Abstract

Internal quality control
The measurement of internal quality-control samples (iQC) is the cornerstone of analytical quality assurance as implemented on a daily basis in medical laboratories. 1 For many tests, iQC is effective in ensuring that analytical performance is adequate. For other tests, however, using iQC alone is insufficient to assure appropriate analytical quality. Reasons include, firstly, the absence of (stable) quality-control materials, secondly, rapid-onset or temporary critical errors (between scheduled iQC measures), thirdly, quality-control materials with non-commutability issues and, fourthly, tests with a sigma value less than or equal to 4.2,3 Although stable, preferably third-party, iQC materials are essential to perform iQC, it can be challenging or even impossible to obtain them for some tests, including those for serum indices, erythrocyte sedimentation rate or mean corpuscular volume (MCV). There may also be non-commutability issues with the iQC materials that do not properly represent for example, lot-to-lot variations of reagent or introduction of a new generation of a specific test. Such a case has been described by Hinge et al. for alkaline phosphatase, 4 and the author has experienced these kinds of issues with several immunoassay-based tests, such as PSA, CEA and vitamin D. Another limiting factor is the scheduling frequency of iQC, which becomes relevant for tests with short turnaround times that may be reported before iQC confirmation of proper analytical performance. The high degree of automation and digitization of modern medical laboratories enables rapid turnaround times for many tests. Although this is extremely valuable for urgent medical diagnostics and patient-friendly healthcare by integrated care pathways, there is always a risk of a rapid-onset of critical error due to a technical failure. When applying iQC alone, there may be a significant delay before this failure is detected. Furthermore, temporary assay failure may be undetectable by iQC due to its scheduling frequency, as has been demonstrated. 5 Finally, given its design, iQC is limited in detecting clinically relevant errors in tests with a low sigma performance value according to the sigma metrics approach. 2 Such tests, characterized by a low ratio of biological variation to analytical variation, generally require stringent control limits or rules and necessitate frequent analysis of iQC samples.6,7 However, even with such a strict iQC set-up, the probability of detecting a clinically relevant error by iQC remains limited.6,7 Despite all the efforts to improve the effectiveness of iQC, for example by introducing sigma-metrics-based iQC to reduce the false-alarm rate6,8 and by designing iQC plans based on patient risk,7,9 the abovementioned issues remain intrinsic limitations of iQC. 2 Furthermore, these more complex mathematical approaches used to increase the performance and efficiency of iQC are not only challenging to understand but also are complicated to implement in clinical practice. This is evident in the extremely low degree of adoption of these approaches, as observed in a recent survey in USA-based university hospitals. 10 Finally, iQC requires the use of iQC materials and the performing of analytical tests and therefore brings significant costs.
Patient-based real-time quality control
Patient-based real-time quality control (PBRTQC) is a generic term for the use of patient results for real-time quality-control purposes. In principle, such results can be used for quality assurance of all phases of the laboratory process (i.e. preanalytical, analytical and postanalytical). The term includes the use of so-called checks (i.e. limit checks, delta checks and multivariable checks) as described by Lindberg and clarified by Cembrowski.11,12 Most, if not all, medical laboratories already use such checks for quality assurance purposes. More recently, this term has been used particularly in the context of moving-average or average-of-normal-related techniques to support preanalytical and analytical quality control. 13 These techniques average multiple obtained results using various types of algorithms to reduce the population variation, thereby enabling the detection of systematic errors. Algorithms used include the mean, the median, the exponentially weighted moving average and the XbarB algorithm described by Bull et al.12,14 Even though these techniques were described many decades ago,14,15 very few laboratories have adopted them, although to some extent the Bull’s algorithm has been used for quality control of blood cell indices. 14 Reasons for this slow adoption include, firstly, the complexity associated with setting up and validating these rather laboratory-specific PBRTQC procedures, secondly, insufficient understanding of how to integrate them with iQC and, thirdly, inferior software features to operate PBRTQC in clinical practice.2,16 There has also been a lack of data demonstrating the added value of these techniques to support analytical quality control.
Recently, however, new methods to obtain optimized and validated PBRTQC have been described. 2 These methods are based on running realistic error detection simulations that reflect true laboratory PBRTQC performance.2,17–19 They require a manageable number of PBRTQC alarms and therefore use much stricter false-alarm criteria than iQC, which is essential for practical application of PBRTQC. A major breakthrough is that one of these methods has been made available online in the MA Generator application, so that laboratories can readily use the advanced simulation techniques to set up and validate their laboratory-specific PBRTQC procedures.20,21 Using these new optimization techniques to design PBRTQC procedures in routine practice, it has been shown that PBRTQC application is feasible and valuable for (rapid) detection of relevant errors and increases the effectiveness and cost-effectiveness of quality control plans.3,16,18,19 Insights and guidance on when to select which type of calculation algorithm for PBRTQC are needed to enhance further the performance and application of PBRTQC. In the present edition of the Annals, Smith et al. have made a first step and identified the patient test result distribution as important parameter to guide algorithm selection. Furthermore, they demonstrated that Box-Cox transformation improves the PBRTQC performance of especially analytes with skewed distributions. 22
Integration of PBRTQC and iQC
In the introduction to this editorial, several limitations of iQC were mentioned. The next pertinent question would be how PBRTQC techniques can be used to support iQC to overcome these limitations and perhaps, to design more effective and cost-effective QC plans. Firstly, since PBRTQC by design is based on patient results, it is always commutable and available, although an increasing number of test results in an analytical run significantly increases the performance of these techniques. 12 Also, when used in continuous mode by calculating a new PBRTQC value for each newly available test result, a continuous and almost real-time QC instrument becomes available. Finally, by design, PBRTQC techniques perform best for assays characterized by a low biological to analytical variation ratio. 12 These are, in fact, the tests with low sigma performance values. Due to these characteristics, PBRTQC is perfectly suitable for supplementing iQC and can be used when iQC itself is considered insufficient. 3 Furthermore, when validated PBRTQC procedures are available, for example if there are simulation analysis results and/or validation charts, PBRTQC can be used to reduce or more efficiently apply iQC when PBRTQC error detection outperforms iQC error detection or offers sufficient error detection.3,17,19 An example of a validated PBRTQC procedure by simulation analysis results presented in a MA Validation chart is shown (Figure 1).
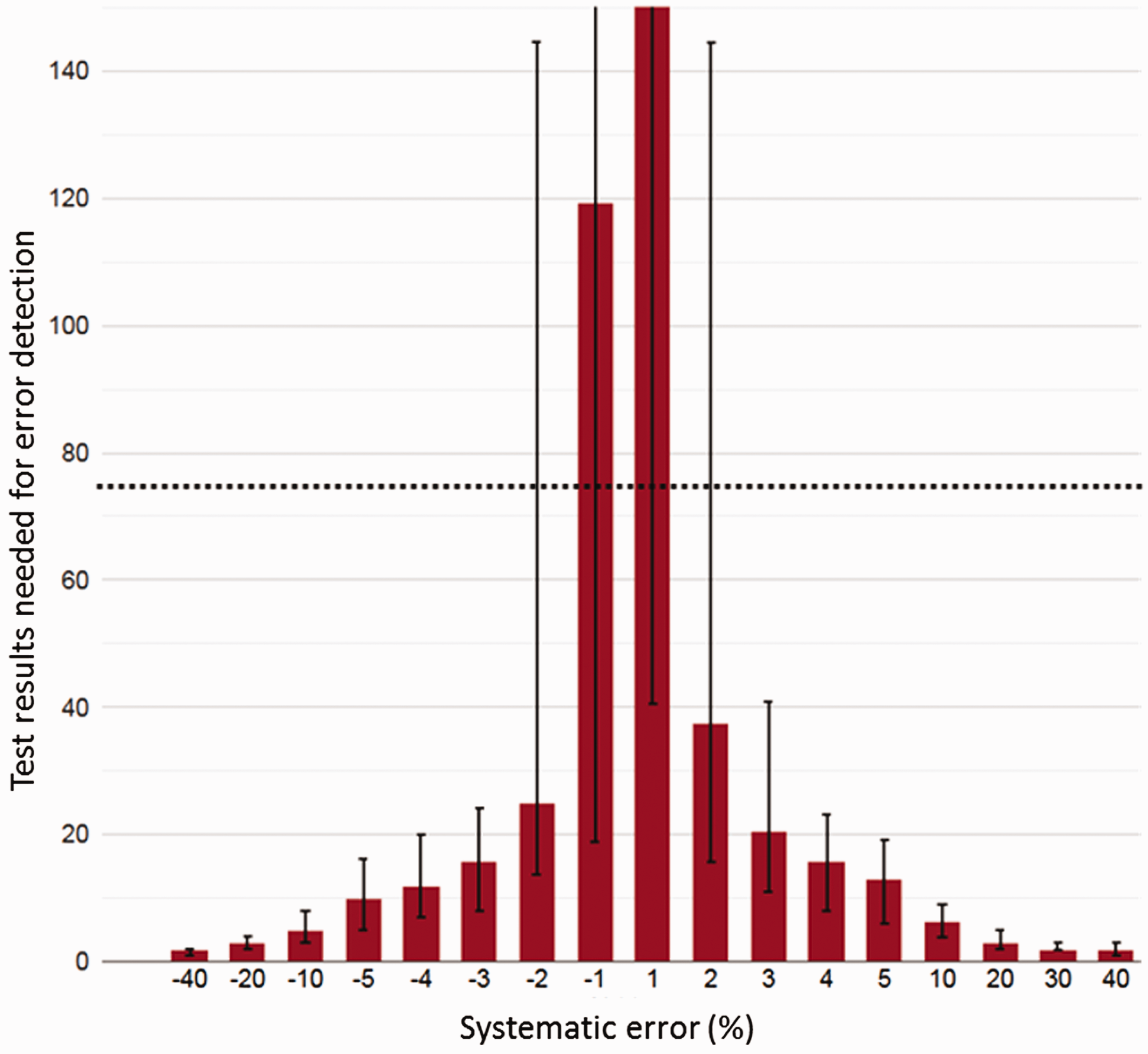
In summary, recent developments and new insights in the field of PBRTQC have made it possible to design more powerful and efficient QC plans by incorporating these techniques. Furthermore, the availability of the necessary optimization and validation methods has paved the way for a more general application of PBRTQC in medical laboratories.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The author is the owner and director of Huvaros that markets the MA Generator application. The author is a member of the IFCC working group on PBRTQC.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
HvR.
Contributorship
HvR is the sole author.
