Abstract

Investigation of cryoproteins requires that serum and plasma samples are maintained at 37°C during collection, transport, clotting and separation. 1 Laboratories use a wide range of approaches including thermos flasks, water, sand, heat blocks and bespoke devices to achieve this. 2 We describe here the use of pipe insulation tubing to create insulation ‘socks’ for the blood tubes, and present data validating this approach.
Insulation ‘socks’ were made by cutting pipe Insulation (9 mm Wall Eurobatex Class 0 Pipe Insulation; Advanced Insulation Manufacturing Ltd, St Helens, UK) approximately 3 cm longer than the vacutainer (3.5 mL; Becton Dickinson, Wokingham, UK), folding one end over and sealing with sticky tape.
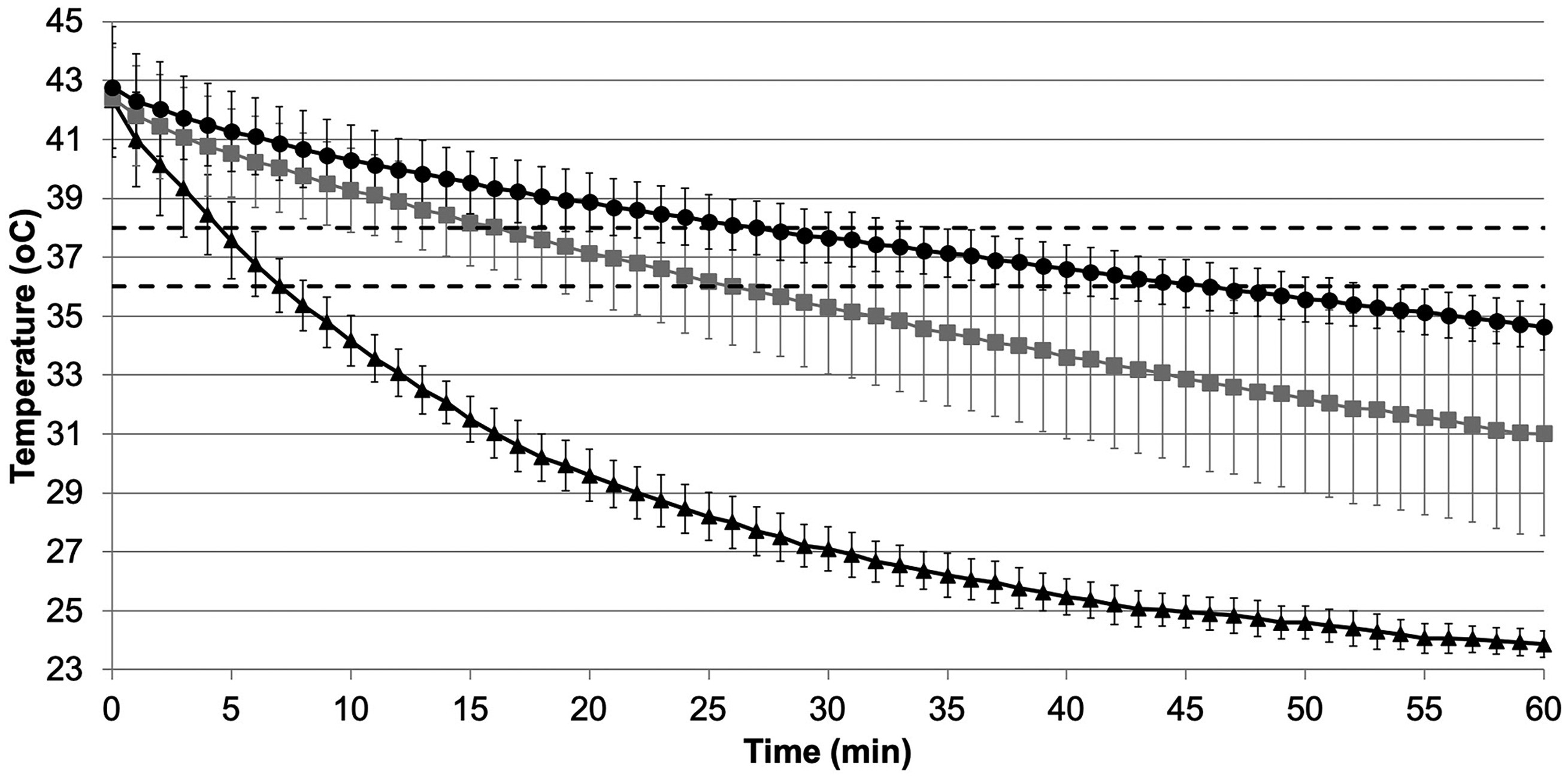
Temperature profile of water placed into prewarmed vacutainers insulated in an insulation sock alone (black triangles), a thermos flask alone (grey squares) or an insulation sock in a thermos flask (black circles). Dotted lines indicate 38°C and 36°C. Data shown is mean ± sd, n = 3.
We assessed the effectiveness of the insulation socks by comparing vacutainers placed in insulation socks alone, vacutainers placed in a thermos flask (Thermos Ltd, Leeds, UK) and vacutainers placed in an insulation sock within a thermos flask (Figure 1). The equipment was stored at 37°C in a calibrated laboratory incubator (SciQuip Incu-80S; SciQuip, Newtown, UK) for 1 h. We then filled the vacutainers with warm water (approx. 3.5 mL), inserted a temperature probe (Thermo Pro TP17; ThermoPro, iTronics, Toronto, Canada), replaced the lids and placed the vacutainers into the insulation sock and/or thermos flask. The water temperature remained within 1°C of 37°C for 20.8 ± 0.8 min, compared with 14.0 ± 7.0 min with only the thermos flask, and 3.3 ± 0.8 min with only the insulation sock (mean ± sd, n = 3). The most effective temperature maintenance was achieved using the insulation sock within a thermos flask, and this showed more consistent performance than using the thermos flask alone.
We tested the performance of the combined (insulation sock+thermos flask) if the equipment was taken out of the incubator and left on the bench for 30 min (to mimic travelling to the patient) before opening up the flask, putting warm water into the vacutainer in the insulation sock as before, adding the temperature probe, re-closing the lid of the vacutainer and thermos flask. Under these conditions, the temperature remained within 1°C of 37°C for 15.0 ± 2.2 min (mean ± sd, n = 6).
We have adopted this approach in our standard protocol. Laboratory staff sign out the equipment, go to the patient, then bring the collected samples back to the laboratory incubator and sign the equipment back in. (It is necessary to briefly remove the vacutainer from the insulation sock to connect to the needle; this was mimicked in the experiments described above.) Any collections that take longer than 20 min in total are discussed and considered when interpreting results.
The results presented here are likely to underestimate the equipment performance because the temperature probe wire prevents the lids of the vacutainer and thermos flask being closed completely. In addition, blood has a slightly lower specific heat capacity than water. However, this validation method provides a simple approach to obtain objective data on the performance of the equipment used to collect samples for cryoprotein studies. Published reports on best practice for investigating cryoproteins state that samples should be maintained at 37°C during collection, transport, clotting and separation, but there are no guidelines or criteria for how this should be done.1,2 If all laboratories validated their collection processes, performance criteria for the collection process could be agreed, and this would greatly aid the standardization of cryoprotein investigations.
Footnotes
Authors' note
Claire J Escaron's current affiliation is Clinical Immunology, Liverpool Clinical Laboratories, Liverpool University Hospitals NHS Foundation Trust, Liverpool L69 3GA, UK.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Declarations
RW.
Contributorship
JS and RDW conceived the study, RDW and CJE did the experimental work and data analysis, RDW wrote the initial draft of the article. All authors reviewed and edited the article and approved the final version.
