Abstract
We report a case of 33-year-old female with underlying genetic susceptibility for familial porphyria cutanea tarda due to novel UROD variant (c.636 + 2 dupT) unmasked by transient exposure to supraphysiological oestrogen concentrations following a single cycle of successful controlled ovarian stimulation for oocyte retrieval. Use of oral oestrogen in the form of oral contraceptive pills and hormone replacement therapy has been well known to trigger active porphyria cutanea tarda phenotype in susceptible women. However, to date, the emergence of clinically overt porphyria cutanea tarda has not been reported in association with fertility treatment in the literature before.
Introduction
Porphyria cutanea tarda (PCT), the most commonly encountered porphyria in clinical practice, results from a deficiency in the hepatic haem biosynthesis enzyme Uroporphyrinogen Decarboxylase (UROD). 1 It is clinically characterized by painful blistering lesions, skin fragility, hyperpigmentation and hypertrichosis affecting sun-exposed areas. 2 While this is primarily an acquired disorder (sporadic PCT) that is very strongly associated with the presence of hepatic iron overload due to susceptibility factors such as Hepatitis C, HIV, excess alcohol and hereditary haemochromatosis, 3 a minority of patients may suffer with an inherited form (familial PCT). 3 Familial PCT is inherited as an autosomal-dominant condition and has low penetrance. 4 Most patients with genetic predisposition for familial PCT do not develop PCT as additional susceptibility factors are also required for the disease to manifest clinically. 3 Moreover, in female patients, long-term oestrogen treatments, e.g. oral contraceptive pills (OCPs), are also well-recognized aetiological factors. 3 Although supraphysiological concentrations of oestrogen are an intrinsic feature of many IVF-related procedures, to our knowledge, there are no reports of PCT following such exposure.
We present the case of a female patient in whom the emergence of clinically overt PCT was triggered by a successful single cycle of ovarian stimulation for oocyte cryopreservation. Furthermore, enhanced susceptibility to PCT in this patient was facilitated by a novel genetic variant causing disruption of gene splicing in the UROD gene, which was revealed during the subsequent investigation of her PCT.
Case report
A 33-year-old white Irish female presented with a four-month history of blistering on her nose and the dorsum of hands (Figure 1). The onset of symptoms occurred while she was on a sun-holiday in Greece, and in general, she was quite anxious about her symptoms. The patient was a non-smoker and had a moderate alcohol intake of approximately 10 units per week. She had been using a transvaginal combined contraceptive ring (NuvaRing®) over the previous 10 years without incident. However, three months prior to the onset of her symptoms, she had undertaken a successful single-cycle of oocyte retrieval procedure, during which she had received a hormonal regimen comprising of Gonal F (recombinant human follicle-stimulating hormone), Luveris (recombinant human luteinizing hormone), Cetrotide (a gonadotrophin releasing hormone antagonist) and Buserelin (gonagotrophin-releasing hormone agonist). Of note, there was no family history of skin photosensitivity, while a hereditary haemochromatosis genetic screen only confirmed a low susceptibility H63D heterozygous state. A comprehensive porphyrin screen reported a pattern consistent with PCT including markedly raised urinary excretion of uroporphyrin and heptacarboxyl porphyrin, positive plasma porphyrin scan at 619 nm and the presence of elevated iso- and heptacoproporhyrin in faeces (Table 1). In addition, hepatology work-up reported unremarkable hepatic autoimmune, HIV and viral hepatitis screens, as well as liver ultrasound.
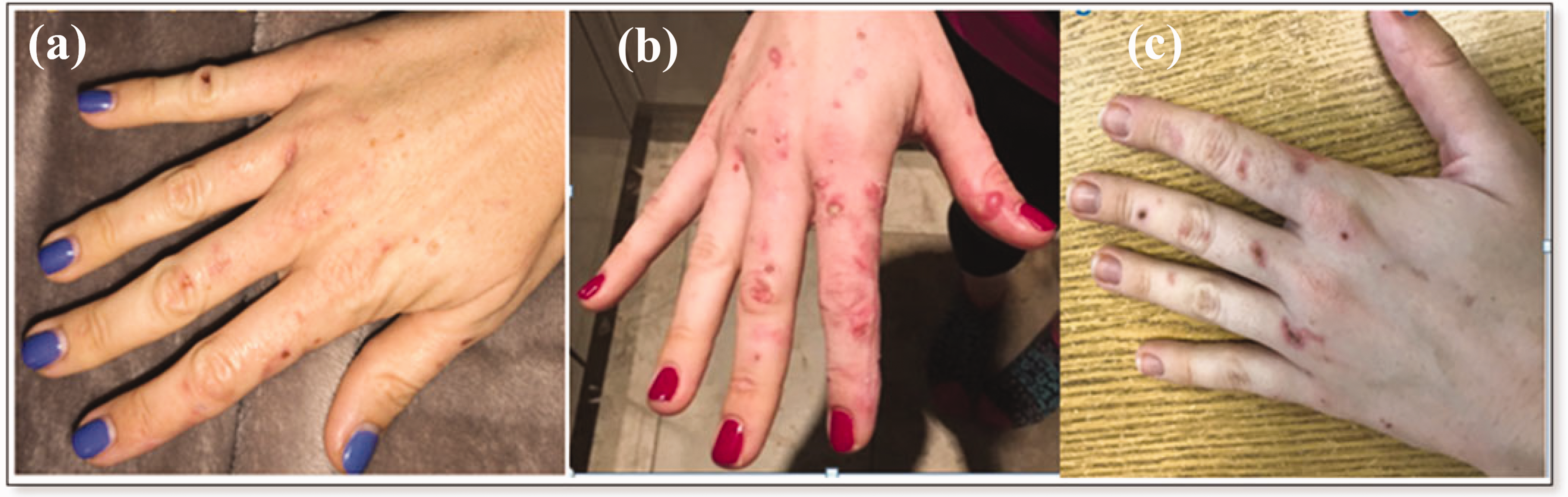
Clinical manifestations of PCT in a 33-year-old female patient showing (a) Early stage of PCT, (b) active blistering phase and (c) erosion and scarring.
Laboratory investigations.
Given the clear cut absence of marked hyperferritinaemia (serum ferritin 223 μg/L [reference interval: 23–393] and transferrin saturation of 44%) as a major susceptibility factor, genetic testing for familial PCT was also undertaken. A novel heterozygous variant c.636 + 2 dupT was identified at the splice donor site of exon 6 in the UROD gene, (Figure 2) and further analysis by reverse transcriptase polymerase chain reaction (RT-PCR) confirmed a complete deletion of this exon (Figure 3 and 4). She was counselled regarding photoprotection measures and specific treatment options for PCT were discussed with her. Venesection was commenced, due to patient preference and she had an excellent clinical and biochemical response with remission of symptoms and a marked reduction in uroporphyrin concentrations (Table 1).
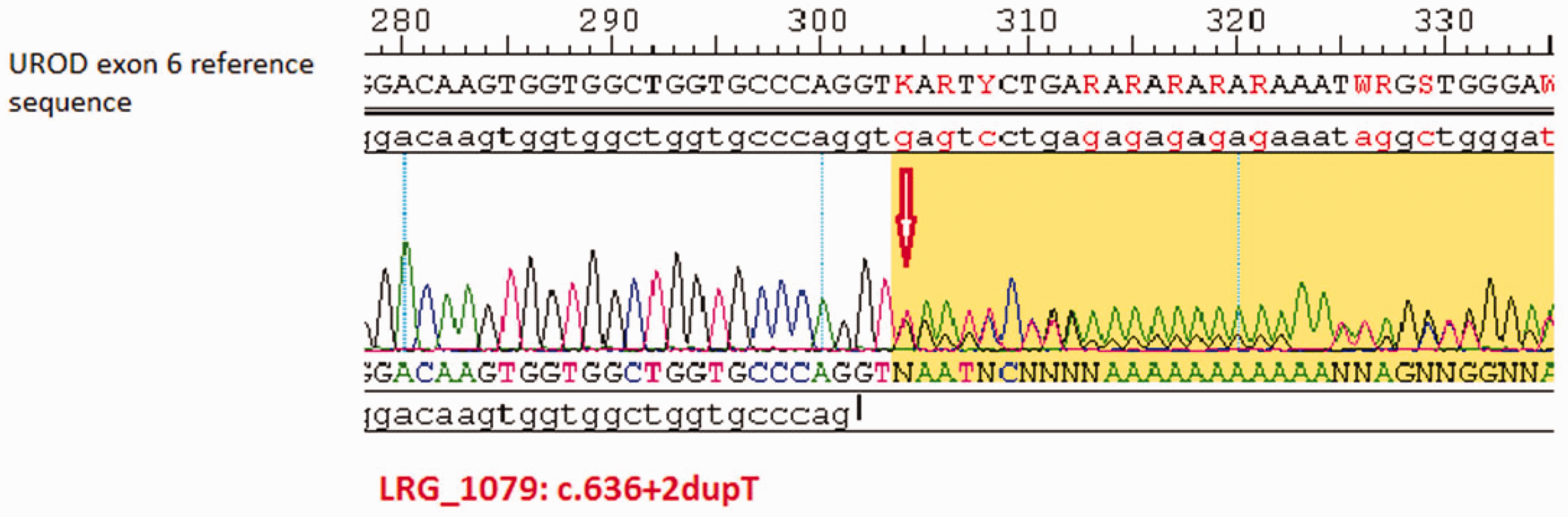
Chromatogram showing heterozygosity for c.636 + 2dupT in UROD gene.
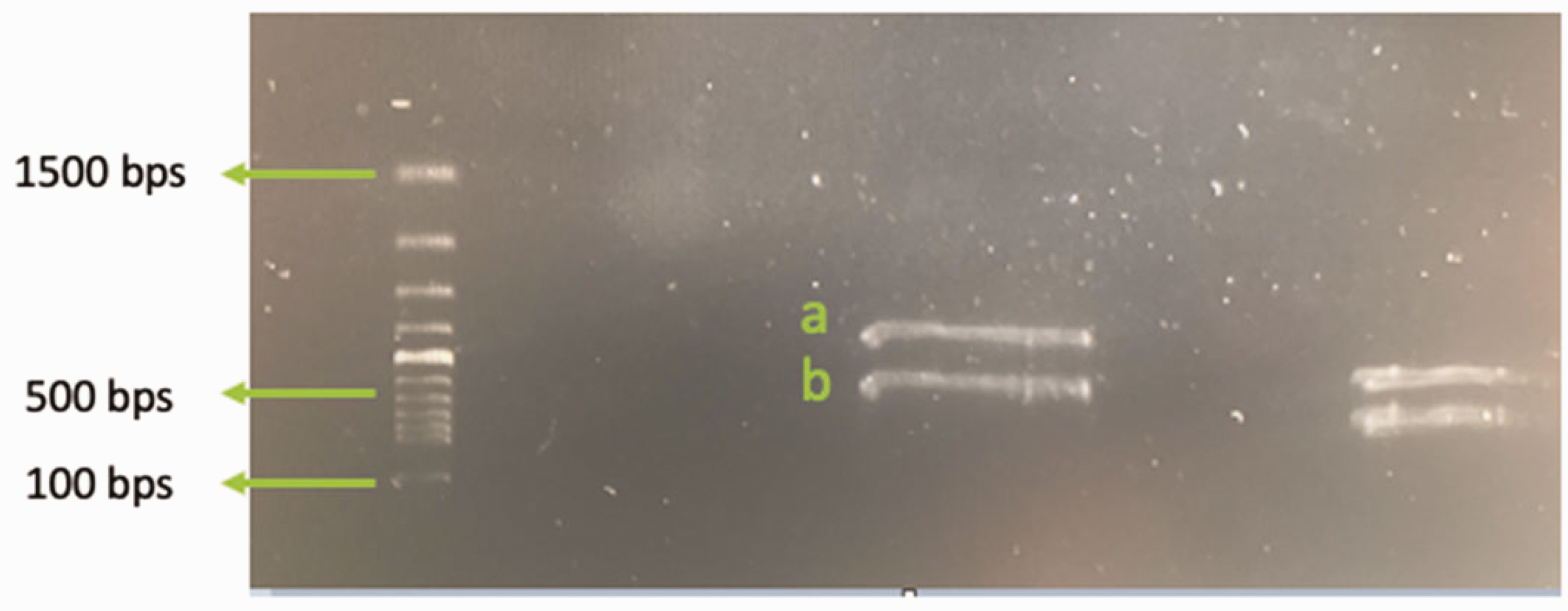
Gel electrophoresis of RT-PCR UROD product showing two different band sizes. Band (a) represents expected Wild type, whereas Band (b) is a variant band with a smaller product size representing deletion of exon 6.
Discussion
It is proposed that various common susceptibility factors for PCT causing liver iron accumulation enable the oxidative formation of uroporphomethene, a UROD inhibitor, 5 which reduces enzyme activity to less than 20% of normal.6,7 Oestrogen is another well-recognized precipitant for PCT, but the exact mechanism underpinning its role in the pathogenesis remains to be determined. Its association with PCT was initially described in men receiving diethylstilbestrol or fosfestrol for prostate cancer 8 and subsequent reports recognized an increased incidence of PCT in women taking oestrogen therapy in the form of OCPs, postmenopausal hormonal replacement and tamoxifen for breast cancer.3,9,10 Oestrogen has been shown to cause oxidative damage to kidneys in rodents and a similar effect has been proposed as a means of triggering an oxidation-mediated UROD inhibition in the liver. 11
In our patient, long-term hormonal contraception was provided using NuvaRing®, which steadily releases low doses of etonogestrel and ethinyl estradiol (EE) transvaginally. A study comparing EE pharmacokinetics between different hormonal contraceptive formulations reported markedly lower systemic exposure to EE with NuvaRing® than either a transdermal patch or OCPs. 12 These data suggest that transvaginal EE has a relatively low systemic absorption and consequently would have a much lower risk of precipitating PCT in general.
This is an important observation, as our patient did not show any clinical features of PCT while on NuvaRing® for over 10 years, despite unwittingly carrying an underlying major susceptibility UROD genetic variant. Of notable significance in this case is the fact that PCT manifested following a successful oocyte retrieval procedure, where oestrogen concentrations up to 10 times those observed during natural menstrual cycles are very commonly attained during controlled ovarian stimulation. 13 The capacity of a short-term surge in oestrogen to trigger PCT has been documented in a recent report relating to a male-to-female transgender subject 14 and also in previous reports relating to first trimester pregnancy. 15 However, in these situations, additional major susceptibility factors including high alcohol intake and heavy smoking were clearly present, and exclusion of familial PCT was not specifically undertaken.
Due to the absence of other notable susceptibility factors and hepatic iron overload in our patient, the question remained whether a single exposure to supraphysiological levels of oestrogen would be sufficient to reduce UROD activity to a level that could activate clinically apparent PCT. While the majority manifest with sporadic PCT, approximately 20% of patients have an inherited familial autosomal-dominant PCT susceptibility. 6 Therefore, UROD genetic variant scanning was performed in this case to further elucidate the pathogenesis. Although the genetic analysis confirmed the presence of a novel splice-site variant, establishing the pathogenic potential of any single nucleotide variant should be a priority before attributing direct causality. 16 In this regard, RT-PCR demonstrated the impact of the c.636 + 2 dupT variant on mRNA processing by confirming a complete deletion of exon 6, which at a minimum predicts a truncated UROD enzyme (Figure 4).
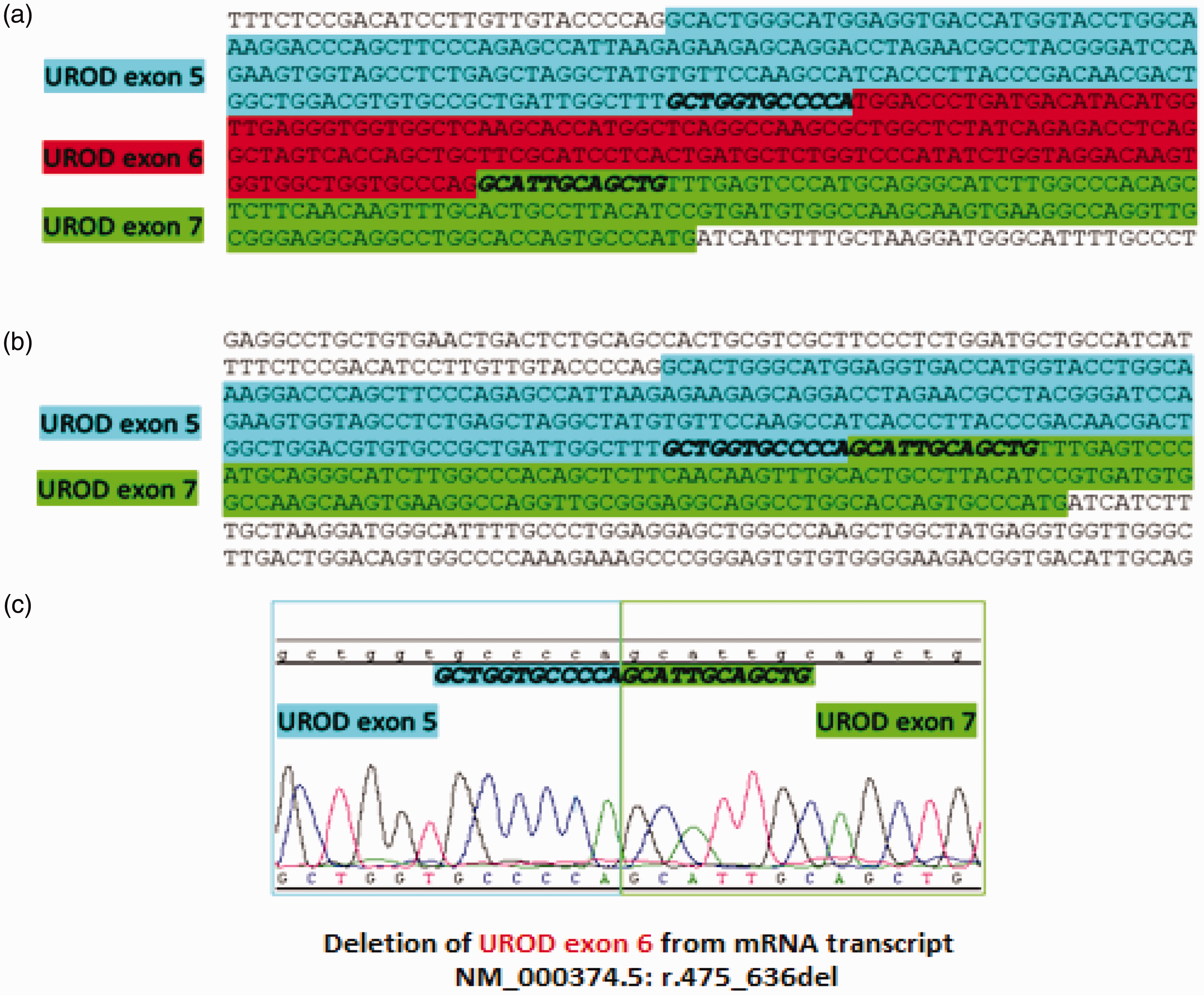
(a) Reference sequence of UROD mRNA (exons 5–7). (b) Reference sequence showing the deletion of exon 6 from UROD mRNA as a result of c.636 + 2dupT splice variant in UROD gene. (c) Chromatogram demonstrating the nucleotide sequencing of the variant RT-PCR cDNA product Band (b) from Figure 3, confirming a deletion of exon 6 from UROD mRNA transcript.
Conclusion
Overall, it is important to note that the diagnosis of PCT is primarily based on the identification of characteristic clinical features and specific findings on a biochemical porphyrin screen. Moreover, the majority of PCT cases are sporadic in nature and genetic testing for UROD mutation would not be routinely undertaken. However, familial PCT should be considered in patients presenting with a family history of PCT, in a younger age group, where there is an absence of significant iron overload or in females in whom transvaginal combined contraception is the only identifiable PCT susceptibility factor. Furthermore, in patients manifesting a photosensitive rash following IVF treatments, PCT should be considered in the differential diagnosis, including the possibility of familial PCT.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Patient gave written informed consent for the publication of this case report.
Guarantor
VC.
Contributorship
ER and VC wrote the paper. All authors reviewed and edited the article and approved the final version of the article. VC, ER, NR and POG were involved in the diagnosis and management of patient. SS, EW and NB carried out analytical work.
