occurs as a syndrome of inappropriate ADH secretion with an estimated duration of 2–3 days. 7 As a result, a small short-lived post-operative drop in s[Na] may be due to a normal physiological response to trauma with duration less than 48 h. 7 Hyponatraemia may also, as we have shown, indicate underlying physiological vulnerabilities, being associated in the literature with increasing age, comorbidity and polypharmacy.12,18–22 Low pre-operative sodium is associated with negative outcomes after surgery, suggesting that both the hyponatraemia itself and the negative outcomes may reflect underlying vulnerabilities.23–25
In contrast to our findings, Leung et al. 23 (2012), in their database study, showed that pre-operative hyponatraemia (s[Na] < 135 mmol/L) predicted longer LOS and increased risk of post-operative complications and 30-day mortality. Abola et al. 24 (2018) found that pre-operative hyponatraemia was associated with longer LOS and greater risk of reoperation. Additionally, McCausland et al. 26 (2014) stated that dysnatraemia (hyper or hyponatraemia) is relatively common in the hospitalised orthopaedic population and is associated with greater LOS and 30‐day mortality. It could be argued that our elective lower limb arthroplasty patient population studied are significantly different to those studied in the above papers (Leung et al. studied all major surgery, Abola et al. studied upper limb arthroplasty and McCauseland et al. studied all major orthopaedic surgery).
Other findings
In keeping with our finding that knee surgery independently predicted post-operative hyponatraemia (2018–2019 data only), Sah 13 (2014) reported that TKA patients may be more susceptible to developing post-operative hyponatraemia compared to THA patients. However, Hennrikus et al. (2015) found that THA patients had an increased risk of developing post-operative hyponatraemia compared to TKA patients. 14
Limitations
The main limitation of this report is that other relevant laboratory analyses and specific treatments administered for post-operative hyponatraemia (e.g. fluid restriction vs. fluid administration) are not described. We cannot say, for example, how many patients were fluid restricted or what the average daily fluid intake was for patients post-operatively.
Over both study periods the total numbers of patients with moderate, and specifically severe hyponatraemia are low (moderate: n = 54, severe: n = 9). The authors have made a number of conclusions regarding hyponatraemia as a whole; any results comparing subgroups of hyponatraemia need to be read taking these small numbers into consideration.
This data are from a single centre, however, the description of two large patient cohorts and the inclusion in the latter of all surgeons arguably enhances generalisability.
Conclusion
Post-operative hyponatraemia is common following elective arthroplasty, and is not independently associated with re-attendance and/or re-admission to hospital within 90 days. We suggest that otherwise well patients with mild (s[Na] 130–135 mmol/L) hyponatraemia can safely be discharged and followed up in the community. It is important to note that this is an observational finding, and studies directly comparing prolonged inpatient management to early discharge with community care would be preferable. Furthermore, it would be desirable to undertake research to ascertain a pre-operative model to predict which patients will develop significant post-operative hyponatraemia.
Footnotes
Acknowledgements
Thank you to Tim Mawhinney (anaesthetist), Sonia Derbyshire (research nurse) and Queen's University Belfast, Medical Students Ioan Hughes, John Vincent, David Elliott and Ryan Doherty who helped with data collection.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DB is a consultant for Depuy Synthes.
Funding
Ethical approval
The data presented in this manuscript were collected within audit and service evaluation which were approved by the relevant Trust audit office. (2012–2013 reference number 4232 and 2018–2019 reference number 5996). Ethical approval is not applicable.
Guarantor
DB.
Contributorship
Conception: DB; Design of the work: EC, DB; Acquisition, analysis or interpretation of data: JM, EC, NG, RC, PH, LB; Drafted the work or substantively revised it: JM, EC, PH, DB.
