Abstract

Dear Sir
Faecal calprotectin is recommended and widely used as a biomarker to differentiate between inflammatory bowel disease and irritable bowel syndrome. 1 Assays require the extraction of calprotectin from stool prior to its measurement, but there is no standardised extraction method. Manual weighing, a time-consuming process, is considered the gold standard extraction method. Commercially available extraction devices are quicker, simple to operate and manufacturers claim comparable performance to manual weighing. It has, however, been recently reported that calprotectin results measured from Calex® Cap extracts were higher (mean bias 33%) compared with weighing method extracts. 2 We, therefore, re-evaluated the BÜHLMANN Calex® Stool Extraction Devices (BÜHLMANN LABORATORIES AG Schönenbuch, Switzerland) 3 and compared it to manual weighing extraction method.
Calprotectin concentrations in extracts following extraction with the Calex® and the manual weighing method were compared in 56 homogenized stool samples and in 11 external quality assurance (EQA) samples. Interbatch imprecision and stability of extracts were also evaluated. Extracts were analysed for calprotectin using BÜHLMANN fCAL® turbo reagent (Alpha Laboratories Ltd, Eastleigh, England) on an Abbott ARCHITECT c16000 (Abbott Diagnostics, Abbott Park, IL, USA).
After exclusion of 14 paired results below the limit of quantitation, Passing-Bablok linear regression of results between 26 μg/g and 1487.9 μg/g showed a constant negative bias (intercept) of –5.53 μg/g (95% confidence interval –18.33 μg/g to 14.42 μg/g) and a proportional bias (slope) of 1.02 μg/g (95% confidence interval 0.84 μg/g to 1.14 μg/g) (Figure 1). Regression analysis contained 0 and 1 for 95% CI for intercept and slope, respectively. A Bland-Altman difference plot demonstrated that the Calex® extraction devices had a mean bias of 1% (95% confidence interval of –9.1 to 11%) relative to the manual weighing method (Figure 1). Interpretation of the results was concordant for 44 (79%) of the 56 samples. There was no negative (≤50 μg/g) to positive (≥100 μg/g) discordance between the results. Result interpretation was altered as follows: 4/56 negative by manual weighing but equivocal (50–100 μ/g) by Calex®, 3/56 equivocal by manual weighing but positive by Calex® and 5/56 were positive by manual weighing and equivocal by Calex®. Eleven EQA samples extracted using Calex® devices compared well to the method laboratory trimmed mean (MLTM) produced from results of 12 laboratories over four distributions. Mean bias for Calex® versus MLTM across the four distributions was 10.7% (UK NEQAS bias limit = 75%). Eight (73%) of the 11 samples were within ± 1 standard deviation of the MLTM. Interbatch imprecision using the Calex® extraction devices was similar to the weighing extraction method (29.9% Calex® vs. 26.5% manual weighing at mean concentration of 29 μg/g and 9.3% Calex® vs. 12.3% manual weighing at mean concentration of 123 μg/g). Imprecision at low concentrations was difficult to quantify as several values were below the limit of quantitation of the assay but all values produced a negative result for both extraction methods. Faecal calprotectin in extracts prepared by Calex® devices was not significantly affected by storage at 4°C over five days. The percentage difference at day 5 compared with day 0 for samples 1, 2 and 3 was 11%, 32% and 22%, respectively. The difference between baseline and repeat at day 5 did not exceed the difference expected due to imprecision of the assay.
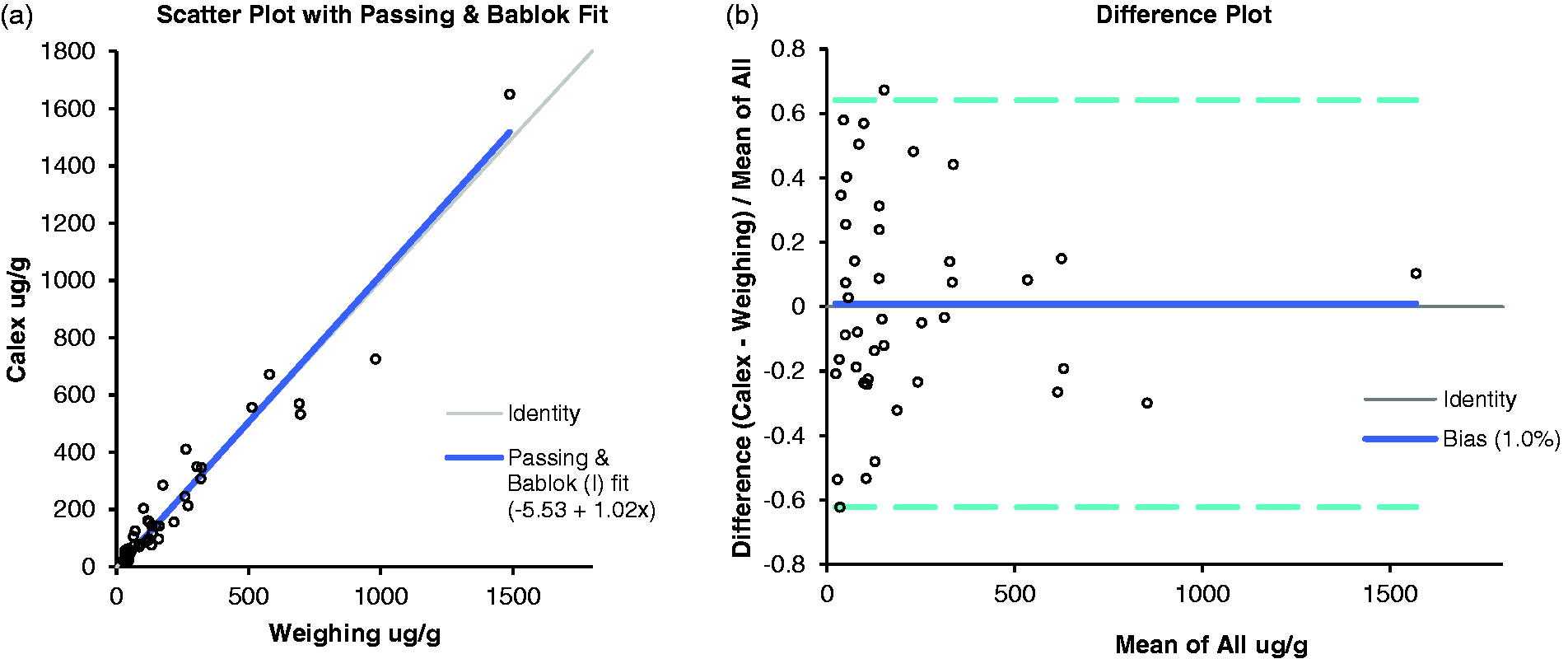
Comparison of manual weighing to commercial Calex® devices for the measurement of calprotectin following extraction from stool samples. (a) Passing-Bablok linear regression analysis and (b) Bland-Altman difference plot.
Considering the non-homogenous nature of stool samples, Calex® devices demonstrate similar accuracy and imprecision to the gold standard manual method for extracting faecal calprotectin. Our results differ from those previously reported 2 but may be explained that all assays in this report were performed by a single operative. Compared with the manual method, the Calex® was easy to use, reduced staff time, used fewer consumables (such as inoculation loops and centrifugation tubes) and improved health and safety by avoiding further direct contact with the specimen after initial sampling.
Calex® devices are fit for purpose, easy to use and offer a quicker extraction process compared with manual weighing and will therefore enable increasing demands of a faecal calprotectin service.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
RG.
Contributorship
SK designed the evaluation study, performed analyses and analysed the data and wrote the first draft. All authors critically revised and approved the final version of the article.
